Abstract
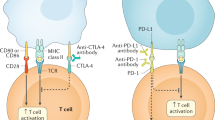
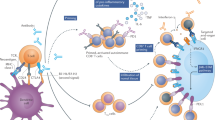
Immune checkpoint inhibitors (ICIs) are monoclonal antibodies that target two key signalling pathways related to T cell activation and exhaustion, by binding to and inhibiting cytotoxic T lymphocyte antigen 4 (CTLA4) or PD1 and its ligand PDL1. ICIs, such as nivolumab, pembrolizumab and ipilimumab, are approved for the treatment of numerous and diverse cancer types, in various combination regimens, and are now an established cornerstone of cancer therapeutics. Toxicities induced by ICIs are autoimmune in nature and are referred to as immune-related adverse events (irAEs); these events can affect any organ system in an unpredictable fashion. Importantly, irAEs can manifest as endocrinopathies involving the thyroid (hypothyroidism or thyrotoxicosis), pituitary (hypophysitis), adrenal glands (adrenal insufficiency) and pancreas (diabetes mellitus). These events are a frequent source of acute and persistent morbidity in patients treated with ICIs and can even be fatal. Over the past few years, there has been a growing understanding of the underlying pathogenesis of irAEs that has led to the development of more effective management strategies. Herein, we review the current understanding of the pathobiology, clinical manifestations and treatment approaches to endocrine toxicities arising from ICIs.
Key points
-
Immune checkpoint inhibitors (ICIs) are now a cornerstone of cancer therapies and are associated with long-term treatment responses, but they can also cause inflammatory toxicities in endocrine and other organs.
-
The most common ICI-induced endocrine toxicity is hypothyroidism, which occurs in 10–20% of treated patients, is often preceded by transient thyrotoxicosis, and usually persists after ICI therapy completion.
-
Hypopituitarism, which might co-occur with hypophysitis, is a condition nearly unique to ICI therapy, and is a clinically relevant cause of morbidity and rarely mortality.
-
ICI-associated diabetes mellitus (ICI-DM) occurs rarely (<1% of treated patients) but resembles type 1 diabetes mellitus, and patients can present with diabetic ketoacidosis.
-
Treatment for ICI-induced endocrinopathies involves hormone replacement therapy; patients can continue or resume their ICI once clinically stable.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Postow, M. A., Sidlow, R. & Hellmann, M. D. Immune-related adverse events associated with immune checkpoint blockade. N. Engl. J. Med. 378, 158–168 (2018). This comprehensive yet accessible review reports the pathobiology, incidence and management of irAEs resulting from ICI.
Johnson, D. B., Chandra, S. & Sosman, J. A. Immune checkpoint inhibitor toxicity in 2018. JAMA 320, 1702–1703 (2018).
Johnson, D. B. et al. Immune checkpoint inhibitor toxicities: systems-based approaches to improve patient care and research. Lancet Oncol. 21, e398–e404 (2020).
Ribas, A. & Wolchok, J. D. Cancer immunotherapy using checkpoint blockade. Science 359, 1350–1355 (2018).
Haslam, A. & Prasad, V. Estimation of the percentage of US patients with cancer who are eligible for and respond to checkpoint inhibitor immunotherapy drugs. JAMA Netw. Open 2, e192535 (2019).
Vilgelm, A. E., Johnson, D. B. & Richmond, A. Combinatorial approach to cancer immunotherapy: strength in numbers. J. Leukoc. Biol. 100, 275–290 (2016).
Antonia, S. J. et al. Overall survival with durvalumab after chemoradiotherapy in stage III NSCLC. N. Engl. J. Med. 379, 2342–2350 (2018).
Weber, J. et al. Adjuvant nivolumab versus ipilimumab in resected stage III or IV melanoma. N. Engl. J. Med. 377, 1824–1835 (2017).
Eggermont, A. M. M. et al. Adjuvant pembrolizumab versus placebo in resected stage III melanoma. N. Engl. J. Med. 378, 1789–1801 (2018).
Hodi, F. S. et al. Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 363, 711–723 (2010).
