Abstract
Background
To evaluate the role of the choroidal vascularity index (CVI) and the tortuosity index (TI), along with choroidal thickness measurements, in follow-up for anteriorly draining carotid cavernous fistulas (CCF) that would otherwise necessitate an invasive carotid angiogram.
Methods
In this longitudinal observational study, analysis of enhanced depth imaging spectral domain optical coherence tomography (SD-EDI-OCT) images of 22 patients with angiographically proven unilateral CCF with anterior drainage was performed for subfoveal choroidal thickness (SFCT), central foveal thickness (CFT), CVI and TI. Baseline measurements were compared with those taken at the last visit after occlusion of the fistula.
Results
Both in the clinically affected and unaffected eyes, there was a significant decrease in CFT (p = 0.015 and p = 0.005, respectively), SFCT (p = 0.000 for both eyes), CVI (p = 0.002 and 0.001, respectively) and all three TI parameters, including inferior (p < 0.001 and p = 0.01, respectively) and superior temporal vein tortuosity index (p = 0.005 and p = 0.02, respectively) as well as total venular tortuosity index (p < 0.001 and p = 0.002, respectively) after successful closure of the fistula compared to first presentation. Changes in all parameters were similar between the D-CCF and I-CCF groups except for CVI.
Conclusions
In this first study concerning follow-up of anteriorly draining unilateral CCFs by SD-EDI-OCT, we not only demonstrated involvement of the clinically unaffected eye but also showed that, this modality, utilizing CVI and TI together with choroidal thickness measurements, can be used as a readily available, noninvasive first-line followup method for evaluating occlusion of fistulas by endovascular treatment or spontaneous resolution.
Similar content being viewed by others
Introduction
Carotid cavernous fistulas (CCFs) are acquired vascular lesions that represent abnormal vascular shunts between the carotid arterial system and cavernous sinus. CCFs are classified according to their anatomy as direct versus indirect (dural) fistulas [1] or posteriorly versus anteriorly draining fistulas [2]. Direct CCFs (D-CCFs) are caused by a direct communication between the cavernous sinus and internal carotid artery. They are commonly traumatic in origin and have acute presentation with symptoms of orbital venous congestion secondary to their high flow. Indirect (dural) fistulas commonly have low flow and represent connections between the small dural branches of the carotid arterial system and the cavernous sinus.
The choroid is mainly comprised of blood vessels with adjacent stromal tissue [3], and the influence of CCF on choroid can be evaluated using enhanced-depth imaging (EDI) spectral domain optical coherence tomography. Increased subfoveal choroidal thickness (SFCT) in CCF was previously reported in six cases in the literature, showing a decline after treatment or spontaneous resolution [4,5,6,7,8]. A recent study investigated both the vascularity and morphology of the choroid in a series of CCFs with anterior and posterior drainage using thickness parameters together with the choroidal vascularity index (CVI) and the tortuosity index (TI), measured by SD-EDI-OCT [8]. This is the only study showing increased CVI and TI in patients with anterior CCFs compared to normal subjects. Our current study aimed to evaluate the role of CVI and TI along with choroidal thickness measurements in follow-up of anterior CCFs that would otherwise require an invasive carotid angiogram. In other words, we intended to evaluate changes in these indices over time after closure of the CCF. This is the first study evaluating the role of choroidal thickness measurements as well as CVI and TI parameters as noninvasive and first-line follow-up SD-EDI-OCT parameters for anterior CCFs.
Materials and methods
Institutional Ethics Committee approval was obtained for this study. This study was conducted in accordance with the Declaration of Helsinki.
In this longitudinal observational study, SD-EDI-OCT images of 22 consecutive patients with symptomatic and angiographically proven unilateral CCFs with anterior drainage admitted to our Interventional Neuroradiology service from February 2017 to December 2019 were included. SD-EDI-OCT images of patients with missing follow-up visits, CCFs with posterior drainage, coexistent macular or choroidal pathologies, and higher than 6 dioptres of myopia or hypermetropia were excluded from the study. Age, gender, clinical findings and angiographic features mainly the venous drainage routes, treatment and outcome were noted. Patients were divided according to CCF anatomy into direct and indirect fistulas for subgroup analysis.
