Abstract
Excess adiposity is a risk factor for several cancers of the gastrointestinal system, specifically oesophageal adenocarcinoma and colorectal, small intestine, pancreatic, liver, gallbladder and stomach cancers. With the increasing prevalence of obesity in nearly all regions of the world, this relationship could represent a growing source of cancers of the digestive system. Experimental and molecular epidemiological studies indicate important roles for alterations in insulin signalling, adipose tissue-derived inflammation and sex hormone pathways in mediating the association between adiposity and gastrointestinal cancer. The intestinal microbiome, gut hormones and non alcoholic fatty liver disease (NAFLD) also have possible roles. However, important gaps remain in our knowledge. For instance, our understanding of how adiposity throughout the life course is related to the risk of gastrointestinal cancer development and of how obesity influences gastrointestinal cancer prognosis and survival is limited. Nonetheless, the increasing use of state-of-the-art analytical methods (such as omics technologies, Mendelian randomization and MRI) in large-scale epidemiological studies offers exciting opportunities to advance our understanding of the complex relationship between adiposity and gastrointestinal cancers. Here, we examine the epidemiology of associations between obesity and gastrointestinal cancer, explore potential mechanisms underlying these relationships and highlight important unanswered research questions.
Key points
-
A large body of epidemiological evidence supports a causal relationship between excess adiposity and elevated risk of developing gastrointestinal cancers.
-
With the rising prevalence of obesity worldwide, this relationship could represent a growing source of cancers of the gastrointestinal system.
-
Experimental and molecular epidemiological studies indicate important roles for alterations in insulin signalling, adipose tissue-derived inflammation and sex hormone pathways in mediating the association between adiposity and gastrointestinal cancer.
-
Emerging evidence suggests that the gut microbiome, gut hormones, and non alcoholic fatty liver disease might also have important roles in mediating the obesity-gastrointestinal cancer relationship.
-
The use of state-of-the-art analytical methods (omics technologies, Mendelian randomization, MRI and molecular pathological epidemiology) in large-scale epidemiological studies will probably offer novel insights into the adiposity-gastrointestinal cancer relationship.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

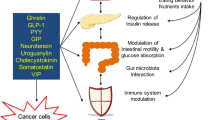
Adapted from ref.8, CC-BY-4.0.

Adapted from ref.8, CC-BY-4.0.

Similar content being viewed by others
References
WCRF-AICR. Diet, nutrition, physical activity and colorectal cancer. Continuous Update Project http://www.wcrf.org/sites/default/files/CUP%20Colorectal%20Report_2017_Digital.pdf (2017).
WCRF-AICR. Diet, nutrition, physical activity and liver cancer. Continuous Update Project http://www.wcrf.org/sites/default/files/Liver-Cancer-2015-Report.pdf (2015).
WCRF-AICR. Diet, nutrition, physical activity and oesophageal cancer. Continuous Update Project http://www.wcrf.org/sites/default/files/Oesophageal-cancer-report.pdf (2016).
WCRF-AICR. Food, nutrition, physical activity, and the prevention of pancreatic cancer. Continuous Update Project http://www.wcrf.org/sites/default/files/Pancreatic-Cancer-2012-Report.pdf (2012).
WCRF-AICR. Diet, nutrition, physical activity and stomach cancer. Continuous Update Project http://www.wcrf.org/sites/default/files/Stomach-Cancer-2016-Report.pdf (2016).
Lauby-Secretan, B. et al. Body fatness and cancer — viewpoint of the IARC Working Group. N. Engl. J. Med. 375, 794–798 (2016).
WCRF-AICR. Diet, nutrition, physical activity and gallbladder cancer. Continuous Update Project http://www.wcrf.org/sites/default/files/Gallbladder-Cancer-2015-Report.pdf (2015).
NCD Risk Factor Collaboration. Trends in adult body-mass index in 200 countries from 1975 to 2014: a pooled analysis of 1698 population-based measurement studies with 19.2 million participants. Lancet 387, 1377–1396 (2016).
