Abstract
We previously demonstrated that engagement of cadherins, cell to cell adhesion molecules, triggers a dramatic increase in levels and activity of the Rac/Cdc42 small GTPases, which is followed by secretion of IL6 family cytokines and activation of their common receptor, gp130, in an autocrine manner. This results in phosphorylation of the Signal Transducer and Activator of Transcription-3 (Stat3) on tyrosine-705, which then dimerizes, migrates to the nucleus, and activates transcription of genes involved in cell division and survival. In the present report we demonstrate that, in mouse Balb/c3T3 fibroblasts, mutationally activated Src527F also increases Rac levels, leading to secretion of IL6 family cytokines and gp130 activation, which triggers the Stat3-ptyr705 increase. Interestingly, our results also demonstrate that cadherin-11 is required to preserve gp130 levels for IL6 family signaling. At the same time, however, activated Src527F downregulates cadherin-11, in a quantitative manner. As a result, Src527F expression to intermediate levels allows sufficient cadherin-11, hence gp130 levels for Stat3 activation, as expected. However, expressed to high levels, Src527F eliminates cadherin-11, hence gp130 signaling, thus abolishing Stat3-ptyr705 stimulation. Taken together, these data establish for the first time a loop between Src, cadherin-11, gp130, and Stat3 activation. This fine balance between Src527F and cadherin-11 levels which is required for Stat3 activation and cellular survival could have significant therapeutic implications.
Similar content being viewed by others
Introduction
The signal transducer and activator of transcription-3 (Stat3) is a latent cytoplasmic transcription factor. Stat3 is activated by receptor tyrosine kinases such as EGFR, cytokine receptors such as the Interleukin-6 receptor (IL6R), and non-receptor tyrosine kinases such as Src. Ligand engagement leads to phosphorylation of specific tyrosine residues on the receptors, which provide docking sites for the Src homology 2 (SH2) domain of Stat3. Thus, Stat3 is recruited to the activated receptors and, in turn, becomes phosphorylated by the receptor itself, or by associated Janus kinase (JAK) or Src family kinases [1, 2]. Following phosphorylation of the critical tyrosine-705 residue (ptyr705), Stat3 monomers associate with each other through reciprocal SH2-ptyr705 interactions to form dimers that translocate to the nucleus. There, Stat3 dimers bind DNA sequences to activate the transcription of specific genes involved in cell division and survival, such as myc, bcl-xL, mcl-1, and survivin while they downregulate the tumor suppressor p53 [3], thus protecting tumor cells from apoptosis [4, 5]. Hyperactivation of Stat3 is present in a large number of cancers and has been reported to be required for tumor cell growth and survival, as well as angiogenesis, metastasis, and immune evasion [6]. The fact that a constitutively active form of Stat3 (Stat3C) alone is sufficient to induce transformation of cultured fibroblasts [7] and epithelial cells [8] points to an etiological role of Stat3 in oncogenesis.
We and others previously demonstrated that engagement of cadherins, cell to cell adhesion molecules [E-, N-cadherin, or cadherin-11 (Cad11)], as occurs with a confluence of cultured cells, causes a dramatic increase in Stat3 activity in breast carcinoma lines as well as normal or transformed epithelial cells and fibroblasts [8,9,10]. This is triggered by a dramatic increase in protein levels and activity of the Rac1 (Rac) and Cdc42 small GTPases [11], which, in turn, causes a transcriptional upregulation of IL6 family cytokines, thus activating Stat3 through the common receptor subunit of the family, gp130, and JAK kinases [8, 12].
The Src family of non-receptor tyrosine kinases is often hyperactive in a variety of cancers [13], and activated Src is frequently associated with worse patient survival [14]. Early results demonstrated that Src activates Stat3 in cultured cells and that transcriptionally active, tyrosine-705-phosphorylated Stat3 is required for neoplastic transformation by the Src oncogene [15, 16]. Since Cad11 is also a potent activator of Stat3 [12], we revisited the question of Stat3 activation by Src, by exploring the role of Cad11 in a defined system of Balb/c3T3 cells expressing different levels of activated Src527F. These cells are not transformed so that, unlike tumor cells, the relationship between Src, cadherin-11, and Stat3 can be examined in the absence of confounding pathways. Our results reveal the importance of a fine balance between Src527F and Cad11 levels for Stat3 activation; expressed to high levels, Src527F eliminates Cad11, which, in turn, is required for gp130 signaling and Stat3 activation. Expressed to intermediate levels, on the other hand, Src527F allows sufficient residual levels of Cad11 and gp130 for Stat3 activation. Taken together, these data establish a loop between Src, Cad11, gp130, and Stat3 activation, a finding which could have significant therapeutic implications.
Materials and methods
Cell lines and culture techniques
The Balb/c3T3 cell line (ATCC) has been described [17]. The rat-F111 line and its mT-expressing derivatives are described in [18]. All cells were grown in Petri dishes in DMEM in a 5% CO2 incubator. Balb/c3T3 cells and derivatives required supplementation with 10% fetal bovine serum (PAA Laboratories, cat. #A15-751), but the rat-F111 cells and derivatives required 5% calf bovine serum. It was especially important to ensure even distribution of the cells during plating, by passing them at subconfluence and pipetting vigorously with a 9” Pasteur pipette. To reduce the variability that might be caused by nutrient depletion in post-confluent cultures, the medium was changed every 24 h. Cell confluence was estimated visually and quantitated by imaging analysis of live cells under phase contrast using a Leitz Diaplan microscope and the MCID-elite software (Imaging Research, St. Catharine’s, ON).
All lines were treated with plasmocin (Invivogen #ant-mpt-1, 10 μg/ml for 2 weeks) periodically and tested for mycoplasma contamination by DAPI staining or by PCR, as described in [19].
Gene transduction
The pWZL-Src527F-hygro plasmid which encodes the constitutively active chicken Src mutant and a hygromycin resistance gene was a gift from Dr. Andrew Craig, Queen’s University and propagated by transfection in Phoenix ecotropic retroviral packaging cells using the PolyJet in vitro DNA transfection reagent (SignaGen, Frederick, MD, cat. #SL100688). Following selection, clones picked were further subcloned at least twice more to eliminate revertants and propagated at subconfluence. A number of vials of each were frozen and used for a maximum of two weeks after thawing. This was especially important for high-Src527F-expressing cells.
Balb-shCad11 and Balb-shRac cells have been described [12, 20]. Cad11 shRNA-encoding retroviral constructs were purchased from Open Biosystems (Huntsville, AL, cat. #RMM4530-NM_009866). For shCad11 we used 4 different sequences (Table S1), with essentially identical results. Rac shRNA was purchased from Open Biosystems (cat. #RMN 1766-97047533). Following infection, cells were selected with 0.5–1 µg/ml puromycin, and individual clones expanded into lines for further experimentation.