All patients with D-CCFs underwent endovascular treatment. Indications for endovascular treatment of indirect CCFs (I-CCFs) included decreased visual acuity, visual field defects, diplopia, ocular hypertension or severe proptosis.
Analysis of SD-EDI-OCT (Heidelberg Spectralis, Heidelberg Engineering, Heidelberg, Germany) images was performed for SFCT, central foveal thickness (CFT), CVI and TI. Measurements were performed using the built-in Heidelberg Eye Explorer software (Heidelberg Eye Explorer version 1.8.6.0; Heidelberg Engineering). All measurements were performed by the same masked ophthalmologist.
Choroidal thickness was measured subfovealy on the transfoveolar scan with a built-in caliper of the software. SFCT was measured from the hyperreflective line of the retinal pigment epithelium (RPE) to the hyperreflective line of the inner scleral border (Supplementary Fig. 1A). CFT was measured from the vitreomacular interface to the anterior surface of the RPE.
CVI was calculated as explained in detail by Agrawal et al. and Inam et al. [8, 9]. (Supplementary Fig. 1 B–D). CVI was defined as the proportion of luminal choroidal area (LCA) to total choroidal area (TCA). Stromal area percent (SAP) was calculated as the proportion of stromal choroidal area (SCA) to TCA. Luminal-to-stromal ratio (LSR) is calculated by dividing CVI by SAP.
TI was calculated as described by Mohsenin et al. and Inam et al. [8, 10] (Supplementary Fig. 2A–D). TI was calculated for the superior temporal vein (SVTI), inferior temporal vein (IVTI) and total venular tortuosity (VTTI).
All measurements at presentation were compared with that at last follow-up after endovascular treatment. Post hoc subgroup analysis was performed to compare changes in the D-CCF and I-CCF groups. To analyse the changes at different time points, the measurements at presentation were compared to those at <4 weeks, 4–8 weeks and >8 weeks follow-up visits after endovascular treatment of CCF. If CCF was observed without intervention for spontaneous resolution, then the OCT measurements at presentation were compared with those taken at the last follow-up visit, as well as those at <4 weeks, 4–8 weeks and >8 weeks follow-up visits after the first presentation. Separate analyses were also conducted for D-CCF and I-CCF groups individually.
All measurements of D-CCF and I-CCF groups were compared with each other at both presentation and last follow-up.
The Shapiro–Wilk test was used to assess the normality of data. P < 0.05 was accepted as significant. The mean and standard deviation were used for continuous variables, and percentages were used for categorical variables. Student’s t test was used for the analysis of normally distributed data. Mann–Whitney U test was used for the analysis of the data that were not normally distributed. Repeated measures ANOVA was used to assess the differences between different time points. Post hoc analyses were performed using the Bonferroni test.