Schadendorf, D. et al. Pooled analysis of long-term survival data from phase II and phase III trials of ipilimumab in unresectable or metastatic melanoma. J. Clin. Oncol. 33, 1889–1894 (2015).
Yarchoan, M., Hopkins, A. & Jaffee, E. M. Tumor mutational burden and response rate to PD-1 inhibition. N. Engl. J. Med. 377, 2500–2501 (2017).
Larkin, J. et al. Five-year survival with combined nivolumab and ipilimumab in advanced melanoma. N. Engl. J. Med. 381, 1535–1546 (2019).
Motzer, R. J. et al. Nivolumab plus ipilimumab versus sunitinib in advanced renal-cell carcinoma. N. Engl. J. Med. 378, 1277–1290 (2018).
Hellmann, M. D. et al. Nivolumab plus ipilimumab in advanced non-small-cell lung cancer. N. Engl. J. Med. 381, 2020–2031 (2019).
Horn, L. et al. First-line atezolizumab plus chemotherapy in extensive-stage small-cell lung cancer. N. Engl. J. Med. 379, 2220–2229 (2018).
Schmid, P. et al. Atezolizumab plus nab-paclitaxel as first-line treatment for unresectable, locally advanced or metastatic triple-negative breast cancer (IMpassion130): updated efficacy results from a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 21, 44–59 (2020).
Burtness, B. et al. Pembrolizumab alone or with chemotherapy versus cetuximab with chemotherapy for recurrent or metastatic squamous cell carcinoma of the head and neck (KEYNOTE-048): a randomised, open-label, phase 3 study. Lancet 394, 1915–1928 (2019).
Rini, B. I. et al. Pembrolizumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N. Engl. J. Med. 380, 1116–1127 (2019).
Berner, F. et al. Association of checkpoint inhibitor-induced toxic effects with shared cancer and tissue antigens in non-small cell lung cancer. JAMA Oncol. 5, 1043–1047 (2019).
Johnson, D. B. et al. Fulminant myocarditis with combination immune checkpoint blockade. N. Engl. J. Med. 375, 1749–1755 (2016).
Dubin, K. et al. Intestinal microbiome analyses identify melanoma patients at risk for checkpoint-blockade-induced colitis. Nat. Commun. 7, 10391 (2016).
Johnson, D. B. et al. A case report of clonal EBV-like memory CD4(+) T cell activation in fatal checkpoint inhibitor-induced encephalitis. Nat. Med. 25, 1243–1250 (2019).
Das, R. et al. Early B cell changes predict autoimmunity following combination immune checkpoint blockade. J. Clin. Invest. 128, 715–720 (2018).
Iwama, S. et al. Pituitary expression of CTLA-4 mediates hypophysitis secondary to administration of CTLA-4 blocking antibody. Sci. Transl. Med. 6, 230ra45 (2014). This paper reports a preclinical model of hypophysitis with translational correlates.
US Department of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE). Version 5.0 https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/CTCAE_v5_Quick_Reference_5x7.pdf (2017).
Lebbe, C. et al. Evaluation of two dosing regimens for nivolumab in combination with ipilimumab in patients with advanced melanoma: results from the phase IIIb/IV checkmate 511 Trial. J. Clin. Oncol. 37, 867–875 (2019).
Tarhini, A. A. et al. Phase III study of adjuvant ipilimumab (3 or 10 mg/kg) versus high-dose interferon alfa-2b for resected high-risk melanoma: North American Intergroup E1609. J. Clin. Oncol. 38, 567–575 (2020).
Wang, D. Y. et al. Fatal toxic effects associated with immune checkpoint inhibitors: a systematic review and meta-analysis. JAMA Oncol. 4, 1721–1728 (2018).
Braaten, T. J. et al. Immune checkpoint inhibitor-induced inflammatory arthritis persists after immunotherapy cessation. Ann. Rheum. Dis. 79, 332–338 (2020).
Johnson, D. B. et al. Survivorship in immune therapy: assessing chronic immune toxicities, health outcomes, and functional status among long-term ipilimumab survivors at a single referral center. Cancer Immunol. Res. 3, 464–469 (2015).