Western blotting
Following electrophoresis and transfer, membranes were cut into strips based on the positions of pre-stained markers and probed for SrcpY416 (Cell Signaling, Danvers, MA, cat. #2101), pan-Src (Cell Signaling, cat. #2109), Cadherin-11 (Cell Signaling, cat. #4442), Stat3-ptyr705 (Cell Signaling, cat. #9145, Rabbit) cleaved PARP (Cell Signaling, cat. #9544), Rac (Millipore #05-389), b-actin (Cell Signaling #3700) and secondary antibodies according to the manufacturer’s protocols. Bands were visualized using the Clarity Western ECL substrate (Bio-Rad, Hercules, CA, cat. #170-5060). Images were developed by exposing the membranes to film or by using the Azure 300 Digital imager (Azure Biosystems, Dublin, CA). Photo-shop or Corel Draw software was used for the organization of non-adjusted, original images. Quantitation was achieved by image analysis using ImageJ (U.S. NIH). In all cases, band intensities were normalized to β-actin or α-tubulin levels of the same samples. Rac activation assays were performed using GST-PAK pull-down assays with the Rac activation kit from Cytoskeleton (Denver, CO, cat. #BK035), using beads coated with glutathione-S-transferase (GST) fused to the binding domain of p21-activated kinase (PAK), followed by probing for Rac by Western blotting, as previously described [8].
Experiments were repeated at least twice and with a number of lines expressing different Src levels. Stat3-ptyr705 levels were examined at different densities spanning 100% confluence, as described in Results.
TUNEL staining
Cells were fixed with 4% paraformaldehyde and assayed for apoptosis by terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) using fluorescein-coupled nucleotides, according to the manufacturer’s instructions (Roche, cat. #11684795910). Cells with apoptotic nuclei were visualized and photographed under fluorescence and phase contrast illumination.
Results
Src527F expression reduces Cadherin-11 protein levels in mouse Balb/c3T3 fibroblasts
Src is known to negatively regulate E-cadherin expression and function [reviewed in ref. [21]]. To examine the effect of Src upon Cad11, a type II classical cadherin, constitutively active Src527F was stably expressed in mouse Balb/c3T3 fibroblasts which possess high Cad11 levels [17], using a pBabeHygro-based retroviral vector and Hygromycin resistance selection (see Materials and Methods). Detergent lysates of resistant clones, together with the parental Balb/c3T3, were probed for SrcpY416 (which correlates with Src activity) by Western blotting (supplementary Fig. S1, A, top panel). Bands were quantitated by image analysis using the value of the highest SrcY416- expressing clone obtained as 100%. A number of individual clones with increasing amounts of Src527F were chosen for further experimentation. Results from representative clones expressing low, medium, or high Src527F levels, named Src-low, Src-med, or Src-high, respectively, are presented in Fig. 1.
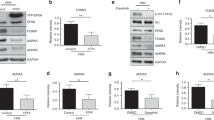
A Parental Balb/c3T3 cells (lane 1) and clones stably expressing low (Src-low, lane 2), medium (Src-med, lane 3), or high (Src-high, lane 4) levels of Src527F were grown to 100% confluence. Detergent lysates were resolved by gel electrophoresis and probed for SrcpY416, Cad11, pan-Src, or β-actin as a loading control, as indicated. Numbers under the lanes refer to band intensities obtained through quantitation by image analysis and normalized to β-actin levels, with the peak values of clone Src-high (lane 4) for SrcpY416, and the parental Balb/c3T3 (lane 1) for Cad11, respectively, taken as 100%. Numbers at the left refer to molecular weight markers. B Quantitation analysis of Cad11 and SrcpY416 protein levels in clones in A. Values are averages of three independent experiments, ±SEM. C Morphology of Balb/c3T3 cells and clones expressing medium, or high Src527F levels. Cells were photographed under phase contrast illumination. Bar: 100 µm. Note the round shape of Src-high cells (c), which lack Cad11.
Examination of Cad11 levels in detergent cell extracts by Western blotting using the parental Balb/c3T3 as a positive control, revealed a quantitative, inverse relationship between SrcpY416 and Cad11 levels (Fig. 1A): Src-high cells had no detectable Cad11 (lane 4), Src-med cells had approximately 42% the Cad11 protein levels of the parental Balb/c3T3 cells (lane 3 vs 1), while Src-low cells had approximately 75% the Cad11 levels of the Balb/c3T3 (lane 2 vs 1). A number of additional clones expressing different Src527F levels gave similar results (supplementary Fig. S1 B, top panel). Collectively, these findings indicate that although Src527F is, in fact, able to dramatically reduce Cad11, the effect is dependent upon the levels of Src expression; elimination of detectable Cad11 required ~90% of the highest Src527F levels obtained in this system.
High Src527F expression reduces Stat3-ptyr705 levels
It has been extensively documented that Src expression increases Stat3-ptyr705 phosphorylation, DNA binding, and transcriptional activity [15, 16]. As shown above, however, Src527F, in a quantitative manner, also downregulates Cad11, which (upon engagement) is a potent Stat3 activator. To examine this apparent paradox, we assessed the effect of different levels of Src527F upon the Cad11/Stat3 axis. Since cadherin engagement, as occurs with a density of cultured cells, causes a dramatic increase in Stat3 activity [[8, 10], reviewed in ref. [22]], experiments were performed at different cell densities. Balb/c3T3, Src-med, and Src-high cells were plated in plastic culture dishes and, when 50% confluent and over several days thereafter (Fig. 2A), detergent cell extracts were probed for the tyr705 phosphorylated, i.e., the activated form of Stat3 by western blotting. As shown in Fig. 2B, the levels of Stat3-ptyr705 increased with density in the parental Balb/c3T3 (lanes 1–5), as previously documented [12]. Levels of Stat3-ptyr705 also increased with activated Src527F expression, from the parental Balb/c3T3 (lanes 1–5) to Src-med cells (Src527F 67% of the highest, lanes 6–10), in agreement with previous findings [15, 16]. Interestingly, however, expression of high Src527F expression (line Src-high) rather than increasing, dramatically decreased Stat3-ptyr705 to undetectable levels (lanes 11–15). A number of additional clones expressing graded Src527F levels gave similar results (Fig. S1, B, bottom panel). Levels of the prominent Stat3 target, survivin, mirrored the levels of Stat3-ptyr705 (Fig. S1, C).
A Balb/c3T3 cells grown to densities of a 75%, b 100%, c 100%+1 day, and d 100%+2 days, respectively, were photographed under phase contrast illumination. Bar: 100 µm. B Parental Balb/c3T3 (lanes 1–5), Src-med (lanes 6–10), and Src-high (lanes 11–15) cells were grown to densities of 50% to 2 days post-confluence. Detergent cell extracts were probed for SrcpY416, Cad11, Stat3-ptyr705, total Stat3, total Src, or β-actin as a loading control, as indicated. Numbers immediately under the lanes refer to band intensities of SrcpY416, Cad11, or Stat3-ptyr705 obtained through quantitation by image analysis and normalized to β-actin levels, with the highest values of each taken as 100%. Numbers at the left refer to molecular weight markers. Note the absence of Stat3-ptyr705 in Src-high cells (lanes 11–15), despite expression of the highest Src527F levels. C Cad11 is required for Stat3, ptyr705 phosphorylation by Src527F. Parental Balb/c3T3 (lanes 1–5), shCad11 (lanes 6–10), and shCad11-Src-med (lanes 11–15) cells were grown to the indicated densities. Detergent cell extracts were probed for Cad11 (A), SrcpY416 (B), Stat3-ptyr705, (C) or β-actin as a loading control. Numbers immediately under the lanes refer to band intensities obtained through quantitation by image analysis and normalized to β-actin levels. Numbers at the left refer to molecular weight markers.