Results
Demographic and clinical findings
Twenty-two patients with angiographically proven unilateral CCF with anterior drainage pattern were included in the study. The mean age of subjects was 55.40 years with a range of 21–91 years (8 males, 14 females). Sixteen patients (4 males, 12 females) had I-CCF, and six patients (4 males, 2 females) had D-CCF. The mean age was 39.83 ± 24.70 (range: 21–82 years) for the D-CCF group and 61.25 ± 15.40 (range: 28–91 years) for the I-CCF group. The left eye was affected in 16 patients (72.7%). Three of six patients in the D-CCF group had a history of trauma to the affected eye. Two patients had spontaneous cavernous aneurysm ruptures as the underlying cause of their direct fistulas. Four of 6 patients (66.7%) in the D-CCF and 5 out of 16 patients (31.2%) in the I-CCF group had decreased vision at presentation due to optic neuropathy. The average duration of symptoms at presentation was 10.16 ± 11.53 months with a range of 2–36 months (Supplementary Table 1). The most common presenting symptom was redness in the affected eye, which was noted in all patients. Nine patients complained of swelling/prominence of the affected eye, and three had complaints of diplopia. On examination, 16 patients were noted to have tortuous corkscrew episcleral vessels. Proptosis was noted in 18 patients with a mean exophthalmometer difference of 5.14 mm (range: 3–12 mm) between the affected and unaffected eyes. Extraocular motility restriction was noted in 14 patients with a limitation in abduction in 13 patients. Nine patients had a limitation in up gaze. Intraocular pressure measured by applanation was elevated in ten patients with an average of 8.3 mmHg (3–14 mmHg) difference between the affected eyes compared to the unaffected eyes. Dilated tortuous vessels were noted on fundus examination in six patients with retinal haemorrhages in all quadrants in one patient. Two patients (one with D-CCF) had macular oedema noted on OCT.
Results of treatment and follow-up
Twenty-one patients with CCF were treated with embolization of the fistula, and one patient with indirect fistula was observed for spontaneous closure.
Endovascular treatments were performed by a single operator, and under general anaesthesia using biplane angiographic systems (Siemens, Erlangen, Germany). All patients with indirect type of fistulas were treated through a transvenous approach. Transvenous access was obtained either via the femoral vein (seven cases) or via percutaneous puncture of the craniofacial veins (eight cases). The draining veins were obliterated with coils (four cases), ethylene vinyl alcohol copolymers including Squid (Balt, Montmorency, France) or Onyx (Covidien, Plymouth, MN, USA) in three cases, a combination of these copolymers and coils (six cases) and cyanoacrylate glue (two cases). The direct fistulas were treated by a combined transarterial–transvenous route with coils or a combination of coils and liquid embolic agents noted above, in a single patient, a detachable balloon was also used.
The mean follow-up was 18 ± 15.48 weeks (range: 4–60 weeks). Optic neuropathy completely resolved in six out of nine patients with complete recovery of vision after embolization. Two patients had significant improvement in vision (3 and 5 Snellen lines), and one patient who had long standing direct fistula with temporal optic disc pallor at presentation did not have significant improvement in vision on his last follow-up visit. Sixteen of 22 patients had a follow-up cerebral angiogram (n = 14) or magnetic resonance angiography (MRA) (n = 2) after the procedure (range: 1.5–10.5 ± 2.20 months, mean: 3.92 months) that showed complete resolution of the CCF. One patient had a recurrent fistula and required a second embolization 2 months after the first procedure. A 3-month follow-up angiogram after the second procedure showed complete resolution of the fistula. The remaining six patients without follow-up imaging experienced complete resolution of symptoms and signs related to CCF at the time of their last clinic visits. None of the patients had complications related to treatment. One patient was noted to have retinal haemorrhages at four quadrants 1 month after treatment, which resolved completely without sequela during follow-up.
Change in choroidal thickness, CVI and TI parameters during follow-up
The mean CFT value of the clinically affected eye in all 22 patients was 212.16 ± 37.27 μm at presentation and 190.92 ± 37.97 μm at the last follow-up. There was a significant decrease in the mean CFT values for both the affected and unaffected eyes (p = 0.015 and p = 0.005, respectively) after closure of the fistula. Mean SFCT of the affected eye was 383.13 ± 113.25 at presentation and 283.08 ± 117.87 at the last follow-up. There was a significant decrease in the mean SFCT values of both the affected (p < 0.001) and unaffected eyes (p < 0.001) after treatment (Table 1 and Fig. 1A, B). In the subgroup analysis, changes in CFT and SFCT in the affected eye were similar in the D-CCF and I-CCF groups (p = 0.68 and p = 0.21, respectively) (Fig. 1I, J).