Patrinely, J. R. Jr. et al. Survivorship in immune therapy: assessing toxicities, body composition and health-related quality of life among long-term survivors treated with antibodies to programmed death-1 receptor and its ligand. Eur. J. Cancer 135, 211–220 (2020).
Topalian, S. L. et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N. Engl. J. Med. 366, 2443–2454 (2012).
Betof, A. S. et al. Impact of age on outcomes with immunotherapy for patients with melanoma. Oncologist 22, 963–971 (2017).
Zamami, Y. et al. Factors associated with immune checkpoint inhibitor-related myocarditis. JAMA Oncol. 5, 1635–1637 (2019).
Shah, K. P. et al. Demographic factors associated with toxicity in patients treated with anti-programmed cell death-1 therapy. Cancer Immunol. Res. 8, 851–855 (2020).
Menzies, A. M. et al. Anti-PD-1 therapy in patients with advanced melanoma and preexisting autoimmune disorders or major toxicity with ipilimumab. Ann. Oncol. 28, 368–376 (2017).
Johnson, D. B. et al. Ipilimumab therapy in patients with advanced melanoma and preexisting autoimmune disorders. JAMA Oncol. 2, 234–240 (2016).
Leonardi, G. C. et al. Safety of programmed death-1 pathway inhibitors among patients with non-small-cell lung cancer and preexisting autoimmune disorders. J. Clin. Oncol. 36, 1905–1912 (2018).
Kanz, B. A. et al. Safety and efficacy of anti-PD-1 in patients with baseline cardiac, renal, or hepatic dysfunction. J. Immunother. Cancer 4, 60 (2016).
Naidoo, J. et al. Pneumonitis in patients treated with anti-programmed death-1/programmed death ligand 1 therapy. J. Clin. Oncol. 35, 709–717 (2017).
Brahmer, J. R. et al. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: American Society of Clinical Oncology clinical practice guideline. J. Clin. Oncol. 36, 1714–1768 (2018). Clinically useful guidelines from the American Society of Clinical Oncology for managing irAEs, including endocrine irAEs.
Puzanov, I. et al. Managing toxicities associated with immune checkpoint inhibitors: consensus recommendations from the Society for Immunotherapy of Cancer (SITC) Toxicity Management Working Group. J. Immunother. Cancer 5, 95 (2017). Clinically useful guidelines from the Society for Immunotherapy of Cancer for managing irAEs, including endocrine irAEs.
Martins, F. et al. Adverse effects of immune-checkpoint inhibitors: epidemiology, management and surveillance. Nat. Rev. Clin. Oncol. 16, 563–580 (2019).
de Filette, J., Andreescu, C. E., Cools, F., Bravenboer, B. & Velkeniers, B. A systematic review and meta-analysis of endocrine-related adverse events associated with immune checkpoint inhibitors. Horm. Metab. Res. 51, 145–156 (2019). A meta-analysis of randomized clinical trials showing the prevalence of ICI-induced endocrinopathies.
Liu, J. et al. Reporting of immune checkpoint inhibitor therapy-associated diabetes, 2015-2019. Diabetes Care 43, e79–e80 (2020).
Winther, K. H. et al. Disease-specific as well as generic quality of life is widely impacted in autoimmune hypothyroidism and improves during the first six months of levothyroxine therapy. PLoS ONE 11, e0156925 (2016).
Ho, W. & Druce, M. Quality of life in patients with adrenal disease: a systematic review. Clin. Endocrinol. 89, 119–128 (2018).
Chen, C., Cohrs, C. M., Stertmann, J., Bozsak, R. & Speier, S. Human beta cell mass and function in diabetes: recent advances in knowledge and technologies to understand disease pathogenesis. Mol. Metab. 6, 943–957 (2017).
Min, L. et al. Systemic high-dose corticosteroid treatment does not improve the outcome of ipilimumab-related hypophysitis: a retrospective cohort study. Clin. Cancer Res. 21, 749–755 (2015).