Total Stat3 was slightly reduced with a reduction in Stat3-ptyr705, as expected from the fact that Stat3 normally activates its own promotor [23], while levels of total Cad11 or Src remained essentially unaffected by cell density. These results demonstrate that, although Src527F, expressed to levels of up to ~60% of the maximum, does increase Stat3-ptyr705, as previously established [15, 16], expressed to high levels Src527F eliminates Stat3-ptyr705. In fact, Stat3-ptyr705 was undetectable in Src-high cells, i.e., dramatically lower than in the parental Balb/c3T3.
Cadherin-11 is required for Stat3 activation by Src527F
The fact that the potent Stat3 activator Cad11 was reduced to undetectable levels in Src-high cells along with Stat3-ptyr705, raises the question of whether Cad11 might, in fact, be needed for Stat3 activation by Src. Therefore, to explore the Cad11 requirement for the Src527F-mediated Stat3-tyr705 phosphorylation, we assessed the ability of Src527F to activate Stat3 in Balb/c3T3 derivatives where Cad11 was knocked down through stable expression of shCad11 [12] (line shCad11). As shown in Fig. 2C, shCad11 cells expressed very low Cad11 levels (lanes 6–10). Src527F was subsequently expressed in shCad11 cells to medium levels (Fig. S1 A, bottom panel, and B top panel), and Stat3-ptyr705 levels were assessed at different densities. The results revealed, as expected, a distinct increase in Stat3-ptyr705 levels with density in the parental Balb/c3T3 cells (Fig. 2C, lanes 1–5). Downregulation of Cad11 resulted in a dramatic reduction in Stat3-ptyr705 levels, at all densities examined (lanes 6–10), consistent with previous data [12]. Interestingly, however, expression of Src527F in shCad11 cells did not trigger an increase in Stat3-ptyr705 levels (lanes 11–15), indicating that although Src527F can activate Stat3 in the parental Balb/c3T3 when expressed to medium levels (Fig. 2B, lanes 6–10), Src527F is unable to do so in the face of a Cad11 deficiency. Similarly, the converse experiment, ie expression of shCad11 in Src-med cells, showed reduced Stat3-ptyr705 levels (Fig. S2). Taken together, the above findings indicate that Cad11 is required for Stat3-tyr705 phosphorylation triggered by activated Src527F expression.
Src527F increases Rac protein levels and requires Rac for Stat3, ptyr705 phosphorylation
We previously demonstrated that Cadherin engagement in both epithelial cells (E-cadherin) [8] and fibroblasts (Cad11) [12] leads to a dramatic increase in Rac1 (Rac) and Cdc42 protein and activity levels, through inhibition of proteasomal degradation [24]. This triggers the transcriptional activation of the IL6 family of cytokines, hence the activation of Stat3 in an autocrine manner [22].
Since, as shown above, Cad11 is required for Stat3 activation by Src527F, we explored whether Rac might actually be involved in Stat3 activation by Src527F as well. At first, the effect of Src upon Rac protein levels was examined. Balb/c3T3, Src-med, and Src-high cells were plated at densities of 100% confluence or 100% plus one day and Rac levels assessed by Western blotting. As shown in Fig. 3A, Rac protein levels increased with cell density in all three lines [22]. Interestingly, Src527F expression triggered an increase in Rac protein levels, which was most prominent at high Src527F levels (Src-high, lanes 3–4 vs Balb/c3T3, lanes 1–2). Activated Rac-GTP mirrored the levels of Rac protein (lanes 7–9). Taken together, these data indicate that Src527F expression is able to increase Rac protein levels and activity. Moreover, this increase must be independent of any effects of Cad11, since in Src-high cells Cad11 is undetectable while Rac is the highest.
A Src527F expression increases Rac protein levels and activity. Detergent lysates from Balb/c3T3, Src-high, or Src-med cells were probed for Rac protein at 100% confluence or at 100% plus one day, as indicated. Parallel lysates were subjected to Rac activity assays (Rac-GTP) as previously described [8]. B Rac is required for Stat3, ptyr705 phosphorylation by Src527F. Parental Balb/c3T3 (lane 1), Src-med (lane 2), shRac (lane 3), and shRac-Src-med (lane 4) cells were grown to 2 days post confluence. Detergent cell extracts were probed for SrcpY416, total Src, Rac, Stat3-ptyr705, or β-actin as a loading control, as indicated. Numbers under the lanes refer to band intensities obtained through quantitation by image analysis and normalization to β-actin levels. Numbers at the left refer to molecular weight markers.
We next examined the Rac requirement for Stat3 activation by Src527F. Rac was downregulated through the expression of shRac with a retroviral vector (see Materials and Methods). As shown in Fig. 3B, shRac expression reduced Rac levels to 15% compared to the parental Balb/c3T3 (lane 3 vs 1). This Rac reduction resulted in a dramatic reduction in Stat3-ptyr705, to undetectable levels (lane 3 vs 1), in keeping with previous reports [11]. Interestingly, Src527F expression in shRac cells caused only a small (12%) increase in Stat3-ptyr705 (lane 4), compared to Src527F expression in the parental Balb/c3T3 (lane 2), revealing a Rac requirement for Stat3 activation by Src527F. It is possible that Src may be using Cdc42 as an alternate branch to transmit the signal to gp130/Stat3, which could explain the background level of Stat3-ptyr705 (12%) in shRac-Src-med cells (lane 4). Taken together, these findings indicate that Src527F increases Rac levels in Balb/c3T3 fibroblasts, by a mechanism that is independent of Cad11. In addition, Rac is, in fact, required for full Stat3, ptyr705 phosphorylation following Src527F expression, consistent with previous reports [25].
Secretion of Stat3-activating cytokines by high-Src527F-expressing cells
Rac was previously shown to trigger the secretion of IL6 family cytokines, through a mechanism requiring NFκB [11]. Since Rac levels are increased upon Src527F expression, we assessed the secretion of cytokines by cells expressing different Src527F levels. Balb/c3T3, Src-med, or Src-high cells were grown to high densities and conditioned medium collected 24 h later. This medium was added to Balb/c3T3 cells grown to a low density (50% of confluence) and Stat3-ptyr705 levels were assessed 30 min later by Western blotting (Fig. 4A). As shown in Fig. 4B, Src-med-conditioned medium stimulated Stat3-ptyr705 expression in sparsely growing, Balb/c3T3 cells, substantially more than medium conditioned by the parental Balb/c3T3 (lane 3 vs 2). These results indicate that Src-med cells may secrete high amounts of cytokines, which, acting in an autocrine manner, may induce the high Stat3-ptyr705 levels observed in Src-med cells, as shown in Fig. 2B (lanes 6–10). Interestingly, however, medium conditioned by Src-high cells also increased Stat3-ptyr705 in sparsely growing Balb/c3T3 cells, pointing to the secretion of high amounts of cytokines (lane 4), despite the fact that their own Stat3-ptyr705 levels were undetectable (Fig. 2B, lanes 11–15). Examination of IL6 levels in a conditioned medium by ELISA testing confirmed high secretion by Src-high cells (Fig. S4). Therefore, Src-high cells, although they do secrete cytokines that are able to activate Stat3 in sparsely growing, Balb/c3T3 cells, Src-high cells themselves are not able to respond to the cytokines they secrete, that is, the block to Stat3 activation by high-Src527F must be at a point downstream from cytokine secretion.