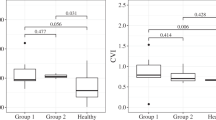
A–H Graphics showing central foveal thickness (CFT), subfoveal choroidal thickness (SFCT), choroidal imaging parameters and tortuosity index values of the clinically affected eye in all patients with CCF at baseline and last visit after occlusion of the fistula. A significant decrease was noted in CFT (A), SFCT (B), CVI (choroidal vascularity index) (C), luminal-to-stromal ratio (LSR) (E) and all tortuosity indices, including inferior temporal vein index (IVTI) (F), superior temporal vein index (SVTI) (G) and total venular tortuosity index (VTTI) (H), with a significant increase in stromal area percent (SAP) (D) after closure of the fistula. I–P Graphics showing CFT, SFCT, choroidal imaging parameters and tortuosity index values of clinically affected eye in the patients with direct (D-CCF) and indirect carotid cavernous fistula (I-CCF) at baseline and the last visit after closure of the fistula. CFT (I), SFCT (J) and all tortuosity indices, including IVTI, SVTI and VTTI (N–P), similarly decreased in patients with D-CCF and I-CCF after closure of the fistula. There was a significant difference in changes of CVI, SAP and LSR (K–M), between the two groups according to post hoc subgroup analysis.
In both the clinically affected and unaffected eyes, there was a significant decrease in CVI (p = 0.010 and 0.043, respectively) and LSR (p = 0.011 and 0.025, respectively) with an increase in SAP (p = 0.010 and 0.043, respectively) at the last follow-up compared to the first presentation (Table 1 and Fig. 1C–E). There was a significant difference in changes of CVI (p = 0.018), SAP (p = 0.018) and LSR (p = 0.029), between the two groups according to post hoc subgroup analysis (Fig. 1K–M).
In both the clinically affected and unaffected eyes, TI values, including IVTI (p < 0.001 both), SVTI (p = 0.002 and p = 0.001, respectively) and VTTI (p < 0.001 both) significantly decreased at the last follow-up compared to first presentation (Table 1 and Fig. 1F–H). In the subgroup analysis, the changes in TI measurements in the affected eye were similar between D-CCF and I-CCF groups (p = 0.57 for IVTI, p = 0.45 for SVTI and p = 0.96 for VTTI) (Fig. 1N–P).
Analysis of the changes at different time points showed a significant decrease in the mean SFCT (p = 0.033), IVTI (p = 0.003) and VTTI (p = 0.012) of the affected eye <4 weeks follow-up after closure of the fistula.
When the I-CCF group was analysed separately, the mean SFCT of the clinically affected eye was 361.10 ± 113.01 at presentation and 249.49 ± 106.22 at the last follow-up. There was a significant decrease in the mean SFCT values of both the clinically affected (p < 0.001) and unaffected eyes (p < 0.001) after treatment. Both in the affected and unaffected eyes, there was a significant decrease in CVI (p = 0.002 and 0.001, respectively) and LSR (p = 0.004 and 0.005, respectively) with an increase in SAP (p = 0.002 and 0.001, respectively) at the last follow-up compared to first presentation. Both in the clinically affected and unaffected eyes, TI values, including IVTI (p < 0.001 and p = 0.01, respectively), SVTI (p = 0.005 and p = 0.02, respectively) and VTTI (p < 0.001 and p = 0.002, respectively), were significantly decreased at last follow-up compared to first presentation.
When the D-CCF group was analysed separately, the mean SFCT of the clinically affected eye was 441.86 ± 99.29 at presentation and 372.65 ± 106.15 at last follow-up, showing a significant decrease in the mean SFCT values in the affected eye (p = 0.037). There was a significant decrease in TI values, including IVTI (p = 0.023) and VTTI (p = 0.046), in the affected eye after treatment. Although there was a trend towards decreased SAP and a corresponding increase in CVI and LSR, these findings were not statistically significant.
