Abstract
Gout is a chronic disease caused by monosodium urate (MSU) crystal deposition. Gout typically presents as an acute, self-limiting inflammatory monoarthritis that affects the joints of the lower limb. Elevated serum urate level (hyperuricaemia) is the major risk factor for MSU crystal deposition and development of gout. Although traditionally considered a disorder of purine metabolism, altered urate transport, both in the gut and the kidneys, has a key role in the pathogenesis of hyperuricaemia. Anti-inflammatory agents, such corticosteroids, NSAIDs and colchicine, are widely used for the treatment of gout flare; recognition of the importance of NLRP3 inflammasome activation and bioactive IL-1β release in initiation of the gout flare has led to the development of anti-IL-1β biological therapy for gout flares. Sustained reduction in serum urate levels using urate-lowering therapy is vital in the long-term management of gout, which aims to dissolve MSU crystals, suppress gout flares and resolve tophi. Allopurinol is the first-line urate-lowering therapy and should be started at a low dose, with gradual dose escalation. Low-dose anti-inflammatory therapies can reduce gout flares during initiation of urate-lowering therapy. Models of care, such as nurse-led strategies that focus on patient engagement and education, substantially improve clinical outcomes and now represent best practice for gout management.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 1 digital issues and online access to articles
$99.00 per year
only $99.00 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Chen-Xu, M., Yokose, C., Rai, S. K., Pillinger, M. H. & Choi, H. K. Contemporary prevalence of gout and hyperuricemia in the United States and decadal trends: the National Health and Nutrition Examination Survey 2007–2016. Arthritis Rheumatol. 71, 991–999 (2019). This article describes the contemporary epidemiology of gout and hyperuricaemia in the USA.
Faires, J. S. & McCarty, D. J. Acute arthritis in man and dog after intrasynovial injection of sodium urate crystals. Lancet 280, 682–685 (1962). This paper proved that monosodium urate crystals cause the gout flare.
Loeb, J. N. The influence of temperature on the solubility of monosodium urate. Arthritis Rheum. 15, 189–192 (1972).
Taylor, W. J. et al. Study for updated gout classification criteria: identification of features to classify gout. Arthritis Care Res. (Hoboken) 67, 1304–1315 (2015). This study describes the major clinical features of gout in a large multicentre study, using monosodium urate crystal identification as the gold standard.
Gutman, A. B. The past four decades of progress in the knowledge of gout, with an assessment of the present status. Arthritis Rheum. 16, 431–445 (1973).
Neogi, T. et al. 2015 gout classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Ann. Rheum. Dis. 74, 1789–1798 (2015). This paper describes the 2015 ACR/EULAR gout classification criteria.
Bursill, D. et al. Gout, Hyperuricemia, and Crystal-Associated Disease Network consensus statement regarding labels and definitions for disease elements in gout. Arthritis Care Res. (Hoboken) 71, 427–434 (2019).
Kuo, C. F., Grainge, M. J., Zhang, W. & Doherty, M. Global epidemiology of gout: prevalence, incidence and risk factors. Nat. Rev. Rheumatol. 11, 649–662 (2015).
Zhu, Y., Pandya, B. J. & Choi, H. K. Prevalence of gout and hyperuricemia in the US general population: the National Health and Nutrition Examination Survey 2007–2008. Arthritis Rheum. 63, 3136–3141 (2011).
Lawrence, R. C. et al. Estimates of the prevalence of selected arthritic and musculoskeletal diseases in the United States. J. Rheumatol. 16, 427–441 (1989).
Choi, H. K. & Curhan, G. Soft drinks, fructose consumption, and the risk of gout in men: prospective cohort study. BMJ 336, 309–312 (2008).
Choi, H. K., Willett, W. & Curhan, G. Fructose-rich beverages and risk of gout in women. JAMA 304, 2270–2278 (2010).
Currie, W. J. Prevalence and incidence of the diagnosis of gout in Great Britain. Ann. Rheum. Dis. 38, 101–106 (1979).
Harris, C. M., Lloyd, D. C. & Lewis, J. The prevalence and prophylaxis of gout in England. J. Clin. Epidemiol. 48, 1153–1158 (1995).
Kuo, C. F., Grainge, M. J., Mallen, C., Zhang, W. & Doherty, M. Rising burden of gout in the UK but continuing suboptimal management: a nationwide population study. Ann. Rheum. Dis. 74, 661–667 (2015).
Zhang, Y. et al. Alcohol consumption as a trigger of recurrent gout attacks. Am. J. Med. 119, 800.e11–800.e16 (2006).
Kuo, C. F. et al. Epidemiology and management of gout in Taiwan: a nationwide population study. Arthritis Res. Ther. 17, 13 (2015).
Roddy, E. & Doherty, M. Epidemiology of gout. Arthritis Res. Ther. 12, 223 (2010).
Arromdee, E., Michet, C. J., Crowson, C. S., O’Fallon, W. M. & Gabriel, S. E. Epidemiology of gout: is the incidence rising? J. Rheumatol. 29, 2403–2406 (2002).
Nakayama, A. et al. GWAS of clinically defined gout and subtypes identifies multiple susceptibility loci that include urate transporter genes. Ann. Rheum. Dis. 76, 869–877 (2017).
Lim, S. Y. et al. Trends in gout and rheumatoid arthritis hospitalizations in the United States, 1993-2011. JAMA 315, 2345–2347 (2016).
Robinson, P. C., Merriman, T. R., Herbison, P. & Highton, J. Hospital admissions associated with gout and their comorbidities in New Zealand and England 1999–2009. Rheumatology 52, 118–126 (2013).
Dehlin, M. & Jacobsson, L. T. H. Trends in gout hospitalization in Sweden. J. Rheumatol. 45, 145–146 (2018).
Edwards, N. L. Quality of care in patients with gout: why is management suboptimal and what can be done about it? Curr. Rheumatol. Rep. 13, 154–159 (2011).
Sarawate, C. A. et al. Gout medication treatment patterns and adherence to standards of care from a managed care perspective. Mayo Clin. Proc. 81, 925–934 (2006).