Lin, K. J., Cheung, W. Y., Lai, J. Y.-C. & Giovannucci, E. L. The effect of estrogen versus combined estrogen-progestogen therapy on the risk of colorectal cancer. Int. J. Cancer 130, 419–430 (2012).
Terry, P. D., Miller, A. B. & Rohan, T. E. Obesity and colorectal cancer risk in women. Gut 51, 191–194 (2002).
Pischon, T. et al. Body size and risk of colon and rectal cancer in the European Prospective Investigation Into Cancer and Nutrition (EPIC). J. Natl Cancer Inst. 98, 920–931 (2006).
Adams, K. F. et al. Body mass and colorectal cancer risk in the NIH–AARP cohort. Am. J. Epidemiol. 166, 36–45 (2007).
Keimling, M. et al. Comparison of associations of body mass index, abdominal adiposity, and risk of colorectal cancer in a large prospective cohort study. Cancer Epidemiol. Biomarkers Prev. 22, 1383–1394 (2013).
Ortega, L. S. et al. A prospective investigation of body size, body fat composition and colorectal cancer risk in the UK Biobank. Sci. Rep. 7, 17807 (2017).
Davey Smith, G. & Hemani, G. Mendelian randomization: genetic anchors for causal inference in epidemiological studies. Hum. Mol. Genet. 23, R89–R98 (2014).
Sheehan, N. A., Didelez, V., Burton, P. R. & Tobin, M. D. Mendelian randomisation and causal inference in observational epidemiology. PLOS Med. 5, e177 (2008).
Jarvis, D. et al. Mendelian randomisation analysis strongly implicates adiposity with risk of developing colorectal cancer. Br. J. Cancer 115, 266–272 (2016).
Campbell, P. T. et al. Case–control study of overweight, obesity, and colorectal cancer risk, overall and by tumor microsatellite instability status. J. Natl Cancer Inst. 102, 391–400 (2010).
Hughes, L. A. E. et al. Body size and risk for colorectal cancers showing BRAF mutations or microsatellite instability: a pooled analysis. Int. J. Epidemiol. 41, 1060–1072 (2012).
Morikawa, T. et al. Prospective analysis of body mass index, physical activity, and colorectal cancer risk associated with β-catenin (CTNNB1) status. Cancer Res. 73, 1600–1610 (2013).
Kuchiba, A. et al. Body mass index and risk of colorectal cancer according to fatty acid synthase expression in the Nurses’ Health Study. J. Natl Cancer Inst. 104, 415–420 (2012).
Zhang, X. et al. Early life body fatness and risk of colorectal cancer in U. S. women and men — results from two large cohort studies. Cancer Epidemiol. Biomarkers Prev. 24, 690–697 (2015).
Arnold, M. et al. Duration of adulthood overweight, obesity, and cancer risk in the Women’s Health Initiative: a longitudinal study from the United States. PLOS Med. 13, e1002081 (2016).
Fedirko, V. et al. Pre-diagnostic anthropometry and survival after colorectal cancer diagnosis in Western European populations. Int. J. Cancer 135, 1949–1960 (2014).
Campbell, P. T. et al. Impact of body mass index on survival after colorectal cancer diagnosis: The Cancer Prevention Study-II Nutrition Cohort. J. Clin. Oncol. 30, 42–52 (2012).
Caan, B. J. et al. Explaining the obesity paradox: the association between body composition and colorectal cancer survival (C-SCANS Study). Cancer Epidemiol. Biomarkers Prev. 26, 1008–1015 (2017).
Cross, A. J., Hollenbeck, A. R. & Park, Y. A large prospective study of risk factors for adenocarcinomas and malignant carcinoid tumors of the small intestine. Cancer Causes Control 24, 1737–1746 (2013).
Lu, Y. et al. Comparison of abdominal adiposity and overall obesity in relation to risk of small intestinal cancer in a European Prospective Cohort. Cancer Causes Control 27, 919–927 (2016).
