Abstract
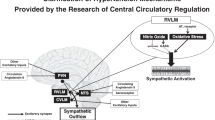
Paroxysmal sympathetic hyperactivity (PSH) is a common clinical feature secondary to ischemic stroke (IS), but its mechanism is poorly understood. We aimed to investigate the role of H2S in the pathogenesis of PSH. IS patients were divided into malignant (MCI) and non-malignant cerebral infarction (NMCI) group. IS in rats was induced by the right middle cerebral artery occlusion (MCAO). H2S donor (NaHS) or inhibitor (aminooxy-acetic acid, AOAA) were microinjected into the hypothalamic paraventricular nucleus (PVN). Compared with the NMCI group, patients in the MCI group showed PSH, including tachycardia, hypertension, and more plasma norepinephrine (NE) that was positively correlated with levels of creatine kinase, glutamate transaminase, and creatinine respectively. The 1-year survival rate of patients with high plasma NE levels was lower. The hypothalamus of rats with MCAO showed increased activity, especially in the PVN region. The levels of H2S in PVN of the rats with MCAO were reduced, while the blood pressure and renal sympathetic discharge were increased, which could be ameliorated by NaHS and exacerbated by AOAA. NaHS completely reduced the disulfide bond of NMDAR1 in PC12 cells. The inhibition of NMDAR by MK-801 microinjected in PVN of rats with MCAO also could lower blood pressure and renal sympathetic discharge. In conclusion, PSH may be associated with disease progression and survival in patients with IS. Decreased levels of H2S in PVN were involved in regulating sympathetic efferent activity after cerebral infarction. Our results might provide a new strategy and target for the prevention and treatment of PSH.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Perkes IE, Menon DK, Nott MT, Baguley IJ. Paroxysmal sympathetic hyperactivity after acquired brain injury: a review of diagnostic criteria. Brain Inj. 2011;25:925–32.
Baguley IJ, Perkes IE, Fernandez-Ortega JF, Rabinstein AA, Dolce G, Hendricks HT, et al. Paroxysmal sympathetic hyperactivity after acquired brain injury: consensus on conceptual definition, nomenclature, and diagnostic criteria. J Neurotrauma. 2014;31:1515–20.
Fernandez-Ortega JF, Baguley IJ, Gates TA, Garcia-Caballero M, Quesada-Garcia JG, Prieto-Palomino MA. Catecholamines and Paroxysmal Sympathetic Hyperactivity after Traumatic Brain Injury. J Neurotrauma. 2017;34:109–14.
Hinson HE, Puybasset L, Weiss N, Perlbarg V, Benali H, Galanaud D, et al. Neuroanatomical basis of paroxysmal sympathetic hyperactivity: a diffusion tensor imaging analysis. Brain Inj. 2015;29:455–61.
Meyfroidt G, Baguley IJ, Menon DK. Paroxysmal sympathetic hyperactivity: the storm after acute brain injury. Lancet Neurol. 2017;16:721–9.
Jafari AA, Shah M, Mirmoeeni S, Hassani MS, Nazari S, Fielder T, et al. Paroxysmal sympathetic hyperactivity during traumatic brain injury. Clin Neurol Neurosurg. 2022;212:107081.
Louraoui SM, Fliyou F, Aasfara J, El Azhari A. Paroxysmal Sympathetic Hyperactivity After Traumatic Brain Injury: What Is Important to Know? Cureus. 2022;14:e24693.
Khalid F, Yang GL, McGuire JL, Robson MJ, Foreman B, Ngwenya LB, et al. Autonomic dysfunction following traumatic brain injury: translational insights. Neurosurg Focus. 2019;47:E8.
Dias C, Gaio AR, Monteiro E, Barbosa S, Cerejo A, Donnelly J, et al. Kidney-brain link in traumatic brain injury patients? A preliminary report. Neurocrit Care. 2015;22:192–201.
Fernandez-Ortega JF, Prieto-Palomino MA, Munoz-Lopez A, Lebron-Gallardo M, Cabrera-Ortiz H, Quesada-Garcia G. Prognostic influence and computed tomography findings in dysautonomic crises after traumatic brain injury. J Trauma. 2006;61:1129–33.
Lv LQ, Hou LJ, Yu MK, Qi XQ, Chen HR, Chen JX, et al. Prognostic influence and magnetic resonance imaging findings in paroxysmal sympathetic hyperactivity after severe traumatic brain injury. J Neurotrauma. 2010;27:1945–50.
Nott MT, Chapparo C, Baguley IJ. Agitation following traumatic brain injury: an Australian sample. Brain Inj. 2006;20:1175–82.