Shiozawa, A., Szabo, S. M., Bolzani, A., Cheung, A. & Choi, H. K. Serum uric acid and the risk of incident and recurrent gout: a systematic review. J. Rheumatol. 44, 388–396 (2017).
Rothenbacher, D., Primatesta, P., Ferreira, A., Cea-Soriano, L. & Rodriguez, L. A. Frequency and risk factors of gout flares in a large population-based cohort of incident gout. Rheumatology 50, 973–981 (2011).
Zhang, Y. et al. Purine-rich foods intake and recurrent gout attacks. Ann. Rheum. Dis. 71, 1448–1453 (2012).
Dubreuil, M. et al. Increased risk of recurrent gout attacks with hospitalization. Am. J. Med. 126, 1138–1141.e1 (2013).
Hak, A. E., Curhan, G. C., Grodstein, F. & Choi, H. K. Menopause, postmenopausal hormone use and risk of incident gout. Ann. Rheum. Dis. 69, 1305–1309 (2010).
Puig, J. G. et al. Female gout. Clinical spectrum and uric acid metabolism. Arch. Intern. Med. 151, 726–732 (1991).
Dalbeth, N. et al. Relationship between serum urate concentration and clinically evident incident gout: an individual participant data analysis. Ann. Rheum. Dis. 77, 1048–1052 (2018).
Campion, E. W., Glynn, R. J. & DeLabry, L. O. Asymptomatic hyperuricemia. Risks and consequences in the Normative Aging Study. Am. J. Med. 82, 421–426 (1987).
Kapetanovic, M. C. et al. The risk of clinically diagnosed gout by serum urate levels: results from 30 years follow-up of the Malmo Preventive Project Cohort in Southern Sweden. Arthritis Res. Ther. 20, 190 (2018).
Dalbeth, N. et al. Urate crystal deposition in asymptomatic hyperuricaemia and symptomatic gout: a dual energy CT study. Ann. Rheum. Dis. 74, 908–911 (2015).
De Miguel, E. et al. Diagnosis of gout in patients with asymptomatic hyperuricaemia: a pilot ultrasound study. Ann. Rheum. Dis. 71, 157–158 (2012).
Dalbeth, N. et al. Cellular characterization of the gouty tophus: a quantitative analysis. Arthritis Rheum. 62, 1549–1556 (2010).
Wu, X. W., Lee, C. C., Muzny, D. M. & Caskey, C. T. Urate oxidase: primary structure and evolutionary implications. Proc. Natl Acad. Sci. USA 86, 9412–9416 (1989).
Ames, B. N., Cathcart, R., Schwiers, E. & Hochstein, P. Uric acid provides an antioxidant defense in humans against oxidant- and radical-caused aging and cancer: a hypothesis. Proc. Natl Acad. Sci. USA 78, 6858–6862 (1981).
Shi, Y., Evans, J. E. & Rock, K. L. Molecular identification of a danger signal that alerts the immune system to dying cells. Nature 425, 516–521 (2003).
Watanabe, S. et al. Uric acid, hominoid evolution, and the pathogenesis of salt-sensitivity. Hypertension 40, 355–360 (2002).
Choi, H. K., Liu, S. & Curhan, G. Intake of purine-rich foods, protein, and dairy products and relationship to serum levels of uric acid: the Third National Health and Nutrition Examination Survey. Arthritis Rheum. 52, 283–289 (2005).
Choi, H. K., Atkinson, K., Karlson, E. W., Willett, W. & Curhan, G. Purine-rich foods, dairy and protein intake, and the risk of gout in men. N. Engl. J. Med. 350, 1093–1103 (2004).
Stirpe, F. et al. Fructose-induced hyperuricaemia. Lancet 2, 1310–1311 (1970).
Bode, C., Schumacher, H., Goebell, H., Zelder, O. & Pelzel, H. Fructose induced depletion of liver adenine nucleotides in man. Hormone Metab. Res. 3, 289–290 (1971).
Choi, J. W., Ford, E. S., Gao, X. & Choi, H. K. Sugar-sweetened soft drinks, diet soft drinks, and serum uric acid level: the Third National Health and Nutrition Examination Survey. Arthritis Rheum. 59, 109–116 (2008).
Faller, J. & Fox, I. H. Ethanol-induced hyperuricemia: evidence for increased urate production by activation of adenine nucleotide turnover. N. Engl. J. Med. 307, 1598–1602 (1982).
Puig, J. G. & Fox, I. H. Ethanol-induced activation of adenine nucleotide turnover. Evidence for a role of acetate. J. Clin. Invest. 74, 936–941 (1984).
Choi, H. K. & Curhan, G. Coffee, tea, and caffeine consumption and serum uric acid level: the Third National Health and Nutrition Examination Survey. Arthritis Rheum. 57, 816–821 (2007).
Gao, X., Curhan, G., Forman, J. P., Ascherio, A. & Choi, H. K. Vitamin C intake and serum uric acid concentration in men. J. Rheumatol. 35, 1853–1858 (2008).
Choi, H. K., Soriano, L. C., Zhang, Y. & Rodriguez, L. A. Antihypertensive drugs and risk of incident gout among patients with hypertension: population based case-control study. BMJ 344, d8190 (2012).
Zhu, Y., Pandya, B. J. & Choi, H. K. Comorbidities of gout and hyperuricemia in the US general population: NHANES 2007-2008. Am. J. Med. 125, 679–687.e1 (2012).
Nakayama, A. et al. Common dysfunctional variants of ABCG2 have stronger impact on hyperuricemia progression than typical environmental risk factors. Sci. Rep. 4, 5227 (2014).
Major, T. J., Topless, R. K., Dalbeth, N. & Merriman, T. R. Evaluation of the diet wide contribution to serum urate levels: meta-analysis of population based cohorts. BMJ 363, k3951 (2018).
Perez-Ruiz, F., Calabozo, M., Erauskin, G. G., Ruibal, A. & Herrero-Beites, A. M. Renal underexcretion of uric acid is present in patients with apparent high urinary uric acid output. Arthritis Rheum. 47, 610–613 (2002).