Aune, D. et al. Body mass index, abdominal fatness and pancreatic cancer risk: a systematic review and non-linear dose–response meta-analysis of prospective studies. Ann. Oncol. 23, 843–852 (2012).
Carreras-Torres, R. et al. The role of obesity, type 2 diabetes, and metabolic factors in pancreatic cancer: a Mendelian randomization study. J. Natl Cancer Inst. 109, djx012 (2017).
Nogueira, L., Stolzenberg-Solomon, R., Gamborg, M., Sørensen, T. I. & Baker, J. L. Childhood body mass index and risk of adult pancreatic cancer. Curr. Dev. Nutr. 1, e001362 (2017).
Stolzenberg-Solomon, R. Z., Schairer, C., Moore, S., Hollenbeck, A. & Silverman, D. T. Lifetime adiposity and risk of pancreatic cancer in the NIH-AARP Diet and Health Study cohort. Am. J. Clin. Nutr. 98, 1057–1065 (2013).
Pang, Y. et al. Young adulthood and adulthood adiposity in relation to incidence of pancreatic cancer: a prospective study of 0.5 million Chinese adults and a meta-analysis. J. Epidemiol. Commun. Health 71, 1059–1067 (2017).
Yuan, C. et al. Prediagnostic body mass index and pancreatic cancer survival. J. Clin. Oncol. 31, 4229–4234 (2013).
Shi, Y.-Q. et al. Effect of body mass index on overall survival of pancreatic cancer: a meta-analysis. Medicine 95, e3305 (2016).
Campbell, P. T. et al. Body mass index, waist circumference, diabetes, and risk of liver cancer for U. S. adults. Cancer Res. 76, 6076–6083 (2016).
Schlesinger, S. et al. Abdominal obesity, weight gain during adulthood and risk of liver and biliary tract cancer in a European cohort. Int. J. Cancer 132, 645–657 (2013).
Marrero, J. A. et al. Alcohol, tobacco and obesity are synergistic risk factors for hepatocellular carcinoma. J. Hepatol. 42, 218–224 (2005).
Venook, A. P., Papandreou, C., Furuse, J. & Ladrón de Guevara, L. The incidence and epidemiology of hepatocellular carcinoma: a global and regional perspective. Oncology 15, 5–13 (2010).
Yang, B. et al. Adiposity across the adult life course and incidence of primary liver cancer: the NIH-AARP cohort. Int. J. Cancer 141, 271–278 (2017).
Campbell, P. T. et al. Body Size Indicators and Risk of Gallbladder Cancer: Pooled Analysis of Individual-Level Data from 19 Prospective Cohort Studies. Cancer Epidemiol. Biomarkers amp; Prevention 26, 597–606 (2017).
Petrick, J. L. et al. Body weight trajectories and risk of oesophageal and gastric cardia adenocarcinomas: a pooled analysis of NIH-AARP and PLCO Studies. Br. J. Cancer 116, 951–959 (2017).
Steffen, A. et al. General and abdominal obesity and risk of esophageal and gastric adenocarcinoma in the European Prospective Investigation into Cancer and Nutrition. Int. J. Cancer 137, 646–657 (2015).
Thrift, A. P. et al. Obesity and Risk of esophageal adenocarcinoma and Barrett’s esophagus: a mendelian randomization study. J. Natl Cancer Inst. 106, dju252 (2014).
Song, M. et al. Trajectory of body shape across the lifespan and cancer risk. Int. J. Cancer 138, 2383–2395 (2016).
Song, H. et al. Body mass index change during adulthood and risk of oesophageal squamous-cell carcinoma in a Japanese population: the Japan Public Health (JPHC)-based prospective study. Br. J. Cancer 117, 1715–1722 (2017).
Bezemer, I. D. et al. C-peptide, IGF-I, sex-steroid hormones and adiposity: a cross-sectional study in healthy women within the European Prospective Investigation into Cancer and Nutrition (EPIC). Cancer Causes Control 16, 561–572 (2005).
Pollak, M. N., Schernhammer, E. S. & Hankinson, S. E. Insulin-like growth factors and neoplasia. Nat. Rev. Cancer 4, 505–518 (2004).
