Abstract
The clinical diagnosis of vascular dementia (VaD) is based on imaging criteria, and specific biochemical markers are not available. Here, we investigated the potential of cerebrospinal fluid (CSF) lipocalin 2 (LCN2), a secreted glycoprotein that has been suggested as mediating neuronal damage in vascular brain injuries. The study included four independent cohorts with a total n = 472 samples. LCN2 was significantly elevated in VaD compared to controls, Alzheimer’s disease (AD), other neurodegenerative dementias, and cognitively unimpaired patients with cerebrovascular disease. LCN2 discriminated VaD from AD without coexisting VaD with high accuracy. The main findings were consistent over all cohorts. Neuropathology disclosed a high percentage of macrophages linked to subacute infarcts, reactive astrocytes, and damaged blood vessels in multi-infarct dementia when compared to AD. We conclude that CSF LCN2 is a promising candidate biochemical marker in the differential diagnosis of VaD and neurodegenerative dementias.
Similar content being viewed by others
Introduction
Vascular dementia (VaD) is considered one of the most common causes of dementia after Alzheimer’s disease (AD)1. Identifying VaD patients for epidemiological or clinical research and clinical trials as well as monitoring of therapeutic interventions in VaD trials is challenging. Despite the importance of the disease, diagnosis has been hampered by the lack of well-defined standardized criteria. Commonly used criteria2 have suboptimal diagnostic accuracy3,4. Unified neuropathological criteria have not been available5 in the past and recently published guidelines6 have not yet been widely adopted. A research framework for AD proposed in 20187 includes cerebrospinal fluid (CSF) and imaging markers of tau and amyloid-pathology. Recent efforts to standardize and harmonize diagnosis in VaD have led to a consensus study on vascular cognitive impairment (VCI)8 and the suggestion of imaging and neuropsychological criteria. However, no specific fluid marker for VaD has been established in the clinical routine to date. Imaging markers related to vascular brain injury (VBI), e.g., white matter hyperintensities (WMH) on magnetic resonance imaging (MRI) are accepted markers for VaD9,10 and provide valuable information about cerebrovascular pathology in morphological terms, but they are not pathognomonic10. The nature of their association with dementia has not been fully clarified and further research is still needed11,12,13. Mixed forms of dementia (such as VaD plus AD) are frequent and differentiation from pure forms of AD and VaD is not always possible14. In addition, WMH are frequent in patients with AD15 and might be caused by neurodegeneration16. In summary, new biomarkers are needed to improve diagnosis, to uncover the pathophysiology, and to monitor clinical trials17,18,19,20.
Lipocalin 2 (LCN2) is a secreted glycoprotein involved in innate immunity and highly expressed in the central nervous system in response to injury and inflammatory stimuli21. It has been discussed as a potential marker for AD22. Studies in humans have shown slightly elevated levels of LCN2 in plasma of patients with mild cognitive impairment (MCI)23 and in CSF of patients with multiple sclerosis24. LCN2 has also been discussed as an attractive blood-based biomarker of inflammation, ischemia25, and, in particular kidney injury26,27. Experimental models of VaD have shown that LCN2 mediates hippocampal damage and that LCN2 deficiency is associated with less white matter damage and cognitive decline28 but LCN2 has not been clinically evaluated in the context of differential diagnosis of VaD.
The primary aim of the study was to investigate the potential of LCN2 as a biomarker for VaD. Therefore, we compared CSF LCN2 levels in different forms of dementia, VBI, and controls. Regarding the importance and known difficulties in the clinical differentiation between AD and VaD, we validated our results using independent cohorts. In addition, neuropathological analyses were performed to provide information on the location of LCN2 in brain tissue of patients with chronic multi-infarct dementia (MID), and those with AD.
Our results show that CSF LCN2 is elevated in patients with VaD compared to controls, cognitively unimpaired patients with VBI, and other forms of dementia. The high diagnostic accuracy highlights its potential as a biomarker for VaD in the differential diagnosis of dementia. Our neuropathological investigations display different expression of LCN2 in brains of patients with VaD and AD.
Results
Group descriptions
Data on age, sex and CSF biomarkers in all cohorts are presented in Table 1. In general, more samples from female patients were analysed (254 female, 218 male). VaD patients were older than those from other diagnostic groups. Patients with VaD, Lewy body dementias (LBD), and fronto-temporal dementia (FTD) showed similar profiles of established CSF biomarkers (t-tau, p-tau and amyloid beta 42). Patients with sporadic Creutzfeldt-Jakob disease (CJD) showed highly elevated CSF t-tau. Patients with AD and mixed dementia (MD, AD plus VaD) showed a typical signature of AD-pathology associated biomarkers7.
LCN2 in the differential diagnosis of dementia
Cohort 1 included VaD (n = 27), MD (n = 31), AD (n = 47), LBD (n = 36), FTD (n = 21), CJD (n = 54), as well as non-primarily neurodegenerative and non-ischemic neuropsychiatric diseases (ND, n = 73). In a multi-comparative analysis corrected for covariates, LCN2 concentrations were significantly increased in VaD and MD compared to ND (p < 0.001, Tukey contrast for multiple comparisons of means) and other diagnostic groups (Table 1, Fig. 1a). No significant difference between VaD and MD could be observed. The diagnostic accuracy of LCN2 in the differentiation from ND is indicated by areas under the curve (AUC) and 95% confidence intervals (95% CI) in Fig. 1b, c. LCN2 concentrations discriminated ND from VaD (AUC = 0.88, 95% CI: 0.80–0.96, p < 0.001) and MD (AUC = 0.85, 95% CI: 0.77–0.93, p < 0.001) with high accuracy. LCN2 was able to discriminate AD from VaD (AUC = 0.9, 95% CI: 0.82–0.98, p < 0.001, z test with H0: AUC = 0.5 in all cases) with a sensitivity of 82% and a specificity of 87%, as well as ND from VaD with a sensitivity of 78% and a specificity of 82%. In contrast, LCN2 levels showed no diagnostic value in distinguishing AD, LBD, FTD, and CJD from ND (p > 0.05, z test with H0: AUC = 0.5).
a LCN2 in non-primarily neurodegenerative and non-ischemic neuropsychiatric diseases (ND, n = 73), Alzheimer’s disease (AD, n = 47), vascular dementia (VaD, n = 27), mixed Alzheimer’s and vascular dementia (MD, n = 31), Lewy body dementias (LBD, n = 36), frontotemporal dementia (FTD, n = 21), and sporadic Creutzfeldt-Jakob disease (CJD, n = 54). Results are shown as mean ± SD for each condition. b Area under the curve (AUC) derived from receiver operating characteristic (ROC) curves, Standard Error (SE), 95% Confidence interval (95% CI) for LCN2 in the comparative analyses. c ROC curves of differentiating ND and VaD as well as AD and VaD. Differences between groups were analysed with Tukey contrasts using linear regression models controlled for age and sex. *p < 0.05, **p < 0.01, ***p < 0.001.
