Abstract
Mucosal tissues act as a barrier throughout the oral, nasopharyngeal, lung, and intestinal systems, offering first-line protection against potential pathogens. Conventionally, vaccines are applied parenterally to induce serotype-dependent humoral response but fail to drive adequate mucosal immune protection for viral infections such as influenza, HIV, and coronaviruses. Oral mucosa, however, provides a vast immune repertoire against specific microbial pathogens and yet is shaped by an ever-present microbiome community that has co-evolved with the host over thousands of years. Adjuvants targeting mucosal T-cells abundant in oral tissues can promote soluble-IgA (sIgA)-specific protection to confer increased vaccine efficacy. Th17 cells, for example, are at the center of cell-mediated immunity and evidence demonstrates that protection against heterologous pathogen serotypes is achieved with components from the oral microbiome. At the point of entry where pathogens are first encountered, typically the oral or nasal cavity, the mucosal surfaces are layered with bacterial cohabitants that continually shape the host immune profile. Constituents of the oral microbiome including their lipids, outer membrane vesicles, and specific proteins, have been found to modulate the Th17 response in the oral mucosa, playing important roles in vaccine and adjuvant designs. Currently, there are no approved adjuvants for the induction of Th17 protection, and it is critical that this research is included in the preparedness for the current and future pandemics. Here, we discuss the potential of oral commensals, and molecules derived thereof, to induce Th17 activity and provide safer and more predictable options in adjuvant engineering to prevent emerging infectious diseases.
Similar content being viewed by others
Introduction
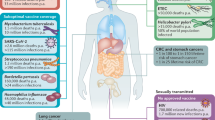
Mucosal barriers throughout the oral, nasopharyngeal, lung, and intestinal systems, offer protection against potential pathogens and exogenous invaders. An abundance of T-helper 17 l (Th17) cells occupy these tissues and mediate serotype-independent immunity and promote mucosal immunoglobulin A (IgA) protection1,2,3,4,5. Traditional vaccines are applied parenterally with adjuvants meant to induce a powerful serotype-dependent response which often fail to drive mucosal immune protection, illustrated in Fig. 1. Alum, the current gold-standard in vaccine adjuvants is known to elicit a Th2 response, some pathogens require different cellular immunity. For example, Bordetella pertussis is re-emerging as an infectious disease despite having a current vaccine. The current adjuvant employed in the B. pertussis vaccine, provided maximum protective immunity requiring Th1 and Th17 responses6, not Th2 response. Accordingly, within the last decade, efforts to induce a more productive mucosal response have targeted therapies toward induction of memory Th17 immunity in hopes of gaining broader protection against pathogens that historically have resisted traditional vaccine strategies; Table 1 provides examples of pathogens that naturally elicit Th17 protection but whose vaccines, if available do not. Experiments targeting Th17 have shown mixed results; success on one hand with confirmed induction of Th17 memory and on the other hand evidence that Th17 protection may come with exacerbated pathology upon rechallenge7,8. Achieving successful Th17 protection will likely require a new vaccine adjuvant. Currently there are no approved adjuvants for Th17 induction and therefore, the research is littered with experimental molecules which stand to have a long regulatory hurdle. Yet, the host microbiome is uniquely adapted to mucosal surfaces, with an ability to modulate the IL-17 environment, making it a potential source of naturally derived mucosal adjuvants.
A Current adjuvanted vaccines are administered subcutaneously and induce primarily a Th2 mediated cellular and pronounced IgG antibody response. B Experimental adjuvants seeking Th17 protection elicit primarily Th1(1) sometimes accompanied by Th17 (2) cell mediated in addition to the IgG response when applied subcutaneously (3). When combined with a mucosal booster, these experimental adjuvants can offer a robust high affinity IgA response that confers additional mucosal protection (1,2). C Experimental adjuvants specific for Th17 can also elicit robust serum IgG when administered via mucosal tissue (4) along with robust Th17 and mucosal IgA with utilization of pIgR (3). Sources194,195,196,197,198.
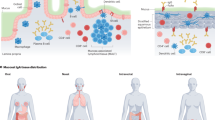
The mucosal tissues are home to constituents of the human microbiome shown to influence the activity of the Th17 response9,10,11 recently reviewed here12. Often overlooked in mucosal immunology is the oral cavity where the microbiome has unique and shifting abilities to shape the immune landscape. Th17 cells appear in response to mastication and are then influenced by the microbial milieu10,13. Additionally, ɣδT-cell-secreting IL-17 cells that contribute to innate homeostasis functions in the oral tissues, have also been found to be regulated by the oral microbiome9. Intriguingly, IL-17 from ɣδT-cells, regulated by microbiota seem critical for induction of Th17 responses in the mouse genital mucosa suggesting the microbiome may help bridge the IL-17 innate immune response to adaptive memory14. More recently, a dysbiotic oral microbiome enhanced intestinal pathology in a mouse model of colitis significantly more than the healthy microbiome3,15. This study underscores the ability of oral microbes to influence the immunological landscape in the gut where the microbiome has been found to tailor the Th17 response in irritable bowel syndrome3,15. Although it is well understood that a dysbiotic oral microbiome can drive IL-17 related gingival pathology, the effects of oral bacteria are less explored16. However, there is a great deal of research targeted at understanding Porphyromonas gingivalis (P. gingivalis), an inflammophillic constituent of the oral microbiome most notable for its low abundance contribution to periodontal disease17. P. gingivalis lipopolysaccharide (LPS) has a unique capacity to induce CD4+ T-cell activity in an ovalbumin-challenged mouse model18 found prior to the discovery of the Th17 cell type. Since then, research has shown that P. gingivalis LPS induces Th17 differentiation via Toll Like Receptor (TLR)-2 activity (Fig. 2)19. This unique activity from P. gingivalis LPS may provide a natural adjuvant quality to the oral microbiome not previously recognized. As research into the host microbiome in the context of IL-17 mucosal regulation and Th17 memory subsets expands, additional preferred induction pathways and additional natural adjuvants will likely be identified.
In the left panel mFA is shown to induce a Th2 response whereas FimA elicits Th1 activity, it is not known if these effects are direct or indirect but FimA can signal through TLR2 to upregulate NO and CD11b/CD18 integrin expression. FimA also acts as a ligand to the CD11b/CD18 integrin. In the right panel, the products that purify out from the LPS-extraction method are shown; antagonist and agonist LPS (pink and purple) and the lipoprotein from gene product PG1828 (orange). Together, this ‘LPS’ can elicit CD4 T-cell, and Th17 expansion as well as IL-17 from γδTc subsets. Although the signaling pathways for T-cell induction are not yet identified, LPS typically signals through TLR4 but can also be presented (lipid A) by DCs via CD1b or CD1c.
In the last ten years have there been efforts to target a memory-Th17 response in the context of vaccine therapy. Th17 protection is particularly attractive because these memory T-cells have been shown to protect against heterologous pathogens in an antibody-independent manner20. This is a particularly pertinent issue as coronavirus vaccine candidates race through clinical trials; the potential protection offered by a Th17 response that may protect against other coronavirus strains/mutations is a very attractive proposition. Protection from vaccine-induced memory Th17 cell response has been described for Candida albicans, Streptococcus pneumoniae, Staphylococcus aureus, Pseudomonas aeruginosa and Mycobacterium tuberculosis21,22,23,24. While the Th17-targeted vaccine protection from fungal and bacterial infections appear successful, the efforts to design similar vaccines for protection from viral infections have been fraught with complications. Specifically, studies investigating Th17 memory response for lasting influenza protection have been successful in producing effective memory response but have shown exacerbation of pathology upon challenge7,8. The route and site of inoculation appear to play a key role in effective induction of Th17 response, indicating that induction of mucosal memory might be more complex than initially thought25. Currently there are efforts to improve our understanding of the numerous pathways of mucosal induction of Th17 and to identify successful adjuvants that will ensure safe, but effective vaccine responses. In an attempt to provide some insight into these issues, we review the role of the oral microbiome, specifically in terms of Th17-immune induction and discuss its potential role in harnessing natural adjuvants that might be utilized for future vaccine therapies.
Mucosal IL-17-producing T-cells
Mucosal tissue is characterized by epithelial cells, connective tissue and resident myeloid and lymphoid cell types. Within these tissues reside IL-17-producing lymphocytes, which can further be divided into ɑβ and ɣδ T-cells (ɑβTc and ɣδTc). The ɣδTc have been described as an innate-type of T-cell that are unrestricted by major histocompatibility complex (MHC) class molecules, that appear required for macrophage recruitment and differentiation upon bacterial challenge26,27,28,29, and can, upon direct stimulation, produce the antibacterial products granulysin and defensin to provide barrier protection26,27. The IL-17+ɑβTc, more commonly known as Th17 mediate a heterologous mucosal immunity that is independent of antibody response20,30. The ɣδTc have been shown to shape the T-cell repertoire in the mucosal tissues and contribute to induction of Th17 memory subsets14,31,32. Although both Th17 and ɣδTc33 can develop memory subsets that respond rapidly to challenges specific to mucosal protection, Th17 immunity is linked to the production of a protective IgA response5 making the Th17 cells attractive targets for vaccine design.