Giorgino, F. et al. Overexpression of insulin receptors in fibroblast and ovary cells induces a ligand-mediated transformed phenotype. Mol. Endocrinol. 5, 452–459 (1991).
Ish-Shalom, D. et al. Mitogenic properties of insulin and insulin analogues mediated by the insulin receptor. Diabetologia 40 (Suppl. 2), S25–S31 (1997).
Tran, T. T. et al. Hyperinsulinemia, but not other factors associated with insulin resistance, acutely enhances colorectal epithelial proliferation in vivo. Endocrinology 147, 1830–1837 (2006).
Chettouh, H. et al. Mitogenic Insulin receptor-A is overexpressed in human hepatocellular carcinoma due to EGFR-mediated dysregulation of RNA splicing factors. Cancer Res. 73, 3974–3986 (2013).
Kiunga, G. A. et al. Elevated insulin receptor protein expression in experimentally induced colonic tumors. Cancer Lett. 211, 145–153 (2004).
Santoro, M. A. et al. Reduced insulin-like growth factor I receptor and altered insulin receptor isoform mRNAs in normal mucosa predict colorectal adenoma risk. Cancer Epidemiol. Biomarkers Prev. 23, 2093–2100 (2014).
Spector, S. A. et al. Human insulin receptor and insulin signaling proteins in hepatic disease. J. Surg. Res. 83, 32–35 (1999).
Belfiore, A. et al. Insulin receptor isoforms in physiology and disease: an updated view. Endocr. Rev. 38, 379–431 (2017).
Schoen, R. E. et al. Increased blood glucose and insulin, body size, and incident colorectal cancer. J. Natl Cancer Inst. 91, 1147–1154 (1999).
Limburg, P. J. et al. Insulin, glucose, insulin resistance and incident colorectal cancer in male smokers. Clin. Gastroenterol. Hepatol. 4, 1514–1521 (2006).
Gunter, M. J. et al. Insulin, insulin-like growth factor-I, endogenous estradiol, and risk of colorectal cancer in postmenopausal women. Cancer Res. 68, 329–337 (2008).
Palmqvist, R. et al. Plasma insulin, IGF-binding proteins-1 and -2 and risk of colorectal cancer: a prospective study in Northern Sweden. Int. J. Cancer 107, 89–93 (2003).
Saydah, S. H. et al. Association of markers of insulin and glucose control with subsequent colorectal cancer risk. Cancer Epidemiol. Biomarkers Prev. 12, 412–418 (2003).
Kaaks, R. et al. Serum C-peptide, insulin-like growth factor (IGF)-I, IGF-binding proteins, and colorectal cancer risk in women. J. Natl Cancer Inst. 92, 1592–1600 (2000).
Otani, T., Iwasaki, M., Sasazuki, S., Inoue, M. & Tsugane, S. Plasma C-peptide, insulin-like growth factor-I, insulin-like growth factor binding proteins and risk of colorectal cancer in a nested case-control study: the Japan public health center-based prospective study. Int. J. Cancer 120, 2007–2012 (2007).
Jenab, M. et al. Serum C-peptide, IGFBP-1 and IGFBP-2 and risk of colon and rectal cancers in the European Prospective Investigation into Cancer and Nutrition. Int. J. Cancer 121, 368–376 (2007).
Wei, E. K. et al. A prospective study of C-peptide, insulin-like growth factor-I, insulin-like growth factor binding protein-1, and the risk of colorectal cancer in women. Cancer Epidemiol. Biomarkers Prev. 14, 850–855 (2005).
Chen, L. et al. Circulating C-peptide level is a predictive factor for colorectal neoplasia: evidence from the meta-analysis of prospective studies. Cancer Causes Control 24, 1837–1847 (2013).
Murphy, N. et al. A nested case-control study of metabolically defined body size phenotypes and risk of colorectal cancer in the European Prospective Investigation into Cancer and Nutrition (EPIC). PLOS Med. 13, e1001988 (2016).