Kottgen, A. et al. Genome-wide association analyses identify 18 new loci associated with serum urate concentrations. Nat. Genet. 45, 145–154 (2013). This landmark paper describes a large GWAS to identify loci associated with serum urate levels and gout, emphasizing the importance of urate transporters and glucose metabolism in regulation of serum urate concentrations.
Nakatochi, M. et al. Genome-wide meta-analysis identifies multiple novel loci associated with serum uric acid levels in Japanese individuals. Commun. Biol. 2, 115 (2019).
Li, C. et al. Genome-wide association analysis identifies three new risk loci for gout arthritis in Han Chinese. Nat. Commun. 6, 7041 (2015).
Phipps-Green, A. J. et al. Twenty-eight loci that influence serum urate levels: analysis of association with gout. Ann. Rheum. Dis. 75, 124–130 (2016).
Matsuo, H. et al. Genome-wide association study of clinically defined gout identifies multiple risk loci and its association with clinical subtypes. Ann. Rheum. Dis. 75, 652–659 (2016).
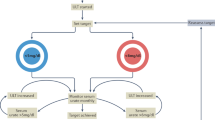
Enomoto, A. et al. Molecular identification of a renal urate anion exchanger that regulates blood urate levels. Nature 417, 447–452 (2002). This paper describes the discovery of URAT1, the major target of uricosuric agents.
Shin, H. J. et al. Interactions of urate transporter URAT1 in human kidney with uricosuric drugs. Nephrology 16, 156–162 (2011).
Miner, J. N. et al. Lesinurad, a novel, oral compound for gout, acts to decrease serum uric acid through inhibition of urate transporters in the kidney. Arthritis Res. Ther. 18, 214 (2016).
Matsuo, H. et al. Mutations in glucose transporter 9 gene SLC2A9 cause renal hypouricemia. Am. J. Hum. Genet. 83, 744–751 (2008).
Dinour, D. et al. Homozygous SLC2A9 mutations cause severe renal hypouricemia. J. Am. Soc. Nephrol. 21, 64–72 (2010).
Kawamura, Y. et al. Pathogenic GLUT9 mutations causing renal hypouricemia type 2 (RHUC2). Nucleosides Nucleotides Nucleic Acids 30, 1105–1111 (2011).
Chiba, T. et al. NPT1/SLC17A1 is a renal urate exporter in humans and its common gain-of-function variant decreases the risk of renal underexcretion gout. Arthritis Rheumatol. 67, 281–287 (2015).
Hollis-Moffatt, J. E. et al. The renal urate transporter SLC17A1 locus: confirmation of association with gout. Arthritis Res. Ther. 14, R92 (2012).
Woodward, O. M. et al. Identification of a urate transporter, ABCG2, with a common functional polymorphism causing gout. Proc. Natl Acad. Sci. USA 106, 10338–10342 (2009).
Matsuo, H. et al. Common defects of ABCG2, a high-capacity urate exporter, cause gout: a function-based genetic analysis in a Japanese population. Sci. Transl Med. 1, 5ra11 (2009).
Matsuo, H. et al. ABCG2 dysfunction causes hyperuricemia due to both renal urate underexcretion and renal urate overload. Sci. Rep. 4, 3755 (2014).
Matsuo, H. et al. Hyperuricemia in acute gastroenteritis is caused by decreased urate excretion via ABCG2. Sci. Rep. 6, 31003 (2016).
Matsuo, H. et al. Common dysfunctional variants in ABCG2 are a major cause of early-onset gout. Sci. Rep. 3, 2014 (2013).
Phipps-Green, A. J. et al. A strong role for the ABCG2 gene in susceptibility to gout in New Zealand Pacific Island and Caucasian, but not Maori, case and control sample sets. Hum. Mol. Genet. 19, 4813–4819 (2010).
Higashino, T. et al. Multiple common and rare variants of ABCG2 cause gout. RMD Open 3, e000464 (2017).
Stiburkova, B. et al. Functional non-synonymous variants of ABCG2 and gout risk. Rheumatology 56, 1982–1992 (2017).
Dehghan, A. et al. Association of three genetic loci with uric acid concentration and risk of gout: a genome-wide association study. Lancet 372, 1953–1961 (2008).
Ichida, K. et al. Decreased extra-renal urate excretion is a common cause of hyperuricemia. Nat. Commun. 3, 764 (2012). This paper describes decreased extra-renal urate excretion caused by ABCG2 dysfunction as a new concept in the pathogenesis of hyperuricaemia.
Becker, M. A. in The Metabolic and Molecular Bases of Inherited Disease (eds Scriver, C. R., Childs, B., Kinzler, K. W., & Vogelstein, B.) Ch. 106, 2513–2535 (McGraw-Hill, 2001).
Wortmann, R. L. in Harrison’s Principles of Internal Medicine (eds Fauci, A. S. et al.) Ch. 353, 2444–2449 (McGraw-Hill, 2008).
Mandel, N. S. & Mandel, G. S. Monosodium urate monohydrate, the gout culprit. J. Am. Chem. Soc. 98, 2319–2323 (1976).
Dalbeth, N. et al. Tendon involvement in the feet of patients with gout: a dual-energy CT study. Ann. Rheum. Dis. 72, 1545–1548 (2013).
Chhana, A., Lee, G. & Dalbeth, N. Factors influencing the crystallization of monosodium urate: a systematic literature review. BMC Musculoskelet. Disord. 16, 296 (2015).
Schumacher, H. R. Pathology of the synovial membrane in gout. Light and electron microscopic studies. Interpretation of crystals in electron micrographs. Arthritis Rheum. 18, 771–782 (1975).
Wood, D. D., Ihrie, E. J., Dinarello, C. A. & Cohen, P. L. Isolation of an interleukin-1-like factor from human joint effusions. Arthritis Rheum. 26, 975–983 (1983).
Di Giovine, F. S., Malawista, S. E., Nuki, G. & Duff, G. W. Interleukin 1 (IL 1) as a mediator of crystal arthritis. Stimulation of T cell and synovial fibroblast mitogenesis by urate crystal-induced IL 1. J. Immunol. 138, 3213–3218 (1987).