The ɣδTc are primarily described as innate responders in the mucosal tissues, which maintain epithelial cell integrity and are also capable of bridging the innate to adaptive immune response34,35,36. Similar to the classical T-helper cell, the ɣδTc can develop into distinct subtypes and specific effector profiles to produce an array of chemokines and cytokines including INF-ɣ, TNF-ɑ, IL-17, IL-21 and Il-2237. When stimulated, the naive ɣδTc will produce IL-17. However, if antigen-experienced, the ɣδTc will secrete INF-ɣ38. The production of different ɣδTc subsets during RAG-mediated recombination of V(D)J in the thymus gives rise to several subtypes most notably the Vγ9Vδ2+ ɣδTc population primarily seen in human peripheral blood and the Vδ1+ which are found in the epithelium36,37,39. The ɣδTc are capable of sensing and responding to self-antigens as well as bacterial antigens both peptide and non-peptide, such as the lipid-A moiety of LPS39,40. ɣδTc activity can elicit a protective Th17 or autoimmune response14,32,41, the difference being likely the result of local cytokine milieu reviewed here42. The authors postulate that induction of INF-ɣ+Th17 is protective in a vaccine model while GM-CSF may confer autoimmune pathology. The stimulation of ɣδTc by bacterial lipids or fungal β-glucan through TLR2 or Dectin-1 respectively produces rapid induction of IL-1743. IL-17 and INF-ɣ production by ɣδTc can also be induced by exposure to IL-1β and IL-23 and contribute to a Th17 response37. Although, the cytokine milieu plays an important role in activation of the ɣδTc, so, too, is the location of the ɣδTc. The subset of ɣδTc found in the dermis seems important for induction of a protective Th17 response. Complete Freund’s Agent (CFA) adjuvant was found to stimulate a dermal subset of ɣδTc to induce a Th17 profile that included IFN-ɣ+ɑβ-Tc and TNF-ɑ+ɑβ-Tc subsets; when the ɣδTc was specifically blocked via Vɣ, the ɑβTc profile shifted to ɑIL-6+βTc32. The circulating ɣδTc did not appear to be effective in this model of Th17 differentiation indicating a unique role for the tissue ɣδTc subset for directing the mucosal IL-17-mediated immune response. The ɣδTc have also been implicated for their role in shaping the natural antibody repertoire that participate in host defense, autophagy, tissue remodeling and immune regulation44. Through a series of ɣ- or δ- subtype T-cell-deficient mouse strains, the authors discovered that the peripheral ɣδTc subset with cooperation from ɑβTc shape the natural antibody response in the non-immunized state. With the understanding that the ɣδTc are influenced by the shifting constituents of the microbiome whose presence can contribute to changes in the cytokine milieu and influence specific adaptive immune memory, a deeper examination of the bacterial influence during both treatment and outcome is warranted.
Heterologous or universal protection is perhaps the “Holy Grail” for lifetime vaccine protection against seasonal viral challenges that result in loss of life rather than the current annual vaccine requirement. Heterologous protection has been demonstrated with bacterial vaccines where Pneumococcal vaccines have been found to protect against more than 90 pneumococcal serotypes. Observations like this one have led to the development of novel methods that leverage the homology between species. Streptococcus mitis, for example, an oral commensal45 that shares immunogenic characteristics with S. pneumoniae was recently utilized as a vaccine candidate. Shekhar et al. evaluated the potential of S. mitis and its mutant expressing the pneumococcal capsule type 4 (S. mitis TIGR4cps) to induce protection against S. pneumoniae lung infection in mice. Their findings demonstrated that intranasal vaccination with S. mitis provided protection against two S. pneumoniae strains in a serotype-independent fashion, which was associated with enhanced antibody and cell-mediated responses, including increased Th17 immunity46. Recent advances in vaccine research have illuminated a role for the induction of Th17+ and Th1+ to confer vaccine protection against multiple influenza strains which indicate that a universal vaccine is possible27,47,48,49. IL-17 plays an integral role in immune protection following viral infection. Th17 differentiation can be induced by IL-17 from ɣδTc whereas IL-17 from Th17 has been shown essential for mounting an effective Th1 response against viral infection50. Th17 cells contribute to a cell-mediated, clade-specific, serotype-independent immunity with the additional ability to induce high-affinity mucosal IgA, heterologous protection and memory Th17 subsets effective for vaccine-induced protection5,20,51,52.
In order to stimulate an effective immune response to viral pathogens, much recent vaccine work has centered around different adjuvant systems targeting Th17. Intriguingly, evidence is mounting in support of two-component adjuvant systems for induction of Th17-immune protection to increase efficacy and improve safety. One adjuvant named CAF01 combines a mycobacterial fusion protein with a cationic liposome that function together to promote antigen presentation cell (APC) uptake and signaling through Dectin-153. Although the signaling pathways utilized by CAF01 are not fully understood, they have been shown to signal in part through the MyD88 pathway54 with likely interactions that include integrin and TRIF/Inflammasome engagement due to the cationic liposome component55. In another study, the adjuvant consisted of a lipidated TLR7/8 agonist and a synthetic TLR4 agonist, that when applied together elicited a more potent Th1+ Th17+ response than when adjuvants were applied individually47. Another study evaluated a nanoemulsion combination of dioacetyl dimethyl ammonium chloride (DODAC) named NE01 as an adjuvant system, and found it to be effective only when a booster dose was added49. A subsequent study found that by substituting cetytlpyridinium chloride (NE02) for DODAC and adding a TLR9 agonist, the vaccine challenge produced a very rapid and specific humoral response that conferred reduced toxicity with improved health outcomes56. NE02 was previously characterized for its ability to induce IL-17, although the signaling pathway was not identified57. This last study underscores the possibility that a second component for induction of a Th17 vaccine response may be required to improve vaccine efficacy and safety. To further illustrate this, in a study of influenza vaccine in a mouse model, the adjuvant CRX-601 (a TLR4 agonist) was used to stimulate a Th17 response; however, results came with a detrimental neutrophilic lung response8. A subsequent study found that by combining CRX-601 with chitosan in liposomes, the safety and vaccine efficacy was improved58. Chitosan is known for utilizing many cellular pathways such as TLR4, Dectin-1, and CR3, reviewed here59. Together, these studies illuminate a distinct role for engagement of multiple pattern recognition receptors (PRR) for the induction of a safe, effective and potentially heterologous mucosal protection driven by IL-17-induced cell-mediated immunity. This is compelling given the fact that the human microbiome is continually modulating PRRs that contribute to an IL-17-response and may likely confer some adjuvant quality, especially for therapies that utilize sublingual, buccal or intranasal inoculation where the bacteria colonize.
Indeed, the host-microbiome has been shown to function as an adjuvant during vaccine treatment. A study to evaluate the nasal microbiome and efficacy of IgA-seroconversion during the challenge of a live-attenuated, seasonal influenza vaccine found that carriage of several different commensal species but perhaps most significantly Lactobacillus helveticus and Bacteroides ovatus, could increase IgA specificity to the vaccine60. Although it is understood that the success of a mucosal vaccine response will involve Th17 to gain IgA protection, the T-cell repertoire was not evaluated in this study so it is unknown how the commensals may have contributed to T-cell activation during seroconversion. The commensal (and sometimes pathogenic) Staphylococcus aureus can stimulate recruitment of CD11b+ CCR2+ monocytes that mature into M2 macrophages and dampen influenza-mediated acute lung injury61. How this outcome may affect the T-cell repertoire is unknown yet: a study of Staphylococcus aureus in persistent carriage compared the IL-17, Th17 and T-regulatory (Treg) response to non-carriage controls62. It was discovered that carriage came with a diminishment of both IL-17 and Th17, with no change in IFN-ɣ. Together, these studies illuminate the potential of Staphylococcus aureus in protection against severe influenza infection potentially arising from a reduced but not ablated Th17 response. The impact of the host-microbiome relationship cannot be understated. Evidence is building around a role for the oral microbiome in health outcomes in which a healthy microbiome and coordinated actions with the immune system can maintain homeostasis. While dysbiotic inflammatory networks63 derived from microbiome induction trigger a cascade of events that lead to oral diseases such as caries64, periodontal diseases17, oral cancer65. In addition, due to this dysbiotic relationship oral-systemic diseases66 have also been implicated including diabetes67,68, cardiovascular disease69, diabetes, and IBS3,15,70. Further, the host-microbiome (both oral and gut) has been implicated in nosocomial and ventilator-associated pneumonia where a dysbiotic microbiome can increase the abundance of respiratory pathogens71,72,73 and contribute to superinfections74. The superinfection in this case was driven by alterations to the host microbiome from influenza infection. Alterations to the host microbiome have been documented during infection with both influenza and coronavirus56,75,76, but the understanding of how microbial members may impart immune protection or contribute to a successful/detrimental vaccine is lacking. A fairly recent review of the microbiome impact on vaccine response examines the issue of protective versus detrimental microbiota in vaccine efficacy77. The authors discuss the health outcome disparity of vaccine response between countries with a focus on factors that influence host-microbial diversity such as diet and gastrointestinal infections. The overwhelming take-home message is that a dysbiotic microbiome can negatively affect vaccine efficacy since microbial diversity and TLR-engagement drive the T-cell activity required for successful therapeutic outcomes. Although these studies provide evidence that the host microbiome can contribute adjuvant activity, the specific pathways for successful antiviral protection remain undefined.
Some significant progress in understanding antiviral T-cell response has emerged from the rapid study of SARS-CoV-2 infection. First, several studies published recently with regard to SARS-CoV-2 indicated that healthy, seronegative donors may have heterologous protection from the current coronavirus strain due to previous coronavirus infections78,79,80. In Braun et al., the T-cells obtained from both recovered (83%) and healthy donors (34%) showed reactivity from the SARS-CoV-2 spike glycoprotein78,79. In depth epitope mapping and characterization provided evidence of cross-reactive immunity to SARS-CoV-2 from prior coronavirus exposure. These data are compelling evidence to suggest a universal coronavirus vaccine might be possible. The cited studies above indicated that both CD4+ (likely Th1 dominant) and CD8+ T-cells were capable of reactivity but no further characterization was provided. A recent study of T-cell characterization for patients with COVID-19 describes a dominant Th1 repertoire with additional but smaller subsets of Th2 and Th1780,81. It remains unclear what type of T-cell repertoire would be found in an asymptomatic person or the patient successfully controlling a SARS-CoV-2 infection which is potentially of greater importance for informing vaccine targeting strategy. There is emerging discussion about IL-17 and Th17 as detrimental in coronavirus immunopathology and concerns around vaccination-induced immune enhancement reviewed here80,81,82. However, an epidemiological study of patients on current IL-17 inhibitors indicated that these patients were at higher risk for respiratory diseases compared to controls83. Given that the dominant T-cell response in SARS-CoV-2 infection seems to be Th1 with a subset of Th17 and, combined with the understanding that the inhibition of IL-17 may lead to increased viral susceptibility, it is attractive to consider that the Th17 may be playing a supportive role for induction of a potent Th1 response. Indeed, there is some additional evidence that the addition of IFN-ɣ can reduce IL-17 mediated pathology84. This is the mechanism by which adjuvant activity is directed towards a Th17 response that confers Th1 memory might improve vaccine design for a safe and effective mucosal immunity with potential heterologous protection, a particularly attractive notion given the current drive for a successful SARS-CoV-2 vaccine. Additionally, it might be important to evaluate the host-microbial influence on disease severity and therapeutic outcome especially if the therapeutic application is at the mucosal interface.