Grote, V. A. et al. Diabetes mellitus, glycated haemoglobin and C-peptide levels in relation to pancreatic cancer risk: a study within the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort. Diabetologia 54, 3037–3046 (2011).
Wolpin, B. M. et al. Hyperglycemia, insulin resistance, impaired pancreatic β-cell function, and risk of pancreatic cancer. J. Natl Cancer Inst. 105, 1027–1035 (2013).
Aleksandrova, K. et al. Inflammatory and metabolic biomarkers and risk of liver and biliary tract cancer. Hepatology 60, 858–871 (2014).
Hidaka, A. et al. Plasma insulin, C-peptide and blood glucose and the risk of gastric cancer: the Japan Public Health Center-based prospective study. Int. J. Cancer 136, 1402–1410 (2015).
Valentinis, B. & Baserga, R. IGF-I receptor signalling in transformation and differentiation. Mol. Pathol. 54, 133–137 (2001).
Rinaldi, S. et al. Serum levels of IGF-I, IGFBP-3 and colorectal cancer risk: results from the EPIC cohort, plus a meta-analysis of prospective studies. Int. J. Cancer 126, 1702–1715 (2010).
Douglas, J. B. et al. Serum IGF-I, IGF-II, IGFBP-3, and IGF-I/IGFBP-3 molar ratio and risk of pancreatic cancer in the prostate, lung, colorectal, and ovarian cancer screening trial. Cancer Epidemiol. Biomarkers Prev. 19, 2298–2306 (2010).
Rohrmann, S. et al. Concentrations of IGF-I and IGFBP-3 and pancreatic cancer risk in the European Prospective Investigation into Cancer and Nutrition. Br. J. Cancer 106, 1004–1010 (2012).
Lukanova, A. et al. Prediagnostic plasma testosterone, sex hormone-binding globulin, IGF-I and hepatocellular carcinoma: etiological factors or risk markers? Int. J. Cancer 134, 164–173 (2014).
Endogenous Hormones and Breast Cancer Collaborative Group. Insulin-like growth factor 1 (IGF1), IGF binding protein 3 (IGFBP3), and breast cancer risk: pooled individual data analysis of 17 prospective studies. Lancet Oncol. 11, 530–542 (2010).
Tannenbaum, G., Guyda, H. & Posner, B. Insulin-like growth factors: a role in growth hormone negative feedback and body weight regulation via brain. Science 220, 77–79 (1983).
Allen, N. E. et al. Lifestyle determinants of serum insulin-like growth-factor-I (IGF-I), C-peptide and hormone binding protein levels in British women. Cancer Causes Control 14, 65–74 (2003).
Lee, K.-W. et al. Rapid apoptosis induction by IGFBP-3 involves an insulin-like growth factor-independent nucleomitochondrial translocation of RXRα/Nur77. J. Biol. Chem. 280, 16942–16948 (2005).
Baxter, R. C. Insulin-like growth factor binding protein-3 (IGFBP-3): novel ligands mediate unexpected functions. J. Cell Commun. Signal. 7, 179–189 (2013).
Crusz, S. M. & Balkwill, F. R. Inflammation and cancer: advances and new agents. Nat. Rev. Clin. Oncol. 12, 584 (2015).
Zhou, B. et al. C-Reactive protein, interleukin-6 and the risk of colorectal cancer: a meta-analysis. Cancer Causes Control 25, 1397–1405 (2014).
Nimptsch, K. et al. Association of CRP genetic variants with blood concentrations of C-reactive protein and colorectal cancer risk. Int. J. Cancer 136, 1181–1192 (2015).
Kant, P. & Hull, M. A. Excess body weight and obesity[mdash]the link with gastrointestinal and hepatobiliary cancer. Nat. Rev. Gastroenterol. Hepatol. 8, 224–238 (2011).
Vaiopoulos, A. G., Marinou, K., Christodoulides, C. & Koutsilieris, M. The role of adiponectin in human vascular physiology. Int. J. Cardiol. 155, 188–193 (2012).