Clinical findings and SD-EDI-OCT parameters of the clinically affected eye of the patients at presentation and after closure of the fistula are shown in Supplementary Table 2.
Comparison of choroidal thickness, CVI and TI parameters in direct versus indirect fistulas
There was no significant difference in SFCT, LCA or TI values of D-CCF and I-CCF groups at presentation. TCA (p = 0.040), SCA (p = 0.008) and SAP (p = 0.001) were all significantly higher and correspondingly, CVI (p = 0.001) and LSR (p = 0.001) were lower in D-CCFs compared to the I-CCF group at presentation. At the last follow-up visit, CVI (p = 0.59), SAP (p = 0.59) and LSR (p = 0.59) became similar in both groups.
Sample pre-procedure and last follow-up SD-EDI-OCT images of patients with D-CCFs and I-CCFs demonstrating clinical findings with SFCT, CVI and TI measurements are shown in Figs. 2–4.
Clinical image of the patient with direct CCF affecting the left eye before (A) and 6 weeks after the endovascular treatment (B). SD-EDI-OCT image of the left eye at presentation with a subfoveal choroidal thickness measurement of 511 mm (C), which decreased to 325 mm 6 weeks after treatment (D). Tortuosity of the vessels is noted at presentation (C) with a total venular tortuosity index (VTTI) value of 1.115, which significantly decreased after treatment (D) to a VTTI value of 1.041.
SD-EDI-OCT image of the left eye at presentation with a subfoveal choroidal thickness measurement of 210 mm (A), which decreased to 85 mm at 9 months after treatment (B). Binarized image of the left eye at presentation (C) with a choroidal vascularity index (CVI) measurement of 78.16, which decreased to 61.15 9 months after the treatment (D). Luminal (black) and stromal (white) parts are seen. Scale-adjusted infra-red fundus image of the left eye at presentation (E) with a total venular tortuosity (VTTI) value of 1.067, which decreased to 1.041 9 months after treatment (F).
After a brief period of clinical improvement following the first endovascular treatment, he presented with deterioration of symptoms and findings 2 months after the treatment that required a second procedure resulting in complete closure of the fistula. Clinical image of the patient with direct CCF affecting the right eye 2 months after the first treatment (A) and 3 months after the second endovascular treatment (B). The left eye became phthisical from the gunshot injury. SD-EDI-OCT image of the right eye 2 months after the first session of endovascular treatment with a subfoveal choroidal thickness (SFCT) measurement of 477 mm (C), which decreased to 402 mm 3 months after the second treatment (D). SFCT was 468 mm at first presentation. E, F Binarized image of the right eye 2 months after the first session endovascular treatment with a choroidal vascularity index (CVI) measurement of 63.07. Three months after the second session treatment, CVI was 63.90 (F).
Discussion
While some I-CCFs can have spontaneous closure, many cases of CCFs require treatment for the occlusion the fistula [11]. Endovascular management is the current treatment modality of choice for CCFs [12,13,14]. Rates of complete occlusion after endovascular treatment range from 70 to 90% for I-CCF and 55 to 99% for D-CCFs [15]. Despite the high success rate of endovascular treatment, recurrence is still possible regardless of complete obliteration in the primary treatment [16]. Due to the rarity of this disease, difference in the types and characteristics of the fistula and the variety of therapeutic strategies, there is no consensus for follow-up of CCFs. Lin et al. [16] recommended close follow-up for 3–6 months after intervention according to their data from 55 patients with traumatic D-CCF, and the median time to recurrence was 3 months. Holland et al. [15] reported that multiple interventions were required in 33% of direct fistulas and in 16% of indirect fistulas in their series, including 39 patients with CCF. Three to six months after endovascular treatment, computed tomography angiogram or MRA could be used in some cases to confirm complete closure of the fistula and exclude recurrence, but cerebral angiogram is still the gold standard for follow-up. Clinical assessment may not always correlate with angiographic findings because successful occlusion results in gradual resolution of orbital signs within days to weeks to sometimes months [17]. Conversely, transient worsening of orbital congestion can also be seen after the closure of the fistula [17]. Our data show that SD-EDI-OCT can be used as a noninvasive, readily available ancillary test for follow-up of CCFs with anterior drainage. We noted a significant decrease in choroid thickness, CVI and TI measurements after successful closure of the fistula. The decreases in choroid thickness, IVTI and VTTI of the affected eye became significant as early as the first month after treatment.