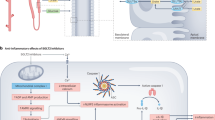
Martinon, F., Petrilli, V., Mayor, A., Tardivel, A. & Tschopp, J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature 440, 237–241 (2006). This paper describes the crucial role of activation of the NLRP3 inflammasome and release of mature IL-1β in the initiation of the gout flare.
Solomon, D. H. et al. Relationship of interleukin-1β blockade with incident gout and serum uric acid levels: exploratory analysis of a randomized controlled trial. Ann. Intern. Med. 169, 535–542 (2018).
Schlesinger, N. et al. Canakinumab reduces the risk of acute gouty arthritis flares during initiation of allopurinol treatment: results of a double-blind, randomised study. Ann. Rheum. Dis. 70, 1264–1271 (2011).
Janssen, C. A. et al. Anakinra for the treatment of acute gout flares: a randomized, double-blind, placebo-controlled, active-comparator, non-inferiority trial. Rheumatology 58, 1344–1352 (2019).
Schlesinger, N. et al. Canakinumab for acute gouty arthritis in patients with limited treatment options: results from two randomised, multicentre, active-controlled, double-blind trials and their initial extensions. Ann. Rheum. Dis. 71, 1839–1848 (2012).
Martinon, F., Burns, K. & Tschopp, J. The inflammasome: a molecular platform triggering activation of inflammatory caspases and processing of proIL-beta. Mol. Cell 10, 417–426 (2002).
Pascual, E., Batlle-Gualda, E., Martinez, A., Rosas, J. & Vela, P. Synovial fluid analysis for diagnosis of intercritical gout. Ann. Intern. Med. 131, 756–759 (1999).
Mangan, M. S. J. et al. Targeting the NLRP3 inflammasome in inflammatory diseases. Nat. Rev. Drug Discov. 17, 588–606 (2018).
Giamarellos-Bourboulis, E. J. et al. Crystals of monosodium urate monohydrate enhance lipopolysaccharide-induced release of interleukin 1 beta by mononuclear cells through a caspase 1-mediated process. Ann. Rheum. Dis. 68, 273–278 (2009).
Joosten, L. A. et al. Engagement of fatty acids with Toll-like receptor 2 drives interleukin-1β production via the ASC/caspase 1 pathway in monosodium urate monohydrate crystal-induced gouty arthritis. Arthritis Rheum. 62, 3237–3248 (2010).
Crisan, T. O. et al. Soluble uric acid primes TLR-induced proinflammatory cytokine production by human primary cells via inhibition of IL-1Ra. Ann. Rheum. Dis. 75, 755–762 (2016).
Mylona, E. E. et al. Enhanced interleukin-1β production of PBMCs from patients with gout after stimulation with Toll-like receptor-2 ligands and urate crystals. Arthritis Res. Ther. 14, R158 (2012).
Crisan, T. O. et al. Uric acid priming in human monocytes is driven by the AKT-PRAS40 autophagy pathway. Proc. Natl Acad. Sci. USA 114, 5485–5490 (2017).
Chen, Y. H. et al. Spontaneous resolution of acute gouty arthritis is associated with rapid induction of the anti-inflammatory factors TGFβ1, IL-10 and soluble TNF receptors and the intracellular cytokine negative regulators CIS and SOCS3. Ann. Rheum. Dis. 70, 1655–1663 (2011).
Yagnik, D. R. et al. Macrophage release of transforming growth factor beta1 during resolution of monosodium urate monohydrate crystal-induced inflammation. Arthritis Rheum. 50, 2273–2280 (2004).
Barden, A. E. et al. Specialised pro-resolving mediators of inflammation in inflammatory arthritis. Prostaglandins Leukot. Essent. Fatty Acids 107, 24–29 (2016).
Liu, L. et al. Interleukin 37 limits monosodium urate crystal-induced innate immune responses in human and murine models of gout. Arthritis Res. Ther. 18, 268 (2016).
Steiger, S. & Harper, J. L. Neutrophil cannibalism triggers transforming growth factor β1 production and self regulation of neutrophil inflammatory function in monosodium urate monohydrate crystal-induced inflammation in mice. Arthritis Rheum. 65, 815–823 (2013).
Schauer, C. et al. Aggregated neutrophil extracellular traps limit inflammation by degrading cytokines and chemokines. Nat. Med. 20, 511–517 (2014). This article describes the role of aggNETs in resolution of the gout flare and also implicates aggNETs in development of the tophus.
Sokoloff, L. The pathology of gout. Metabolism 6, 230–243 (1957).
Schweyer, S., Hemmerlein, B., Radzun, H. J. & Fayyazi, A. Continuous recruitment, co-expression of tumour necrosis factor-alpha and matrix metalloproteinases, and apoptosis of macrophages in gout tophi. Virchows Archiv. 437, 534–539 (2000).
Dalbeth, N. et al. Mechanisms of bone erosion in gout: a quantitative analysis using plain radiography and computed tomography. Ann. Rheum. Dis. 68, 1290–1295 (2009).
Towiwat, P. et al. Urate crystal deposition and bone erosion in gout: ‘inside-out’ or ‘outside-in’? A dual-energy computed tomography study. Arthritis Res. Ther. 18, 208 (2016).
Dalbeth, N. et al. Enhanced osteoclastogenesis in patients with tophaceous gout: urate crystals promote osteoclast development through interactions with stromal cells. Arthritis Rheum. 58, 1854–1865 (2008).
Chhana, A. et al. Monosodium urate monohydrate crystals inhibit osteoblast viability and function: implications for development of bone erosion in gout. Ann. Rheum. Dis. 70, 1684–1691 (2011).
Chhana, A. et al. Monosodium urate crystals reduce osteocyte viability and indirectly promote a shift in osteocyte function towards a proinflammatory and proresorptive state. Arthritis Res. Ther. 20, 208 (2018).
Janssens, H. J. et al. A diagnostic rule for acute gouty arthritis in primary care without joint fluid analysis. Arch. Intern. Med. 170, 1120–1126 (2010). This article describes a diagnostic rule for gout in patients presenting with monoarthritis in clinical practice, which has subsequently been validated in a number of different clinical situations.