Ascierto, P. A. et al. Adjuvant nivolumab versus ipilimumab in resected stage IIIB-C and stage IV melanoma (CheckMate 238): 4-year results from a multicentre, double-blind, randomised, controlled, phase 3 trial. Lancet Oncol. 21, 1465–1477 (2020).
Eggermont, A. M. M. et al. Longer follow-up confirms recurrence-free survival benefit of adjuvant pembrolizumab in high-risk stage III melanoma: updated results from the EORTC 1325-MG/KEYNOTE-054 Trial. J. Clin. Oncol. 38, 3925–3936 (2020).
Brancatella, A. et al. Graves’ disease induced by immune checkpoint inhibitors: a case report and review of the literature. Eur. Thyroid. J. 8, 192–195 (2019).
Kotwal, A., Kottschade, L. & Ryder, M. PD-L1 inhibitor-induced thyroiditis is associated with better overall survival in cancer patients. Thyroid 30, 177–184 (2020).
Sakakida, T. et al. Clinical features of immune-related thyroid dysfunction and its association with outcomes in patients with advanced malignancies treated by PD-1 blockade. Oncol. Lett. 18, 2140–2147 (2019).
Delivanis, D. A. et al. Pembrolizumab-induced thyroiditis: comprehensive clinical review and insights into underlying involved mechanisms. J. Clin. Endocrinol. Metab. 102, 2770–2780 (2017).
Okada, N. et al. Anti-thyroid antibodies and thyroid echo pattern at baseline as risk factors for thyroid dysfunction induced by anti-programmed cell death-1 antibodies: a prospective study. Br. J. Cancer 122, 771–777 (2020).
Kobayashi, T. et al. Patients with antithyroid antibodies are prone to develop destructive thyroiditis by nivolumab: a prospective study. J. Endocr. Soc. 2, 241–251 (2018). This paper provides clinical characterization of thyroiditis associated with ICIs, and demonstrates that high-dose steroids do not affect clinical outcomes.
Toi, Y. et al. Profiling preexisting antibodies in patients treated with anti-PD-1 therapy for advanced non-small cell lung cancer. JAMA Oncol. 5, 376–383 (2019).
Koyama, J. et al. Correlation between thyroid transcription factor-1 expression, immune-related thyroid dysfunction, and efficacy of anti-programmed cell death protein-1 treatment in non-small cell lung cancer. J. Thorac. Dis. 11, 1919–1928 (2019).
Kurimoto, C. et al. Predictive and sensitive biomarkers for thyroid dysfunctions during treatment with immune-checkpoint inhibitors. Cancer Sci. 111, 1468–1477 (2020).
Angell, T. E., Min, L., Wieczorek, T. J. & Hodi, F. S. Unique cytologic features of thyroiditis caused by immune checkpoint inhibitor therapy for malignant melanoma. Genes. Dis. 5, 46–48 (2018).
Tan, M. H. et al. Spectrum of immune checkpoint inhibitors-induced endocrinopathies in cancer patients: a scoping review of case reports. Clin. Diabetes Endocrinol. 5, 1 (2019). A systematic review of case reports of ICI-induced endocrinopathies.
Yu, C., Chopra, I. J. & Ha, E. A novel melanoma therapy stirs up a storm: ipilimumab-induced thyrotoxicosis. Endocrinol. Diabetes Metab. Case Rep. 2015, 140092 (2015).
Yonezaki, K. et al. Combination therapy of ipilimumab and nivolumab induced thyroid storm in a patient with Hashimoto’s disease and diabetes mellitus: a case report. J. Med. Case Rep. 12, 171 (2018).
Min, L., Vaidya, A. & Becker, C. Thyroid autoimmunity and ophthalmopathy related to melanoma biological therapy. Eur. J. Endocrinol. 164, 303–307 (2011).
Gan, E. H., Mitchell, A. L., Plummer, R., Pearce, S. & Perros, P. Tremelimumab-induced Graves hyperthyroidism. Eur. Thyroid. J. 6, 167–170 (2017).
Borodic, G., Hinkle, D. M. & Cia, Y. Drug-induced Graves disease from CTLA-4 receptor suppression. Ophthalmic Plast. Reconstr. Surg. 27, e87–e88 (2011).