Baguley IJ, Nott MT, Slewa-Younan S, Heriseanu RE, Perkes IE. Diagnosing dysautonomia after acute traumatic brain injury: evidence for overresponsiveness to afferent stimuli. Arch Phys Med Rehabil. 2009;90:580–6.
Samuel S, Lee M, Brown RJ, Choi HA, Baguley IJ. Incidence of paroxysmal sympathetic hyperactivity following traumatic brain injury using assessment tools. Brain Inj. 2018;32:1115–21.
Mathew MJ, Deepika A, Shukla D, Devi BI, Ramesh VJ. Paroxysmal sympathetic hyperactivity in severe traumatic brain injury. Acta Neurochir. 2016;158:2047–52.
Ammar MA, Hussein NS. Using propranolol in traumatic brain injury to reduce sympathetic storm phenomenon: A prospective randomized clinical trial. Saudi J Anaesth. 2018;12:514–20.
Jang SH, Kwon HG. Injury of the Hypothalamus in Patients With Hypoxic-Ischemic Brain Injury: A Diffusion Tensor Imaging Study. Am J Phys Med Rehabil. 2018;97:160–3.
Zhao ZD, Yang WZ, Gao C, Fu X, Zhang W, Zhou Q, et al. A hypothalamic circuit that controls body temperature. Proc Natl Acad Sci USA. 2017;114:2042–7.
Lee SJ, Jang SH. Hypothalamic injury in spontaneous subarachnoid hemorrhage: a diffusion tensor imaging study. Clin Auton Res. 2021;31:321–2.
Jang SH, Yi JH, Kim SH, Kwon HG. Relation between injury of the hypothalamus and subjective excessive daytime sleepiness in patients with mild traumatic brain injury. J Neurol Neurosurg Psychiatry. 2016;87:1260–1.
Jang SH, Seo YS. Neurogenic fever due to injury of the hypothalamus in a stroke patient: Case report. Medicine. 2021;100:e24053.
Jang SH, Choi KH. Paroxysmal sympathetic hyperactivity concurrent with hypothalamic injury in a patient with intracerebral hemorrhage: A case report. Medicine. 2022;101:e30058.
Ding JS, Zhang Y, Wang TY, Li X, Ma C, Xu ZM, et al. Therapeutic applications of hydrogen sulfide and novel donors for cerebral ischemic stroke: a narrative review. Med Gas Res. 2023;13:7–9.
Fan J, Du J, Zhang Z, Shi W, Hu B, Hu J, et al. The Protective Effects of Hydrogen Sulfide New Donor Methyl S-(4-Fluorobenzyl)-N-(3,4,5-Trimethoxybenzoyl)-l-Cysteinate on the Ischemic Stroke. Molecules. 2022;27:1554.
Pomierny B, Krzyzanowska W, Jurczyk J, Skorkowska A, Strach B, Szafarz M, et al. The Slow-Releasing and Mitochondria-Targeted Hydrogen Sulfide (H(2)S) Delivery Molecule AP39 Induces Brain Tolerance to Ischemia. Int J Mol Sci. 2021;22:7816.
Yu Q, Guo Q, Jin S, Gao C, Zheng P, Li DP, et al. Melatonin suppresses sympathetic vasomotor tone through enhancing GABA(A) receptor activity in the hypothalamus. Front Physiol. 2023;14:1166246.
Messmer SJ, Salmeron KE, Frank JA, McLouth CJ, Lukins DE, Hammond TC, et al. Extended Middle Cerebral Artery Occlusion (MCAO) Model to Mirror Stroke Patients Undergoing Thrombectomy. Transl Stroke Res. 2022;13:604–15.
Zhang Q, Yin J, Xu F, Zhai J, Yin J, Ge M, et al. Isoflurane post-conditioning contributes to anti-apoptotic effect after cerebral ischaemia in rats through the ERK5/MEF2D signaling pathway. J Cell Mol Med. 2021;25:3803–15.
Chen Z, Zhang Y, Wu X, Huang H, Chen W, Su Y. Characteristics and Outcomes of Paroxysmal Sympathetic Hyperactivity in Anti-NMDAR Encephalitis. Front Immunol. 2022;13:858450.
Feng S, Chen JX, Liu S, Zheng P, Sun J, Zhang X, et al. Clinical and prognostic study of anti-N-methyl-D-aspartate receptor encephalitis children with paroxysmal sympathetic hyperactivity syndrome. Zhonghua Yi Xue Za Zhi. 2021;101:3600–3..
Xu L, Qiu X, Wang S, Wang Q, Zhao XL. NMDA Receptor Antagonist MK801 Protects Against 1-Bromopropane-Induced Cognitive Dysfunction. Neurosci Bull. 2019;35:347–61.