Vazquez-Mellado, J. et al. Intradermal tophi in gout: a case-control study. J. Rheumatol. 26, 136–140 (1999).
El-Zawawy, H. & Mandell, B. F. Crystal-induced arthritides in the elderly: an update. Rheum. Dis. Clin. North Am. 44, 489–499 (2018).
Zhang, W. et al. EULAR evidence based recommendations for gout. Part I: Diagnosis. report of a task force of the Standing Committee for International Clinical Studies Including Therapeutics (ESCISIT). Ann. Rheum. Dis. 65, 1301–1311 (2006).
Perez-Ruiz, F., Castillo, E., Chinchilla, S. P. & Herrero-Beites, A. M. Clinical manifestations and diagnosis of gout. Rheum. Dis. Clin. North Am. 40, 193–206 (2014).
Neogi, T., Krasnokutsky, S. & Pillinger, M. H. Urate and osteoarthritis: evidence for a reciprocal relationship. Joint Bone Spine https://doi.org/10.1016/j.jbspin.2018.11.002 (2018).
Forbess, L. J. & Fields, T. R. The broad spectrum of urate crystal deposition: unusual presentations of gouty tophi. Semin. Arthritis Rheum. 42, 146–154 (2012).
Roddy, E., Zhang, W. & Doherty, M. Are joints affected by gout also affected by osteoarthritis? Ann. Rheum. Dis. 66, 1374–1377 (2007).
Perez Ruiz, F., Ruiz Lopez, J. & Herrero Beites, A. M. Influence of the natural history of disease on a previous diagnosis in patients with gout [Spanish]. Reumatol. Clin. 5, 248–251 (2009).
Taylor, W. J. et al. Performance of classification criteria for gout in early and established disease. Ann. Rheum. Dis. 75, 178–182 (2016).
Janssens, H. et al. Performance of the 2015 ACR-EULAR classification criteria for gout in a primary care population presenting with monoarthritis. Rheumatology 56, 1335–1341 (2017).
Kienhorst, L. B., Janssens, H. J., Fransen, J. & Janssen, M. The validation of a diagnostic rule for gout without joint fluid analysis: a prospective study. Rheumatology 54, 609–614 (2015).
Lee, K. H., Choi, S. T., Lee, S. K., Lee, J. H. & Yoon, B. Y. Application of a novel diagnostic rule in the differential diagnosis between acute gouty arthritis and septic arthritis. J. Korean Med. Sci. 30, 700–704 (2015).
Perez-Ruiz, F., Martin, I. & Canteli, B. Ultrasonographic measurement of tophi as an outcome measure for chronic gout. J. Rheumatol. 34, 1888–1893 (2007).
Taylor, W. J. et al. Diagnostic arthrocentesis for suspicion of gout is safe and well tolerated. J. Rheumatol. 43, 150–153 (2016).
Ankli, B. et al. Calcium pyrophosphate deposition disease: a frequent finding in patients with long-standing erosive gout. Scand. J. Rheumatol. 47, 127–130 (2018).
Filippucci, E. et al. Ultrasound imaging for the rheumatologist. XLVII. Ultrasound of the shoulder in patients with gout and calcium pyrophosphate deposition disease. Clin. Exp. Rheumatol. 31, 659–664 (2013).
Reuss-Borst, M. A., Pape, C. A. & Tausche, A. K. Hidden gout- Ultrasound findings in patients with musculo-skeletal problems and hyperuricemia. Springerplus 3, 592 (2014).
Choi, H. K. et al. Dual energy computed tomography in tophaceous gout. Ann. Rheum. Dis. 68, 1609–1612 (2009).
Wang, Y., Deng, X., Xu, Y., Ji, L. & Zhang, Z. Detection of uric acid crystal deposition by ultrasonography and dual-energy computed tomography: a cross-sectional study in patients with clinically diagnosed gout. Medicine 97, e12834 (2018).
Ogdie, A. et al. Performance of ultrasound in the diagnosis of gout in a multicenter study: comparison with monosodium urate monohydrate crystal analysis as the gold standard. Arthritis Rheumatol. 69, 429–438 (2017).
Logan, J. A., Morrison, E. & McGill, P. E. Serum uric acid in acute gout. Ann. Rheum. Dis. 56, 696–697 (1997).
Kang, E. H., Lee, E. Y., Lee, Y. J., Song, Y. W. & Lee, E. B. Clinical features and risk factors of postsurgical gout. Ann. Rheum. Dis. 67, 1271–1275 (2008).
Kannangara, D. R. et al. Fractional clearance of urate: validation of measurement in spot-urine samples in healthy subjects and gouty patients. Arthritis Res. Ther. 14, R189 (2012).
Perez-Ruiz, F., Hernandez-Baldizon, S., Herrero-Beites, A. M. & Gonzalez-Gay, M. A. Risk factors associated with renal lithiasis during uricosuric treatment of hyperuricemia in patients with gout. Arthritis Care Res. 62, 1299–1305 (2010).
Stamp, L. & Dalbeth, N. Screening for hyperuricaemia and gout: a perspective and research agenda. Nat. Rev. Rheumatol. 10, 752–756 (2014).
Khanna, D. et al. 2012 American College of Rheumatology guidelines for management of gout. Part 1: systematic nonpharmacologic and pharmacologic therapeutic approaches to hyperuricemia. Arthritis Care Res. 64, 1431–1446 (2012).
Richette, P. et al. 2016 updated EULAR evidence-based recommendations for the management of gout. Ann. Rheum. Dis. 76, 29–42 (2017).
Abhishek, A. et al. Monosodium urate monohydrate crystal deposits are common in asymptomatic sons of patients with gout: the Sons of Gout Study. Arthritis Rheumatol. 70, 1847–1852 (2018).
Howard, R. G. et al. Reproducibility of musculoskeletal ultrasound for determining monosodium urate deposition: concordance between readers. Arthritis Care Res. 63, 1456–1462 (2011).