LCN2 discriminates VaD from AD in validation cohorts
To validate the presence of elevated LCN2 concentrations in VaD, two independent cohorts (cohort 2 and 3) including ND, AD, and VaD cases were analysed with linear regression models adjusted for covariates and posterior multiple comparisons of means were performed with Tukey contrasts. In cohort 2, LCN2 was significantly increased in VaD (n = 10) compared to ND (n = 24, p < 0.01) and AD (n = 15, p < 0.01). It discriminated VaD from AD with an AUC of 0.88 (Fig. 2a). In cohort 3, LCN2 was significantly increased in VaD (n = 16) compared to ND (n = 15, p < 0.05) and AD (n = 27, p < 0.001). It discriminated VaD from AD with an AUC of 0.83 (Fig. 2b). AUCs derived from ND vs. VaD and AD vs. VaD comparisons were not significantly different (z test) between the cohorts (Supplementary Table 1). In cohort 4, LCN2 was significantly increased in VaD (n = 10) compared to AD (n = 28, p < 0.05, Tukey contrast) but no control group (ND) was available (Fig. 3b). In a separate statistical model including Mini Mental Status Examination (MMSE) score in the group of covariates, VaD groups showed significantly higher LCN2 levels than AD groups in all four cohorts, indicating that dementia stage does not significantly alter the results (Supplementary Table 2).
a Cohort 2: LCN2 concentrations in non-primarily neurodegenerative and non-ischemic neuropsychiatric diseases (ND, n = 24), Alzheimer’s disease (AD, n = 15), and vascular dementia (VaD, n = 10). Area under the curve (AUC) derived from receiver operating characteristic (ROC) curves, Standard Error, and 95% CI in the comparative analysis of VaD versus ND and AD. b Cohort 3: LCN2 concentrations in ND (n = 15), AD (n = 27), and VaD (n = 16). AUC derived from ROC curves, Standard Error, 95% CI in the comparative analysis of VaD versus ND and AD. Differences between groups were analysed with Tukey contrasts using linear regression models controlled for age and sex. *p < 0.05, **p < 0.01, ***p < 0.001.
a Cohort 1: LCN2 levels in Alzheimer’s disease (AD, n = 47), small vessel disease no dementia (SVDND, n = 20), vascular cognitive impairment no dementia (VCIND, n = 7), and vascular dementia (VaD, n = 27). b Cohort 4: LCN2 levels in AD (n = 28), SVDND (n = 3), VCIND (n = 8), and VaD (n = 10). Differences between groups were analysed with Tukey contrasts using linear regression models controlled for age and sex. *p < 0.05, **p < 0.01, ***p < 0.001. a, b Mean ± SD is represented in the graphs. c, d Correlation analysis of LCN2 and Mini Mental Status Examination (MMSE) scores in SVDND, VCIND, and VaD in cohort 1 (n = 48) (c) and cohort 4 (n = 21) (d). Spearman correlation test, correlation coefficients (cc) and associated two-tailed p values. e CSF LCN2 and age-related white matter changes (ARWMC) in SVDND, VCIND, and VaD in cohort 1 (n = 50). Spearman correlation test, correlation coefficients (cc), and associated two-tailed p values. f LCN2 and Fazekas scale in SVDND, VCIND, and VaD in cohort 4 (n = 21). Spearman correlation test, correlation coefficients (cc), and associated two-tailed p values.
VaD types, dementia stage, WMH, and albumin ratio
Regarding different types of VaD, LCN2 levels seemed to be similar in subcortical ischemic vascular dementia (SID) and multi-infarct (cortical) dementia (MID) while being lower in post-(single)stroke dementia (PSD) (Table 2). We could not investigate this observation further because case numbers were too low after building the diagnostic subgroups. Especially for PSD, only few cases were identified.
To assess the association between LCN2 and cognitive impairment in patients with cerebrovascular pathology, LCN2 concentrations were quantified in two cohorts that included cerebral small vessel disease but no dementia (SVDND), vascular cognitive impairment but no dementia (VCIND), and VaD (cohort 1 and 4). AD cases were included in the multi-comparative analysis (linear regression adjusted for covariates) in order to compare baseline LCN2 in non-vascular pathology. In cohort 1, LCN2 concentrations in VaD (n = 27) were significantly different from AD (n = 47), SVDND (n = 20), and VCIND (n = 7) (p < 0.01, Tukey contrast for multiple comparisons of means) (Fig. 3a). In cohort 4, VaD (n = 10) displayed higher LCN2 concentrations than AD (n = 28), SVDND (n = 3), and VCIND (n = 8), although significant differences were only detected between AD and VaD groups (p < 0.05) (Supplementary Table 1, Fig. 3b). Additional association analyses of LCN2 and MMSE scores including all groups with cerebrovascular disease (SVDND, VCIND, and VaD) showed highly significant negative correlations in cohort 1 (cc = −0.55, p < 0.0001, Fig. 3c, Spearman correlation test) and cohort 4 (cc = −0.56, p = 0.008, Fig. 3d, Spearman correlation test). In AD groups, we observed no significant correlation (cohort 1: cc = 0.16, cohort 4: cc = −0.06, Supplementary Fig. 1a, Supplementary Fig. 1b).
We investigated the association of white matter changes and LCN2 in patients with cerebrovascular disease (SVDND, VCIND, and VaD) in cohorts 1 and 4 excluding patients with radiological evidence for cortical infarctions (Fig. 3e, f). LCN2 levels showed a significant positive correlation with the Age-Related White Matter Changes (ARWMC) scale in cohort 1 (cc = 0.33, p = 0.037, Spearman correlation test) and a non-significant positive correlation with the Fazekas scale in cohort 4 (cc = 0.38, p = 0.15, Spearman correlation test).
The CSF/serum albumin ratio (QAlb) is a well-known marker of blood-brain-barrier (BBB) function and was measured in the continuum of vascular pathology in cohort 1 and 4. It was significantly elevated in VaD compared to SVDND (p < 0.05 One-way ANOVA Bonferroni’s post hoc) (Supplementary Fig. 2a), presenting a strong positive correlation with LCN2 in SVDND and VaD and non-significant positive correlation in VCIND (Supplementary Fig. 2b).