Wei, E. K., Giovannucci, E., Fuchs, C. S., Willett, W. C. & Mantzoros, C. S. Low Plasma adiponectin levels and risk of colorectal cancer in men: a prospective study. J. Natl Cancer Inst. 97, 1688–1694 (2005).
Aleksandrova, K. et al. Leptin and soluble leptin receptor in risk of colorectal cancer in the European Prospective Investigation into Cancer and Nutrition Cohort. Cancer Res. 72, 5328–5337 (2012).
Stattin, P. et al. Obesity and colon cancer: does leptin provide a link? Int. J. Cancer 109, 149–152 (2004).
Ho, G. Y. F. et al. Adipokines linking obesity with colorectal cancer risk in postmenopausal women. Cancer Res. 72, 3029–3037 (2012).
Bao, Y. et al. A prospective study of plasma adiponectin and pancreatic cancer risk in five US cohorts. J. Natl Cancer Inst. 105, 95–103 (2013).
Bao, Y. et al. Inflammatory plasma markers and pancreatic cancer risk: a prospective study of five U. S. cohorts. Cancer Epidemiol. Biomarkers Prev. 22, 855–861 (2013).
Ohishi, W. et al. Serum interleukin-6 associated with hepatocellular carcinoma risk: a nested case–control study. Int. J. Cancer 134, 154–163 (2014).
Endogenous Hormones and Breast Cancer Collaborative Group. Circulating sex hormones and breast cancer risk factors in postmenopausal women: reanalysis of 13 studies. Br. J. Cancer 105, 709–722 (2011).
Wallace, I. R., McKinley, M. C., Bell, P. M. & Hunter, S. J. Sex hormone binding globulin and insulin resistance. Clin. Endocrinol. 78, 321–329 (2013).
Hartman, J. et al. Tumor repressive functions of estrogen receptor β in SW480 colon cancer cells. Cancer Res. 69, 6100–6106 (2009).
Konduri, S. & Schwarz, R. E. Estrogen receptor β/α ratio predicts response of pancreatic cancer cells to estrogens and phytoestrogens. J. Surg. Res. 140, 55–66 (2007).
Waliszewski, P. et al. Molecular study of sex steroid receptor gene expression in human colon and in colorectal carcinomas. J. Surg. Oncol. 64, 3–11 (1997).
Castiglione, F. et al. Expression of estrogen receptor beta in colon cancer progression. Diagn. Mol. Pathol. 17, 231–236 (2008).
Murphy, N. et al. A prospective evaluation of endogenous sex hormone levels and colorectal cancer risk in postmenopausal women. J. Natl Cancer Inst. 107, djv210 (2015).
Clendenen, T. V. et al. Postmenopausal levels of endogenous sex hormones and risk of colorectal cancer. Cancer Epidemiol. Biomarkers Prev. 18, 275–281 (2009).
Lin, J. H. et al. Association between sex hormones and colorectal cancer risk in men and women. Clin. Gastroenterol. Hepatol. 11, 419–424 (2013).
Hampel, H., Abraham, N. S. & El-Serag, H. B. Meta-analysis: obesity and the risk for gastroesophageal reflux disease and its complications. Ann. Internal Med. 143, 199–211 (2005).
Friedenberg, F. K., Xanthopoulos, M., Foster, G. D. & Richter, J. E. The association between gastroesophageal reflux disease and obesity. Am. J. Gastroenterol. 103, 2111 (2008).
Lagergren, J., Bergström, R., Lindgren, A. & Nyrén, O. Symptomatic gastroesophageal reflux as a risk factor for esophageal adenocarcinoma. N. Engl. J. Med. 340, 825–831 (1999).
Rubenstein, J. H. & Taylor, J. B. Meta-analysis: the association of oesophageal adenocarcinoma with symptoms of gastro-oesophageal reflux. Aliment. Pharmacol. Ther. 32, 1222–1227 (2010).
