Abstract
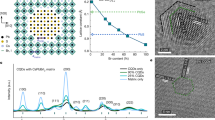
The mixed caesium and formamidinium lead triiodide perovskite system (Cs1−xFAxPbI3) in the form of quantum dots (QDs) offers a pathway towards stable perovskite-based photovoltaics and optoelectronics. However, it remains challenging to synthesize such multinary QDs with desirable properties for high-performance QD solar cells (QDSCs). Here we report an effective oleic acid (OA) ligand-assisted cation-exchange strategy that allows controllable synthesis of Cs1−xFAxPbI3 QDs across the whole composition range (x = 0–1), which is inaccessible in large-grain polycrystalline thin films. In an OA-rich environment, the cross-exchange of cations is facilitated, enabling rapid formation of Cs1−xFAxPbI3 QDs with reduced defect density. The hero Cs0.5FA0.5PbI3 QDSC achieves a certified record power conversion efficiency (PCE) of 16.6% with negligible hysteresis. We further demonstrate that the QD devices exhibit substantially enhanced photostability compared with their thin-film counterparts because of suppressed phase segregation, and they retain 94% of the original PCE under continuous 1-sun illumination for 600 h.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the plots within this paper and other findings of this study are available from the corresponding authors on reasonable request.
References
Kojima, A., Teshima, K., Shirai, Y. & Miyasaka, T. Organometal halide perovskites as visible-light sensitizers for photovoltaic cells. J. Am. Chem. Soc. 131, 6050–6051 (2009).
Kim, H.-S. et al. Lead iodide perovskite sensitized all-solid-state submicron thin film mesoscopic solar cell with efficiency exceeding 9%. Sci. Rep. 2, 591 (2012).
Lee, M. M., Teuscher, J., Miyasaka, T., Murakami, T. N. & Snaith, H. J. Efficient hybrid solar cells based on meso-superstructured organometal halide perovskites. Science 338, 643–647 (2012).
Burschka, J. et al. Sequential deposition as a route to high-performance perovskite-sensitized solar cells. Nature 499, 316–319 (2013).
Lin, K. et al. Perovskite light-emitting diodes with external quantum efficiency exceeding 20 per cent. Nature 562, 245–248 (2018).
Cao, Y. et al. Perovskite light-emitting diodes based on spontaneously formed submicrometre-scale structures. Nature 562, 249–253 (2018).
Xing, G. et al. Low-temperature solution-processed wavelength-tunable perovskites for lasing. Nat. Mater. 13, 476–480 (2014).
Zhu, H. et al. Lead halide perovskite nanowire lasers with low lasing thresholds and high quality factors. Nat. Mater. 14, 636–642 (2015).
Jeon, N. J. et al. Compositional engineering of perovskite materials for high-performance solar cells. Nature 517, 476–480 (2015).
Jiang, Q. et al. Surface passivation of perovskite film for efficient solar cells. Nat. Photonics 13, 460–466 (2019).
Best research-cell efficiencies chart. NREL https://www.nrel.gov/pv/cell-efficiency.html (2019).
Lee, J.-W. et al. Formamidinium and cesium hybridization for photo- and moisture-stable perovskite solar cell. Adv. Energy Mater. 5, 1501310 (2015).
Huang, J., Xu, P., Liu, J. & You, X.-Z. Sequential introduction of cations deriving large-grain CsxFA1−xPbI3 thin film for planar hybrid solar cells: insight into phase-segregation and thermal-healing behavior. Small 13, 1603225 (2017).
Turren-Cruz, S.-H., Hagfeldt, A. & Saliba, M. Methylammonium-free, high-performance, and stable perovskite solar cells on a planar architecture. Science 362, 449–453 (2018).
Saliba, M. et al. Incorporation of rubidium cations into perovskite solar cells improves photovoltaic performance. Science 354, 206–209 (2016).
Li, Z. et al. Stabilizing perovskite structures by tuning tolerance factor: formation of formamidinium and cesium lead iodide solid-state alloys. Chem. Mater. 28, 284–292 (2016).
Kubicki, D. J. et al. Phase segregation in potassium-doped lead halide perovskites from 39K solid-state NMR at 21.1 T. J. Am. Chem. Soc. 140, 7232–7238 (2018).
Domanski, K. et al. Migration of cations induces reversible performance losses over day/night cycling in perovskite solar cells. Energy Environ. Sci. 10, 604–613 (2017).
Christians, J. A. et al. Tailored interfaces of unencapsulated perovskite solar cells for >1,000 hour operational stability. Nat. Energy 3, 68–74 (2018).