Azmat, U., Liebner, D., Joehlin-Price, A., Agrawal, A. & Nabhan, F. Treatment of ipilimumab induced Graves’ disease in a patient with metastatic melanoma. Case Rep. Endocrinol. 2016, 2087525 (2016).
Iadarola, C. et al. Nivolumab induced thyroid dysfunction: unusual clinical presentation and challenging diagnosis. Front. Endocrinol. 9, 813 (2018).
Hollowell, J. G. et al. Serum TSH, T(4), and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J Clin. Endocrinol. Metab. 87, 489–499 (2002).
Walsh, J. P. et al. Thyrotropin and thyroid antibodies as predictors of hypothyroidism: a 13-year, longitudinal study of a community-based cohort using current immunoassay techniques. J. Clin. Endocrinol. Metab. 95, 1095–1104 (2010).
Mariotti, S., Caturegli, P., Piccolo, P., Barbesino, G. & Pinchera, A. Antithyroid peroxidase autoantibodies in thyroid diseases. J. Clin. Endocrinol. Metab. 71, 661–669 (1990).
Lee, H. et al. Characterization of thyroid disorders in patients receiving immune checkpoint inhibition therapy. Cancer Immunol. Res. 5, 1133–1140 (2017).
Iyer, P. C. et al. Immune-related thyroiditis with immune checkpoint inhibitors. Thyroid 28, 1243–1251 (2018).
Ross, D. S. et al. 2016 American Thyroid Association guidelines for diagnosis and management of hyperthyroidism and other causes of thyrotoxicosis. Thyroid 26, 1343–1421 (2016).
Thompson, J. A. et al. Management of immunotherapy-related toxicities, version 1.2019. J. Natl Compr. Canc Netw. 17, 255–289 (2019).
Garber, J. R. et al. Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Thyroid 22, 1200–1235 (2012).
Bekkering, G. E. et al. Thyroid hormones treatment for subclinical hypothyroidism: a clinical practice guideline. BMJ 365, l2006 (2019).
Pearce, S. H. et al. 2013 ETA guideline: management of subclinical hypothyroidism. Eur. Thyroid. J. 2, 215–228 (2013).
Ma, C. et al. The impact of high-dose glucocorticoids on the outcome of immune-checkpoint inhibitor-related thyroid disorders. Cancer Immunol. Res. 7, 1214–1220 (2019).
Sjolin, G. et al. The long-term outcome of treatment for Graves’ hyperthyroidism. Thyroid 29, 1545–1557 (2019).
Faje, A. T. et al. Ipilimumab-induced hypophysitis: a detailed longitudinal analysis in a large cohort of patients with metastatic melanoma. J. Clin. Endocrinol. Metab. 99, 4078–4085 (2014).
Tahir, S. A. et al. Autoimmune antibodies correlate with immune checkpoint therapy-induced toxicities. Proc. Natl Acad. Sci. USA 116, 22246–22251 (2019). This paper provides a translational assessment of the mechanisms of ICI-induced hypophysitis.
Faje, A. et al. Hypophysitis secondary to nivolumab and pembrolizumab is a clinical entity distinct from ipilimumab-associated hypophysitis. Eur. J. Endocrinol. 181, 211–219 (2019). This study provides a clinical description of hypophysitis associated with different ICI regimens.
Joseph, R. M., Hunter, A. L., Ray, D. W. & Dixon, W. G. Systemic glucocorticoid therapy and adrenal insufficiency in adults: a systematic review. Semin. Arthritis Rheum. 46, 133–141 (2016).
Broersen, L. H., Pereira, A. M., Jorgensen, J. O. & Dekkers, O. M. Adrenal insufficiency in corticosteroids use: systematic review and meta-analysis. J. Clin. Endocrinol. Metab. 100, 2171–2180 (2015).
Garon-Czmil, J. et al. Immune check point inhibitors-induced hypophysitis: a retrospective analysis of the French Pharmacovigilance database. Sci. Rep. 9, 19419 (2019).
Bornstein, S. R. et al. Diagnosis and treatment of primary adrenal insufficiency: an Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 101, 364–389 (2016).
