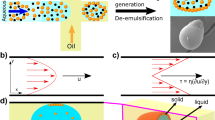
Abstract
Membrane-less organelles resulting from liquid–liquid phase separation of biopolymers into intracellular condensates control essential biological functions, including messenger RNA processing, cell signalling and embryogenesis1,2,3,4. It has recently been discovered that several such protein condensates can undergo a further irreversible phase transition, forming solid nanoscale aggregates associated with neurodegenerative disease5,6,7. While the irreversible gelation of protein condensates is generally related to malfunction and disease, one case where the liquid-to-solid transition of protein condensates is functional, however, is that of silk spinning8,9. The formation of silk fibrils is largely driven by shear, yet it is not known what factors control the pathological gelation of functional condensates. Here we demonstrate that four proteins and one peptide system, with no function associated with fibre formation, have a strong propensity to undergo a liquid-to-solid transition when exposed to even low levels of mechanical shear once present in their liquid–liquid phase separated form. Using microfluidics to control the application of shear, we generated fibres from single-protein condensates and characterized their structural and material properties as a function of shear stress. Our results reveal generic backbone–backbone hydrogen bonding constraints as a determining factor in governing this transition. These observations suggest that shear can play an important role in the irreversible liquid-to-solid transition of protein condensates, shed light on the role of physical factors in driving this transition in protein aggregation-related diseases and open a new route towards artificial shear responsive biomaterials.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All relevant data are included in the manuscript and Supplementary Information. More detailed protocols, calculation and analysis are available from the authors upon request.
Change history
29 December 2020
Supplementary Videos 1–6 were missing when this Letter was originally published online; they have now been uploaded.
References
Brangwynne, C. P. et al. Germline P granules are liquid droplets that localize by controlled dissolution/condensation. Science 324, 1729–1732 (2009).
Wippich, F. et al. Dual specificity kinase DYRK3 couples stress granule condensation/dissolution to MTORC1 signaling. Cell 152, 791–805 (2013).
Strzelecka, M. et al. Coilin-dependent SnRNP assembly is essential for zebrafish embryogenesis. Nat. Struct. Mol. Biol. 17, 403–409 (2010).
Patel, A. et al. A liquid-to-solid phase transition of the ALS protein FUS accelerated by disease mutation. Cell 162, 1066–1077 (2015).
Murakami, T. et al. ALS/FTD mutation-induced phase transition of FUS liquid droplets and reversible hydrogels into irreversible hydrogels impairs RNP granule function. Neuron 88, 678–690 (2015).
Qamar, S. et al. FUS phase separation is modulated by a molecular chaperone and methylation of arginine cation-π interactions. Cell 173, 720–734 (2018).
Jin, H.-J. & Kaplan, D. L. Mechanism of silk processing in insects and spiders. Nature 424, 1057–1061 (2003).
Heim, M., Keerl, D. & Scheibel, T. Spider silk: from soluble protein to extraordinary fiber. Angew. Chem. Int. Ed. Engl. 48, 3584–3596 (2009).
de Kruif, C. G., Weinbreck, F. & de Vries, R. Complex coacervation of proteins and anionic polysaccharides. Curr. Opin. Colloid Interface Sci. 9, 340–349 (2004).
Ianiro, A. et al. Liquid–liquid phase separation during amphiphilic self-assembly. Nat. Chem. 11, 320–328 (2019).
Vollrath, F., Porter, D. & Holland, C. The science of silks. MRS Bull. 38, 73–80 (2013).
Holland, C., Vollrath, F., Ryan, A. J. & Mykhaylyk, O. O. Silk and synthetic polymers: reconciling 100 degrees of separation. Adv. Mater. 24, 105–109 (2012).
Vollrath, F. & Knight, D. P. Liquid crystalline spinning of spider silk. Nature 410, 541–548 (2001).
Weber, S. C. & Brangwynne, C. P. Getting RNA and protein in phase. Cell 149, 1188–1191 (2012).
Shin, Y. & Brangwynne, C. P. Liquid phase condensation in cell physiology and disease. Science 357, eaaf4382 (2017).