Derakhshan, M. H. et al. Combination of gastric atrophy, reflux symptoms and histological subtype indicates two distinct aetiologies of gastric cardia cancer. Gut 57, 298–305 (2008).
Figueroa, J. D. et al. Cigarette smoking, body mass index, gastro-esophageal reflux disease, and non-steroidal anti-inflammatory drug use and risk of subtypes of esophageal and gastric cancers by P53 overexpression. Cancer Causes Control 20, 361–368 (2009).
Karimi, P., Islami, F., Anandasabapathy, S., Freedman, N. D. & Kamangar, F. Gastric cancer: descriptive epidemiology, risk factors, screening, and prevention. Cancer Epidemiol. Biomarkers Prev. 23, 700–713 (2014).
Turnbaugh, P. J. et al. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 444, 1027–1031 (2006).
Turnbaugh, P. J. et al. A core gut microbiome in obese and lean twins. Nature 457, 480–484 (2009).
Boulangé, C. L., Neves, A. L., Chilloux, J., Nicholson, J. K. & Dumas, M.-E. Impact of the gut microbiota on inflammation, obesity, and metabolic disease. Genome Med. 8, 42 (2016).
Cani, P. D. et al. Changes in gut microbiota control metabolic endotoxemia-induced inflammation in high-fat diet-induced obesity and diabetes in mice. Diabetes 57, 1470–1481 (2008).
Higgins, S. C., Gueorguiev, M. & Korbonits, M. Ghrelin, the peripheral hunger hormone. Ann. Med. 39, 116–136 (2007).
Baatar, D., Patel, K. & Taub, D. D. The effects of ghrelin on inflammation and the immune system. Mol. Cell. Endocrinol. 340, 44–58 (2011).
Shiiya, T. et al. Plasma ghrelin levels in lean and obese humans and the effect of glucose on ghrelin secretion. J. Clin. Endocrinol. Metab. 87, 240–244 (2002).
Murphy, G. et al. The relationship between serum ghrelin and the risk of gastric and esophagogastric junctional adenocarcinomas. J. Natl Cancer Inst. 103, 1123–1129 (2011).
Murphy, G. et al. Serum ghrelin is associated with risk of colorectal adenocarcinomas in the ATBC study. Gut https://doi.org/10.1136/gutjnl-2016-313157 (2017).
Sundkvist, A., Myte, R., Palmqvist, R., Harlid, S. & Van Guelpen, B. Plasma ghrelin is probably not a useful biomarker for risk prediction or early detection of colorectal cancer. Gut https://doi.org/10.1136/gutjnl-2018-316110 (2018).
Ruhl, C. E. & Everhart, J. E. Determinants of the association of overweight with elevated serum alanine aminotransferase activity in the United States. Gastroenterology 124, 71–79 (2003).
Fabbrini, E., Sullivan, S. & Klein, S. Obesity and Nonalcoholic fatty liver disease: biochemical, metabolic and clinical implications. Hepatology 51, 679–689 (2010).
Michelotti, G. A., Machado, M. V. & Diehl, A. M. NAFLD, NASH and liver cancer. Nat. Rev. Gastroenterol. Hepatol. 10, 656 (2013).
Renehan, A. G., Zwahlen, M. & Egger, M. Adiposity and cancer risk: new mechanistic insights from epidemiology. Nat. Rev. Cancer 15, 484–498 (2015).
Kim, G.-A. et al. Association between non-alcoholic fatty liver disease and cancer incidence rate. J. Hepatol. 68, 140–146 (2018).
Wong, V. W.-S. et al. High prevalence of colorectal neoplasm in patients with non-alcoholic steatohepatitis. Gut 60, 829–836 (2011).
Pearson-Stuttard, J. et al. Worldwide burden of cancer attributable to diabetes and high body mass index: a comparative risk assessment. Lancet Diabetes Endocrinol. 6, 95–104 (2017).
Arnold, M. et al. Global patterns and trends in colorectal cancer incidence and mortality. Gut 66, https://doi.org/10.1136/gutjnl-2015-310912 (2016).