Association with demographics and other biomarkers
LCN2 showed a weak non-significant positive correlation with age in all cohorts when all diagnostic groups were analysed (Supplementary Table 3). Yet, all group comparisons were adjusted for age and sex (Fig. 1, Fig. 2, Supplementary Fig. 2, Supplementary Table 2) to exclude that these factors behave as relevant confounders. Associations between LCN2 concentrations and demographic parameters as well as CSF total-Tau, p-Tau, and amyloid beta 42 were analysed in all VaD (n = 62) and AD (n = 117) cases. In VaD, LCN2 showed a significant positive correlation with total-Tau (cc = 0.34, p = 0.006, Spearman correlation test) but not with other studied parameters (Table 3). In contrast, no significance was observed in AD (cc = 0.05, p = 0.57, Spearman correlation test). Since LCN2 has been discussed as a blood-based marker for kidney injury, we retrospectively reviewed medical files of all AD and VBI cases to identify those with kidney injury and included this condition in the regression model as an additional covariate. It was not a significant variable in the model (p = 0.711 for the significance of the β estimate) and differences of LCN2 levels between groups were not affected. Therefore, we did not consider the presence of kidney injury to be a significant confounder.
LCN2 expression in the brain of AD and VaD
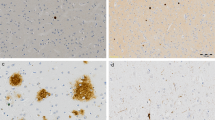
We included controls (n = 11, mean age ± SD, 56.3 ± 8.4), “pure” AD (co-morbidities were restricted to minimal small vessel disease, n = 10, mean age ± SD: 76.6 ± 6.0), and MID cases (n = 11, mean age ± SD: 72.5 ± 11.4) for neuropathological studies. In control brains, we detected LCN2 immunoreactivity in capillaries and resting microglia (Fig. 4a, upper- left). In AD cases (stages V and VI), LCN2 immunoreactivity was preserved in capillaries but increased in a subpopulation of astrocytes localized around β-amyloid plaques or layered in the cerebral cortex, and in reactive microglia (Fig. 4a, upper-right). Lesions at different evolution states were observed in all cases with MID. These lesions included rare acute infarcts in which acute necrosis was the predominant alteration, subacute infarcts with abundant macrophages and variable peripheral reactive astrocytes, and old, often cystic, infarcts with a necrotic center surrounded by a scar of astrocytes (chronic reactive astrocytosis). Massive LCN2 immunoreactivity was observed in subacute infarct areas (MID-SAI area) and in macrophages (Fig. 4a, bottom- left insert). Additional LCN2 immunoreactivity occurred in reactive astrocytes as seen in small numbers in the peripheral region of subacute infarcts (Fig. 4a, bottom left) and in the astrocytic scar area surrounding a cystic area in large infarcts or replacing deceased neurons in small infarcts (MID-AS area) (Fig. 4a bottom right). Total LNC2 staining was analysed by Kruskal-Wallis followed by Dunn's Multiple Comparison test showing an increased immunoreactivity in MID in comparison with AD (p < 0.05) and controls (p < 0.001, Fig. 4b).
a LCN2 immunohistochemistry in the cerebral cortex of control, Alzheimer’s disease (AD), and Multi-Infarct Chronic Encephalopathy (MID) cases, including subacute infarct areas (MID-SAI area) and astrocytic scar areas (MID-AS area). LCN2 staining is observed in intact blood vessels in controls, AD and MID cases (arrow-heads). Increased LCN2 expression is observed in reactive astrocytes in AD and MID (arrows) and in monocyte/macrophage cells of the MID-SAI area (empty arrow-heads). Paraffin sections counterstained with hematoxylin. Bar: 25 µm; insert-bar 50 µm. b Quantification of LCN2 positive cells area in cerebral cortex and striatum in µm2. Significant increase of LCN2 positive cells area in MID cases with respect to controls (***p < 0.001) and AD cases (*p < 0.05). Shown results are means (±SD) of controls (n = 11), AD (n = 10), and MID (n = 11) cases. In MID cases, area of subacute infraction (n = 6) and chronic infarcts (n = 11) were analysed. Results were analysed by Kruskal-Wallis followed by Dunn's Multiple Comparison test.
Double-labeling immunofluorescence to GFAP and LCN2 showed that about 58% of astrocytes in AD, 12% of astrocytes in the peripheral region of subacute infarcts, and 32% of astrocytes at the periphery of chronic infarcts expressed LCN2 (Fig. 5a, c, e). In contrast, double-labeling immunofluorescence disclosed that about 75% of Iba1-positive cells in subacute infarcts expressed LCN2. About the same percentage or even more LCN2-immunoreactive cells were macrophages (Fig. 5b, d, e).
a LCN2 and GFAP double-immunostaining in the cerebral cortex of control, AD, and MID cases. LCN2 co-localises with GFAP immunoreactive astrocytes (arrows) in AD, with surrounding plaque like structures, and in MID in astrocytic scar area (MID-AS). Abundant LCN2+GFAP- cells are observed in MID at the subacute infarction area (MID-SAI; arrow-heads), where there is little astrocyte presence. Bar = 50 µm. b LCN2 and Iba1 double-immunostaining in the cerebral cortex of control, AD, and MID cases. LCN2 co-localises with Iba1 immunoreactive microglia in AD and MID-AS (middle panels, arrows). In MID-SAI, predominant Iba1 positive staining was observed, displaying almost entire colocalisation with LCN2+ cells (bottom panel, arrows). LCN2+Iba1- cells are observed in MID-AS slices (arrow-heads). Bar = 50 µm. c, d Quantification of (c) LCN2 and GFAP, and (d) LCN2 and Iba1 double-immunostainings. Tables show single- and double-stained area (µm2), percentage of cells and Pearson’s colocalisation coefficient in Control, AD, and MID (total and divided in MID-SAI and MID-AS). e Graphic representation of double-stained area (left panel) and percentage of double-stained cells (right panel) in control, AD, MID-SAI, and MID-AS. Data are shown as mean ± SD of control (n = 4), AD (n = 4), and MID (n = 10). MID-AS areas were quantified in 10 cases and MID-SAI areas in 6 cases. Mean ± SD is included for each graph. Two-way ANOVA followed by Bonferroni’s post hoc test were used to analyse results *p < 0.05, **p < 0.01, ***p < 0.001.
IgG and Fibrinogen immunoreactivity was detected in the perivascular space in AD and VaD, but not in controls (Supplementary Fig. 3), suggesting BBB impairment in both diseases and the absence of differential contribution of BBB alteration to the CSF concentrations of LCN2 in AD and VaD.
Discussion
Imaging markers like N-acetyl aspartate (NAA) in proton magnetic resonance spectroscopy for Binswanger’s disease29 and candidate CSF markers like Qalb30,31, neurofilament light32, and matrix metalloproteinases33 for VaD have been suggested in recent years. These markers have shown uncertain or at best moderate diagnostic accuracy34,35.
The results from cohort 1 show that LCN2 levels might allow accurate differentiation between VaD and neurological controls as well as all other investigated causes of dementia. The broad spectrum of different dementia types analysed underlines the potential clinical value of the marker. However, cases numbers were rather low. Therefore, we investigated two independent cohorts (cohort 2 and 3) and were able to validate our findings through multi-national European cooperation. The results represent a very robust proof of concept.