Yuan, C. et al. Nucleation and growth of amino acid and peptide supramolecular polymers through liquid–liquid phase separation. Angew. Chem. Int. Ed. Engl. 131, 18284–18291 (2019).
Iserman, C. et al. Condensation of Ded1p promotes a translational switch from housekeeping to stress protein production. Cell 181, 818–831 (2020).
Liao, Y.-C. et al. RNA granules hitchhike on lysosomes for long-distance transport, using annexin A11 as a molecular tether. Cell 179, 147–164 (2019).
Ruggeri, F. S. et al. Infrared nanospectroscopy characterization of oligomeric and fibrillar aggregates during amyloid formation. Nat. Commun. 6, 1–9 (2015).
Ruggeri, F. et al. Identification of oxidative stress in red blood cells with nanoscale chemical resolution by infrared nanospectroscopy. Int. J. Mol. Sci. 19, 2582 (2018).
Ruggeri, F. S. et al. Concentration-dependent and surface-assisted self-assembly properties of a bioactive estrogen receptor α-derived peptide. J. Pept. Sci. 21, 95–104 (2015).
Boeynaems, S. et al. Phase separation of C9orf72 dipeptide repeats perturbs stress granule dynamics. Mol. Cell 65, 1044–1055 (2017).
Ke, H. et al. Shear-induced assembly of a transient yet highly stretchable hydrogel based on pseudopolyrotaxanes. Nat. Chem. 11, 470–477 (2019).
Zebrowski, J., Prasad, V., Zhang, W., Walker, L. & Weitz, D. Shake-Gels: shear-induced gelation of laponite–PEO mixtures. Colloids Surf. A 213, 189–197 (2003).
Axelrod, D., Koppel, D. E., Schlessinger, J., Elson, E. & Webb, W. W. Mobility measurement by analysis of fluorescence photobleaching recovery kinetics. Biophys. J. 16, 1055–1069 (1976).
Niwayama, R. et al. Bayesian inference of forces causing cytoplasmic streaming in Caenorhabditis elegans embryos and mouse oocytes. PLoS ONE 11, e0159917 (2016).
Goldstein, R. E. & van de Meent, J.-W. A physical perspective on cytoplasmic streaming. Interface Focus 5, 20150030 (2015).
Brown, A. Axonal transport of membranous and nonmembranous cargoes: a unified perspective. J. Cell Biol. 160, 817–821 (2003).
Brown, A. Slow axonal transport: stop and go traffic in the axon. Nat. Rev. Mol. Cell Biol. 1, 153–156 (2000).
Kaether, C., Skehel, P. & Dotti, C. G. Axonal membrane proteins are transported in distinct carriers: a two-color video microscopy study in cultured hippocampal neurons. Mol. Biol. Cell 11, 1213–1224 (2000).
Ochs, S. Fast axoplasmic transport of materials in mammalian nerve and its integrative role. Ann. N. Y. Acad. Sci. 193, 43–58 (1972).
Roy, S., Zhang, B., Lee, V. M.-Y. & Trojanowski, J. Q. Axonal transport defects: a common theme in neurodegenerative diseases. Acta Neuropathol. 109, 5–13 (2005).
Li, X. et al. 3D culture of chondrocytes in gelatin hydrogels with different stiffness. Polymers (Basel). 8, 269 (2016).
Vigolo, D., Ramakrishna, S. N. & deMello, A. J. Facile tuning of the mechanical properties of a biocompatible soft material. Sci. Rep. 9, 7125 (2019).
Chao, P.-H. G. et al. Silk hydrogel for cartilage. J. Biomed. Mater. Res. B 95B, 84–90 (2010).
Partlow, B. P. et al. Highly tunable elastomeric silk biomaterials. Adv. Funct. Mater. 24, 4615–4624 (2014).
Ashby, M. F., Gibson, L. J., Wegst, U. & Olive, R. The mechanical properties of natural materials. I. Material property charts. Proc. R. Soc. A 450, 123–140 (1995).