A Flow-sheet: medium conditioned by confluent Balb/c3T3, Src-med or Src-high cells (3mls serum-free DMEM medium/6 cm petri, 24 h) was collected, filtered, and added to sparsely growing, Balb/c3T3 cells for 30 min. These cells were subsequently lysed and Stat3-ptyr705 examined by western blotting. B Src-high cells do secrete Stat3-activating cytokines. Growth medium conditioned by Src-med (lanes 3, 7) or Src-high (lanes 4, 8) cells or the parental Balb/c3T3 (lanes 2, 6), was added to Balb/c3T3 (lanes 1–4), or shCad11 (lanes 5–8) cells growing to 50% confluence. Cells were subsequently lysed and Stat3-ptyr705 or Cad11 levels determined by Western blotting, with tubulin as a loading control. Note the dramatic increase in Stat3-ptyr705 upon treatment of Balb/c3T3 cells with medium conditioned by Src-med or Src-high cells (lanes 3 and 4 vs 2) and the absence of Stat3-ptyr705 increase by the same conditioned medium in shCad11 cells (lanes 7–8 vs 3, 4).
To explore the possibility that the low Cad11 levels in Src-high cells (Fig. 2B, lanes 11–15) may be responsible for the absence of response to the conditioned medium, the experiment was repeated by stimulating Cad11-deficient, shCad11 cells with medium conditioned by the three lines above, in a similar manner. As shown in Fig. 4B (lanes 5–8), shCad11 cells did not display any increase in Stat3-ptyr705 upon stimulation with medium conditioned by Src-med or Src-high cells. Taken together, the above data indicate that Src527F-expressing cells secrete cytokines that can activate their receptor, hence Stat3, but only under conditions where Cad11 is present.
High Src527F expression or Cadherin-11 knockdown results in gp130 and Jak-p1022/1023 downregulation
Cytokines of the IL6 family normally activate Stat3 following attachment to their receptor. This receptor consists of two subunits, an extracellular one which is responsible for ligand binding and is specific for each member of the family, and a common intracellular subunit, gp130. Following ligand binding, the Jak kinase is recruited to the receptor, and this is necessary for Stat3 activation [26]. Since Src-med and Src-high cells do secrete high amounts of IL6 family cytokines, we examined the levels of gp130 in these cells by Western blotting. As shown in Fig. 5A, expression of medium Src527F levels substantially reduced gp130 protein levels (lanes 5–8 vs 1–4), while high Src expression levels eliminated gp130 altogether (lanes 9–12), concomitant with the elimination of Stat3-ptyr705. Interestingly, Stat3-ptyr705 levels were higher in Src-med cells (lanes 5–8) than Balb/c3T3, despite their lower gp130 levels, presumably due to higher Rac and IL6 family cytokines secreted by Src-med than the parental Balb/c3T3. However, high Src527F levels that eliminated Cad11 and gp130, eliminated Stat3-ptyr705 as well (lanes 9–12). Levels of Jak1-p1022/1023 mirrored levels of gp130 and Stat3 (Fig. S3) in all three lines.
A High Src527F expression results in gp130 downregulation. Balb/c3T3 (lanes 1–4), Src-med (lanes 5–8), or Src-high (lanes 9–12) cells were grown to the indicated densities and detergent extracts probed for Cad11, gp130, Stat3-ptyr705, or tubulin as a loading control, as indicated. B Cadherin-11 knockdown results in gp130 downregulation. Balb/c3T3 (lanes 1–4) or shCad11 (lanes 5–8) cells were grown to the indicated densities and detergent extracts probed for gp130 or tubulin, as indicated. C Dasatinib treatment of Src-high cells restores gp130 and Stat3-ptyr705 levels Src-med (lanes 1–5) or Src-high (lanes 6–10) cells grown to a density of one day after confluence were untreated (lanes 1 and 6), treated with 5 µM (lanes 3, 8) or 7.5 µM (lanes 5, 10) Dasatinib or the corresponding amounts of the DMSO carrier alone, as indicated. Detergent extracts were subsequently probed for Src-ptyr416, Stat3-ptyr705, gp130, or tubulin as a loading control, as indicated.
To examine whether the absence of gp130 in Src-high cells could, in fact, be attributed to the absence of Cad11, we examined gp130 levels in shCad11 cells. As shown in Fig. 5B, gp130 was undetectable in shCad11 cells (lanes 5–8), pointing to the possibility that Cad11 is, in fact, required for the integrity of the gp130 receptor. Similar results were obtained with rat-F111 fibroblasts expressing the middle Tumor Antigen of polyomavirus [mT [18]], which induces transformation by binding to and activating c-Src (Fig. S7). Taken together, the above data reveal that reducing the levels of Cad11 either through the expression of high Src527F levels or shRNA, results in downregulation of the gp130 receptor and this prevents Stat3 phosphorylation by Src527F/Jak1. It is possible that the role of Cad11 is to stabilize the gp130 receptor at the membrane, in a manner reminiscent of the E-cadherin requirement for proper activation of the gp130 receptor in mouse embryonic stem cells [27].
Dasatinib treatment of Src-high cells restores gp130 and Stat3-ptyr705 levels
Despite the fact that Src has been found to be hyperactive in a number of cancers, Src inhibitors such as Dasatinib were found to be largely ineffective in clinical trials [28, 29]. It is certainly possible that other, Src-independent pathways were active in these tumors. However, a fundamental inability of Src inhibitors to reduce Stat3 activity in cells expressing high Src activity cannot be excluded. To explore the possibility that an increase (rather than decrease) in Stat3 activity upon Src inhibition might explain this apparent paradox, Src-med and Src-high cells were treated with the inhibitor Dasatinib or the DMSO carrier and detergent lysates probed for gp130 and Stat3-ptyr705. As shown in Fig. 5C, Dasatinib treatment of Src-med cells caused a slight increase in gp130 (lanes 3 and 5 vs 2 and 4) above the DMSO controls. Interestingly, however, treatment of Src-high cells with Dasatinib caused a prominent gp130 increase, which translated into a robust increase in Stat3-ptyr705 (lanes 8 and 10 vs 7 and 9). These results confirm that expressed to high levels, Src actually reduces gp130 and Stat3 activity. Thus, the exact level of Src expression in a given cancer could offer a potential explanation for the inconsistent results of Dasatinib or other Src inhibitors in clinical trials; treatment of high-Src cancers would actually increase Stat3 activity, leading to metastasis.
Cadherin-11 and Rac are required for survival of Src527F-expressing cells
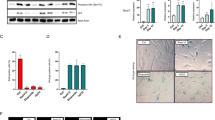
Previous results have shown that Stat3 induces anti-apoptotic genes such as bcl-xL, mcl-1, and survivin [4, 30, 31], it downregulates the p53 promoter [3], while it can affect the cellular metabolism through its ser727-phosphorylated form, in a way that it protects tumor cells from apoptosis further [32,33,34]. On the other hand, Src527F, besides Stat3, is known to activate the E2F transcription factor family, which actually promotes apoptosis, through the Ras/Raf/Erk pathway [35]. Therefore, the question arises as to the net effect of Src527F expression upon apoptosis vs survival, which could be different depending upon Src activity levels. To answer this question, cells were grown to 1 day post-confluence and apoptosis examined by TUNEL staining and analysis of poly (ADP-ribose) polymerase (PARP) cleavage by Western blotting. As shown in Fig. 6B, Src-med cells displayed a very low level of apoptosis compared to the parental Balb/c3T3 (Fig. 6B, b vs d), suggesting that the pro-apoptotic effect of E2F activation by Src527F may be prevailing over the anti-apoptotic effect of Stat3 (Fig. 7).