Immune landscape of the oral mucosa
Humans are host to a multitude of microorganisms that co-develop from birth, and are dependent on the host genome, nutrition, and lifestyle85. The oral microbes have evolved with host tissues over thousands of years and are specifically adapted to the mucosal tissues86. The human oral cavity is home to one of the most complex microbial ecosystems within the body. Estimates suggest there are more than 750 bacterial species in the human oral cavity, many of which have been implicated in local and systemic diseases. The sequential organization of the oral microbiome is complex, niche-dependent, and distinct in health and disease. Salivary mucins and proline-rich proteins help initiate bacterial colonization and biofilm formation87, but also act to aggregate and clear the bacteria from oral surfaces88. Although biomechanical and biochemical cues control initial adhesion of microbial biofilms on hard and soft tissue surfaces, multiple signaling molecules facilitate, such as cytokines, protect microbial colonization as well as participate in the development and maintenance of a healthy immunity at the mucosal border. For example, the gingival crevice has been shown to express an interleukin 8 (IL-8) gradient in the junctional epithelium which serves to recruit neutrophils into the tooth pocket where bacteria naturally accumulate89. Once in the gingival sulcus/pocket, neutrophils form a barrier between the junctional epithelium and the subgingival biofilm. The neutrophil-wall prevents the apical migration of the bacteria deeper into tissues, which is essential to maintain periodontal homeostasis90. Thus, preventing microbial access to connective tissue, major blood vessels and migration to systemic sites.
Out of all immune cells present in the oral mucosa and gingival tissue, neutrophils constitute 95% of total leukocytes present in oral tissues. Transmigration of neutrophils is continuous through gingival and buccal mucosal tissues, with 30,000 neutrophils per minute passing through the highly permeable epithelium, namely the junctional epithelium91. The neutrophil is the main immune cell that coordinates anti-bacterial responses in the gingival tissues primarily by utilizing proteases, defensins, cytokines, phagocytosis and neutrophil-extracellular-nets (NET); its function is intimately tied to IL-17 production92,93. Although few in number, other important immune cells reside in the gingival tissues: these include resident T- and B-cells (rare in healthy tissues), innate lymphoid cells (ILCs), macrophages, and dendritic cells (DCs). CD4+ and CD8+ T-cells produce specific types of cytokines, including canonical types 1 and 17. Additionally, novel patterns of B-cells and plasmacytes have been identified in gingival tissues94,95. T regulatory cells (Tregs) also reside in the gingival tissue and provide immunological tolerance94,96,97. M2 macrophages and DCs are found in small numbers in the healthy gingiva, with a subset of CD103+ dendritic cells that provide yet another level of barrier function, while M1 macrophages increase during inflammatory insult98.
The neutrophil interplay with the macrophage is a central component to mucosal tissue homeostasis. The tissue macrophage produces signals, including IL-23 which is followed by an induction of IL-17 from nearby T-cells, shown to regulate neutrophil granulopoiesis92. When sequestered into the tissue by damage or infection, the otherwise short-lived neutrophil is activated by microbes and/or local chemokines (e.g., IL-8), thereby inhibiting apoptosis programs, which allows the neutrophil to coordinate an appropriate cellular response99,100. Once phagocytosis of the infectious particle occurs, the neutrophil typically undergoes apoptosis101. The apoptotic neutrophil then signals to the monocyte, triggering the second wave of host-mediated immune responses102. Endocytosis of the apoptotic neutrophil by the monocyte or macrophage downregulates IL-23 and IL-17 production92, reduces inflammation and initiates pathways of wound healing102,103. If neutrophils are unable to move into tissues to coordinate cleanup as in the case of Leukocyte Adhesion Deficiency (LAD)-1 or if neutrophil apoptosis is delayed, the IL-23 and IL-17 cytokines persist and contribute to exacerbated tissue inflammation104,105. The IL-23-IL-17 tissue program has been shown to be essential for tissue homeostasis in gingival mucosa and, if disrupted genetically by bacterial manipulation or hormonal changes, IL-17 can contribute to inflammatory pathology104,106,107.
Germ-free animals have been utilized to uncouple the gingival tissue program from microbial influence. These studies have shown that as the gingival tissue develops early in life, microbial independent tissue sequestration drives an influx of IL-17 secreting-γδT and chemokine C-X-C-ligand (CXCL)-1-dependant neutrophil recruitment to basal and junctional epithelium while the acquisition of the oral microbiome alters the γδT subtype and coordinates additional neutrophil recruitment via CXCL29,108. Microbial-independent mastication forces drive Th17 cells into the gingival tissues while a distinct dysbiotic microbial community can initiate Th17-dependent oral pathology10,13. During the aging process, increases in the number of Th17 are found with apparent simultaneous decreases to the γδT in the gingival tissues9,10,13,108. These IL-17-producing T-cell profile alterations coincide with significant bacterial burden, IL-17-related bone loss pathology109 marked by a distinct and significant rise in neutrophil activity. Combined, these studies illustrate a very unique tissue program, which underscores a role for γδT-IL-17 in homeostasis that is heavily influenced by age and the bacterial community.
The neutrophil has been shown to regulate ɣδTc. Two neutrophil-derived products, neutrophil elastase (NE) and reactive oxygen species (ROS) have been found to exert opposing effects on ɣδTc; activating the T-cells with NE and inhibiting with ROS110. Both of these factors have been found in the gingival crevice and shown to be stimulated by oral bacteria111,112. When NE is specifically inhibited during sublingual immunization, significant increases are found for Th1, Th2, Th17, and for antigen-specific IgG and mucosal IgA when compared to controls113. This study underscores the impact of the neutrophil in relation to T-cell activity and B-cell class switch. A prior study by the same laboratory found neutrophils inhibited the B-cell activity mainly through IKKβ. When the IKKβ signal was depleted, IL-17RA+ B-cells increased significantly114, which is compelling given that IL-17A has been found to trigger class switch via Th17 helper cell activity115. Th17 cells are required for induction of a long-lasting protective IgA response which is an important mediator for the first line of defense in mucosal tissues5. Further, a comparison between sublingual and intramuscular influenza vaccine application found that sublingual routes elicited systemic immunity similar to that from intramuscular routes, but only the sublingual application leads to a protective Th17 with mucosal IgA protection116.
The interaction between γδTc and neutrophils seems intimately tied. An examination of γδTc found they were capable of inducing antigen-presenting cell (APC)-functions in the neutrophil117,118. After exposure (and phagocytosis) to bacterial metabolites, the neutrophils differentiated into APC upon co-culture with the γδTc. Once activated, the neutrophils could present antigens to both CD4+ and CD8+ T-cells. In this scenario, the neutrophils had the ability to flexibly activate either CD4 or CD8 T-cells. To substantiate the finding, study participants with sepsis were shown to have highly activated γδTc, APC-like neutrophils, and activated CD8+ T-cells117,118. Neutrophils have been shown to either inhibit or activate T-cell mediated functions depending on cellular responses to the microenvironment. Since neutrophil activity in the mucosal tissue is directly tied to and influenced by bacterial products as well as IL-17 regulation, any vaccines that require the induction of Th17 for protective response may need to be designed around or informed by neutrophil activity.
Oral microbiome: naturally derived adjuvants
Both the γδTc and neutrophils are essential for maintenance of the mucosal border where the host-microbiome community resides and can respond accordingly through TLRs and complement receptors. The γδTc is also capable of sensing phospho-antigens like LPS-derived Lipid A via γδ-TCR cross-presentation of CD1b or CD1c on APCs or directly through TLR2119,120. Much of the interaction between the γδTc and neutrophil is to coordinate the management of a symbiotic relationship with the microbial community, modulating between tolerance and removal121,122. It is here where the adjuvant quality of the oral microbiome may impart therapeutic IL-17 activity. Indeed, the IL-17 program is intimately tied to microbial activity with γδTc providing an IL-17 environment that supports homeostasis9 while the neutrophil is often associated with IL-17 pathology16,123.
Bacteria-derived proteins, polysaccharides and lipids have been utilized as adjuvants to induce specific immune responses and tailor antibody specificity and have been reviewed extensively124,125. TLR ligands have long been examined for their adjuvant activity and are currently under investigation for therapies in allergy, cancer, vaccine and autoimmune dysfunction126,127,128,129. Monophosphoryl lipid A (MPL) is one example of an FDA-approved Toll-like receptor 4 (TLR4) adjuvant. The lipid A structure is derived from the LPS of Salmonella minnesota and is currently used in the human papillomavirus vaccine, Cervarix Ⓡ. The lipid A structure of MPL shares some similar features to that of the oral microbe P. gingivalis. This oral derived adjuvant is capable of activating cell receptors such as TLR4130. The vaccine is now use for HPV-16 and 18 associated cervical cancers131. P. gingivalis has heterogeneous TLR4 activity found to be caused by the ability to shift its LPS structure, specifically the lipid-A moiety, to that of TLR4 antagonist or agonist132,133. P. gingivalis can alter its lipid-A structure during changes to environmental conditions such as levels of hemin or temperature, both important features of inflammation activation134,135. Two well-characterized lipid-A moieties of the P. gingivalis LPS molecule have been shown to exert opposing effects on neutrophil recruitment and are likely the culprits of P. gingivalis-induced “chemokine paralysis”: P. gingivalis has been found to disrupt epithelial cell expression of interleukin 8 (IL-8), an important chemokine for neutrophil recruitment into gingival tissues89. The tetra-acylated structure, characterized as a potent TLR4 antagonist, can reduce recruitment of neutrophils by blocking IL-8 chemokine production in gingival tissues while the penta-acylated structure is an agonist to TLR4 that increases IL-8 production and subsequent neutrophil traffic89,134,136,137,138,139. LPS activity in the gingival tissues has been implicated in periodontal disease. The LPS activity from the oral bacteria seems to play a role in the progression to periodontal disease, moving from TLR4 antagonist activity in healthy sites towards agonist activity in sites with active disease140 and may take on different adjuvant activity in the context of vaccine therapeutics.