Akkerman, Q. A., Rainò, G., Kovalenko, M. V. & Manna, L. Genesis, challenges and opportunities for colloidal lead halide perovskite nanocrystals. Nat. Mater. 17, 394–405 (2018).
Li, M. et al. Low threshold and efficient multiple exciton generation in halide perovskite nanocrystals. Nat. Commun. 9, 4197 (2018).
Pan, J. et al. Bidentate ligand-passivated CsPbI3 perovskite nanocrystals for stable near-unity photoluminescence quantum yield and efficient red light-emitting diodes. J. Am. Chem. Soc. 140, 562–565 (2018).
Zheng, W. et al. Near-infrared-triggered photon upconversion tuning in all-inorganic cesium lead halide perovskite quantum dots. Nat. Commun. 9, 3462 (2018).
Zhou, D. et al. Cerium and ytterbium codoped halide perovskite quantum dots: a novel and efficient downconverter for improving the performance of silicon solar cells. Adv. Mater. 29, 1704149 (2017).
Shockley, W. & Queisser, H. J. Detailed balance limit of efficiency of p-n junction solar cells. J. Appl. Phys. 32, 510–519 (1961).
Swarnkar, A. et al. Quantum dot-induced phase stabilization of α-CsPbI3 perovskite for high-efficiency photovoltaics. Science 354, 92–95 (2016).
Xue, J. et al. Surface ligand management for stable FAPbI3 perovskite quantum dot solar cells. Joule 2, 1866–1878 (2018).
Yuan, J. et al. Band-aligned polymeric hole transport materials for extremely low energy loss α-CsPbI3 perovskite nanocrystal solar cells. Joule 2, 2450–2463 (2018).
Sanehira, E. M. et al. Enhanced mobility CsPbI3 quantum dot arrays for record-efficiency, high-voltage photovoltaic cells. Sci. Adv. 3, eaao4204 (2017).
Wheeler, L. M. et al. Targeted ligand-exchange chemistry on cesium lead halide perovskite quantum dots for high-efficiency photovoltaics. J. Am. Chem. Soc. 140, 10504–10513 (2018).
Lin, J. et al. Thermochromic halide perovskite solar cells. Nat. Mater. 17, 261–267 (2018).
Kubicki, D. J. et al. Cation dynamics in mixed-cation (MA)x(FA)1−xPbI3 hybrid perovskites from solid-state NMR. J. Am. Chem. Soc. 139, 10055–10061 (2017).
Zhu, H. et al. Screening in crystalline liquids protects energetic carriers in hybrid perovskites. Science 353, 1409–1413 (2016).
Protesescu, L. et al. Dismantling the ‘red wall’ of colloidal perovskites: highly luminescent formamidinium and formamidinium–cesium lead iodide nanocrystals. ACS Nano 11, 3119–3134 (2017).
Lignos, I. et al. Exploration of near-infrared-emissive colloidal multinary lead halide perovskite nanocrystals using an automated microfluidic platform. ACS Nano 12, 5504–5517 (2018).
Hazarika, A. et al. Perovskite quantum dot photovoltaic materials beyond the reach of thin films: full-range tuning of A-site cation composition. ACS Nano 12, 10327–10337 (2018).
Suri, M. et al. Enhanced open-circuit voltage of wide-bandgap perovskite photovoltaics by using alloyed (FA1−xCsx)Pb(I1−xBrx)3 quantum dots. ACS Energy Lett. 4, 1954–1960 (2019).
Zhao, Q. et al. High efficiency perovskite quantum dot solar cells with charge separating heterostructure. Nat. Commun. 10, 2842 (2019).
Meggiolaro, D. et al. Iodine chemistry determines the defect tolerance of lead-halide perovskites. Energy Environ. Sci. 11, 702–713 (2018).
Meggiolaro, D. & De Angelis, F. First-principles modeling of defects in lead halide perovskites: best practices and open issues. ACS Energy Lett. 3, 2206–2222 (2018).
Stolterfoht, M. et al. The impact of energy alignment and interfacial recombination on the internal and external open-circuit voltage of perovskite solar cells. Energy Environ. Sci. 12, 2778–2788 (2019).
Boyd, C. C., Cheacharoen, R., Leijtens, T. & McGehee, M. D. Understanding degradation mechanisms and improving stability of perovskite photovoltaics. Chem. Rev. 119, 3418–3451 (2019).
Draguta, S. et al. Rationalizing the light-induced phase separation of mixed halide organic–inorganic perovskites. Nat. Commun. 8, 200 (2017).