A Parental Balb/c3T3 (lane 1), Src-med (lane 2), shCad11 (lane 3), and shCad11-Src-med (lane 4) cells were grown to 1 day post-confluence. Detergent cell extracts were probed for SrcpY416, Cad11, Stat3-ptyr705, cleaved PARP, total Src or β-actin as a loading control, as indicated. Numbers under the lanes of the upper panels refer to band intensities obtained through quantitation by image analysis and normalized to β-actin levels, with the peak values of Src-med (lane 2) for SrcpY416 and Stat3-ptyr705, Balb/c3T3 (lane 1) for Cad11, and shCad11-Src (lane 4) for cleaved PARP taken as 100%, respectively. Numbers at the left refer to molecular weight markers. B Balb/c3T3 (a, b), Src-med (c, d), shCad11 (e, f), and shCad11-Src-med (g, h) cells were grown to 1 day post confluence and apoptosis examined by TUNEL staining (see Materials and Methods). Cells were photographed under phase contrast and fluorescence illumination. Bar: 100 µm.
A Medium Src activity levels: activated Src increases Rac levels and activity in a Cad11-independent manner. This leads to transcriptional upregulation of IL6 family cytokines, followed by activation of the gp130 receptor subunit, then Jak and Stat3. Although Src at the same time downregulates Cad11, which is required for gp130 function, the residual gp130 levels are sufficient to allow Stat3, ptyr705 phosphorylation, and activation. At the same time, Src527F, through the Ras/Raf/Erk/Rb pathway activates the transcription factor E2F which leads to apoptosis. However, Stat3 inhibits the apoptosis triggered by high E2F activity, with cell proliferation as a result. B High Src activity levels: paradoxically perhaps, upon high Src527F expression (or upon genetic downregulation of Cad11), Cad11 levels are reduced to non-detectable and this leads to gp130 downregulation, with a dramatic reduction in Jak and Stat3 activity, so that cells succumb to apoptosis as a result. Engagement of Cad11 also increases Rac protein levels and activity, independent of Src [8].
We next examined the role of Cad11 in the survival of Src527F-expressing cells; as shown in Fig. 6A, shCad11 cells had a notably higher level of cleaved PARP compared to Balb/c3T3 (Fig. 6A, lanes 3 vs 1) and higher TUNEL staining (Fig. 6B, panels f vs b), indicating that knockdown of Cad11 reduces Stat3-ptyr705 levels and leads to apoptosis. Interestingly, expression of Src527F in shCad11 cells triggered a further increase in cleaved PARP (Fig. 6A lane 4 vs 3) and TUNEL staining (Fig. 6B, panels h vs f). Similar results were obtained when the sequence of Src527F and shCad11 expression was reversed, i.e. with Src-med vs Src-med-shCad11 cells (Supplementary data, Fig. S5). Taken together, these results indicate that Cad11 deficiency triggers apoptosis in Src527F cells, that is, the Cad11/Stat3 axis is a crucial determinant of survival of Src527F-transformed mouse fibroblasts. These findings may explain the inability to express high Src527F levels in shCad11 cells (Fig. S1 A bottom panel) since high Src527F levels would promote apoptosis in shCad11 cells, presumably due to high E2F levels combined with very low Stat3-ptyr705. As expected, Src527F expression triggered apoptosis in Rac-deficient, shRac cells as well (Fig. S6). Taken together, our results point to the Cad11/Rac/Stat3 axis as a crucial signaling pathway for Src-transformed cell survival.
Discussion
Cadherin-11 is abundant in osteoblasts, and it has been demonstrated to promote metastasis primarily to the bone, at least in breast [36] and prostate [37] cancer [38, 39]. Furthermore, high Cad11 expression is associated with invasive human breast cancer [40] and poor overall survival in gastric cancer [41]. Such tumors invariably express high levels of activated Src or other oncogenes to drive the transformed phenotype [9]. Interestingly, our results now indicate that Src expression per se leads to a dramatic reduction or elimination of Cad11, the main cadherin expressed in Balb/c3T3 fibroblasts and that higher Src levels, rather than increase as expected, actually reduce, or eliminate Stat3-ptyr705 and activity. This apparent paradox raises the question of the actual role of Cad11 in the Src-induced Stat3 activation and metastasis. Therefore, we attempted to explore the following questions:
-
1.
How can Cad11 promote the metastasis of tumor cells, where the Src oncogene downregulates or eliminates Cad11?
-
2.
What is the role of Cad11 in Stat3 activation by Src?
-
3.
What is the biological role of Cad11 in the survival of Src527F-expressing cells?
These are critical questions regarding Cad11/Stat3 in Src Biology and neoplasia.
Activated Src527F expression downregulates Cadherin-11 in a quantitative manner
Expression of activated Src was previously shown to disrupt E-cadherin engagement and the cadherin/catenin complex in both tumor cells and transformed fibroblasts [42]. This negative regulation of E-cadherin occurs through several different mechanisms, including alterations in E-cadherin turnover and expression [reviewed in [21]]. Our results now reveal that Cad11 also is negatively regulated by Src. Furthermore, by stably expressing different levels of activated Src527F in Balb/c3T3 cells, we demonstrated a quantitative negative effect of Src upon Cad11 protein levels; low Src527F expression, ~21% of the highest achieved, reduced Cad11 to 75% of Balb/c3T3, medium Src expression, ~67% of the highest, reduced Cad11 to 42%, while high Src levels eliminated Cad11 entirely.
The quantitative effect of Src upon Cad11 may be a result of threshold effects of some or all of the different effectors causing Cad11 downregulation. The exact mechanism is currently under investigation. Since Balb/c3T3 cells, in addition to Cad11, also express N-cadherin [12], which also activates Stat3, the apparent absence of Stat3-ptyr705 in Src-high cells may be an indication that N-cadherin as well is downregulated by Src. In any event, these findings broaden the scope of known Src effectors to include mesenchymal cadherins.
Since high Src levels were found to completely eliminate Cad11, and Cad11 is required for metastasis [37, 43, 44], it appears that actual metastatic tumors cannot possibly possess such high levels of Src activity; that is, a fine balance between Src and Cad11 levels may be required for the formation of a metastatic tumor, as observed in medium-Src-expressing cells, i.e. this level of Src activity may be more relevant to cancer in vivo.
Role of Cadherin-11 in gp130 signaling and Stat3 activation by Src527F
Previous reports have extensively documented that Src activates Stat3 [15, 16]. Our results now further demonstrate that progressively higher Src527F expression in individual clones, up to ~60% of the highest levels achieved, results in proportionally higher Stat3-ptyr705 levels, as expected. Interestingly, however, expression of Src527F levels above 90% of the highest (where Cad11 was eliminated), rather than resulting in even higher Stat3-ptyr705 levels, was found for the first time to eliminate Stat3-ptyr705 entirely. The fact that Cad11 downregulation indeed prevented Stat3, ptyr705 phosphorylation by Src527F (Fig. 2C), indicates that Src527F requires Cad11 to achieve Stat3-ptyr705 phosphorylation, despite the fact that Src527F downregulates Cad11 at the same time. This is further reinforced by the fact that Dasatinib treatment of Src-high cells, rather than reducing as expected, dramatically increased Stat3-ptyr705 levels (Fig. 5C). These results also offer an explanation for the slight increase in Stat3 activity previously observed upon Src downregulation [45].