Clinical and MRI data from cohort 1 and 4 suggest that higher LCN2 levels are associated with the degree of cognitive impairment in VBI, but not in AD. Regarding the different types of VaD, LCN2 is possibly lower in patients with PSD, but low case numbers precluded validating this observation statistically. Only in cohort 1, the correlation of high LCN2 levels and WMH load showed (borderline) significance. All this may indicate that LCN2 is closely associated with clinical disease stage in VaD rather than with common imaging markers. Further investigations and deeper analyses of MRI markers (atrophy, infarct volume, WMH volume, etc.) will need to explore these assumptions and the presence of VaD patients with unaltered or only slightly elevated CSF LCN2 levels in all cohorts. Here, we focused on the association between LCN2 levels and clinical diagnosis.
A previous study showed elevation of plasma LCN2 in patients with amnestic MCI but, in line with our results, not in demented patients with AD23. Inflammatory processes in early AD stages were suggested as a possible cause but concomitant vascular pathology was not excluded and diagnostic accuracy was not evaluated. Another study reported an elevation of CSF LCN2 and a positive correlation with neurofilament light in patients with progressive multiple sclerosis, but the increase was moderate and the authors stated that LCN2 was likely not a suitable biomarker for the clinical diagnosis of this condition24.
Considering this, LCN2 may be a valuable biomarker for VaD in the context of differential diagnosis. Such a marker has long been pursued17,18,34. Besides new imaging markers that display functional consequences of vascular lesions29,36, fluid markers like LCN2 are candidates to contribute to a biomarker-based definition of VaD as has recently been proposed for AD7.
LCN2 is synthesized and secreted as an inducible factor from activated microglia, reactive astrocytes, neurons, and endothelial cells in response to inflammatory, infectious, and injurious insults, in all of which it plays varied functions37. Several lines of evidence implicate LCN2 in the progression of cerebral infarcts38.
Because the pathophysiology of LCN2 in VaD has not yet been resolved, caution is necessary when disclosing the molecular causes of LCN2 alterations in biological fluids. In cohort 1 and cohort 4, Qalb was strongly associated with LCN2 levels. Since disturbance of the BBB is a core feature of the pathophysiology of VaD39, elevated CSF LCN2 might be related to impaired BBB function like it has been assumed to be the case for Qalb. However, LCN2 seems to differentiate VaD from other forms of dementia better than what has been shown for Qalb30,31,34. Furthermore, LCN2 also displays significant positive correlation with CSF total-Tau in VaD but not in AD (Table 3), which might indicate an association with the extent of neuronal damage.
Neuropathological examination of control brains indicates that LCN2 is expressed in capillaries and microglia, but rarely in astrocytes. In AD brains, LCN2 is expressed in reactive microglia and reactive astrocytes. In MID brains, LCN2 is expressed in macrophages located in subacute infarcts, in reactive astrocytes at the periphery of subacute infarcts, and in chronic infarcts. Previous studies associated LCN2 with inflammation and cellular damage in cerebral inflammatory diseases such as multiple sclerosis and neuropsychiatric lupus24,40,41. LCN2 is involved in the inflammatory activation of astrocytes42. Hypoxia induces astrocyte-derived LCN2 in ischemic stroke43 while astrocyte-derived LCN2 mediates hippocampal damage and cognitive deficits in experimental models of VaD28.
It is tempting to speculate that high LCN2 levels in the brain may be expected in conditions with robust astrocytic gliosis such as CJD. However, CSF LCN2 in CJD was not significantly altered in multi-comparative analyses. Therefore, other scenarios may be considered. VaD may present with a particular profile of reactive astrocytes. Microglia and macrophages may be additional sources of LCN2, and areas of subacute infarcts in MID are particularly rich in macrophages. Unfortunately, comprehensive studies combining clinical data, high sensitivity MRI, and CSF LCN2 levels in the progression of ischemic infarcts are not available. In addition to alterations of the BBB, cerebrovascular diseases, and particularly MID in the present context, show disruption of the interface between blood vessels and Virchow-Robin spaces as well as between Virchow-Robin spaces and brain tissue, which adjoin to the subventricular space and the CSF. This represents another considerable mechanism regarding abnormal protein extravasation of secreted brain proteins to the CSF.
Our study concerns an important area of research with major socioeconomic relevance. The participating centres are specialized institutions capable of defining patient groups with great expertise in applying clinical and para-clinical assessments. The multi-centre setting also allowed for demonstration of the reproducibility of our findings.
The absence of a reliable gold standard for the clinical diagnosis of VaD is a limitation of our study. The reference standard2 and its key markers probably lack high accuracy3,11,12. We assume that the inclusion of independent cohorts with independent diagnostic evaluations is an appropriate way to regard this problem and to validate the clinical reliability of the results. The frequent occurrence of concomitant pathologies (VaD and AD) is a problem that we tried to address by differentiating (cohort 1) or excluding (cohorts 2, 3, 4) patients with clinical evidence of possible MD. A biomarker for “pure” VaD may also be useful in the differential diagnosis of MD when used in combination with AD-related biomarkers. Since these biomarkers were part of the reference standard for the diagnosis of AD, we could investigate this further. In general, clinical evaluations of biomarkers of VaD have to be interpreted with caution.
The retrospective study design and low case numbers in each cohort have several limitations. We could not investigate relations between kidney disease (for which LCN2 is considered to be a promising biomarker), peripheral LCN2, and CSF LCN2 further. Thus, we cannot be sure that elevated CSF LCN2 is brain-derived and we cannot state a specific causal relationship between LCN2 and VBI.
No sample-size calculation was performed. Different forms of VaD8,44 were stratified but not statistically analysed. Advanced neuropathological differentiation of cases was not available for the clinical cohorts and it cannot be ruled out that other subtypes of VaD (e.g. multi-infarct encephalopathy vs. sole diffuse alteration of the white matter) may account for differences of CSF LCN2 levels within the VaD groups. However, this did not result in low overall diagnostic accuracy. We performed basic correlation analyses of LCN2 and MRI markers as well as neuropathological investigation of certain VaD subtypes, but detailed data will have to be acquired through prospective studies to validate the findings.
Due to the case-control design, the cohorts may be biased. Especially in cohort 1, many samples were collected in the framework of differential diagnosis of atypical or rapidly progressive dementia, which may explain higher LCN2 levels compared to the other cohorts. Non-identic population characteristics and pre-analytical conditions in the study centres cannot be ruled out. By contrast, the design and the heterogeneity of cohorts is also a strength of the study. We could show that different LCN2 levels in AD and VaD were consistent even though diagnostic procedures and case selection were not performed in the same centre.