Pineda, C. et al. Joint and tendon subclinical involvement suggestive of gouty arthritis in asymptomatic hyperuricemia: an ultrasound controlled study. Arthritis Res. Ther. 13, R4 (2011).
Fisher, M. C., Rai, S. K., Lu, N., Zhang, Y. & Choi, H. K. The unclosing premature mortality gap in gout: a general population-based study. Ann. Rheum. Dis. 76, 1289–1294 (2017).
Andres, M. et al. Cardiovascular risk of patients with gout seen at rheumatology clinics following a structured assessment. Ann. Rheum. Dis. 76, 1263–1268 (2017).
Maglio, C. et al. Effects of bariatric surgery on gout incidence in the Swedish Obese Subjects study: a non-randomised, prospective, controlled intervention trial. Ann. Rheum. Dis. 76, 688–693 (2017).
Waldman, B. et al. Effect of fenofibrate on uric acid and gout in type 2 diabetes: a post-hoc analysis of the randomised, controlled FIELD study. Lancet Diabetes Endocrinol. 6, 310–318 (2018).
Kimura, K. et al. Febuxostat therapy for patients with stage 3 CKD and asymptomatic hyperuricemia: a randomized trial. Am. J. Kidney Dis. 72, 798–810 (2018).
Khanna, D. et al. 2012 American College of Rheumatology guidelines for management of gout. Part 2: therapy and antiinflammatory prophylaxis of acute gouty arthritis. Arthritis Care Res. 64, 1447–1461 (2012).
Rubin, B. R. et al. Efficacy and safety profile of treatment with etoricoxib 120 mg once daily compared with indomethacin 50 mg three times daily in acute gout: a randomized controlled trial. Arthritis Rheum. 50, 598–606 (2004).
Janssens, H. J., Janssen, M., van de Lisdonk, E. H., van Riel, P. L. & van Weel, C. Use of oral prednisolone or naproxen for the treatment of gout arthritis: a double-blind, randomised equivalence trial. Lancet 371, 1854–1860 (2008).
Terkeltaub, R. et al. High versus low dosing of oral colchicine for early acute gout flare. Arthritis Rheum. 62, 1060–1068 (2010).
Schlesinger, N. et al. Canakinumab relieves symptoms of acute flares and improves health-related quality of life in patients with difficult to treat gouty arthritis by suppressing inflammation: results of a randomized, dose-ranging study. Arthritis Res. Ther. 13, R53 (2011).
Dalbeth, N. et al. Effects of allopurinol dose escalation on bone erosion and urate volume in gout: a dual energy CT imaging study of a randomized controlled trial. Arthritis Rheumatol. https://doi.org/10.1002/art.40929 (2019).
Doherty, M. et al. Efficacy and cost-effectiveness of nurse-led care involving education and engagement of patients and a treat-to-target urate-lowering strategy versus usual care for gout: a randomised controlled trial. Lancet 392, 1403–1412 (2018). This large, randomized controlled trial demonstrated major improvements in clinical outcomes in patients receiving nurse-led gout care according to rheumatology guidelines for gout management compared with usual care by a general practitioner.
Hui, M. et al. The British Society for Rheumatology guideline for the management of gout. Rheumatology 56, 1056–1059 (2017).
Qaseem, A., Harris, R. P. & Forciea, M. A. Management of acute and recurrent gout: a clinical practice guideline from the American College of Physicians. Ann. Intern. Med. 166, 58–68 (2017).
Taylor, T. H., Mecchella, J. N., Larson, R. J., Kerin, K. D. & Mackenzie, T. A. Initiation of allopurinol at first medical contact for acute attacks of gout: a randomized clinical trial. Am. J. Med. 125, 1126–1134 (2012). e1127.
Hill, E. M., Sky, K., Sit, M., Collamer, A. & Higgs, J. Does starting allopurinol prolong acute treated gout? A randomized clinical trial. J. Clin. Rheumatol. 21, 120–125 (2015).
Yamanaka, H. et al. Stepwise dose increase of febuxostat is comparable with colchicine prophylaxis for the prevention of gout flares during the initial phase of urate-lowering therapy: results from FORTUNE-1, a prospective, multicentre randomised study. Ann. Rheum. Dis. 77, 270–276 (2018). This clinical trial showed that gradual escalation of the febuxostat dose reduces the risk of gout flares compared with full-dose febuxostat when starting urate-lowering therapy; this study also demonstrated the efficacy of low-dose colchicine (0.5 mg daily) in reducing gout flares when initiating urate-lowering therapy.
Becker, M. et al. Febuxostat compared with allopurinol in patients with hyperuricaemia and gout. N. Engl. J. Med. 353, 2450–2461 (2005).
Becker, M., Schumacher, H. R., MacDonald, P., Lloyd, E. & Lademacher, C. Clinical efficacy and safety of successful longterm urate lowering with febuxostat or allopurinol in subjects with gout. J. Rheumatol. 36, 1273–1282 (2009).
Stamp, L. et al. A randomised controlled trial of the efficacy and safety of allopurinol dose escalation to achieve target serum urate in people with gout. Ann, Rheum, Dis. 76, 1522–1528 (2017). This randomized controlled trial showed that allopurinol dose escalation can be used to attain a target serum urate concentration in most patients with gout.
Stamp, L. et al. Allopurinol dose escalation to achieve serum urate below 6 mg/dl: an open label extension study. Ann. Rheum. Dis. 76, 2065–2070 (2017).
Stamp, L. K. et al. How much allopurinol does it take to get to target urate? Comparison of actual dose with creatinine clearance-based dose. Arthritis Res. Ther. 20, 255 (2018).
Wallace, M. C. et al. Association between ABCG2 rs2231142 and poor response to allopurinol: replication and meta-analysis. Rheumatology 57, 656–660 (2018).
Stamp, L., Day, R. & Yun, J. Allopurinol hypersensitivity: investigating the cause and minimizing the risk. Nat. Rev. Rheumatol. 12, 235–242 (2016).
Hung, S. I. et al. HLA-B*5801 allele as a genetic marker for severe cutaneous adverse reactions caused by allopurinol. Proc. Natl Acad. Sci. USA 102, 4134–4139 (2005). This was the original description of HLA-B*5801 as a genetic marker for allopurinol-induced SCAR.