Faje, A. T. et al. High-dose glucocorticoids for the treatment of ipilimumab-induced hypophysitis is associated with reduced survival in patients with melanoma. Cancer 124, 3706–3714 (2018).
Grouthier, V. et al. Immune checkpoint inhibitor-associated primary adrenal insufficiency: WHO VigiBase report analysis. Oncologist 25, 696–701 (2020).
Quandt, Z., Young, A. & Anderson, M. Immune checkpoint inhibitor diabetes mellitus: a novel form of autoimmune diabetes. Clin. Exp. Immunol. 200, 131–140 (2020). This review provides an in-depth discussion on clinical presentation and mechanistic understanding of ICI-DM.
Stamatouli, A. M. et al. Collateral damage: insulin-dependent diabetes induced with checkpoint inhibitors. Diabetes 67, 1471–1480 (2018). This paper provides a clinical description of ICI-DM in the largest series to date.
Wright, J. J. et al. Increased reporting of immune checkpoint inhibitor-associated diabetes. Diabetes Care 41, e150–e151 (2018).
Tsang, V. H. M. et al. Checkpoint inhibitor-associated autoimmune diabetes is distinct from type 1 diabetes. J. Clin. Endocrinol. Metab. 104, 5499–5506 (2019). This article provides a comparison of T1DM and ICI-DM.
de Filette, J. M. K. et al. Immune checkpoint inhibitors and type 1 diabetes mellitus: a case report and systematic review. Eur. J. Endocrinol. 181, 363–374 (2019).
Osum, K. C. et al. Interferon-gamma drives programmed death-ligand 1 expression on islet β cells to limit T cell function during autoimmune diabetes. Sci. Rep. 8, 8295 (2018).
Rui, J. et al. β Cells that resist immunological attack develop during progression of autoimmune diabetes in NOD mice. Cell Metab. 25, 727–738 (2017).
Ansari, M. J. et al. The programmed death-1 (PD-1) pathway regulates autoimmune diabetes in nonobese diabetic (NOD) mice. J. Exp. Med. 198, 63–69 (2003).
Martinov, T., Spanier, J. A., Pauken, K. E. & Fife, B. T. PD-1 pathway-mediated regulation of islet-specific CD4(+) T cell subsets in autoimmune diabetes. Immunoendocrinology 3, e1164 (2016).
Pauken, K. E., Jenkins, M. K., Azuma, M. & Fife, B. T. P. D.-1 but not PD-L1, expressed by islet-reactive CD4+ T cells suppresses infiltration of the pancreas during type 1 diabetes. Diabetes 62, 2859–2869 (2013).
Yoneda, S. et al. T-lymphocyte infiltration to islets in the pancreas of a patient who developed type 1 diabetes after administration of immune checkpoint inhibitors. Diabetes Care 42, e116–e118 (2019).
Lowe, J. R. et al. Genetic risk analysis of a patient with fulminant autoimmune type 1 diabetes mellitus secondary to combination ipilimumab and nivolumab immunotherapy. J. Immunother. Cancer 4, 89 (2016).
American Diabetes Association. 15. Diabetes care in the hospital: standards of medical care in diabetes-2021. Diabetes Care 44, S211–S220 (2021).
Hansen, E., Sahasrabudhe, D. & Sievert, L. A case report of insulin-dependent diabetes as immune-related toxicity of pembrolizumab: presentation, management and outcome. Cancer Immunol. Immunother. 65, 765–767 (2016).
Trinh, B., Donath, M. Y. & Laubli, H. Successful treatment of immune checkpoint inhibitor-induced diabetes with infliximab. Diabetes Care 42, e153–e154 (2019).
Piranavan, P., Li, Y., Brown, E., Kemp, E. H. & Trivedi, N. Immune checkpoint inhibitor-induced hypoparathyroidism associated with calcium-sensing receptor-activating autoantibodies. J. Clin. Endocrinol. Metab. 104, 550–556 (2019).