Gualdrón-Reyes, A. F. et al. Controlling the phase segregation in mixed halide perovskites through nanocrystal size. ACS Energy Lett. 4, 54–62 (2019).
Protesescu, L. et al. Nanocrystals of cesium lead halide perovskites (CsPbX3, X = Cl, Br, and I): Novel optoelectronic materials showing bright emission with wide color gamut. Nano Lett. 15, 3692–3696 (2015).
Grabolle, M. et al. Determination of the fluorescence quantum yield of quantum dots: suitable procedures and achievable uncertainties. Anal. Chem. 81, 6285–6294 (2009).
Chen, P. et al. In situ growth of 2D perovskite capping layer for stable and efficient perovskite solar cells. Adv. Funct. Mater. 28, 1706923 (2018).
Shi, D. et al. Low trap-state density and long carrier diffusion in organolead trihalide perovskite single crystals. Science 347, 519–522 (2015).
Mizusaki, J., Arai, K. & Fueki, K. Ionic conduction of the perovskite-type halides. Solid State Ion. 11, 203–211 (1983).
Acknowledgements
Financial support from the Australian Research Council Discovery Projects (ARC DPs) is appreciated. Y.B. acknowledges the support from UQ Development Fellowship and ARC DECRA Fellowship (DE190101351). We acknowledge the facilities and the scientific support from the Queensland node of the Australian National Fabrication Facility and Australian Microscopy & Microanalysis Research Facility at the Centre for Microscopy and Microanalysis, The University of Queensland. We also acknowledge the use of the facilities at the University of Wollongong Electron Microscopy Centre funded by the ARC (grant nos. LE0882813 and LE120100104). Y.D. acknowledges financial support from the ARC (grant nos. DP160102627, DP170101467 and FT180100585). All computations were undertaken on the supercomputers in National Computational Infrastructure (NCI) in Canberra, which is supported by the Australian Commonwealth Government, and Pawsey Supercomputing Centre in Perth, with funding from the Australian Government and the Government of Western Australia. P.M. is a Sêr Cymru II National Research Chair and A.A. a Sêr Cymru II Rising Star Fellow. The work at Swansea University was funded through the Sêr Cymru II (Welsh European Funding Office and European Regional Development Fund) Program ‘Sustainable Advanced Materials’. Financial support from National Natural Science Foundation of China (grant nos. 51629201 and 51825204) is also appreciated.
Author information
Authors and Affiliations
Contributions
Y.B., M.H. and L.W. conceived the project. L.W. and Y.B. supervised the work. M.H. synthesized the perovskite quantum dot materials, fabricated and characterized the QD thin films and devices with the assistance of S.D. Y.B. designed the experiments for understanding the ligand-assisted cation-exchange reaction mechanism and the role of surface ligands. S.Z., N.Z., A.A. and P.M. conducted the electrical and photophysical characterization of QD devices. L.R., N.C. and Y.D. performed the STEM study and analysis. J.L. and Y.W. conducted DFT calculations. M.G., M.L. and J.-H.Y. performed the cross-sectional TEM characterization. Y.Y. conducted the ion conductivity measurements. P.C. and D.H. fabricated perovskite thin films and devices. Y.B. drafted the manuscript with help from M.H., A.A., L.R., and J.L. L.W., P.M., G.L. and H.M.-C. revised the manuscript. All the authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–22, Tables 1–4, Notes 1–6 and refs 1–7.
Rights and permissions
About this article
Cite this article
Hao, M., Bai, Y., Zeiske, S. et al. Ligand-assisted cation-exchange engineering for high-efficiency colloidal Cs1−xFAxPbI3 quantum dot solar cells with reduced phase segregation. Nat Energy 5, 79–88 (2020). https://doi.org/10.1038/s41560-019-0535-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41560-019-0535-7
This article is cited by
-
Alkyl ammonium iodide-based ligand exchange strategy for high-efficiency organic-cation perovskite quantum dot solar cells
Nature Energy (2024)
-
Unveiling non-radiative center control in CsPbBr3 nanocrystals: A comprehensive comparative analysis of hot injection and ligand-assisted reprecipitation approaches
Nano Research (2024)
-
The role of ion migration, octahedral tilt, and the A-site cation on the instability of Cs1-xFAxPbI3
Nature Communications (2023)
-
Comprehensive guidance for optimizing the colloidal quantum dot (CQD) Perovskite solar cells: experiment and simulation
Scientific Reports (2023)
-
A soft-chemistry route to prepare halide perovskite nanocrystals with tunable emission and high optical performance
Journal of Sol-Gel Science and Technology (2023)