Ko, T. M. et al. Use of HLA-B*58:01 genotyping to prevent allopurinol induced severe cutaneous adverse reactions in Taiwan: national prospective cohort study. BMJ 351, h4848 (2015).
Chung, W.-H. et al. Insights into the poor prognosis of allopurinol-induced severe cutaneous adverse reactions: the impact of renal insufficiency, high plasma levels of oxypurinol and granulysin. Ann. Rheum. Dis. 74, 2157–2164 (2015).
Shibagaki, Y., Ohno, I., Hosoya, T. & Kimura, K. Safety, efficacy and renal effect of febuxostat in patients with moderate-to-severe kidney dysfunction. Hypertens. Res. 37, 919–925 (2014).
Saag, K. et al. Impact of febuxostat on renal function in gout subjects with moderate-to-severe renal impairment. Arthritis Rheum. 68, 2035–2043 (2016).
White, W. et al. Cardiovascular safety of febuxostat or allopurinol in patients with gout. N. Engl. J. Med. 378, 1200–1210 (2018). The published results of the large, randomized controlled CARES trial show increased cardiovascular and all-cause mortality from febuxostat treatment compared with allopurinol treatment in individuals with gout and established cardiovascular disease.
Choi, H., Neogi, T., Stamp, L., Dalbeth, N. & Terkeltaub, R. Implications of the cardiovascular safety of febuxostat and allopurinol in patients with gout and cardiovascular morbidities (CARES) trial and associated FDA public safety alert. Arthritis Rheumatol. 70, 1702–1709 (2018).
Pui, K., Gow, P. & Dalbeth, N. Efficacy and tolerability of probenecid as urate-lowering therapy in gout; clinical experience in high-prevalence population. J. Rheumatol. 40, 872–876 (2013).
Bardin, T. et al. Lesinurad in combination with allopurinol: a randomised, double-blind, placebo-controlled study in patients with gout with inadequate response to standard of care (the multinational CLEAR 2 study). Ann. Rheum. Dis. 76, 811–820 (2016).
Saag, K. et al. Lesinurad combined with allopurinol: randomized, double-blind, placebo-controlled study in gout subjects with inadequate response to standard of care allopurinol (a US-based study). Arthritis Rheum. 69, 203–212 (2017).
Dalbeth, N. et al. Lesinurad, a selective uric acid reabsorption inhibitor, in combination with febuxostat in patients with tophaceous gout. Arthritis Rheum. 69, 1903–1913 (2017).
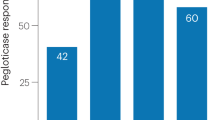
Sundy, J. et al. Efficacy and tolerability of pegloticase for the treatment of chronic gout in patients refractory to conventional treatment: two randomized controlled trials. JAMA 306, 711–720 (2011).
Becker, M. et al. Long-term safety of pegloticase in chronic gout refractory to conventional treatment. Ann. Rheum. Dis. 72, 1469–1474 (2013).
Lipsky, P. et al. Pegloticase immunogenicity: the relationship between efficacy and antibody development in patients treated for refractory chronic gout. Arthritis Res. Ther. 16, R60 (2014).
Becker, M. et al. The urate-lowering efficacy and safety of febuxostat in the treatment of the hyperuricaemia of gout: the CONFIRMS trial. Arthritis Res. Ther. 12, R63 (2010).
Li, X. et al. Serum uric acid levels and multiple health outcomes: umbrella review of evidence from observational studies, randomised controlled trials, and Mendelian randomisation studies. BMJ 357, j2376 (2017).
Terkeltaub, R., Furst, D., DiGiacinto, J., Kook, K. & Davis, M. Novel evidence-based colchicine dose-reduction algorithm to predict and prevent colchicine toxicity in the presence of cytochrome P450 3A4/P-glycoprotein inhibitors. Arthritis Rheum. 63, 2226–2237 (2011).
Jeyaruban, A., Soden, M. & Larkins, S. General practitioners’ perspectives on the management of gout: a qualitative study. Postgrad. Med. J. 92, 603–607 (2016).
Nielsen, S. et al. Weight loss for overweight and obese individuals with gout: a systematic review of longitudinal studies. Ann. Rheum. Dis. 76, 1870–1882 (2017).
Holland, R. & McGill, N. Comprehensive dietary education in treated gout patients does not further improve serum urate. Int. Med. J. 45, 189–189 (2015).
Becker, M. A. et al. Quality of life and disability in patients with treatment-failure gout. J. Rheumatol. 36, 1041–1048 (2009).
Kleinman, N. L. et al. The impact of gout on work absence and productivity. Value Health 10, 231–237 (2007).
Khanna, P. P. et al. Tophi and frequent gout flares are associated with impairments to quality of life, productivity, and increased healthcare resource use: results from a cross-sectional survey. Health Qual. Life Outcomes 10, 117 (2012).
Chandratre, P. et al. Health-related quality of life in gout in primary care: baseline findings from a cohort study. Semin. Arthritis Rheum. 48, 61–69 (2018).
Chandratre, P. et al. Health-related quality of life in gout: a systematic review. Rheumatology 52, 2031–2040 (2013).
Khanna, P. P. et al. Health-related quality of life and treatment satisfaction in patients with gout: results from a cross-sectional study in a managed care setting. Patient Prefer. Adherence 9, 971–981 (2015).
Shields, G. E. & Beard, S. M. A systematic review of the economic and humanistic burden of gout. Pharmacoeconomics 33, 1029–1047 (2015).
Wertheimer, A., Morlock, R. & Becker, M. A. A revised estimate of the burden of illness of gout. Curr. Ther. Res. Clin. Exp. 75, 1–4 (2013).
Flores, N. M., Nuevo, J., Klein, A. B., Baumgartner, S. & Morlock, R. The economic burden of uncontrolled gout: how controlling gout reduces cost. J. Med. Econ. 22, 1–6 (2018).