Lipton SA, Choi YB, Takahashi H, Zhang D, Li W, Godzik A, et al. Cysteine regulation of protein function-as exemplified by NMDA-receptor modulation. Trends Neurosci. 2002;25:474–80.
Siefferman JW, Lai G. Propranolol for Paroxysmal Sympathetic Hyperactivity with Lateralizing Hyperhidrosis after Stroke. Case Rep Neurol Med. 2015;2015:421563.
Du Y, Demillard LJ, Ren J. Catecholamine-induced cardiotoxicity: A critical element in the pathophysiology of stroke-induced heart injury. Life Sci. 2021;287:120106.
Sternberg Z, Schaller B. Central Noradrenergic Agonists in the Treatment of Ischemic Stroke-an Overview. Transl Stroke Res. 2020;11:165–84.
Oto J, Suzue A, Inui D, Fukuta Y, Hosotsubo K, Torii M, et al. Plasma proinflammatory and anti-inflammatory cytokine and catecholamine concentrations as predictors of neurological outcome in acute stroke patients. J Anesth. 2008;22:207–12.
Chamorro A, Amaro S, Vargas M, Obach V, Cervera A, Gomez-Choco M, et al. Catecholamines, infection, and death in acute ischemic stroke. J Neurol Sci. 2007;252:29–35.
Strittmatter M, Meyer S, Fischer C, Georg T, Schmitz B. Location-dependent patterns in cardio-autonomic dysfunction in ischaemic stroke. Eur Neurol. 2003;50:30–8.
Huerta de la Cruz S, Rodriguez-Palma EJ, Santiago-Castaneda CL, Beltran-Ornelas JH, Sanchez-Lopez A, Rocha L, et al. Exogenous hydrogen sulfide restores CSE and CBS but no 3-MST protein expression in the hypothalamus and brainstem after severe traumatic brain injury. Metab Brain Dis. 2022;37:1863–74.
Coletti R, Almeida-Pereira G, Elias LL, Antunes-Rodrigues J. Effects of hydrogen sulfide (H2S) on water intake and vasopressin and oxytocin secretion induced by fluid deprivation. Horm Behav. 2015;67:12–20.
Liang YF, Zhang DD, Yu XJ, Gao HL, Liu KL, Qi J, et al. Hydrogen sulfide in paraventricular nucleus attenuates blood pressure by regulating oxidative stress and inflammatory cytokines in high salt-induced hypertension. Toxicol Lett. 2017;270:62–71.
Liao Y, Fan Y, He Q, Li Y, Wu D, Jiang E. Exogenous H(2)S Ameliorates High Salt-Induced Hypertension by Alleviating Oxidative Stress and Inflammation in the Paraventricular Nucleus in Dahl S Rats. Cardiovasc Toxicol. 2022;22:477–91.
Liu CW, Liao KH, Tseng H, Wu CM, Chen HY, Lai TW. Hypothermia but not NMDA receptor antagonism protects against stroke induced by distal middle cerebral arterial occlusion in mice. PLoS One. 2020;15:e0229499.
Tao BB, Liu SY, Zhang CC, Fu W, Cai WJ, Wang Y, et al. VEGFR2 functions as an H2S-targeting receptor protein kinase with its novel Cys1045-Cys1024 disulfide bond serving as a specific molecular switch for hydrogen sulfide actions in vascular endothelial cells. Antioxid Redox Signal. 2013;19:448–64.
Huang Y, Zhang Z, Huang Y, Mao Z, Yang X, Nakamura Y, et al. Induction of inactive TGF-beta1 monomer formation by hydrogen sulfide contributes to its suppressive effects on Ang II- and TGF-beta1-induced EMT in renal tubular epithelial cells. Biochem Biophys Res Commun. 2018;501:534–40.
Wang R, Tao B, Fan Q, Wang S, Chen L, Zhang J, et al. Fatty-acid receptor CD36 functions as a hydrogen sulfide-targeted receptor with its Cys333-Cys272 disulfide bond serving as a specific molecular switch to accelerate gastric cancer metastasis. EBioMedicine. 2019;45:108–23.
Mao Z, Yang X, Mizutani S, Huang Y, Zhang Z, Shinmori H, et al. Hydrogen Sulfide Mediates Tumor Cell Resistance to Thioredoxin Inhibitor. Front Oncol. 2020;10:252.
Funding
This work is supported by the National Natural Science Foundation of China (32271155, 81770499, 31871154), and the Natural Science Foundation of Hebei [CN] (H2023206367, H2020206350).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sun, J., Miao, Y., Wang, P. et al. Decreased levels of hydrogen sulfide in the hypothalamic paraventricular nucleus contribute to sympathetic hyperactivity induced by cerebral infarction. Hypertens Res 47, 1323–1337 (2024). https://doi.org/10.1038/s41440-024-01643-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-024-01643-5