The presented clinical data indicate that CSF LCN2 is a marker for dementia associated with VBI and that it has the potential to become a complementary tool in the differential diagnosis of VaD and AD. Since this is the first clinical evaluation of LCN2 in this context, further investigation is needed before it can be included in the clinical work-up. Our findings set the hallmark for further neuropathological and prospective clinical investigations.
Methods
Study design and study centres
CSF analyses had been planned in Göttingen before 472 samples were collected from four European centres in the framework of a retrospective case-control study design. The case selection was based on the availability of samples from patients with the target conditions (VaD and important differential diagnoses) and complete information for clinical criteria application (including basic CSF analyses and neuroimaging). Patients with central nervous system inflammation and neoplasia (possible causes for white matter pathology) as well as intermediate diagnostic categories (e.g. possible AD, CJD, DLB etc.) were excluded. CSF LCN2 analyses (index test) were performed in Göttingen blind to diagnoses in 2018.
Cohort 1 was recruited at the Clinical Dementia Center and the National Reference Center for Creutzfeldt-Jakob disease surveillance at the University Medical Center of Göttingen (Germany) and included ND, VaD, AD and other neurodegenerative dementias as well as patients with VCIND and SVDND. Cohort 2 (ND, AD, and VaD) was recruited at the Dementia Clinic, Neurology Department of Coimbra-University Hospital (Portugal). Cohort 3 (ND, AD, and VaD) was recruited at the Geriatric Clinic, Linköping University Hospital, Linköping (Sweden). Cohort 4 (AD, VaD, VCIND, and SVDND) was recruited at the Center of Cognitive Neurology and Inserm, Lariboisière Hospital, University Paris Diderot (France). Case numbers are shown in Table 1.
Diagnostic criteria
The clinical classification of patients and assessment of imaging data was performed in their respective study centres by specialized neurologists and neuroradiologists. Probable AD was diagnosed according to the National Institute on Aging—Alzheimer's Association workgroups (NIA-AA) criteria45 (cohort 1,2 and 4) and National Institute of Neurological and Communicative Disorders and Stroke and the AD and Related Disorders Association (NINCDS-ADRDA) criteria46 (cohort 3). Patients with coexisting pathology (AD plus VBI) were excluded from the AD group as much as possible. Only patients without significant vascular brain lesions (on MRI, rated by the local neuroradiologists) and no specific clinical signs for cerebrovascular disease (e.g. strokes, stroke-like episodes, stepwise worsening of symptoms, etc.) were included.
VaD diagnosis in all four centres was based on clinical and radiological criteria described by the National Institute of Neurological and Communicative Disorders and Stroke and the AD and Related Disorders Association (NINDS-AIREN2). VaD diagnosis also included a complete clinical work up showing no evidence for other than vascular pathology of the brain.
ND patients were diagnosed according to acknowledged standard neurologic clinical and para-clinical findings based on the International Classification of Diseases (ICD) 10 definitions. On MRI or CT, these patients did not show any cerebrovascular lesions other than normal age-related WMH (as rated by neuroradiologists during the diagnostic process). Neuroimaging and lumbar puncture were performed for various reasons (e.g. headaches, affective disorders etc.). ND patients did not show any clinical signs of cognitive impairment but a detailed neuropsychological test battery had not been performed in many of them.
Data from patients with MD, LBD, FTD, and CJD was only available in cohort 1. The MD group included patients according to clinical International Working Group (IWG-2) criteria47 and also patients with VaD according to NINDS-AIREN criteria plus at least one AD-typical CSF biomarker (elevated phosphorylated-tau or low amyloid beta 1-42/1-40 ratio). The LBD group included dementia with Lewy bodies48 and Parkinson’s disease dementia49. The FTD group included only behavioral variant FTD (bvFTD) and was diagnosed according to the International Behavioral Variant FTD Criteria Consortium for bvFTD50. Patients with Sporadic Creutzfeldt-Jakob disease (CJD) were classified as probable or definite according to WHO criteria51.
Classification of cerebrovascular disease
All clinical cohorts included different forms of VaD while for morphological studies and immunohistochemistry, only brains with chronic multi-infarct encephalopathy were available.
We analysed clinical and imaging data from all VaD patients to assign disease type (PSD, SID, and MID) according to the Vascular Impairment of Cognition Classification Consensus Study8. SID includes cases with severe damage of the cerebral white matter often accompanied by status cribosus and lacunar infarcts, MID is characterized by multiple infarcts involving the cerebral cortex but also other parts of the brain, PSD refers to cases of dementia resulting from a single stroke.
Cohort 1 and cohort 4 also inlcuded patients with SVDND and VCIND. Patients with VCIND showed cognitive impairment but no significant impairment of activities of daily living. Neuropsychological assessment (Cambridge Cognitive Examination battery in cohort 1 and Consortium to Establish a Registry for AD battery in cohort 4) did not reveal reduced total or subscale scores in patients with SVDND (>−1.5 SD, matched for sex, age, and education). The diagnosis of subcortical small vessel disease was based on MRI (FLAIR or T2 weighted images) showing ≥4 points on the ARWMC52 scale in cohort 1 and either rank 3 on the Fazekas scale or multiple lacunae in cohort 4. Patients received MRI for various reasons (e.g. headaches, affective disorders etc.).
CSF analyses
CSF LCN2 was quantified using the human LCN2/NGAL (Neutrophil Gelatinase-Associated Lipocalin) Quantikine Enzyme-linked Immunosorbent Assay (ELISA) Kit from R&D according to the manufacturer’s instructions (R&D Systems, Inc. Minneapolis, MN). CSF samples were diluted 1:2. Inter- and intra-assay coefficients of variation were below 12 and 10%, respectively. The limit of quantification was 0.052 (52 pg/ml) and the limit of detection was 0.023 (23 pg/ml). Total-Tau, phospho-Tau (p-Tau), and amyloid beta 42 were quantitatively measured using ELISA kits from Fujirebio (Fujirebio, Ghent, Belgium). QAlb (CSF albumin/serum albumin*103) was measured by local neurochemistry laboratories (cohort 1 and cohort 4) according to standard methodology. Test performers were blind to clinical information and clinical investigators vice versa.