What is the role of Cad11? It was previously reported that Src requires Rac function for Stat3 activation, while Src activates Rac through the exchange factors, Tiam and Vav2 [25]. Our results now further demonstrate that Src527F (in proportion to its levels) increases Rac protein levels as well. Interestingly, we previously demonstrated that Rac, upregulated either through cadherin engagement [8, 10, 12], or mutational activation (RacV12) increases transcription of IL6 family cytokines which stimulate the gp130 receptor in an autocrine manner, leading to an increase in activity of Jak and Stat3 [11]. Since the Rac increase is the highest in the Src-high cells (Fig. 3A), when Cad11 is absent, the Src527F-stimulated, Rac increase must occur in a Cad11-independent manner. Our results further show that in the absence of Cad11, either because of shCad11 knockdown, or high-Src expression, gp130 levels are undetectable. That is, Cad11 is required to maintain sufficiently high gp130 levels to allow signaling from Rac to gp130, then through Jak to Stat3. This is reminiscent of a previous report [27] indicating that E-cadherin is required for proper activation of the IL6/gp130 signaling pathway in mouse Embryonal Stem cells. Therefore, it appears that at intermediate Src527F levels, the residual Cad11 levels are sufficient to maintain gp130, while the total absence of Cad11 in Src-high cells results in the downregulation or degradation of gp130, hence the absence of Jak and Stat3 activation. The fact that Src cannot fully activate Stat3 in gp130 KD cells as previously demonstrated [46], further reinforces the importance of gp130. That is, a fine balance between Cad11/gp130 and Src/Rac is apparently required to induce IL6 family secretion, and preserve sufficient gp130 levels to allow signaling through the Src/Rac/IL6/gp130/Jak/Stat3 axis (Fig. 7, A vs B).
Our results are consistent with earlier findings indicating that activated Src requires Jak1 function to activate Stat3, aided by receptors such as PDGFR, acting as a scaffold [47]. Since in Src-high cells Stat3-ptyr705 is undetectable, it is possible that Cad11 is required for the function of other receptors such as PDGFR as well, even when they merely have a scaffolding role in Jak/Stat3 activation. It is also possible that other cadherins, such as N-cadherin which is also present in Balb/c3T3 cells [12], can function in a manner similar to Cad11 in preserving receptor function. Experiments are underway to resolve these possibilities.
Interestingly, two other signal transducers known to be activated by IL6, Erk, and Akt are not affected by cell density [12, 22]. We recently demonstrated that at low densities, engagement of integrins with the focal adhesion kinase (FAK) activates the FAK/Src complex, and this results in activation of tyrosine-kinase receptors, Erk1/2 and Akt [48]. Interestingly, the FAK/Src complex cannot activate Stat3 in the absence of Cad11 engagement (i.e., in sparsely growing cells), possibly required to maintain gp130 function.
Role of the Cad11/Stat3 axis in Src527F-induced apoptosis
Since the initial discovery that Stat3 is required for transformation by activated Src [15, 16], it has become evident that Stat3 inhibition in Src-transformed cells induces apoptosis, not simply reversion of the cell to a normal phenotype. This observation provided the first clue as to the important role of Stat3 in the survival of Src-transformed cells.
A variety of receptor or non-receptor tyrosine-kinase oncogenes, trigger the activation of the E2F family of transcription factors (the “activating” E2Fs, E2F1-3a [49]), which was shown to occur in the majority of tumors. E2F activation occurs through phosphorylation and inactivation of the Rb (retinoblastoma susceptibility) family of nuclear phosphoproteins. E2F targets include genes involved in DNA synthesis as well as growth factor and receptor genes [50]. Interestingly, at the same time, E2F activates apoptosis through both p53-dependent and independent pathways [[51, 52]; reviewed in [53, 54]]. Certainly, apoptosis is normally prevented due to the activation of survival factors such as PI3k or Stat3 by tyrosine-kinase receptors induced by E2F itself, or directly by Src or other kinases which had activated E2F in the first place, so that transformation does occur. In this schema, upon inhibition of Stat3 activity, tumor cells, having high E2F levels, may succumb to apoptosis [55]. Most importantly, inhibition of Cad11 would induce apoptosis (through Stat3 inhibition) in metastatic tumor cells specifically, since normal cells would have low E2F activity, hence would be spared (Fig. 7). In fact, our results indicated that Cad11-deficient cells succumbed to apoptosis upon Src527F expression; levels of cleaved PARP and TUNEL staining were higher in shCad11-Src-med cells than in Src-med cells which express similar levels of Src527F.
Conclusion
Targeting Cad11 in cancer
The observation that in cultured cells, and potentially in vivo, high Src levels downregulate Cad11, which, in turn, may be required for metastasis, especially to the bone, may signify that actual metastatic tumors cannot possibly express such high Src levels as to eliminate Cad11. Apparently, a fine balance between Src and Cad11 levels is required, as to allow both transformation (through Src) and metastasis (through Cad11), for the formation of a metastatic tumor to occur. As a result, pharmacological inhibition of Src would increase Cad11/gp130/Stat3 levels in tumors with high Src activity, hence increase, rather than decrease, the metastatic ability of the tumor, leading to a deterioration in clinical outcome. The poor performance of Src inhibitors including Dasatinib in clinical trials is consistent with these findings [56,57,58,59].
Our results also stress for the first time the importance of Cad11, Rac, and IL6/gp130 signaling in Src-triggered induction of Stat3, tyr705 phosphorylation, and activation. As such, Cad11 and likely other cadherins are emerging as not only powerful Stat3 activators, but also as critical mediators of Stat3 signaling from Src and possibly other oncogenes, despite the fact that, at the same time, Src downregulates cadherins. Conversely, downregulation of Stat3-ptyr705 by caveolin-1, the main protein of caveolae, has been shown to be mediated through the downregulation of Cad11 [20]. Further investigations will clarify the role of the Cad11/Rac/gp130 axis, to unearth druggable targets. Being on the cell surface, Cad11 should be especially accessible.
Data availability
Unfortunately, the corresponding author recently retired from her position as professor and had to clean out and close down her laboratory. Because they did not anticipate this request they did not keep any materials. They do have the original scans of western blotting experiments performed by the first author, i.e., Figs. 3A, 4, and 5, using Azure imaging technology. Unfortunately, the rest of the western blots were done using film and by co-authors that have left the laboratory several years ago, when this was not a requirement.
References
Ma JH, Qin L, Li X. Role of STAT3 signaling pathway in breast cancer. Cell Commun Signal. 2020;18:33.
Johnson DE, O’Keefe RA, Grandis JR. Targeting the IL-6/JAK/STAT3 signalling axis in cancer. Nat Rev Clin Oncol. 2018;15:234–48.
Niu G, Wright KL, Ma Y, Wright GM, Huang M, Irby R, et al. Role of Stat3 in regulating p53 expression and function. Mol Cell Biol. 2005;25:7432–40.