The oral microbiome has been implicated in induction of IL-17-related pathology. However, the role of the microbiome also seems to be a factor in γδTc IL-17 homeostasis9,10. A dysbiotic oral microbiome has been shown to trigger Th17 activity which resulted in IL-17 associated pathology, whereas ablation of γδTc gave rise to alterations to the microbiome that resulted in IL-17 inflammation and gingival pathology. Although these studies implicate the microbiome in inflammatory pathology, an IL-17-related homeostasis also exists, the difference likely culminating around the activity of the oral neutrophil. In an elegant series of experiments using integrin and developmental endothelial locus-1 (DEL-1)-deficient mice, it was recently shown that apoptotic neutrophil efferocytosis is essential for regulation of IL-17 homeostasis and disruption to this function can result in periodontal disease and dysbiosis104,141. During oral dysbiosis, an outgrowth of anaerobic bacteria is described which are often referred to as perio-pathogens. However, these bacteria are also present in healthy tissues albeit at much lower concentrations142,143. Many of these ‘pathobionts’ have been studied in terms of their ability to cause tissue destruction. Yet, co-evolutionarily they may contribute some benefit when in low abundance. In support of this hypothesis, a recent study illuminated a role for LPS derived from the oral microbiome and released into gingival tissue that was specifically responsible for the ecological maintenance and balance of the mesenchymal stem cells that reside in the mucosal tissues144. In this scenario, the anaerobic bacteria are providing a benefit to host tissues. P. gingivalis, a member of the anaerobic community has been shown to modulate IL-17 activity via LPS, fimbriae and the proteases termed ‘gingipains’145,146,147,148, and induce IL-17 from both Th17 and γδTc149. Together, these data provide evidence that P. gingivalis has the ability to impact the IL-17 program in multiple ways and may be found to participate in IL-17 homeostasis under normal, healthy conditions.
Although there are many oral microbes that appear to contribute to IL-17 activity in the oral mucosa, P. gingivalis and its microbial products are perhaps the most studied in terms of oral mucosal IL-17, T-cell activation, and neutrophil manipulation, reviewed here123,150. Both the P. gingivalis LPS and gingipain proteases have been implicated in IL-17 and Th17 response146,147,151 whereas the fimbrial proteins have been shown to activate a Th1 response152 despite the ability to block IL-12153 required for Th1 activation. IL-12 has also been found to expand Th17154 in which case, the fimbrial proteins may be controlling the amount of Th17 expansion. The focus of potential adjuvant qualities of P. gingivalis will remain on LPS and fimbrial proteins due to the association between gingipains and the development of autoimmunity, cardiovascular disease and diabetes155,156. The LPS and fimbrial proteins have been primarily described as TLR2 agonists and while true, each pathogen-associated molecular pattern (PAMP) has additional signaling capacity that lends further immune modulatory activities.
P. gingivalis LPS
Depending on the cytokine milieu, P. gingivalis derived LPS can contribute to a multitude of host-immune responses that culminate in a specific T-cell response. The purified LPS, described above as having heterogeneous lipid A structures that can modulate TLR4 receptors, and also contains a lipoprotein contaminant encoded by gene product PG1828 which has been evaluated as the main TLR2 agonist157. P. gingivalis LPS has been shown to expand CD4+ T-cells18 and induce Th17 activity in a TLR2-dependent manner19. During a footpad challenge with ovalbumin, P. gingivalis LPS significantly enhanced antigen specific CD4+ T-cells that released IL-10, IL-2 and IL-5 upon stimulation18. IL-2 has been found to modulate tissue-specific γδTc to favor short-term effector activity and preserve survival and plasticity158. Further, P. gingivalis LPS has been shown to induce CD69 on the γδTc159, a molecule required for tissue retention and immune modulation160. Repeated exposure of P. gingivalis LPS tolerized monocytes which then significantly increased ROS production in neutrophils and inhibited neutrophil chemotaxis161. While ROS has been found to inhibit γδTc activity110, nitric oxide (NO) has been shown to protect γδTc from apoptosis162. P. gingivalis LPS can elicit the production of NO in vivo163 with a subsequent study showing the involvement of TLR9164. Notably, NO from macrophages has been shown to confer γδTc protection in mucosal candidiasis165. Although it seems plausible that P. gingivalis LPS is likely involved in γδTc activity it is unclear whether the effect is direct or indirect. γδTc can recognize lipid A from LPS via dendritic cell presentation40,164 or directly through TLR2120 but it is unknown how the heterogeneous lipid A structures of P. gingivalis LPS might alter γδTc activity. It should also be noted that P. gingivalis LPS can induce Natural Killer T-cell (NK) activity in vivo with less cytotoxicity than Escherichia coli LPS166. This activity was not shown to be either direct or indirect but it is interesting that the γδTc has been shown to induce NK activity166,167 and may be involved during P. gingivalis LPS induction due to its ability to sense lipid A. Together, these studies illustrate the capacity of P. gingivalis LPS to modulate the T-cell repertoire and drive an antigen-specific response.
P. gingivalis fimbriae
P. gingivalis fimbriae are expressed as two distinct proteins, the long, major (FimA) and short, minor (Mfa) fimbriae. Both are required for attachment during colonization168 and can differentially modulate T-cell responses. The FimA and accessory proteins (FimCDE) are considered the main colonization factors169 and engage TLR2 in combination with CXCR4 or the complement receptors, C3R or C5aR to modulate integrin function and elicit IL-1ɑ, IL-1β, TNFd, IL-6 cytokines, reviewed here169,170. Integrin function is intimately tied to T-cell activity required for T-cell translocation from vasculature to periphery, chemotaxis through tissues, and APC-T-cell signaling synapse for initiation of adaptive immune response. The minor and major fimbriae appear to direct differential T-cell activity. In a series of fimbriae-deficient bacterial strains, the minor fimbriae were attributed to a Th2 response whereas the major fimbriae induced Th1 response171. The fimbriae activate TLR2 which induces the high-affinity conformation of CD11b/CD18 (aka CR3, MAC-1). The fimbriae can then function as a ligand for CD11b/CD18-blocking integrin activity which includes IL-12 down regulation172. CD11b expression on APCs is required for induction of peripheral oral tolerance and suppression of Th17 differentiation173. P. gingivalis fimbriae may be capable of breaking oral tolerance to promote inflammatory response that can mount an efficient and specific immune response with coordinating antigens. The fimbriae of P. gingivalis can direct specific T-cell activation with additional potential to break tolerance. This would likely improve mucosal adjuvant qualities and enhance specificity for therapeutic design, depending on the antigen and protection requirements.
The PAMPs of P. gingivalis are perhaps the most studied of all the human oral bacteria and provide a unique opportunity to evaluate host immune modulation during vaccine response. The full list of potential functions is far from adequately investigated. Although the specific PAMPs are not yet identified, P. gingivalis has also been shown to induce both Nod-Like Receptors (NOD)-1 and -2174,175 as well as TLR7176. The NLRs are involved in T-cell activation and when paired with TLRs can be utilized to activate both Th1 and Th17 simultaneously177. TLR7 suppresses the Th17 autoimmune response178. While the mechanisms at play here require further investigation of specific PAMP activity, the ability of P. gingivalis to exert a seemingly unlimited number of options for modulating the immune response is enticing. Certainly, there is an opportunity to utilize specific combinations of both P. gingivalis LPS and fimbriae since each contains components with unique capabilities to induce different types of immune response qualities that lend a highly specific adjuvant quality to a given vaccine formulation.
Currently it is unknown whether direct induction of Th17 or γδTc-induced Th17 memory is more beneficial for antiretroviral vaccine response. However, there is evidence that the use of adjuvants that stimulate multiple PRRs can confer higher specificity and increase the safety profile during a Th17-based vaccine response. The range of PRRs that are utilized by P. gingivalis PAMPs present an opportunity to coordinate a very specific T-cell response and may be utilized to tailor more effective vaccine therapies. Since P. gingivalis is a co-evolutionary member of the host oral microbiome, its PAMPs are likely to have a relatively safe immune profile. The unique pathways of IL-17 regulation during homeostasis in the oral cavity are intimately tied to the oral microbiome. Accordingly, the evaluation of P. gingivalis PAMPs, and other oral microbiome derived constituents, in vaccine immune response may very well lead to a new understanding of the natural adjuvanticity of the host-microbiome.
Conclusions
The diversity of the microbiome has been implicated as a driver for differences in vaccine efficacy across geographical regions. In this vein, the use of probiotics has been investigated as a method to improve vaccine response with some successes. In a meta-analysis of probiotic-use in vaccine clinical study, the authors found that while there was significant variability, the probiotic effect was most notable for orally applied vaccines120,179. Some of the studies that were highlighted as successful showcased outcomes that included increased IgA responses and elicited cell-mediated and humoral immunity. It is not known whether the successes of the probiotic applications were due to direct effect of the probiotic or the specific activity of the host-microbiome. Together, these studies underscore the potential of the host-bacterial constituents as a powerful source of adjuvant-activity that could be harnessed for immune-modulation of the vaccine response and may offer a path around the steep regulatory hurdle that most adjuvants incur during development. The oral microbiome is a rich source of potential with regard to adjuvant activity which is convenient since the oral cavity is also the ideal site for vaccine inoculation. Investigation into the microbial products, metabolites responsible for coordinating specific immune protection will likely be a fruitful endeavor for identifying adjuvants, and will also provide a better understanding of how immunity is shaped by our microbiome.
Data availability
There were no datasets generated and/or analyzed during the current study.
References
Konkel, J. E. & Moutsopoulos, N. M. Unique tailoring of Th17 at the gingival oral mucosal barrier. J. Dent. Res. 97, 128–131 (2018).
Mubarak, A. et al. A dynamic relationship between mucosal T helper type 17 and regulatory T-cell populations in nasopharynx evolves with age and associates with the clearance of pneumococcal carriage in humans. Clin. Microbiol. Infect. 22, 736.e1–736.e7 (2016).
Omenetti, S. & Pizarro, T. T. The Treg/Th17 axis: a dynamic balance regulated by the gut microbiome. Front. Immunol. 6, 639 (2015).
Kumar, P., Chen, K. & Kolls, J. K. Th17 cell based vaccines in mucosal immunity. Curr. Opin. Immunol. 25, 373–380 (2013).
Christensen, D., Mortensen, R., Rosenkrands, I., Dietrich, J. & Andersen, P. Vaccine-induced Th17 cells are established as resident memory cells in the lung and promote local IgA responses. Mucosal Immunol. 10, 260–270 (2017).