Morphological studies and immunohistochemistry
Morphological studies were carried out in post mortem human brains obtained from the Institute of Neuropathology brain bank, HUB-ICO-IDIBELL Biobank. One hemisphere was immediately cut in coronal sections, 1 cm thick. Selected areas of the encephalon were rapidly dissected, frozen on metal plates over dry ice, placed in individual air-tight plastic bags, and stored at −80 °C until use for biochemical studies. The other hemisphere was fixed by immersion in 4% buffered formalin for morphological studies; sections from representative regions were stained with hematoxylin/eosin, periodic acid-Schiff and Klüver-Barrera, or processed for immunohistochemistry analysis. Cases were categorized as controls (cerebral cortex frontal area 8 and striatum, n = 11), AD (cerebral cortex frontal area 8, AD stage V-VI/C, n = 10), and MID (cerebral cortex, temporal cortex, striatum, including areas with acute, subacute, and chronic infarcts, n = 11). Control cases had not suffered from neurologic or psychiatric diseases, infections of the nervous system, brain neoplasms, or systemic and central immune diseases, and did not have abnormalities in the neuropathological examination. Mixed pathologies were excluded and cases with circulatory/vascular-linked diffuse white matter encephalopathy and cases with PSD were not included in the study. CSF was not available in any of the studied post-mortem brain series.
De-waxed sections, 4-μm thick, were processed for immunohistochemistry. The sections were boiled in citrate buffer (20 min) to retrieve antigenicity. Endogenous peroxidases were blocked by incubation in Dako Real Peroxidase blocking solution (Dako, S2023). Then, sections were incubated at 4 °C overnight with one of the primary antibodies properly diluted in Dako Real Antibody Diluent (Dako, S2022) and afterwards incubated with MultiLink biotinylated antibody followed by HRP-conjugated streptavidin (BioGenex QP9009L-E). The peroxidase reaction was visualized with diaminobenzidine (DAB; Sigma-Aldrich, D5637) and H2O2. Control of the immunostaining included omission of the primary antibody. Nucleus labeling was obtained by Hematoxylin staining. For immunofluorescence, 4-μm-thick de-waxed sections were used. After antigen retrieval, slices were incubated in Sudan Black for 15 min to reduce lipofuscin autofluorescence. Unspecific bonding was blocked by Fetal Bovine Serum (FBS) 10% 1h; afterwards sections were incubated at 4 °C overnight with one of the primary antibodies properly diluted in FBS 10%. Appropriate Alexa-fluor 488 and 555 were used as secondary antibodies. Nucleus labeling was performed using DAPI. LCN2 antibody (RD Systems MAB1757) was used at 1:50 dilution, GFAP antibody (Dako Z0334) was used at 1:400 dilution, Iba1 antibody (Wako 019-19741) was used at 1:1000 dilution, IgG heavy chain antibody (Proteintech 16402-1-AP) was used at 1:200 dilution, and Fibrinogen FGL2 antibody (Proteintech 11827-1-AP) was used at 1:100 dilution.
LCN2 quantification in brain tissue
Immunohistochemistry and immunofluorescence pictures were taken with a Nikon Eclipse E-800 microscope and ProgRes Capture Pro 2.7.7 software. LCN2 immunostaining was quantified in 11 controls, 10 AD (stage V-VI/C), and 11 MID cases. Ten images of each slice were taken throughout the tissue, including areas of subacute and chronic infarcts in MID slices. Pictures were analysed using Fiji ImageJ software; LCN2 positive area was measured in µm2 only in particles >25 µm2 by stablishing a fixed positive staining threshold.
For immunofluorescence, 4 controls, 4 AD, and 10 MID cases were tested. From MID, six slices were quantified in subacute infarctions (MID-SAI) and 10 in the astrocytic scar in chronic infarcts (MID-AS). Six pictures of each slice were analysed using Fiji ImageJ software. Three different methods were applied. The first one analysed number of green, red and yellow pixel areas using colocalization thresholded plugin. The second one used Coloc2 plugin to obtain Pearson’s colocalization coefficients of all pictures. In addition, two independent researchers counted the number of green, red and yellow cells manually in a double blind study.
Statistical analysis
Group differences of LCN2 levels were assessed through linear regression analyses. We log-transformed LCN2 concentration and built models adjusting for age and sex as potential confounders. Multiple comparisons among diagnostic groups were made with multcomp package in R53. We performed additional comparisons of LCN2 levels between AD and VaD in the four cohorts, controlling for dementia stage through models that include age, sex, and MMSE score as covariates. Models including kidney disease in the group of covariates were built in order to analyse the potential role of kidney injury as a confounder of LCN2 levels.
Spearman rank correlation coefficients were used to assess associations between continuous biomarker levels, ARWMC, and MMSE scores. LCN2 level association with demographic data was investigated using Pearson correlation (age) or Mann–Whitney test (sex).
To determine biomarker diagnostic accuracy, ROC curve analyses were carried out and AUC with 95% CI were calculated. Values of sensitivity and specificity were derived from best cut-off points in cohort 1 according to Youden’s index. Comparisons between AUC values were performed with DeLong’s test available in pROC R package54.
For group comparisons of immunohistochemistry and immunofluorescence results, Kruskal-Wallis followed by Dunn’s Multiple Comparison test and two way-ANOVA followed by Bonferroni’s post hoc test were applied respectively, using GraphPad Prismv5 software. In all analyses, statistical significance was considered at p < 0.05.
Ethics
The study was conducted according to the revised Declaration of Helsinki and Good Clinical Practice guidelines. Informed written consent was obtained from participants and/or their relatives. The study of CSF samples and case data was approved by the ethics committees of the University Medical Center Göttingen (Germany), Linköpping (Sweden), Paris University Hospitals (France), and the Ethics Board of Coimbra University Hospital (Portugal).
Post mortem brain tissue was obtained from the Institute of Neuropathology brain bank, HUB-ICO-IDIBELL biobank following the guidelines of Spanish legislation (Real Decreto de Biobancos 1716/2011).
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
The datasets generated during and/or analysed during the current study contain patient-related clinical information and are not publicly available. However, anonymised raw data to generate figures and tables are available from the corresponding authors on reasonable request.
References
Ferrer, I. Cognitive impairment of vascular origin: Neuropathology of cognitive impairment of vascular origin. J. Neurol. Sci. 299, 139–149 (2010).
Román, G. C. et al. Vascular dementia: diagnostic criteria for research studies. Report of the NINDS-AIREN International Workshop. Neurology 43, 250–260 (1993).
Pohjasvaara, T., Mantyla, R., Ylikoski, R., Kaste, M. & Erkinjuntti, T. Comparison of Different Clinical Criteria (DSM-III, ADDTC, ICD-10, NINDS-AIREN, DSM-IV) for the Diagnosis of Vascular Dementia. Stroke 31, 2952–2957 (2000).
Perneczky, R. et al. Is the time ripe for new diagnostic criteria of cognitive impairment due to cerebrovascular disease? Consensus report of the International Congress on Vascular Dementia working group. BMC Med. 14, 162 (2016).
Jellinger, K. A. Pathology and pathogenesis of vascular cognitive impairment-a critical update. Front. Aging Neurosci. 5, 17 (2013).