Gritsko T, Williams A, Turkson J, Kaneko S, Bowman T, Huang M, et al. Persistent activation of stat3 signaling induces survivin gene expression and confers resistance to apoptosis in human breast cancer cells. Clin Cancer Res. 2006;12:11–9.
Yu H, Pardoll D, Jove R. STATs in cancer inflammation and immunity: a leading role for STAT3. Nat Rev Cancer. 2009;9:798–809.
Bharadwaj U, Kasembeli MM, Robinson P, Tweardy DJ. Targeting janus kinases and signal transducer and activator of transcription 3 to treat inflammation, fibrosis, and cancer: rationale, progress, and caution. Pharm Rev. 2020;72:486–526.
Bromberg JF, Wrzeszczynska MH, Devgan G, Zhao Y, Pestell RG, Albanese C, et al. Stat3 as an oncogene. Cell 1999;98:295–303.
Arulanandam R, Vultur A, Cao J, Carefoot E, Elliott BE, Truesdell PF, et al. Cadherin-cadherin engagement promotes cell survival via Rac1/Cdc42 and signal transducer and activator of transcription-3. Mol Cancer Res. 2009;7:1310–27.
Vultur A, Cao J, Arulanandam R, Turkson J, Jove R, Greer P, et al. Cell-to-cell adhesion modulates Stat3 activity in normal and breast carcinoma cells. Oncogene 2004;23:2600–16.
Raptis L, Arulanandam R, Vultur A, Geletu M, Chevalier S, Feracci H. Beyond structure, to survival: Stat3 activation by cadherin engagement. Biochem Cell Biol. 2009;87:835–43.
Arulanandam R, Geletu M, Feracci H, Raptis L. Activated Rac1 requires gp130 for Stat3 activation, cell proliferation and migration. Exp Cell Res. 2010;316:875–86.
Geletu M, Arulanandam R, Chevalier S, Saez B, Larue L, Feracci H, et al. Classical cadherins control survival through the gp130/Stat3 axis. BBA-Molecular. Cell Res. 2013;1833:1947–59.
Espada J, Martin-Perez J. An update on Src family of nonreceptor tyrosine kinases biology. Int Rev Cell Mol Biol. 2017;331:83–122.
Elsberger B, Tan BA, Mitchell TJ, Brown SB, Mallon EA, Tovey SM, et al. Is expression or activation of Src kinase associated with cancer-specific survival in ER-, PR- and HER2-negative breast cancer patients? Am J Pathol. 2009;175:1389–97.
Turkson J, Bowman T, Garcia R, Caldenhoven E, De Groot RP, Jove R. Stat3 activation by Src induces specific gene regulation and is required for cell transformation. Mol Cell Biol. 1998;18:2545–52.
Bromberg JF, Horvath CM, Besser D, Lathem WW, Darnell JE Jr. Stat3 activation is required for cellular transformation by v-src. Mol Cell Biol. 1998;18:2553–8.
Orlandini M, Oliviero S. In fibroblasts Vegf-D expression is induced by cell-cell contact mediated by cadherin-11. J Biol Chem. 2001;276:6576–81.
Raptis L, Lamfrom H, Benjamin TL. Regulation of cellular phenotype and expression of polyomavirus middle T antigen in rat fibroblasts. Mol Cell Biol. 1985;5:2476–86.
van Kuppeveld FJ, Johansson KE, Galama JM, Kissing J, Bolske G, van der Logt JT, et al. Detection of mycoplasma contamination in cell cultures by a mycoplasma group-specific PCR. Appl Environ Microbiol. 1994;60:149–52.
Geletu M, Mohan R, Arulanandam R, Berger-Becvar A, Nabi IR, Gunning PT, et al. Reciprocal regulation of the Cadherin-11/Stat3 axis by caveolin-1 in mouse fibroblasts and lung carcinoma cells. Biochim Biophys Acta. 2018;1865:794–802.
Wadhawan A, Smith C, Nicholson RI, Barrett-Lee P, Hiscox S. Src-mediated regulation of homotypic cell adhesion: implications for cancer progression and opportunities for therapeutic intervention. Cancer Treat Rev. 2011;37:234–41.
Geletu M, Guy S, Arulanandam R, Feracci H, Raptis L. Engaged for survival; from cadherin ligation to Stat3 activation. Jaks-Stat 2013;2:e27363.
Narimatsu M, Maeda H, Itoh S, Atsumi T, Ohtani T, Nishida K, et al. Tissue-specific autoregulation of the stat3 gene and its role in interleukin-6-induced survival signals in T cells. Mol Cell Biol. 2001;21:6615–25.
Lynch EA, Stall J, Schmidt G, Chavrier P, D’Souza-Schorey C. Proteasome-mediated degradation of Rac1-GTP during epithelial cell scattering. Mol Biol Cell. 2006;17:2236–42.
Servitja JM, Marinissen MJ, Sodhi A, Bustelo XR, Gutkind JS. Rac1 function is required for Src-induced transformation. Evidence of a role for Tiam1 and Vav2 in Rac activation by Src. J Biol Chem. 2003;278:34339–46.
Rose-John S. Interleukin-6 signalling in health and disease. F1000Res. 2020;9.
del Valle I, Rudloff S, Carles A, Li Y, Liszewska E, Vogt R, et al. E-cadherin is required for the proper activation of the Lifr/Gp130 signaling pathway in mouse embryonic stem cells. Development 2013;140:1684–92.
Creedon H, Brunton VG. Src kinase inhibitors: promising cancer therapeutics? Crit Rev Oncog. 2012;17:145–59.
Zhang S, Yu D. Targeting Src family kinases in anti-cancer therapies: turning promise into triumph. Trends Pharm Sci. 2012;33:122–8.
Grandis JR, Drenning SD, Zeng Q, Watkins SC, Melhem MF, Endo S, et al. Constitutive activation of Stat3 signaling abrogates apoptosis in squamous cell carcinogenesis in vivo. Proc Nat Acad Sci USA. 2000;97:4227–32.
Epling-Burnette PK, Liu JH, Catlett-Falcone R, Turkson J, Oshiro M, Kothapalli R, et al. Inhibition of STAT3 signaling leads to apoptosis of leukemic large granular lymphocytes and decreased Mcl-1 expression. J Clin Invest. 2001;107:351–62.
Wegrzyn J, Potla R, Chwae YJ, Sepuri NB, Zhang Q, Koeck T, et al. Function of mitochondrial Stat3 in cellular respiration. Science 2009;323:793–7.
Gough DJ, Corlett A, Schlessinger K, Wegrzyn J, Larner AC, Levy DE. Mitochondrial STAT3 supports Ras-dependent oncogenic transformation. Science 2009;324:1713–6.
Demaria M, Poli V. From the nucleus to the mitochondria and back: the odyssey of a multitask STAT3. Cell Cycle. 2011;10:3221–2.
Sears RC, Nevins JR. Signaling networks that link cell proliferation and cell fate. J Biol Chem. 2002;277:11617–20.
Feltes CM, Kudo A, Blaschuk O, Byers SW. An alternatively spliced cadherin-11 enhances human breast cancer cell invasion. Cancer Res. 2002;62:6688–97.