Ross, P. J. et al. Relative contribution of Th1 and Th17 cells in adaptive immunity to Bordetella pertussis: towards the rational design of an improved acellular pertussis vaccine. PLoS Pathog. 9, e1003264 (2013).
Gopal, R. et al. Mucosal pre-exposure to Th17-inducing adjuvants exacerbates pathology after influenza infection. Am. J. Pathol. 184, 55–63 (2014).
Maroof, A., Yorgensen, Y. M., Li, Y. & Evans, J. T. Intranasal vaccination promotes detrimental Th17-mediated immunity against influenza infection. PLoS Pathog. 10, e1003875 (2014).
Wilharm, A. et al. Mutual interplay between IL-17–producing γδT cells and microbiota orchestrates oral mucosal homeostasis. Proc. Natl Acad. Sci. USA 116, 2652–2661 (2019).
Dutzan, N. et al. A dysbiotic microbiome triggers TH17 cells to mediate oral mucosal immunopathology in mice and humans. Sci. Transl. Med. 10, eaat0797 (2018).
Ivanov, I. I. et al. Specific microbiota direct the differentiation of IL-17-producing T-helper cells in the mucosa of the small intestine. Cell Host Microbe 4, 337–349 (2008).
Pandiyan, P. et al. Microbiome dependent regulation of Tregs and Th17 cells in mucosa. Front. Immunol. 10, 426 (2019).
Dutzan, N. et al. On-going mechanical damage from mastication drives homeostatic Th17 cell responses at the oral barrier. Immunity 46, 133–147 (2017).
Anipindi, V. C. et al. IL-17 production by γδ T cells is critical for inducing Th17 responses in the female genital tract and regulated by estradiol and microbiota. ImmunoHorizons 3, 317–330 (2019).
Kitamoto, S. et al. The intermucosal connection between the mouth and gut in commensal pathobiont-driven colitis. Cell, https://doi.org/10.1016/j.cell.2020.05.048 (2020).
Abusleme, L. & Moutsopoulos, N. M. IL-17: overview and role in oral immunity and microbiome. Oral. Dis. 23, 854–865 (2017).
Hajishengallis, G. et al. Low-abundance biofilm species orchestrates inflammatory periodontal disease through the commensal microbiota and complement. Cell Host Microbe 10, 497–506 (2011).
Pulendran, B. et al. Lipopolysaccharides from distinct pathogens induce different classes of immune responses in vivo. J. Immunol. 167, 5067–5076 (2001).
Zhang, L. et al. Porphyromonas gingivalis lipopolysaccharide promotes T- helper 17 cell differentiation from human CD4 naïve T cells via toll-like receptor-2 in vitro. Arch. Oral Biol. 107, 104483 (2019).
Chen, K. et al. Th17 cells mediate clade-specific, serotype-independent mucosal immunity. Immunity 35, 997–1009 (2011).
Lin, L. et al. Th1-Th17 cells mediate protective adaptive immunity against Staphylococcus aureus and Candida albicans infection in mice. PLoS Pathog. 5, e1000703 (2009).
Kleinnijenhuis, J. et al. Long-lasting effects of BCG vaccination on both heterologous Th1/Th17 responses and innate trained immunity. J. Innate Immun. 6, 152–158 (2014).
Moffitt, K. L. et al. TH17-based vaccine design for prevention of Streptococcus pneumoniae colonization. Cell Host Microbe 9, 158–165 (2011).
Wu, W. et al. Th17-stimulating protein vaccines confer protection against Pseudomonas aeruginosa pneumonia. Am. J. Respir. Crit. Care Med. 186, 420–427 (2012).
Lin, Y., Slight, S. R. & Khader, S. A. Th17 cytokines and vaccine-induced immunity. Semin. Immunopathol. 32, 79–90 (2010).
Dieli, F. et al. Granulysin-dependent killing of intracellular and extracellular Mycobacterium tuberculosis by Vgamma9/Vdelta2 T lymphocytes. J. Infect. Dis. 184, 1082–1085 (2001).
Dudal, S. et al. Release of LL-37 by activated human Vγ9Vδ2 T cells: a microbicidal weapon against Brucella suis. J. Immunol. 177, 5533–5539 (2006).
DiTirro, J. et al. Disruption of the cellular inflammatory response to Listeria monocytogenes infection in mice with disruptions in targeted genes.Infect. Immun. 66, 2284–2289 (1998).
Skeen, M. J., Freeman, M. M. & Ziegler, H. K. Changes in peritoneal myeloid populations and their proinflammatory cytokine expression during infection with Listeria monocytogenes are altered in the absence of gamma/delta T cells. J. Leukoc. Biol. 76, 104–115 (2004).
Bi, S. et al. A multicomponent vaccine provides Immunity against local and systemic infections by group a Streptococcus across serotypes. MBio 10, 02600–19 (2019).
Phalke, S. P. et al. γδ T cells shape memory-phenotype αβ T cell populations in non-immunized mice. PLoS ONE 14, e0218827 (2019).
Roark, C. L. et al. A canonical Vγ4Vδ4+ γδ T cell population with distinct stimulation requirements which promotes the Th17 response. Immunol. Res. 55, 217–230 (2013).
Sheridan, B. S. et al. γδ T cells exhibit multifunctional and protective memory in intestinal tissues. Immunity 39, 184–195 (2013).
Bonneville, M., O’Brien, R. L. & Born, W. K. Gammadelta T cell effector functions: a blend of innate programming and acquired plasticity. Nat. Rev. Immunol. 10, 467–478 (2010).
Hoytema van Konijnenburg, D. P. et al. Intestinal epithelial and intraepithelial T cell crosstalk mediates a dynamic response to infection. Cell 171, 783–794.e13 (2017).
Toulon, A. et al. A role for human skin-resident T cells in wound healing. J. Exp. Med 206, 743–750 (2009).
Sutton, C. E. et al. Interleukin-1 and IL-23 induce innate IL-17 production from gammadelta T cells, amplifying Th17 responses and autoimmunity. Immunity 31, 331–341 (2009).
Jensen, K. D. C. et al. Thymic selection determines gammadelta T cell effector fate: antigen-naive cells make interleukin-17 and antigen-experienced cells make interferon gamma. Immunity 29, 90–100 (2008).
Vantourout, P. & Hayday, A. Six-of-the-best: unique contributions of γδ T cells to immunology. Nat. Rev. Immunol. 13, 88–100 (2013).
Cui, Y., Kang, L., Cui, L. & He, W. Human γδ T cell Recognition of lipid A is predominately presented by CD1b or CD1c on dendritic cells. Biol. Direct 4, 47 (2009).
Shiromizu, C. M. & Jancic, C. C. γδ T lymphocytes: an effector cell in autoimmunity and infection. Front. Immunol. 9, 2389 (2018).
Bystrom, J. et al. Harnessing the therapeutic potential of Th17 cells. Mediators Inflamm. 2015, 205156 (2015).
Martin, B., Hirota, K., Cua, D. J., Stockinger, B. & Veldhoen, M. Interleukin-17-producing γδ T cells selectively expand in response to pathogen products and environmental signals. Immunity 31, 321–330 (2009).
Born, W. K., Huang, Y., Zeng, W., Torres, R. M. & O’Brien, R. L. A special connection between γδ T cells and natural antibodies? Arch. Immunol. Ther. Exp. 64, 455–462 (2016).
Engen, S. A. et al. The oral commensal Streptococcus mitis shows a mixed memory Th cell signature that is similar to and cross-reactive with Streptococcus pneumoniae. PLoS ONE 9, e104306 (2014).
Shekhar, S., Khan, R., Schenck, K. & Petersen, F. C. Intranasal immunization with the commensal Streptococcus mitis confers protective immunity against pneumococcal lung infection. Appl. Environ. Microbiol. 85, 02235–18 (2019).
Miller, S. M. et al. Novel lipidated imidazoquinoline TLR7/8 adjuvants elicit influenza-specific Th1 immune responses and protect against heterologous H3N2 influenza nhallenge in mice. Front. Immunol. 11, 406 (2020).
Christensen, D. et al. Seasonal influenza split vaccines confer partial cross-protection against heterologous influenza virus in ferrets when combined with the CAF01 adjuvant. Front. Immunol. 8, 1928 (2017).
Smith, D., Streatfield, S. J., Acosta, H., Ganesan, S. & Fattom, A. A nanoemulsion-adjuvanted intranasal H5N1 influenza vaccine protects ferrets against homologous and heterologous H5N1 lethal challenge. Vaccine 37, 6162–6170 (2019).
Anipindi, V. C. et al. Estradiol enhances CD4 T-cell anti-viral immunity by priming vaginal DCs to induce Th17 responses via an IL-1-dependent pathway. PLOS Pathog. 12, e1005589 (2016).
Hirota, K. et al. Plasticity of TH17 cells in Peyer’s patches is responsible for the induction of T cell–dependent IgA responses. Nat. Immunol. 14, 372–379 (2013).
Lindenstrøm, T. et al. Vaccine-induced th17 cells are maintained long-term postvaccination as a distinct and phenotypically stable memory subset. Infect. Immun. 80, 3533–3544 (2012).
Christensen, D. Development and evaluation of CAF01. Immunopotent. Modern Vaccines 333–345, https://doi.org/10.1016/b978-0-12-804019-5.00017-7 (2017).
Agger, E. M. et al. Cationic liposomes formulated with synthetic mycobacterial cordfactor (CAF01): a versatile adjuvant for vaccines with different immunological requirements. PLoS ONE 3, e3116 (2008).
Lonez, C., Vandenbranden, M. & Ruysschaert, J.-M. Cationic lipids activate intracellular signaling pathways. Adv. Drug Deliv. Rev. 64, 1749–1758 (2012).
Wang, S. H. et al. A novel combination of intramuscular vaccine adjuvants, nanoemulsion and CpG produces an effective immune response against influenza A virus. Vaccine 38, 3537–3544 (2020).
Wong, P. T. et al. Formulation, high throughput in vitro screening and in vivo functional characterization of nanoemulsion-based intranasal vaccine adjuvants. PLoS ONE 10, e0126120 (2015).
Oberoi, H. S., Yorgensen, Y. M., Morasse, A., Evans, J. T. & Burkhart, D. J. PEG modified liposomes containing CRX-601 adjuvant in combination with methylglycol chitosan enhance the murine sublingual immune response to influenza vaccination. J. Control. Release 223, 64–74 (2016).