Skrobot, O. A. et al. Vascular cognitive impairment neuropathology guidelines (VCING): the contribution of cerebrovascular pathology to cognitive impairment. Brain 139, 2957–2969 (2016).
Jack, C. R. et al. NIA-AA research framework: toward a biological definition of Alzheimer’s disease. Alzheimer’s Dement. 14, 535–562 (2018).
Skrobot, O. A. et al. Progress toward standardized diagnosis of vascular cognitive impairment: Guidelines from the Vascular Impairment of Cognition Classification Consensus Study. Alzheimer’s Dement 14, 280–292 (2018).
Gorelick, P. et al. Vascular Contributions to Cognitive Impairment and Dementia: A Statement for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke 42, 2672–2713 (2011).
Sachdev, P. et al. Diagnostic criteria for vascular cognitive disorders: a VASCOG statement. Alzheimer Dis. Assoc. Disord. 28, 206–218 (2014).
Prins, N. D. et al. Cerebral small-vessel disease and decline in information processing speed, executive function and memory. Brain 128, 2034–2041 (2005).
Kantarci, K. et al. Risk of dementia in MCI: Combined effect of cerebrovascular disease, volumetric MRI, and 1H MRS. Neurology 72, 1519–1525 (2009).
Alber, J. et al. White matter hyperintensities in vascular contributions to cognitive impairment and dementia (VCID): knowledge gaps and opportunities. Alzheimer’s Dement. Transl. Res. Clin. Inter. 5, 107–117 (2019).
Cavalieri, M. et al. Vascular dementia and Alzheimer’s disease—Are we in a dead-end road?. Neurodegener. Dis. 7, 122–126 (2010).
Lee, S. et al. White matter hyperintensities are a core feature of Alzheimer’s disease: evidence from the dominantly inherited Alzheimer network. Ann. Neurol. 79, 929–939 (2016).
McAleese, K. E. et al. Parietal white matter lesions in Alzheimer’s disease are associated with cortical neurodegenerative pathology, but not with small vessel disease. Acta Neuropathol. 134, 459–473 (2017).
Llorens, F. et al. Cerebrospinal fluid biomarkers of Alzheimer’s disease show different but partially overlapping profile compared to vascular dementia. Front. Aging Neurosci. 9, 1–9 (2017).
Roseborough, A., Ramirez, J., Black, S. E. & Edwards, J. D. Associations between amyloid β and white matter hyperintensities: a systematic review. Alzheimer’s Dement. 13, 1154–1167 (2017).
Wallin, A. et al. Biochemical markers in vascular cognitive impairment associated with subcortical small vessel disease—A consensus report. BMC Neurol. 17, 102 (2017).
Skrobot, O. A. et al. The vascular impairment of cognition classification consensus study. Alzheimer’s Dement 13, 624–633 (2017).
Xiao, X., Yeoh, B. S. & Vijay-Kumar, M. Lipocalin 2: an emerging player in iron homeostasis and inflammation. Annu. Rev. Nutr. 37, 103–130 (2017).
Song, J. & Kim, O. Y. Perspectives in Lipocalin-2: emerging biomarker for medical diagnosis and prognosis for Alzheimer’s disease. Clin. Nutr. Res. 7, 1 (2018).
Choi, J., Lee, H. W. & Suk, K. Increased plasma levels of lipocalin 2 in mild cognitive impairment. J. Neurol. Sci. 305, 28–33 (2011).
Al Nimer, F. et al. Lipocalin-2 is increased in progressive multiple sclerosis and inhibits remyelination. Neurol. —Neuroimmunol. Neuroinflammation 3, e191 (2016).
Moschen, A. R., Adolph, T. E., Gerner, R. R., Wieser, V. & Tilg, H. Lipocalin-2: a master mediator of intestinal and metabolic inflammation. Trends Endocrinol. Metab. 28, 388–397 (2017).
Gan, J. & Zhou, X. Comparison of urine neutrophil gelatinase-associated lipocalin and interleukin-18 in prediction of acute kidney injury in adults. Medicine (United States) 97 (2018).
Krstic, D. et al. Biochemical markers of renal function. Curr. Med. Chem. 23, 2018–2040 (2016).
Kim, J. H. et al. Astrocyte-derived lipocalin-2 mediates hippocampal damage and cognitive deficits in experimental models of vascular dementia. Glia 65, 1471–1490 (2017).
Barry Erhardt, E. et al. Biomarkers identify the Binswanger type of vascular cognitive impairment. J. Cereb. Blood Flow. Metab. https://doi.org/10.1177/0271678X18762655 (2018).
Hermann, P., Romero, C., Schmidt, C., Reis, C. & Zerr, I. CSF biomarkers and neuropsychological profiles in patients with cerebral small-vessel disease. PLoS ONE 9, e105000 (2014).
Skillbäck, T. et al. CSF/serum albumin ratio in dementias: a cross-sectional study on 1861 patients. Neurobiol. Aging 59, 1–9 (2017).
Zerr, I. et al. Cerebrospinal fluid neurofilament light levels in neurodegenerative dementia: Evaluation of diagnostic accuracy in the differential diagnosis of prion diseases. Alzheimer’s Dement. 9, 751–763 (2018).
Bjerke, M. et al. Cerebrospinal fluid matrix metalloproteinases and tissue inhibitor of metalloproteinases in combination with subcortical and cortical biomarkers in vascular dementia and Alzheimer’s disease. J. Alzheimer’s Dis. 27, 665–676 (2011).
Skrobot, O. A. et al. The Vascular Impairment of Cognition Classification Consensus Study. Alzheimer’s Dement. 1–10, https://doi.org/10.1016/j.jalz.2016.10.007 (2016).
Rosenberg, G. A. et al. Validation of biomarkers in subcortical ischaemic vascular disease of the Binswanger type: approach to targeted treatment trials. J. Neurol. Neurosurg. Psychiatry 86, 1324–1330 (2015).
Rosenberg, G. A. Binswanger’s disease: biomarkers in the inflammatory form of vascular cognitive impairment and dementia. J. Neurochemistry 144, 634–643 (2018).
Jha, M. K. et al. Diverse functional roles of lipocalin-2 in the central nervous system. Neurosci. Biobehav. Rev. 49, 135–156 (2015).
Wang, G. et al. Lipocalin-2 released in response to cerebral ischaemia mediates reperfusion injury in mice. J. Cell. Mol. Med. 19, 1637–1645 (2015).
Farrall, A. J. & Wardlaw, J. M. Blood-brain barrier: ageing and microvascular disease—systematic review and meta-analysis. Neurobiol. Aging 30, 337–352 (2009).
Berard, J. L. et al. Lipocalin 2 is a novel immune mediator of experimental autoimmune encephalomyelitis pathogenesis and is modulated in multiple sclerosis. Glia 60, 1145–1159 (2012).
