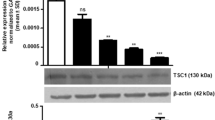
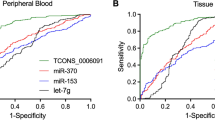
Abstract
microRNAs (miRNAs) have been revealed to participate in some oral cancers and are proved to be effective. In the present study, we tried to explore the biological function of miR-133a in oral squamous cell carcinoma (OSCC) cells. The relationship that C-terminal-binding proteins 2 (CTBP2) was the putative target gene of miR-133a revealed from bioinformatics analysis was further was further validated by dual-luciferase reporter gene assay. In total, 40 patients with OSCC were enrolled for characterization of miR-133a, CTBP2, and Notch signaling pathway-related gene expression in clinical OSCC tissues. Low expression of miR-133a and high expression of CTBP2, Hes1, Notch-1, and Notch-3 were determined in OSCC tissues. OSCC cell lines were transfected with miR-133a inhibitor, miR-133a mimic, or shRNA targeting CTBP2, in response to which cell proliferation, migration, invasion, cell cycle, and apoptosis were evaluated. Transfection of miR-133a mimic induced apoptosis and inhibited OSCC cell proliferation, migration, and invasion and this was demonstrated to be attributable to decreased CTBP2 expression and suppression of the Notch signaling pathway. Taken together, we concluded that miR-133a acted as a tumor suppressor in OSCC through inhibition of the Notch signaling pathway via binding to CTBP2.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
Change history
31 July 2023
This article has been retracted. Please see the Retraction Notice for more detail: https://doi.org/10.1038/s41417-023-00649-4
References
Markopoulos AK. Current aspects on oral squamous cell carcinoma. Open Dent J 2012;6:126–30.
Thomson PJ. Perspectives on oral squamous cell carcinoma prevention-proliferation, position, progression and prediction. J Oral Pathol Med. 2018;47:803–7.
Gharat SA, Momin M, Bhavsar C. Oral squamous cell carcinoma: current treatment strategies and nanotechnology-based approaches for prevention and therapy. Crit Rev Ther Drug Carrier Syst. 2016;33:363–400.
Aali M, Mesgarzadeh AH, Najjary S, Abdolahi HM, Kojabad AB, Baradaran B. Evaluating the role of microRNAs alterations in oral squamous cell carcinoma. Gene. 2020;757:144936
Mazumder S, Datta S, Ray JG, Chaudhuri K, Chatterjee R. Liquid biopsy: miRNA as a potential biomarker in oral cancer. Cancer Epidemiol. 2019;58:137–45.
Gong Y, Ren J, Liu K, Tang LM. Tumor suppressor role of miR-133a in gastric cancer by repressing IGF1R. World J Gastroenterol. 2015;21:2949–58.
Sui Y, Zhang X, Yang H, Wei W, Wang M. MicroRNA-133a acts as a tumour suppressor in breast cancer through targeting LASP1. Oncol Rep. 2018;39:473–82.
Nohata N, Hanazawa T, Kikkawa N, Mutallip M, Fujimura L, Yoshino H, et al. Caveolin-1 mediates tumor cell migration and invasion and its regulation by miR-133a in head and neck squamous cell carcinoma. Int J Oncol. 2011;38:209–17.
He B, Lin X, Tian F, Yu W, Qiao B. MiR-133a-3p inhibits oral squamous cell carcinoma (OSCC) proliferation and invasion by suppressing COL1A1. J Cell Biochem. 2018;119:338–46.
Wang Y, Che S, Cai G, He Y, Chen J, Xu W. Expression and prognostic significance of CTBP2 in human gliomas. Oncol Lett. 2016;12:2429–34.
Dai F, Xuan Y, Jin JJ, Yu S, Long ZW, Cai H, et al. CtBP2 overexpression promotes tumor cell proliferation and invasion in gastric cancer and is associated with poor prognosis. Oncotarget. 2017;8:28736–49.
Wang Y, Liu F, Mao F, Hang Q, Huang X, He S, et al. Interaction with cyclin H/cyclin-dependent kinase 7 (CCNH/CDK7) stabilizes C-terminal binding protein 2 (CtBP2) and promotes cancer cell migration. J Biol Chem. 2013;288:9028–34.
Zhang G, Kang L, Chen J, Xue Y, Yang M, Qin B, et al. CtBP2 regulates TGFbeta2-induced epithelial-mesenchymal transition through notch signaling pathway in lens epithelial cells. Curr Eye Res. 2016;41:1057–63.
Torres A, Erices JI, Sanchez F, Ehrenfeld P, Turchi L, Virolle T, et al. Extracellular adenosine promotes cell migration/invasion of Glioblastoma Stem-like Cells through A3 Adenosine Receptor activation under hypoxia. Cancer Lett. 2019;446:112–22.
Aquino G, Pannone G, Santoro A, Liguori G, Franco R, Serpico R, et al. pEGFR-Tyr 845 expression as prognostic factors in oral squamous cell carcinoma: a tissue-microarray study with clinic-pathological correlations. Cancer Biol Ther. 2012;13:967–77.
Yu XD, Yang JL, Zhang WL, Liu DX. Resveratrol inhibits oral squamous cell carcinoma through induction of apoptosis and G2/M phase cell cycle arrest. Tumour Biol. 2016;37:2871–7.