Farhadihosseinabadi, B. et al. Crosstalk between chitosan and cell signaling pathways. Cell. Mol. Life Sci. 76, 2697–2718 (2019).
Salk, H. M. et al. Taxa of the nasal microbiome are associated with influenza-specific IgA response to live attenuated influenza vaccine. PLoS ONE 11, e0162803 (2016).
Wang, J. et al. Bacterial colonization dampens influenza-mediated acute lung injury via induction of M2 alveolar macrophages. Nat. Commun. 4, 2106. https://doi.org/10.1038/ncomms3106 (2013).
Reiss-Mandel, A., Rubin, C., Zayoud, M., Rahav, G. & Regev-Yochay, G. Staphylococcus aureus colonization induces strain-specific suppression of interleukin-17. Infect. Immun. 86, 00834–17 (2018).
Kleinstein, S. E., Nelson, K. E. & Freire, M. Inflammatory networks linking oral microbiome with systemic health and disease. J. Dent. Res. 99, 1131–1139 (2020).
Freire, M. et al. Longitudinal study of oral microbiome variation in twins. Sci. Rep. 10, 7954 (2020).
Torralba, M. G. et al. Oral microbial species and virulence factors associated with oral squamous cell carcinoma. Microb. Ecol. https://doi.org/10.1007/s00248-020-01596-5 (2020).
Perera, D. et al. Impaired host response and the presence of Acinetobacter baumannii in the serum microbiome of type-II diabetic patients. iScience 24, 101941 (2021).
Freire, M. O., Dalli, J., Serhan, C. N. & Van Dyke, T. E. Neutrophil resolvin E1 receptor expression and function in type 2 diabetes. J. Immunol. 198, 718–728 (2017).
Kleinstein, S. E. et al. Transcriptomics of type 2 diabetic and healthy human neutrophils. In Review, https://doi.org/10.21203/rs.2.21951/v1 (2020).
Kholy, K. E., Genco, R. J. & Van Dyke, T. E. Oral infections and cardiovascular disease. Trends Endocrinol. Metab. 26, 315–321 (2015).
Glick, M. The Oral-systemic Health Connection: A Guide to Patient Care. (Quintessence Publishing Company Incorporated, 2019).
Marco, A. C. D. et al. Oral condition of critical patients and its correlation with ventilator-associated pneumonia: a pilot study. Rev. de. Odontologia da UNESP 42, 182–187 (2013).
Marino, P. J. et al. Comparison of foam swabs and toothbrushes as oral hygiene interventions in mechanically ventilated patients: a randomised split mouth study. BMJ Open Respiratory Res. 3, e000150 (2016).
Linden, G. J., Lyons, A. & Scannapieco, F. A. Periodontal systemic associations: review of the evidence. J. Clin. Periodontol. 40, S8–S19 (2013).
Sencio, V. et al. Gut dysbiosis during influenza contributes to pulmonary pneumococcal superinfection through altered short-chain fatty acid production. Cell Rep. 30, 2934–2947.e6 (2020).
Zuo, T. et al. Alterations in gut microbiota of patients with COVID-19 during time of hospitalization. Gastroenterology, https://doi.org/10.1053/j.gastro.2020.05.048 (2020).
Gu, S. et al. Alterations of the gut microbiota in patients with coronavirus disease 2019 or H1N1 influenza. Clin. Infect. Dis. https://doi.org/10.1093/cid/ciaa709 (2020).
Bhattacharjee, A. & Hand, T. W. Role of nutrition, infection, and the microbiota in the efficacy of oral vaccines. Clin. Sci. 132, 1169–1177 (2018).
Braun, J. et al. SARS-CoV-2-reactive T cells in healthy donors and patients with COVID-19. Nature, https://doi.org/10.1038/s41586-020-2598-9 (2020).
Le Bert, N. et al. SARS-CoV-2-specific T cell immunity in cases of COVID-19 and SARS, and uninfected controls. Nature, https://doi.org/10.1038/s41586-020-2550-z (2020).
Grifoni, A. et al. Targets of T cell responses to SARS-CoV-2 coronavirus in humans with COVID-19 disease and unexposed individuals. Cell 181, 1489–1501.e15 (2020).
Weiskopf, D. et al. Phenotype and kinetics of SARS-CoV-2-specific T cells in COVID-19 patients with acute respiratory distress syndrome. Sci Immunol. 5, eabd2071, https://doi.org/10.1126/sciimmunol (2020).
Hotez, P. J., Bottazzi, M. E. & Corry, D. B. The potential role of Th17 immune responses in coronavirus immunopathology and vaccine-induced immune enhancement. Microbes Infect. 22, 165–167 (2020).
Wan, M. T., Shin, D. B., Winthrop, K. L. & Gelfand, J. M. The risk of respiratory tract infections and symptoms in psoriasis patients treated with interleukin 17 pathway-inhibiting biologics: a meta-estimate of pivotal trials relevant to decision making during the COVID-19 pandemic. J. Am. Acad. Dermatol. 83, 677–679 (2020).
Savarin, C. et al. IFN-γ protects from lethal IL-17 mediated viral encephalomyelitis independent of neutrophils. J. Neuroinflammation 9, 104 (2012).
Sampaio-Maia, B. & Monteiro-Silva, F. Acquisition and maturation of oral microbiome throughout childhood: an update. Dent. Res. J. 11, 291–301 (2014).
Freire, M., Nelson, K. E. & Edlund, A. The oral host–microbial interactome: an ecological chronometer of health? Trends Microbiol. https://doi.org/10.1016/j.tim.2020.11.004 (2020).
Douglas, C. W. Bacterial-protein interactions in the oral cavity. Adv. Dent. Res. 8, 254–262 (1994).
Tabak, L. A. In defense of the oral cavity: structure, biosynthesis, and function of salivary mucins. Annu. Rev. Physiol. 57, 547–564 (1995).
Darveau, R. P., Belton, C. M., Reife, R. A. & Lamont, R. J. Local chemokine paralysis, a novel pathogenic mechanism for Porphyromonas gingivalis. Infect. Immun. 66, 1660–1665 (1998).
Delima, A. J. & Van Dyke, T. E. Origin and function of the cellular components in gingival crevice fluid. Periodontol 2000 31, 55–76 (2003).
Schiott, C. R., Rindom Schiott, C. & Loe, H. The origin and variation in number of leukocytes in the human saliva. J. Periodontal Res. 5, 36–41 (1970).
Stark, M. A. et al. Phagocytosis of apoptotic neutrophils regulates granulopoiesis via IL-23 and IL-17. Immunity 22, 285–294 (2005).
Mei, J. et al. Cxcr2 and Cxcl5 regulate the IL-17/G-CSF axis and neutrophil homeostasis in mice. J. Clin. Invest. 122, 974–986 (2012).
Ebersole, J. L. et al. Transcriptome analysis of B cell immune functions in periodontitis: mucosal tissue responses to the oral microbiome in aging. Front. Immunol. 7, 272 (2016).
Dutzan, N., Konkel, J. E., Greenwell-Wild, T. & Moutsopoulos, N. M. Characterization of the human immune cell network at the gingival barrier. Mucosal Immunol. 9, 1163–1172 (2016).
Moutsopoulos, N. M. & Konkel, J. E. Tissue-specific immunity at the oral mucosal barrier. Trends Immunol. 39, 276–287 (2018).
Alvarez, C. et al. Regulatory T lymphocytes in periodontitis: a translational view. Mediators Inflamm. 2018, 7806912 (2018).
Yu, T. et al. Enhanced activity of the macrophage M1/M2 phenotypes and phenotypic switch to M1 in periodontal infection. J. Periodontol. 87, 1092–1102 (2016).
Colotta, F., Re, F., Polentarutti, N., Sozzani, S. & Mantovani, A. Modulation of granulocyte survival and programmed cell death by cytokines and bacterial products. Blood 80, 2012–2020 (1992).
Lee, A., Whyte, M. K. B. & Haslett, C. Inhibition of apoptosis and prolongation of neutrophil functional longevity by inflammatory mediators. J. Leukoc. Biol. 54, 283–288 (1993).
Kobayashi, S. D., Malachowa, N. & DeLeo, F. R. Influence of Microbes on Neutrophil Life and Death. Front. Cell. Infect Microbiol. 7, 159, https://doi.org/10.3389/fcimb.2017.00159 (2017).
Soehnlein, O., Lindbom, L. & Weber, C. Mechanisms underlying neutrophil-mediated monocyte recruitment. Blood 114, 4613–4623 (2009).
Minutti, C. M., Knipper, J. A., Allen, J. E. & Zaiss, D. M. W. Tissue-specific contribution of macrophages to wound healing. Semin. Cell Dev. Biol. 61, 3–11 (2017).
Moutsopoulos, N. M. et al. Defective neutrophil recruitment in leukocyte adhesion deficiency type I disease causes local IL-17-driven inflammatory bone loss. Sci. Transl. Med. 6, 229ra40 (2014).
El Kebir, D., József, L., Pan, W. & Filep, J. G. Myeloperoxidase delays neutrophil apoptosis through CD11b/CD18 integrins and prolongs inflammation. Circ. Res. 103, 352–359 (2008).
Bär, E., Whitney, P. G., Moor, K., Reis e Sousa, C. & LeibundGut-Landmann, S. IL-17 regulates systemic fungal immunity by controlling the functional competence of NK cells. Immunity 40, 117–127 (2014).
Bagri, P. et al. Novel role for interleukin-17 in enhancing type 1 helper T cell immunity in the female genital tract following mucosal herpes simplex virus 2 vaccination. J. Virol. 91, 01234–17 (2017).
Zenobia, C. et al. Commensal bacteria-dependent select expression of CXCL2 contributes to periodontal tissue homeostasis. Cell. Microbiol. 15, 1419–1426 (2013).
Eskan, M. A. et al. The leukocyte integrin antagonist Del-1 inhibits IL-17-mediated inflammatory bone loss. Nat. Immunol. 13, 465–473 (2012).
Towstyka, N. Y. et al. Modulation of γδ T-cell activation by neutrophil elastase. Immunology 153, 225–237 (2018).
Liu, C. et al. The role of reactive oxygen species and autophagy in periodontitis and their potential linkage. Front. Physiol. 8, 439 (2017).