Mike, E. V. et al. Lipocalin-2 is a pathogenic determinant and biomarker of neuropsychiatric lupus. J. Autoimmun. 96, 59–73 (2019).
Lee, S., Jha, M. K. & Suk, K. Lipocalin-2 in the inflammatory activation of brain astrocytes. Crit. Rev. Immunol. 35, 77–84 (2015).
Taklimie, F. R. et al. Hypoxia induces astrocyte-derived lipocalin-2 in ischemic stroke. Int. J. Mol. Sci. 20, 1271 (2019).
Jellinger, Ka The pathology of ‘vascular dementia’: a critical update. J. Alzheimers Dis. 14, 107–123 (2008).
McKhann, G. M. et al. The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 7, 263–269 (2011).
Dubois, B. et al. Research criteria for the diagnosis of Alzheimer’s disease: revising the NINCDS-ADRDA criteria. Lancet Neurol. 6, 734–746 (2007).
Dubois, B. et al. Advancing research diagnostic criteria for Alzheimer’s disease: The IWG-2 criteria. Lancet Neurol. 13, 614–629 (2014).
McKeith, I. G. et al. Diagnosis and management of dementia with Lewy bodies: fourth consensus report of the DLB Consortium. Neurology 89, 88–100 (2017).
Emre, M. et al. Clinical diagnostic criteria for dementia associated with Parkinson’s disease. Mov. Disord. 22, 1689–1707 (2007).
Rascovsky, K. et al. Sensitivity of revised diagnostic criteria for the behavioural variant of frontotemporal dementia. Brain 134, 2456–2477 (2011).
Zerr, I. et al. Updated clinical diagnostic criteria for sporadic Creutzfeldt-Jakob disease. Brain 132, 2659–2668 (2009).
Wahlund, L. O. et al. A new rating scale for age-related white matter changes. Stroke 32, 1318–1322 (2001).
Hothorn, T., Bretz, F. & Westfall, P. Simultaneous inference in general parametric models. Biometrical J. 50, 346–363 (2008).
Robin, X. et al. pROC: an open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinform. 12, 77 (2011).
Acknowledgements
The authors wish to acknowledge Prof. Jan Marcusson from the Linköping University (Sweden) for his assistance with sample collection and Silja Köchy from the Universitätsmedizin Göttingen (Germany) for technical assistance. We would like to acknowledge the patients and the Biobank HUB-ICO-IDIBELL (PT17/0015/0024) integrated in the Spanish Biobank Network for their collaboration. This study was funded by the ADDF (Alzheimer’s Drug Discovery Foundation—Grant 201810-2017419) to F.L. and I.Z., the Instituto Carlos III (grants CP16/00041 and PI19/00144) to F.L., the Robert Koch Institute through funds from the German Federal Ministry of Health (grant no. 1369–341) to I.Z., and the Spanish Ministry of Health, Instituto Carlos III (Fondo de Investigación Sanitaria—FIS PI14/00757) to I.F. H.Z. is a Wallenberg Academy Fellow supported by grants from the Swedish Research Council, the European Research Council, Swedish State Support for Clinical Research (ALFGBG), and the UK Dementia Research Institute at UCL. K.B. holds the Torsten Söderberg Professorship of Medicine and is supported by grants from the Swedish Research Council, the Swedish Brain Foundation, the Swedish Alzheimer Foundation, and Swedish State Support for Clinical Research (ALFGBG). The funders of the study had no role in the design or conduct of the study, the collection, management, analysis, and interpretation of the data, the preparation, review, or approval of the manuscript, or and decision to submit the manuscript for publication. We wish to thank T. Yohannan for editorial assistance.
Author information
Authors and Affiliations
Contributions
F.L., P.H. and I.Z. designed the study. F.L., P.H., A.V.-P., D.D.-L., and I.F. performed experiments and collected, analysed, and interpreted data. K.N., O.H., I.S., M.S., P.L., D.V., S.G., J.D., H.Z., K.B., C.P., and I.B. collected data and contributed to data interpretation. F.L., P.H., and I.F. wrote the manuscript. F.L., P.H. and A.V.-P. contributed equally to the work. All authors critically revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
K.B. has served as a consultant or on the advisory boards for Alzheon, BioArctic, Biogen, Eli Lilly, Fujirebio Europe, IBL International, Merck, Novartis, Pfizer, and Roche Diagnostics, all un-related to the data presented in the present paper. O.H. has acquired research support (for the institution) from Roche, GE Healthcare, Biogen, AVID Radiopharmaceuticals, Fujirebio, and Euroimmun. In the past 2 years, he has received consultancy/speaker fees (paid to the institution) from Biogen, Roche, and Fujirebio. H.Z. has served on scientific advisory boards for Eli Lilly, Roche Diagnostics, CogRx, Samumed, and Wave, has received travel support from Teva and is a co-founder of Brain Biomarker Solutions in Gothenburg AB, a GU Ventures-based platform company at the University of Gothenburg. C.P. is member of the International Advisory Boards of Lilly, is consultant of Fujiribio, ALZOHIS, NEUROIMMUNE, and GILEAD and is involved as investigator in several clinical trials for Roche, Esai, Lilly, Biogen, Astra-Zeneca, Lundbeck, and Neuroimmune. J.D. is an investigator in several passive anti-amyloid immunotherapies and other clinical trials for Roche, Eisai, Lilly, Biogen, Astra-Zeneca, Lundbeck. The remaining authors report no biomedical financial interests or potential conflicts of interest.
Additional information
Peer review information Nature Communications thanks Terry Quinn and other, anonymous, reviewers for their contributions to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Llorens, F., Hermann, P., Villar-Piqué, A. et al. Cerebrospinal fluid lipocalin 2 as a novel biomarker for the differential diagnosis of vascular dementia. Nat Commun 11, 619 (2020). https://doi.org/10.1038/s41467-020-14373-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-020-14373-2
This article is cited by
-
Comparative evaluation of clinical and cerebrospinal fluid biomarker characteristics in rapidly and non-rapidly progressive Alzheimer’s disease
Alzheimer's Research & Therapy (2023)
-
Reciprocal interactions between innate immune cells and astrocytes facilitate neuroinflammation and brain metastasis via lipocalin-2
Nature Cancer (2023)
-
Molecular biomarkers for vascular cognitive impairment and dementia
Nature Reviews Neurology (2023)
-
Nerve growth factor receptor (Ngfr) induces neurogenic plasticity by suppressing reactive astroglial Lcn2/Slc22a17 signaling in Alzheimer’s disease
npj Regenerative Medicine (2023)
-
Plasma Lipocalin 2 in Alzheimer’s disease: potential utility in the differential diagnosis and relationship with other biomarkers
Alzheimer's Research & Therapy (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.