CVI is a new imaging parameter allowing the assessment of changes in choroidal vascularity and is affected by fewer physiological factors compared to those of choroidal thickness [18]. CVI allows quantification of stromal and luminal vascular components whereas choroidal thickness measurement reflects the total choroidal stromal and vascular system without distinction [18]. CVI and TI were found to increase in patients with CCFs with an anterior drainage pattern compared to normal subjects and were suggested as helpful markers for diagnosis [8]. TI is also a very useful and reliable marker that allows quantitative assessment of tortuosity. This is particularly helpful in patients with CCF since retinal venous tortuosity and dilatation are frequently noted findings in these patients for which subjective evaluation has high interobserver variability [19]. Our results show that CVI and TI, together with choroidal thickness measurements, can be used in follow-up for CCFs in patients who undergo endovascular treatment or conservative treatment by close observation for spontaneous closure of the fistula.
Another distinctive finding of our study was the significant decreases observed in CFT, SFCT, CVI, LSR and all venular TI indices, as well as increases in SAP both in the clinically affected and unaffected eyes. Although uncommon, due to intercavernous communication and variations in drainage pattern, contralateral eye signs can be present [20,21,22]. Unilateral fistulas with bilateral symptoms are uncommon because, despite communication between the cavernous sinus, there is a major route for decompression through the ophthalmic veins [20]. Our data show for the first time that, although clinically not evident, anterior drainage CCFs affect both eyes with increased subfoveal retinal and choroidal thickness, tortuosity of retinal veins and change in CVI.
Another interesting finding of our study noted in post hoc analysis is that even though CFT, SFCT and all TI measurements were similarly decreased in both subgroups, there was a difference in changes of CVI, SAP and LSR between direct and indirect fistulas after closure of the fistula. In accordance with this finding, TCA, SCA and SAP were significantly higher and correspondingly CVI and LSR were lower in D-CCFs compared to I-CCFs at presentation. This difference in CVI at presentation disappears after closure of the fistula. These findings can be explained by the fact that direct fistulas are high-flow lesions resulting in a higher degree of orbital and choroidal congestion compared to indirect fistulas. This congestion may result in an increase not only in the luminal component of the choroid but also the stroma. Since indirect fistulas have a lower flow, they cause a lesser degree of orbital congestion. Hence, they may not have as much stromal oedema. Even though there was no difference in LCA in between the groups, TCA and SCA were higher in the D-CCF group than the I-CCF group at presentation, supporting this hypothesis. Since CVI, SAP and LSR are ratios and not actual values, differences in these parameters between the D-CCF and I-CCF groups indicate that the D-CCF group has more differential stromal thickening at presentation compared to that of the I-CCF group. This is also a new finding, which needs to be supported by further studies with a higher number of D-CCF patients. SD-EDI-OCT may be a useful noninvasive supplementary test to differentiate direct and indirect fistulas. It is important to distinguish between direct and indirect fistulas due to therapeutic and prognostic implications [23].
Since post hoc analysis showed a difference between the D-CCF and the I-CCF groups with respect to changes in CVI, SAP and LSR after occlusion of the fistula, we decided to conduct individual analyses for D-CCFs and I-CCFs in addition to analysis of the entire group. In the I-CCF group (n = 16), there was a significant decrease in subfoveal retinal and choroidal thickness, all TI values, CVI and LSR with an increase in SAP in both the affected and unaffected eyes after the closure of the fistula. These findings are in accordance with results for the entire group. In the D-CCF (n = 6) group, there was a significant decrease in SFCT, TI values, including IVTI and VTTI, of the affected eye after treatment. Although there was a trend towards decreased SAP and a corresponding increase in CVI and LSR, these were not statistically significant. This result is possibly due to the small number of cases in the D-CCF group.