Win, M. A., Thein, K. Z., Qdaisat, A. & Yeung, S. J. Acute symptomatic hypocalcemia from immune checkpoint therapy-induced hypoparathyroidism. Am. J. Emerg. Med. 35, 1039.e5–1039.e7 (2017).
Trinh, B., Sanchez, G. O., Herzig, P. & Laubli, H. Inflammation-induced hypoparathyroidism triggered by combination immune checkpoint blockade for melanoma. J. Immunother. Cancer 7, 52 (2019).
El Kawkgi, O. M., Li, D., Kotwal, A. & Wermers, R. A. Hypoparathyroidism: an uncommon complication associated with immune checkpoint inhibitor therapy. Mayo Clin. Proc. Innov. Qual. Outcomes 4, 821–825 (2020).
Nallapaneni, N. N. et al. Ipilimumab-induced hypophysitis and uveitis in a patient with metastatic melanoma and a history of ipilimumab-induced skin rash. J. Natl Compr. Canc Netw. 12, 1077–1081 (2014).
Zhao, C. et al. Anti-PD-L1 treatment induced central diabetes insipidus. J. Clin. Endocrinol. Metab. 103, 365–369 (2018).
Gunawan, F., George, E. & Roberts, A. Combination immune checkpoint inhibitor therapy nivolumab and ipilimumab associated with multiple endocrinopathies. Endocrinol. Diabetes Metab. Case Rep. 2018, 17-0146 (2018).
Deligiorgi, M. V., Siasos, G., Vergadis, C. & Trafalis, D. T. Central diabetes insipidus related to anti-programmed cell-death 1 protein active immunotherapy. Int. Immunopharmacol. 83, 106427 (2020).
Lupu, J. et al. Transient pituitary ACTH-dependent Cushing syndrome caused by an immune checkpoint inhibitor combination. Melanoma Res. 27, 649–652 (2017).
Acknowledgements
The authors acknowledge the support of NIH/NCI K23 CA204726 (D.B.J.), NIH R01CA227481 (D.B.J.), DK20593 (A.C.P.), NIDDK T32DK007061 (J.J.W.), the Doris Duke Charitable Foundation, the James C. Bradford Jr. Melanoma Fund (D.B.J.), and the Melanoma Research Foundation (D.B.J.).
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
D.B.J. serves on advisory boards for Array Biopharma, Bristol Myers Squibb, Catalyst Biopharma, Iovance, Jansen, Merck, Novartis and Oncosec, and receives research funding from Bristol Myers Squibb and Incyte. All other authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Endocrinology thanks J.-J. Grob, M. Ryder and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Type II hypersensitivity reaction
-
An antibody-mediated immune reaction in which antibodies (IgG or IgM) are directed against cellular or extracellular matrix antigens with the resultant cellular destruction, functional loss or damage to tissues.
- Complement
-
A system of small proteins that enhance phagocytic and antibody-mediated clearance of microorganisms.
- Cosyntropin stimulation testing
-
Determination of adrenal cortisol secretory function by measuring cortisol levels at baseline and 30 or 60 minutes after injection of 250 µg of synthetic ACTH, cosyntropin.
Rights and permissions
About this article
Cite this article
Wright, J.J., Powers, A.C. & Johnson, D.B. Endocrine toxicities of immune checkpoint inhibitors. Nat Rev Endocrinol 17, 389–399 (2021). https://doi.org/10.1038/s41574-021-00484-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41574-021-00484-3
This article is cited by
-
Rare, late onset of immune checkpoint inhibitor-induced type 1 diabetes mellitus in a patient with small-cell lung cancer treated with serplulimab: a case report and review of the literature
Journal of Medical Case Reports (2024)
-
Profound postinduction hypotension precipitated by immune checkpoint inhibitors: a case report
Journal of Medical Case Reports (2024)
-
Thyroid disorders induced by immune checkpoint inhibitors
Endocrine (2024)
-
Wechselwirkung zwischen eingeschränkter Insulinsekretionskapazität und Insulinresistenz
Die Diabetologie (2024)
-
A case of rapidly progressive insulin-dependent diabetes mellitus without islet autoantibodies developed over two years after the first dose of nivolumab
Diabetology International (2024)