Knowles, T. P. J. & Buehler, M. J. Nanomechanics of functional and pathological amyloid materials. Nat. Nanotechnol. 6, 469–479 (2011).
Rockwood, D. N. et al. Materials fabrication from bombyx mori silk fibroin. Nat. Protoc. 6, 1612–1631 (2011).
Ruggeri, F. S., Šneideris, T., Chia, S., Vendruscolo, M. & Knowles, T. P. J. Characterizing individual protein aggregates by infrared nanospectroscopy and atomic force microscopy. J. Vis. Exp. 12, e60108 (2019).
Müller, J. et al. High-resolution CMOS MEA platform to study neurons at subcellular, cellular, and network levels. Lab Chip 15, 2767–2780 (2015).
Ruggeri, F. S. et al. Nanoscale studies link amyloid maturity with polyglutamine diseases onset. Sci. Rep. 6, 31155 (2016).
Shimanovich, U. et al. Silk micrococoons for protein stabilisation and molecular encapsulation. Nat. Commun. 8, 1–9 (2017).
Yang, H. & Wang, K. Genomic variant annotation and prioritization with ANNOVAR and WANNOVAR. Nat. Protoc. 10, 1556–1566 (2015).
Xia, Y. & Whitesides, G. M. Soft lithography. Annu. Rev. Mater. Sci. 28, 153–184 (1998).
Levin, A. et al. Ostwald’s rule of stages governs structural transitions and morphology of dipeptide supramolecular polymers. Nat. Commun. 5, 5219 (2014).
Acknowledgements
This work is supported by the Welcome Trust, ERC, Alzheimer Association Zenith, ALS Canada–Brain Canada, Canadian Institutes of Health Research and the Cambridge Centre for Misfolding Diseases. We thank S. Zhang, Y. Lu and K.L. Saar for assistance with the design and fabrication of the microfluidic devices; K.H. Muller for help with flash-freezing and SEM imaging; and A. Alexiadis for discussions regarding fluid mechanics calculations.
Author information
Authors and Affiliations
Contributions
Y.S. and T.P.J.K. conceived and designed the study. Y.S., F.S.R., A.K. and A.L. performed the experiments. Y.S., S.Q., P.S.G.-H., C.I., S.A. and A.K. produced the materials. F.S.R. performed AMF-IR and analysed the data. D.V. simulated the flow field. Y.S. imaged samples under SEM and ran microfluidic experiments and FRAP analysis. A.K. performed tensile strength measurements. Y.S., F.S.R., D.V., A.K. P.S.G.-H., S.A. and T.P.J.K. analysed the data. All authors contributed to the writing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–5, Table 1, Movie descriptions and refs. 1–5.
Supplementary Video 1
LLPS of FUS by lowering the salt concentration.
Supplementary Video 2
LLPS of Ded1 by lowering the pH value.
Supplementary Video 3
LLPS of A11 by mixing with 10% dextran.
Supplementary Video 4
LLPS of zFF by mixing with 10% dextran.
Supplementary Video 5
LLPS of reconstituted silk fibroin by mixing with 10% dextran.
Supplementary Video 6
A single fibre was formed under shear stress 2.2 Pa and retracted when the shear was removed in the microfluidic channel.
Rights and permissions
About this article
Cite this article
Shen, Y., Ruggeri, F.S., Vigolo, D. et al. Biomolecular condensates undergo a generic shear-mediated liquid-to-solid transition. Nat. Nanotechnol. 15, 841–847 (2020). https://doi.org/10.1038/s41565-020-0731-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41565-020-0731-4
This article is cited by
-
Dipeptide coacervates as artificial membraneless organelles for bioorthogonal catalysis
Nature Communications (2024)
-
Replicating shear-mediated self-assembly of spider silk through microfluidics
Nature Communications (2024)
-
High-throughput and proteome-wide discovery of endogenous biomolecular condensates
Nature Chemistry (2024)
-
Self-assembly of stabilized droplets from liquid–liquid phase separation for higher-order structures and functions
Communications Chemistry (2024)
-
Fluid protein condensates for bio-inspired applications
Nature Reviews Bioengineering (2023)