Our study has the following limitations. Follow-up dates for patients were not standardized, potentially introducing bias. This is reflective of daily clinical practice in a tertiary care hospital setting where most patients are coming from different parts of the country. For this reason, follow-up dates were grouped by certain set intervals for evaluation. Likewise, patients with more complex CCFs that are more difficult to treat might have been included in the study, potentially introducing a selection bias. In addition, not infrequently, these complicated cases especially the ones with D-CCFs, have more severe ophthalmic findings that make imaging more difficult at presentation affecting the quality of images. Another limitation is the small number of patients in the D-CCF group, which prevents us from reaching definitive conclusions regarding D-CCFs. This limitation is primarily due to the rarity of this disease. It was also more difficult to take good quality images of the affected eye especially in the D-CCF group due to media opacities and poor visual acuity.
In conclusion, in patients with unilateral CCFs with anterior drainage, we demonstrated subclinical involvement of the clinically unaffected eye with the disease process. Moreover, we showed that SD-EDI-OCT, using CVI and TI together with choroidal thickness measurements, could be used as a noninvasive ancillary follow-up modality for evaluating occlusion of fistulas by endovascular treatment or spontaneous resolution.
Summary
What was known before
-
SFCT, CVI and the TI could potentially be used to aid in the diagnosis of CCFs with anterior drainage (A-CCF).
What this study adds
-
SD-EDI-OCT, using CVI and TI together with choroidal thickness measurements, could be used as a noninvasive ancillary follow-up modality for evaluating occlusion of fistulas by endovascular treatment or spontaneous resolution.
-
In patients with unilateral CCFs with anterior drainage, there is subclinical involvement of the clinically unaffected eye with the disease process.
References
Barrow DL, Spector RH, Braun IF, Landman JA, Tindall SC, Tindall GT. Classification and treatment of spontaneous carotid-cavernous sinus fistulas. J Neurosurg. 1985;62:248–56.
Stiebel-Kalish H, Setton A, Nimii Y, Kalish Y, Hartman J, Huna Bar-On R, et al. Cavernous sinus dural arteriovenous malformations: patterns of venous drainage are related to clinical signs and symptoms. Ophthalmology. 2002;109:1685–91.
Nickla DL, Wallman J. The multifunctional choroid. Prog Retin Eye Res. 2010;29:144–68.
Gonzalez Martin-Moro J, Sales-Sanz M, Oblanca-Llamazares N, Bustos-Garcia A, Mendez-Cendon JC, Fandino E. Choroidal thickening in a case of carotid cavernous fistula. Orbit. 2018;37:306–8.
Shinohara Y, Kashima T, Akiyama H, Kishi S. Alteration of choroidal thickness in a case of carotid cavernous fistula: a case report and a review of the literature. BMC Ophthalmol. 2013;13:75.
Rey A, Castillo L, Dyrda A, Maseras X, Jurgens I. Subfoveal choroidal thickness changes in carotid cavernous fistula following spontaneous resolution. BMC Ophthalmol. 2016;16:63.
Pascual-Camps I, Martinez-Rubio C, Gallego-Pinazo R, Espana-Gregori E. Optical coherence tomography characterization of macular changes secondary to arteriovenous fistula. Neuroophthalmology. 2018;42:182–6.
Inam O, Arat YO, Yavas GF, Arat A. Retinal and choroidal optical coherence tomography findings of carotid cavernous fistula. Am J Ophthalmol. 2019;206:264–73.