Strand, V., Khanna, D., Singh, J. A., Forsythe, A. & Edwards, N. L. Improved health-related quality of life and physical function in patients with refractory chronic gout following treatment with pegloticase: evidence from phase III randomized controlled trials. J. Rheumatol. 39, 1450–1457 (2012).
Dalbeth, N. et al. Effects of skim milk powder enriched with glycomacropeptide and G600 milk fat extract on frequency of gout flares: a proof-of-concept randomised controlled trial. Ann. Rheum. Dis. 71, 929–934 (2012).
Stamp, L. et al. Clinically insignificant effect of supplemental vitamin C on serum urate in patients with gout; a pilot randomised controlled trial. Arthritis Rheum. 65, 1636–1642 (2013).
Rai, S. K. et al. Key barriers to gout care: a systematic review and thematic synthesis of qualitative studies. Rheumatology 57, 1282–1292 (2018).
Robinson, P. C., Taylor, W. J. & Dalbeth, N. An observational study of gout prevalence and quality of care in a national Australian general practice population. J. Rheumatol. 42, 1702–1707 (2015).
FitzGerald, J. D. et al. Development of the American College of Rheumatology electronic clinical quality measures for gout. Arthritis Care Res. 70, 659–671 (2018).
Roddy, E., Zhang, W. & Doherty, M. Concordance of the management of chronic gout in a UK primary-care population with the EULAR gout recommendations. Ann. Rheum. Dis. 66, 1311–1315 (2007).
Scheepers, L. et al. Medication adherence among patients with gout: a systematic review and meta-analysis. Semin. Arthritis Rheum. 47, 689–702 (2018).
Kuo, C. F., Grainge, M. J., Mallen, C., Zhang, W. & Doherty, M. Eligibility for and prescription of urate-lowering treatment in patients with incident gout in England. JAMA 312, 2684–2686 (2014).
Duyck, S. D., Petrie, K. J. & Dalbeth, N. “You don’t have to be a drinker to get gout, but it helps”: a content analysis of the depiction of gout in popular newspapers. Arthritis Care Res. 68, 1721–1725 (2016).
Bulbin, D. et al. Improved gout outcomes in primary care using a novel disease management program: a pilot study. Arthritis Care Res. 70, 1679–1685 (2018).
Goldfien, R., Pressman, A., Jacobson, A., Ng, M. & Avins, A. A pharmacist-staffed, virtual gout management clinic for achieving target serum uric acid levels: a randomized clinical trial. Perm. J. 20, 18–23 (2016).
Mikuls, T. R. et al. Adherence and outcomes with urate-lowering therapy: a site-randomized trial. Am. J. Med. 132, 354–361 (2018).
Reginato, A. M., Mount, D. B., Yang, I. & Choi, H. K. The genetics of hyperuricaemia and gout. Nat. Rev. Rheumatol. 8, 610–621 (2012).
Acknowledgements
The authors are grateful to A. Nakayama for editing assistance. H.K.C. is supported by the US National Institutes of Health (AR060772).
Author information
Authors and Affiliations
Contributions
Introduction (N.D.); Epidemiology (H.K.C.); Mechanisms/pathophysiology (H.M. and L.A.B.J.); Diagnosis, screening and prevention (F.P.-R. and N.D.); Management (L.K.S.); Quality of life (P.P.K.); Outlook (N.D.); overview of the Primer (N.D.).
Corresponding author
Ethics declarations
Competing interests
N.D. has received speaking fees from Pfizer, Horizon, Janssen and AbbVie; consulting fees from Horizon, Hengrui and Kowa; and research funding from Amgen and AstraZeneca; and is currently principal investigator on a clinical trial of intensive urate-lowering therapy (funded by the Health Research Council of New Zealand). Within the past 5 years, N.D. has been principal investigator on a clinical trial of febuxostat in early gout, and received consulting or speaking fees from Takeda, Menarini and Teijin. H.K.C. received research support from AstraZeneca and consulting fees from Takeda, Selecta, GSK and Horizon. L.A.B.J. has received a speaking fee from Novartis, research funding from Ardea Biosciences/AstraZeneca, and is Scientific Advisory Board member of Olatec Therapeutics LLC. P.P.K. has received research grants from Ironwood, Sobi and Horizon. F.P.-R. has been an advisor for Amgen, Horizon, Grünenthal, Menarini, Sanofi and Syneos-Health; a speaker for Amgen, Astellas, Grünenthal, Lilly, Logarithm, Menarini and the Spanish Foundation for Rheumatology; received investigation grants from Cruces Rheumatology Association and the Spanish Foundation for Rheumatology; and received congress funding from Lilly, Novartis and the European League Against Rheumatism (EULAR). L.K.S. has received speaking fees from Amgen and is currently principal investigator on a clinical trial of colchicine prophylaxis (funded by the Health Research Council of New Zealand). L.K.S. is a member of the Medicines Adverse Reaction Committee of New Zealand and the New Zealand Pharmaceutical Management Agency (PHARMAC) Rheumatology Subcommittee. H.M. declares no competing interests.
Additional information
Peer review information
Nature Reviews Disease Primers thanks J.G. Puig, M. Pillinger, P. Richette, M. Andrés and R. Burgos-Vargas for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dalbeth, N., Choi, H.K., Joosten, L.A.B. et al. Gout. Nat Rev Dis Primers 5, 69 (2019). https://doi.org/10.1038/s41572-019-0115-y
Accepted:
Published:
DOI: https://doi.org/10.1038/s41572-019-0115-y
This article is cited by
-
Clinical characteristics and risk factors associated with bone erosion in patients with tophi
Advances in Rheumatology (2024)
-
Crosstalk between metabolism and cell death in tumorigenesis
Molecular Cancer (2024)
-
The clinical benefits of sodium–glucose cotransporter type 2 inhibitors in people with gout
Nature Reviews Rheumatology (2024)
-
CXCL5 activates CXCR2 in nociceptive sensory neurons to drive joint pain and inflammation in experimental gouty arthritis
Nature Communications (2024)
-
Nuclear receptor coactivator 6 is a critical regulator of NLRP3 inflammasome activation and gouty arthritis
Cellular & Molecular Immunology (2024)