Giannopoulou, C., Andersen, E., Demeurisse, C. & Cimasoni, G. Neutrophil elastase and its inhibitors in human gingival crevicular fluid during experimental gingivitis. J. Dent. Res. 71, 359–363 (1992).
Rowe, J. C., Attia, Z., Kim, E., Cormet-Boyaka, E. & Boyaka, P. N. A novel supplementation approach to enhance host response to sublingual vaccination. Sci. Rep. 9, 715 (2019).
Jee, J. et al. Neutrophils negatively regulate induction of mucosal IgA responses after sublingual immunization. Mucosal Immunol. 8, 735–745 (2015).
Mitsdoerffer, M. et al. Proinflammatory T helper type 17 cells are effective B-cell helpers. Proc. Natl Acad. Sci. USA 107, 14292–14297 (2010).
Gallorini, S. et al. Sublingual immunization with a subunit influenza vaccine elicits comparable systemic immune response as intramuscular immunization, but also induces local IgA and TH17 responses. Vaccine 32, 2382–2388 (2014).
Yang, C.-W., Strong, B. S. I., Miller, M. J. & Unanue, E. R. Neutrophils influence the level of antigen presentation during the immune response to protein antigens in adjuvants. J. Immunol. 185, 2927–2934 (2010).
Davey, M. S. et al. Microbe-specific unconventional T cells induce human neutrophil differentiation into antigen cross-presenting cells. J. Immunol 193, 3704–3716 (2014).
Champagne, E. γδ T cell receptor ligands and modes of antigen recognition. Archivum Immunol. et. Therapiae Exp. 59, 117–137 (2011).
Mokuno, Y. et al. Expression of toll-like receptor 2 on γδ T cells bearing invariant Vγ6/Vδ1 induced by Escherichia coli infection in mice. J. Immunol. 165, 931–940 (2000).
Uriarte, S. M., Edmisson, J. S. & Jimenez-Flores, E. Human neutrophils and oral microbiota: a constant tug-of-war between a harmonious and a discordant coexistence. Immunol. Rev. 273, 282–298 (2016).
Nielsen, M. M., Witherden, D. A. & Havran, W. L. γδ T cells in homeostasis and host defence of epithelial barrier tissues. Nat. Rev. Immunol. 17, 733–745 (2017).
Zenobia, C. & Hajishengallis, G. Basic biology and role of interleukin-17 in immunity and inflammation. Periodontology 2000 69, 142–159 (2015).
Kumar, S., Sunagar, R. & Gosselin, E. Bacterial protein toll-like-receptor agonists: a novel perspective on vaccine adjuvants. Front. Immunol. 10, 1144 (2019).
Stephen, T. L., Groneck, L. & Kalka-Moll, W. M. The modulation of adaptive immune responses by bacterial zwitterionic polysaccharides. Int. J. Microbiol. 2010, 917075 (2010).
Vacchelli, E. et al. Trial watch: FDA-approved Toll-like receptor agonists for cancer therapy. Oncoimmunology 1, 894–907 (2012).
Wang, D. B. Monophosphoryl lipid A is an lipopolysaccharide-derived Toll-like receptor 4 agonist which may improve Alzheimer’s disease pathology. Expert Opin. Biol. Ther. 13, 1639–1641 (2013).
Gregg, K. A. et al. Rationally designed TLR4 ligands for vaccine adjuvant discovery. MBio 8, 00492–17 (2017).
Fukasaka, M. et al. A lipopolysaccharide from pantoea agglomerans Is a promising adjuvant for sublingual vaccines to induce systemic and mucosal immune responses in mice via TLR4 pathway. PLoS ONE 10, e0126849 (2015).
Han, J. E. et al. Characterization of the structure and immunostimulatory activity of a vaccine adjuvant, de-O-acylated lipooligosaccharide. PLoS ONE 9, e85838 (2014).
Monie, A., Hung, C.-F., Roden, R. & Wu, T.-C. Cervarix: a vaccine for the prevention of HPV 16, 18-associated cervical cancer. Biologics 2, 97–105 (2008).
Coats, S. R. et al. The lipid A phosphate position determines differential host Toll-like receptor 4 responses to phylogenetically related symbiotic and pathogenic bacteria. Infect. Immun. 79, 203–210 (2011).
Darveau, R. P., Arbabi, S., Garcia, I., Bainbridge, B. & Maier, R. V. Porphyromonas gingivalis lipopolysaccharide is both agonist and antagonist for p38 mitogen-activated protein kinase activation. Infect. Immun. 70, 1867–1873 (2002).
Al-Qutub, M. N. et al. Hemin-dependent modulation of the lipid A structure of Porphyromonas gingivalis lipopolysaccharide. Infect. Immun. 74, 4474–4485 (2006).
Curtis, M. A. et al. Temperature-dependent modulation of Porphyromonas gingivalis lipid A structure and interaction with the innate host defenses. Infect. Immun. 79, 1187–1193 (2011).
Dixon, D. R. & Darveau, R. P. Lipopolysaccharide heterogeneity: innate host responses to bacterial modification of lipid a structure. J. Dent. Res. 84, 584–595 (2005).
Bainbridge, B. W., Coats, S. R., Pham, T.-T. T., Reife, R. A. & Darveau, R. P. Expression of a Porphyromonas gingivalis lipid A palmitylacyltransferase in Escherichia coli yields a chimeric lipid A with altered ability to stimulate interleukin-8 secretion. Cell. Microbiol. 8, 120–129 (2006).
Reife, R. A. et al. Porphyromonas gingivalis lipopolysaccharide lipid A heterogeneity: differential activities of tetra- and penta-acylated lipid A structures on E-selectin expression and TLR4 recognition. Cell. Microbiol. 8, 857–868 (2006).
Zaric, S. et al. Impaired immune tolerance to porphyromonas gingivalis lipopolysaccharide promotes neutrophil migration and decreased apoptosis. Infect. Immun. 78, 4151–4156 (2010).
To, T. T., Gümüş, P., Nizam, N., Buduneli, N. & Darveau, R. P. Subgingival plaque in periodontal health antagonizes at toll-Like receptor 4 and inhibits E-Selectin expression on endothelial cells. Infect. Immun. 84, 120–126 (2016).
Kajikawa, T. et al. Frontline science: activation of metabolic nuclear receptors restores periodontal tissue homeostasis in mice with leukocyte adhesion deficiency-1. J. Leukoc. Biol. https://doi.org/10.1002/JLB.5HI0420-648R (2020).
Griffen, A. L., Becker, M. R., Lyons, S. R., Moeschberger, M. L. & Leys, E. J. Prevalence of Porphyromonas gingivalis and periodontal health status. J. Clin. Microbiol 36, 3239–3242 (1998).
McClellan, D. L., Griffen, A. L. & Leys, E. J. Age and prevalence of Porphyromonas gingivalis in children. J. Clin. Microbiol. 34, 2017–2019 (1996).
Su, Y. et al. Ecological balance of oral microbiota is required to maintain oral mesenchymal stem cell homeostasis. Stem Cells 36, 551–561 (2018).
Park, Y.-D. et al. Porphyromonas gingivalis lipopolysaccharide regulates interleukin (IL)-17 and IL-23 expression via SIRT1 modulation in human periodontal ligament cells. Cytokine 60, 284–293 (2012).
Glowczyk, I. et al. Inactive Gingipains from P. gingivalis Selectively Skews T Cells toward a Th17 Phenotype in an IL-6 Dependent Manner. Front. Cell. Infect Microbiol. 7, 140, https://doi.org/10.3389/fcimb.2017.00140 (2017).
Monteiro, A. C. et al. Kinin danger signals proteolytically released by gingipain induce fimbriae-specific IFN-gamma- and IL-17-producing T cells in mice infected intramucosally with Porphyromonas gingivalis. J. Immunol. 183, 3700–3711 (2009).
Zhou, Q., Desta, T., Fenton, M., Graves, D. T. & Amar, S. Cytokine profiling of macrophages exposed to porphyromonas gingivalis, its lipopolysaccharide, or its FimA protein. Infect. Immun. 74, 1995–1995 (2006).
Bittner-Eddy, P. D., Fischer, L. A. & Costalonga, M. Transient Expression of IL-17A in Foxp3 Fate-Tracked Cells in Porphyromonas gingivalis-Mediated Oral Dysbiosis. Front. Immunol. 11, 677, https://doi.org/10.3389/fimmu.2020.00677 (2020).
Zenobia, C. & Hajishengallis, G. Porphyromonas gingivalis virulence factors involved in subversion of leukocytes and microbial dysbiosis. Virulence 6, 236–243 (2015).
Khader, S. & Das, S. Faculty Opinions recommendation of Periodontitis-associated pathogens P. gingivalis and A. actinomycetemcomitans activate human CD14() monocytes leading to enhanced Th17/IL-17 responses. Faculty Opinions – Post-Publication Peer Review of the Biomedical Literature, https://doi.org/10.3410/f.726443835.793532082 (2017).
Jotwani, R. & Cutler, C. W. Fimbriated Porphyromonas gingivalis is more efficient than fimbria-deficient P. gingivalis in entering human dendritic cells in vitro and induces an inflammatory Th1 effector response. Infect. Immun. 72, 1725–1732 (2004).
Wang, M. et al. Fimbrial proteins of porphyromonas gingivalis mediate in vivo virulence and exploit TLR2 and complement receptor 3 to persist in macrophages. J. Immunol. 179, 2349–2358 (2007).
Nady, S., Ignatz-Hoover, J. & Shata, M. T. Interleukin-12 is the optimum cytokine to expand human Th17 cells in vitro. Clin. Vaccin. Immunol. 16, 798–805 (2009).
Dominy, S. S. et al. In Alzheimer’s disease brains: evidence for disease causation and treatment with small-molecule inhibitors. Sci. Adv. 5, eaau3333 (2019).
Olsen, I. & Potempa, J. Strategies for the inhibition of gingipains for the potential treatment of periodontitis and associated systemic diseases. J. Oral Microbiol. 6, https://doi.org/10.3402/jom.v6.24800 (2014).
Jain, S., Coats, S. R., Chang, A. M. & Darveau, R. P. A novel class of lipoprotein lipase-sensitive molecules mediates Toll-like receptor 2 activation by Porphyromonas gingivalis. Infect. Immun. 81, 1277–1286 (2013).