Agrawal R, Gupta P, Tan KA, Cheung CM, Wong TY, Cheng CY. Choroidal vascularity index as a measure of vascular status of the choroid: measurements in healthy eyes from a population-based study. Sci Rep. 2016;6:21090.
Mohsenin A, Mohsenin V, Adelman RA. Retinal vascular tortuosity in obstructive sleep apnea. Clin Ophthalmol. 2013;7:787–92.
de Keizer R. Carotid-cavernous and orbital arteriovenous fistulas: ocular features, diagnostic and hemodynamic considerations in relation to visual impairment and morbidity. Orbit. 2003;22:121–42.
Ellis JA, Goldstein H, Connolly ES Jr, Meyers PM. Carotid-cavernous fistulas. Neurosurg Focus. 2012;32:E9.
Zanaty M, Chalouhi N, Tjoumakaris SI, Hasan D, Rosenwasser RH, Jabbour P. Endovascular treatment of carotid-cavernous fistulas. Neurosurg Clin N Am. 2014;25:551–63.
Alexander MD, Halbach VV, Hallam DK, Cooke DL, Ghodke BV, Dowd CF, et al. Long-term outcomes of endovascular treatment of indirect carotid cavernous fistulae: superior efficacy, safety, and durability of transvenous coiling over other techniques. Neurosurgery. 2019;85:E94–100.
Holland LJ, Mitchell Ranzcr K, Harrison JD, Brauchli D, Wong Y, Sullivan TJ. Endovascular treatment of carotid-cavernous sinus fistulas: ophthalmic and visual outcomes. Orbit. 2019;38:290–9.
Lin TC, Mao SH, Chen CH, Chen YL, Wong HF, Chang CJ, et al. Systematic analysis of the risk factors affecting the recurrence of traumatic carotid-cavernous sinus fistula. World Neurosurg. 2016;90:539–45.e1.
Quiros PA, Wang M. Vascular disorders. In: Yanoff M, Duker JS, editors. Ophthalmology. 9. 5th ed. Elsevier; Edinburgh; 2019. p. 995–1002.e1.
Iovino C, Pellegrini M, Bernabei F, Borrelli E, Sacconi R, Govetto A, et al. Choroidal vascularity index: an in-depth analysis of this novel optical coherence tomography parameter. J Clin Med. 2020;9(2):595.
Kagan A, Aureli E, Dobree J. A note on signs in the fundus oculi and arterial hypertension: conventional assessment and significance. Bull World Health Organ. 1966;34:955–60.
Demartini Z Jr., Liebert F, Gatto LA, Jung TS, Rocha C Jr., Santos AM, et al. Unilateral direct carotid cavernous fistula causing bilateral ocular manifestation. Case Rep Ophthalmol. 2015;6:482–7.
Zhu L, Liu B, Zhong J. Post-traumatic right carotid-cavernous fistula resulting in symptoms in the contralateral eye: a case report and literature review. BMC Ophthalmol. 2018;18:183.
Santhosh M, Joseph S, Doctor P. Right traumatic carotico-cavernous fistula with bilateral eye signs and post-treatment right pseudo Argyll Robertson pupil. Indian J Ophthalmol. 2008;56:253–5.
Kaplan JB, Bodhit AN, Falgiani ML. Communicating carotid-cavernous sinus fistula following minor head trauma. Int J Emerg Med. 2012;5:10.
Author information
Authors and Affiliations
Contributions
YOA conceived and designed the work, acquired data, interpreted the results, drafted and revised the manuscript. OI acquired data, analysed the results and drafted the manuscript. GFY and AA acquired data and revised the manuscript. YOA, OI, GFY and AA approved the final version and agreed to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Arat, Y.O., İnam, O., Yavas, G.F. et al. The role of choroidal optical coherence tomography for follow-up of carotid cavernous fistula with anterior drainage. Eye 36, 2020–2027 (2022). https://doi.org/10.1038/s41433-021-01744-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-021-01744-2