Chu K, Cheng CJ, Ye X, Lee YC, Zurita AJ, Chen DT, et al. Cadherin-11 promotes the metastasis of prostate cancer cells to bone. Mol Cancer Res. 2008;6:1259–67.
Carmona FJ, Villanueva A, Vidal A, Munoz C, Puertas S, Penin RM, et al. Epigenetic disruption of cadherin-11 in human cancer metastasis. J Pathol. 2012;228:230–40.
Niit M, Hoskin V, Geletu M, Arulanandam R, BE E, Raptis L. Cell-cell and cell-matrix adhesion in survival and metastasis: Stat3 vs Akt. Biomolecular Concepts. 2015;6:383–99.
Pohlodek K, Tan YY, Singer CF, Gschwantler-Kaulich D. Cadherin-11 expression is upregulated in invasive human breast cancer. Oncol Lett. 2016;12:4393–8.
Wang Q, Jia Y, Peng X, Li C. Clinical and prognostic association of oncogene cadherin 11 in gastric cancer. Oncol Lett. 2020;19:4011–23.
Irby RB, Yeatman TJ. Increased Src activity disrupts cadherin/catenin-mediated homotypic adhesion in human colon cancer and transformed rodent cells. Cancer Res. 2002;62:2669–74.
Pishvaian MJ, Feltes CM, Thompson P, Bussemakers MJ, Schalken JA, Byers SW. Cadherin-11 is expressed in invasive breast cancer cell lines. Cancer Res. 1999;59:947–52.
Tamura D, Hiraga T, Myoui A, Yoshikawa H, Yoneda T. Cadherin-11-mediated interactions with bone marrow stromal/osteoblastic cells support selective colonization of breast cancer cells in bone. Int J Oncol. 2008;33:17–24.
Garcia R, Bowman TL, Niu G, Yu H, Minton S, Muro-Cacho CA, et al. Constitutive activation of Stat3 by the Src and JAK tyrosine kinases participates in growth regulation of human breast carcinoma cells. Oncogene 2001;20:2499–513.
D’Abreo C. Role of Cadherin-11 and gp130 in transformation by activated Src. MSc thesis, Queen’s University. 2011.
Zhang Y, Turkson J, Carter-Su C, Smithgall T, Levitzki A, Kraker A, et al. Activation of Stat3 in v-Src transformed fibroblasts requires cooperation of Jak1 kinase activity. J Biol Chem. 2000;275:24935–44.
Geletu M, Adan H, Niit M, Arulanandam R, Carefoot E, Hoskin V, et al. Modulation of Akt vs Stat3 activity by the focal adhesion kinase in non-neoplastic mouse fibroblasts. Exp Cell Res. 2021;404:112601.
Wong JV, Dong P, Nevins JR, Mathey-Prevot B, You L. Network calisthenics: control of E2F dynamics in cell cycle entry. Cell Cycle. 2011;10:3086–94.
Young AP, Nagarajan R, Longmore GD. Mechanisms of transcriptional regulation by Rb-E2F segregate by biological pathway. Oncogene 2003;22:7209–17.
Tsai KY, Hu Y, Macleod KF, Crowley D, Yamasaki L, Jacks T. Mutation of E2f-1 suppresses apoptosis and inappropriate S phase entry and extends survival of Rb-deficient mouse embryos. Mol Cell. 1998;2:293–304.
Moroni MC, Hickman ES, Lazzerini Denchi E, Caprara G, Colli E, Cecconi F, et al. Apaf-1 is a transcriptional target for E2F and p53. Nat Cell Biol. 2001;3:552–8.
Leone G, DeGregori J, Sears R, Jakoi L, Nevins JR. Myc and Ras collaborate in inducing accumulation of active cyclin E/Cdk2 and E2F. Nature 1997;387:422–6.
Irwin M, Marin MC, Phillips AC, Seelan RS, Smith DI, Liu W, et al. Role for the p53 homologue p73 in E2F-1-induced apoptosis. Nature 2000;407:645–8.
Vultur A, Arulanandam R, Turkson J, Niu G, Jove R, Raptis L. Stat3 is required for full neoplastic transformation by the Simian Virus 40 large tumor antigen. Mol Biol Cell. 2005;16:3832–46.
Herold CI, Chadaram V, Peterson BL, Marcom PK, Hopkins J, Kimmick GG, et al. Phase II trial of dasatinib in patients with metastatic breast cancer using real-time pharmacodynamic tissue biomarkers of Src inhibition to escalate dosing. Clin Cancer Res. 2011;17:6061–70.
Finn RS, Bengala C, Ibrahim N, Roche H, Sparano J, Strauss LC, et al. Dasatinib as a single agent in triple-negative breast cancer: results of an open-label phase 2 study. Clin Cancer Res. 2011;17:6905–13.
Mayer EL, Baurain JF, Sparano J, Strauss L, Campone M, Fumoleau P, et al. A phase 2 trial of dasatinib in patients with advanced HER2-positive and/or hormone receptor-positive breast cancer. Clin Cancer Res. 2011;17:6897–904.
Tian J, Raffa FA, Dai M, Moamer A, Khadang B, Hachim IY, et al. Dasatinib sensitises triple negative breast cancer cells to chemotherapy by targeting breast cancer stem cells. Br J Cancer. 2018;119:1495–507.
Acknowledgements
The authors are grateful to Dr. Andrew Craig of Queen’s University for many valuable reagents and advice, and Dr. Bruce Elliott for a critical reading of the manuscript and numerous suggestions. This work constituted part of the requirements for the M.Sc. degree for H.A. and Ph.D. degree for S.G., respectively. The financial assistance of the Natural Sciences and Engineering Research Council of Canada (NSERC), the Canadian Institutes of Health Research (CIHR), the Canadian Breast Cancer Foundation (CBCF, Ontario Chapter), and the Canadian Breast Cancer Research Alliance through grants to L.R. is gratefully acknowledged. H.A. was supported by a Canada Graduate Scholarships Masters award and a Canada Graduate Scholarships Doctoral Award from NSERC and an Ontario Graduate Scholarship. S.G. was supported by Alexander Graham Bell Canada Graduate Scholarships from NSERC and Queen’s Graduate Awards. M.G. was supported by postdoctoral fellowships from the US Army Breast Cancer Program, the Ministry of Research and Innovation of the Province of Ontario, and the Advisory Research Committee of Queen’s University. R.A. was supported by a Canada Graduate Scholarships Doctoral Award from CIHR, the Ontario Women’s Health Scholars Award from the Ontario Council on Graduate Studies, and a Queen’s graduate award. J.D. is supported by funding from CIHR and NSERC.
Author information
Authors and Affiliations
Contributions
H.A. performed most of the experiments and had original ideas. S.G. performed some of the experiments regarding Src expression. R.A. performed mostly experiments regarding the role of Rac. M.G. performed experiments regarding shCad11 and Src. J.D. co-supervised the work of the first author, offered advice and resources. L.R. conceived of the ideas, demonstrated key experiments, wrote the manuscript and supervised throughout.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Adan, H., Guy, S., Arulanandam, R. et al. Activated Src requires Cadherin-11, Rac, and gp130 for Stat3 activation and survival of mouse Balb/c3T3 fibroblasts. Cancer Gene Ther 29, 1502–1513 (2022). https://doi.org/10.1038/s41417-022-00462-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41417-022-00462-5