Corpuz, T. M. et al. IL-2 shapes the survival and plasticity of IL-17-producing γδ T cells. J. Immunol. 199, 2366–2376 (2017).
Mahanonda, R. et al. Monocyte activation by Porphyromonas gingivalis LPS in aggressive periodontitis with the use of whole-blood cultures. J. Dent. Res. 83, 540–545 (2004).
Cibrián, D. & Sánchez-Madrid, F. CD69: from activation marker to metabolic gatekeeper. Eur. J. Immunol. 47, 946–953 (2017).
Zhu, X.-Q. et al. Effects of Porphyromonas gingivalis lipopolysaccharide tolerized monocytes on inflammatory responses in neutrophils. PLoS ONE 11, e0161482 (2016).
Sciorati, C. et al. Autocrine nitric oxide modulates CD95-induced apoptosis in γδ T lymphocytes. J. Biol. Chem. 272, 23211–23215 (1997).
Frolov, I., Houri-Hadad, Y., Soskolne, A. & Shapira, L. In vivo exposure to Porphyromonas gingivalis up-regulates nitric oxide but suppresses tumour necrosis factor-α production by cultured macrophages. Immunology 93, 323–328 (2001).
Pudla, M., Srisatjaluk, R. & Utaisincharoen, P. Induction of inducible nitric oxide synthase (iNOS) in Porphyromonas gingivalis LPS-treated mouse macrophage cell line (RAW264.7) requires Toll-like receptor 9. Inflamm. Res. 67, 723–726 (2018).
Jones-Carson, J. The role of T cells in mucosal immunity to murine candidiasis. p. 274 (University of Wisconsin–Madison, 1996).
Wang, Y., Zhang, W., Xu, L. & Jin, J. O. Porphyromonas gingivalis Lipopolysaccharide Induced Proliferation and Activation of Natural Killer Cells in Vivo. Molecules 21, 1086, https://doi.org/10.3390/molecules21081086 (2016).
Maniar, A. et al. Human γδ T lymphocytes induce robust NK cell–mediated antitumor cytotoxicity through CD137 engagement. Blood 116, 1726–1733 (2010).
Enersen, M., Nakano, K. & Amano, A. Porphyromonas gingivalis fimbriae. J. Oral Microbiol. 5, https://doi.org/10.3402/jom.v5i0.20265 (2013).
Lamont, R. J. & Jenkinson, H. F. Life below the gum line: pathogenic mechanisms of Porphyromonas gingivalis. Microbiol. Mol. Biol. Rev. 62, 1244–1263 (1998).
Jia, L. et al. Pathogenesis of Important Virulence Factors of Porphyromonas gingivalis via Toll-Like Receptors. Front. Cell. Infect. Microbiol 9, 262, https://doi.org/10.3389/fcimb.2019.00262 (2019).
Zeituni, A. E., Jotwani, R., Carrion, J. & Cutler, C. W. Targeting of DC-SIGN on human dendritic cells by minor fimbriated Porphyromonas gingivalis strains elicits a distinct effector T cell response. J. Immunol. 183, 5694–5704 (2009).
Hajishengallis, G., Wang, M., Harokopakis, E., Triantafilou, M. & Triantafilou, K. Porphyromonas gingivalis fimbriae proactively modulate β2 integrinadhesive activity and promote binding to and internalization by macrophages. Infect. Immun. 74, 5658–5666 (2006).
Ehirchiou, D. et al. CD11b facilitates the development of peripheral tolerance by suppressing Th17 differentiation. J. Exp. Med. 204, 1519–1524 (2007).
Liu, J., Wang, Y. & Ouyang, X. Beyond toll-like receptors: Porphyromonas gingivalis induces IL-6, IL-8, and VCAM-1 expression through NOD-mediated NF-κB and ERK signaling pathways in periodontal fibroblasts. Inflammation 37, 522–533 (2014).
Okugawa, T., Kaneko, T., Yoshimura, A., Silverman, N. & Hara, Y. NOD1 and NOD2 mediate sensing of periodontal pathogens. J. Dent. Res. 89, 186–191 (2010).
Zhou, Q. & Amar, S. Identification of signaling pathways in macrophage exposed to Porphyromonas gingivalis or to its purified cell wall components. J. Immunol. 179, 7777–7790 (2007).
Maisonneuve, C., Bertholet, S., Philpott, D. J. & De Gregorio, E. Unleashing the potential of NOD- and Toll-like agonists as vaccine adjuvants. Proc. Natl Acad. Sci. USA 111, 12294–12299 (2014).
Ye, J. et al. TLR7 signaling regulates Th17 cells and autoimmunity: novel potential for autoimmune therapy. J. Immunol. 199, 941–954 (2017).
Zimmermann, P. & Curtis, N. The influence of probiotics on vaccine responses – a systematic review. Vaccine 36, 207–213 (2018).
Higgins, S. C., Jarnicki, A. G., Lavelle, E. C. & Mills, K. H. G. TLR4 mediates vaccine-induced protective cellular immunity to Bordetella pertussis: role of IL-17-producing T cells. J. Immunol. 177, 7980–7989 (2006).
Lambert, E. E., Buisman, A.-M. & van Els, C. A. C. M. Superior B. pertussis specific CD4+ T-cell immunity imprinted by natural infection. Adv. Exp. Med. Biol. 1183, 81–98 (2019).
Conti, H. R. et al. Th17 cells and IL-17 receptor signaling are essential for mucosal host defense against oral candidiasis. J. Exp. Med 206, 299–311 (2009).
Lin, J.-S., Kummer, L. W., Szaba, F. M. & Smiley, S. T. IL-17 contributes to cell-mediated defense against pulmonary Yersinia pestis infection. J. Immunol. 186, 1675–1684 (2011).
Hemmi, M. et al. T Helper 17 Promotes Inductionof Antigen-Specific Gut-Mucosal Cytotoxic T Lymphocytes following Adenovirus Vector Vaccination. Front. Immunol. 8, 1456, https://doi.org/10.3389/fimmu.2017.01456 (2017).
Khader, S. A. et al. IL-23 and IL-17 in the establishment of protective pulmonary CD4+ T cell responses after vaccination and during Mycobacterium tuberculosis challenge. Nat. Immunol. 8, 369–377 (2007).
Luzza, F. et al. Up-regulation of IL-17 is associated with bioactive IL-8 expression in Helicobacter pylori-infected human gastric mucosa. J. Immunol. 165, 5332–5337 (2000).
Bao, J. et al. Decreased frequencies of Th17 and Tc17 cells in patients infected with avian influenza A (H7N9) virus. J. Immunol. Res. 2019, 1418251 (2019).
Liu, J. et al. Early production of IL-17 protects against acute pulmonary Pseudomonas aeruginosa infection in mice. FEMS Immunol. Med. Microbiol. 61, 179–188 (2011).
Brown, J., Hammerschmidt, S. & Orihuela, C. Streptococcus Pneumoniae: Molecular Mechanisms of Host-Pathogen Interactions. (Academic Press, 2015).
Zelante, T. et al. IL-23 and the Th17 pathway promote inflammation and impair antifungal immune resistance. Eur. J. Immunol. 37, 2695–2706 (2007).
Wüthrich, M. et al. Vaccine-induced protection against 3 systemic mycoses endemic to North America requires Th17 cells in mice. J. Clin. Invest. 121, 554–568 (2011).
Ma, W.-T., Yao, X.-T., Peng, Q. & Chen, D.-K. The protective and pathogenic roles of IL-17 in viral infections: friend or foe? Open Biol. 9, 190109 (2019).
Hartigan-O’Connor, D. J., Hirao, L. A., McCune, J. M. & Dandekar, S. Th17 cells and regulatory T cells in elite control over HIV and SIV. Curr. Opin. HIV AIDS 6, 221–227 (2011).
Van Dis, E. et al. STING-activating adjuvants elicit a Th17 immune response and protect against Mycobacterium tuberculosis infection. Cell Rep. 23, 1435–1447 (2018).
Wern, J. E., Sorensen, M. R., Olsen, A. W., Andersen, P., & Follmann, F. Simultaneous subcutaneous and intranasal administration of a CAF01-adjuvanted vaccine elicits elevated IgA and protective Th1/Th17 responses in the genital tract. Front. Immunol. 8, 569 (2017).
Boyaka, P. N. Inducing mucosal IgA: a challenge for vaccine adjuvants and delivery systems. J. Immunol. 199, 9–16 (2017).
Bielinska, A. U. et al. Induction of Th17 cellular immunity with a novel nanoemulsion adjuvant. Crit. Rev. Immunol. 30, 189–199 (2010).
Leliefeld, P. H. C., Koenderman, L. & Pillay, J. How neutrophils shape adaptive immune responses. Front. Immunol. 6, 471, https://doi.org/10.3389/fimmu.2015.00471 (2015).
Acknowledgements
We would like to thank Marlene Blessing for editorial support. This work was supported in part by The Conrad Prebys Foundation Grant (20-122), U.S. Public Health Service Grants (R00 DE0234804) from the National Institute of Dental and Cranial Research (given to MF).
Author information
Authors and Affiliations
Contributions
M.F. and C.Z. have developed the initial draft. M.F., K.L.H. and C.Z. have revised the entire manuscript and all authors have reviewed the final version of the manuscript. C.Z. developed the figures and all authors revised and finalized the figures and table. There are no competing interests. We agree to be accountable for all aspects of the review in ensuring that questions related to the accuracy or integrity of any part of the review are appropriately investigated and resolved. We confirm that all authors read and approved the final manuscript version to be published.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zenobia, C., Herpoldt, KL. & Freire, M. Is the oral microbiome a source to enhance mucosal immunity against infectious diseases?. npj Vaccines 6, 80 (2021). https://doi.org/10.1038/s41541-021-00341-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41541-021-00341-4
This article is cited by
-
Oral microbiome dysbiosis among cigarette smokers and smokeless tobacco users compared to non-users
Scientific Reports (2024)
-
Helicobacter pylori in oral cavity: current knowledge
Clinical and Experimental Medicine (2024)
-
Oral or intranasal immunization with recombinant Lactobacillus plantarum displaying head domain of Swine Influenza A virus hemagglutinin protects mice from H1N1 virus
Microbial Cell Factories (2022)
-
Targeting the gut microbiota for cancer therapy
Nature Reviews Cancer (2022)