Würtz, P. et al. Metabolic signatures of adiposity in young adults: Mendelian randomization analysis and effects of weight change. PLOS Med. 11, e1001765 (2014).
Stepien, M. et al. Alteration of amino acid and biogenic amine metabolism in hepatobiliary cancers: findings from a prospective cohort study. Int. J. Cancer 138, 348–360 (2016).
Keum, N., Lee, D. H., Kim, R., Greenwood, D. C. & Giovannucci, E. L. Visceral adiposity and colorectal adenomas: dose-response meta-analysis of observational studies. Ann. Oncol. 26, 1101–1109 (2015).
UK-Biobank. Protocol for a large-scale prospective epidemiological resource. UK Biobank http://www.ukbiobank.ac.uk/wp-content/uploads/2011/11/UK-Biobank-Protocol.pdf (2010).
Allen, N. et al. UK Biobank: Current status and what it means for epidemiology. Health Policy Technol. 1, 123–126 (2012).
German National Cohort Consortium. The German National Cohort: aims, study design and organization. Eur. J. Epidemiol. 29, 371–382 (2014).
Ogino, S. et al. The role of molecular pathological epidemiology in the study of neoplastic and non-neoplastic diseases in the era of precision medicine. Epidemiology 27, 602–611 (2016).
Ogino, S., Chan, A. T., Fuchs, C. S. & Giovannucci, E. Molecular pathological epidemiology of colorectal neoplasia: an emerging transdisciplinary and interdisciplinary field. Gut 60, 397–411 (2011).
Colussi, D., Brandi, G., Bazzoli, F. & Ricciardiello, L. Molecular Pathways involved in colorectal cancer: implications for disease behavior and prevention. Int. J. Mol. Sci. 14, 16365 (2013).
Nishihara, R. et al. Molecular pathological epidemiology gives clues to paradoxical findings. Eur. J. Epidemiol. 30, 1129–1135 (2015).
Holmes, M. V., Ala-Korpela, M. & Smith, G. D. Mendelian randomization in cardiometabolic disease: challenges in evaluating causality. Nat. Rev. Cardiol. 14, 577 (2017).
Bowden, J., Davey Smith, G. & Burgess, S. Mendelian randomization with invalid instruments: effect estimation and bias detection through Egger regression. Int. J. Epidemiol. 44, 512–525 (2015).
Bowden, J., Davey Smith, G., Haycock, P. C. & Burgess, S. Consistent estimation in Mendelian randomization with some invalid instruments using a weighted median estimator. Genet. Epidemiol. 40, 304–314 (2016).
O’Doherty, M. G., Freedman, N. D., Hollenbeck, A. R., Schatzkin, A. & Abnet, C. C. A prospective cohort study of obesity and risk of oesophageal and gastric adenocarcinoma in the NIH–AARP Diet and Health Study. Gut 61, 1261–1268 (2012).
Author information
Authors and Affiliations
Contributions
M.J.G. and N.M. researched data for the article. All authors made substantial contributions to discussion of content, wrote the article and reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Murphy, N., Jenab, M. & Gunter, M.J. Adiposity and gastrointestinal cancers: epidemiology, mechanisms and future directions. Nat Rev Gastroenterol Hepatol 15, 659–670 (2018). https://doi.org/10.1038/s41575-018-0038-1
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41575-018-0038-1
This article is cited by
-
Allometric versus traditional body-shape indices and risk of colorectal cancer: a Mendelian randomization analysis
International Journal of Obesity (2024)
-
The prevalence and determinant of overweight and obesity among residents aged 40–69 years in high-risk regions for upper gastrointestinal cancer in southeast China
Scientific Reports (2023)
-
Association of sugar-sweetened beverages with the risk of colorectal cancer: a systematic review and meta-analysis
European Journal of Clinical Nutrition (2023)
-
Modulation of rectal cancer stemness, patient outcome and therapy response by adipokines
Journal of Physiology and Biochemistry (2023)
-
Circulating hormones and risk of gastric cancer by subsite in three cohort studies
Gastric Cancer (2023)