Gkouveris I, Nikitakis N, Karanikou M, Rassidakis G, Sklavounou A. Erk1/2 activation and modulation of STAT3 signaling in oral cancer. Oncol Rep. 2014;32:2175–82.
Salta E, Lau P, Sala Frigerio C, Coolen M, Bally-Cuif L, De, et al. A self-organizing miR-132/Ctbp2 circuit regulates bimodal notch signals and glial progenitor fate choice during spinal cord maturation. Dev Cell. 2014;30:423–36.
Yoshida R, Ito T, Hassan WA, Nakayama H. Notch1 in oral squamous cell carcinoma. Histol Histopathol. 2017;32:315–23.
Weaver AN, Burch MB, Cooper TS, Della Manna DL, Wei S, Ojesina AI, et al. Notch signaling activation is associated with patient mortality and increased FGF1-mediated invasion in squamous cell carcinoma of the oral cavity. Mol Cancer Res. 2016;14:883–91.
El Arifeen S, Masanja H, Rahman AE. Child mortality: the challenge for India and the world. Lancet. 2017;390:1932–3.
Dumache R. Early diagnosis of oral squamous cell carcinoma by salivary microRNAs. Clin Lab. 2017;63:1771–6.
Schneider A, Victoria B, Lopez YN, Suchorska W, Barczak W, Sobecka A, et al. Tissue and serum microRNA profile of oral squamous cell carcinoma patients. Sci Rep. 2018;8:675.
Yan Y, Wang X, Veno MT, Bakholdt V, Sorensen JA, Krogdahl A, et al. Circulating miRNAs as biomarkers for oral squamous cell carcinoma recurrence in operated patients. Oncotarget. 2017;8:8206–14.
Ries J, Vairaktaris E, Kintopp R, Baran C, Neukam FW, Nkenke E. Alterations in miRNA expression patterns in whole blood of OSCC patients. Vivo. 2014;28:851–61.
Gao SH, Liu J, Zhang HJ, Zhao N, Zhang J. Low miR-133a expression is a predictor of outcome in patients with esophageal squamous cell cancer. Eur Rev Med Pharmacol Sci. 2016;20:3788–92.
Xuan Q, Zhong X, Li W, Mo Z, Huang Y, Hu Y. CtBP2 is associated with angiogenesis and regulates the apoptosis of prostate cancer cells. Oncol Rep. 2017;38:1259–67.
Li Y, Chen D, Gao X, Li X, Shi G. LncRNA NEAT1 regulates cell viability and invasion in esophageal squamous cell carcinoma through the miR-129/CTBP2 Axis. Dis Markers. 2017;2017:5314649.
Wei X, Yang Y, Jiang YJ, Lei JM, Guo JW, Xiao H. Relaxin ameliorates high glucose-induced cardiomyocyte hypertrophy and apoptosis via the Notch1 pathway. Exp Ther Med. 2018;15:691–8.
Yoshida R, Nagata M, Nakayama H, Niimori-Kita K, Hassan W, Tanaka T, et al. The pathological significance of Notch1 in oral squamous cell carcinoma. Lab Invest. 2013;93:1068–81.
Dou XW, Liang YK, Lin HY, Wei XL, Zhang YQ, Bai JW, et al. Notch3 maintains luminal phenotype and suppresses tumorigenesis and metastasis of breast cancer via trans-activating estrogen receptor-alpha. Theranostics. 2017;7:4041–56.
Li S, Qin X, Li Y, Zhang X, Niu R, Zhang H, et al. MiR-133a suppresses the migration and invasion of esophageal cancer cells by targeting the EMT regulator SOX4. Am J Transl Res. 2015;7:1390–403.
Guan C, Shi H, Wang H, Zhang J, Ni W, Chen B, et al. CtBP2 contributes to malignant development of human esophageal squamous cell carcinoma by regulation of p16INK4A. J Cell Biochem. 2013;114:1343–54.
Subramaniam D, Ponnurangam S, Ramamoorthy P, Standing D, Battafarano RJ, Anant S, et al. Curcumin induces cell death in esophageal cancer cells through modulating Notch signaling. PLoS ONE. 2012;7:e30590.
Chen G, Fang T, Huang Z, Qi Y, Du S, Di T, et al. MicroRNA-133a inhibits osteosarcoma cells proliferation and invasion via targeting IGF-1R. Cell Physiol Biochem. 2016;38:598–608.
Yan ZY, Luo ZQ, Zhang LJ, Li J, Liu JQ. Integrated analysis and microRNA expression profiling identified seven miRNAs associated with progression of oral squamous cell carcinoma. J Cell Physiol. 2017;232:2178–85.
Moriya Y, Nohata N, Kinoshita T, Mutallip M, Okamoto T, Yoshida S, et al. Tumor suppressive microRNA-133a regulates novel molecular networks in lung squamous cell carcinoma. J Hum Genet. 2012;57:38–45.
Kinoshita T, Nohata N, Watanabe-Takano H, Yoshino H, Hidaka H, Fujimura L, et al. Actin-related protein 2/3 complex subunit 5 (ARPC5) contributes to cell migration and invasion and is directly regulated by tumor-suppressive microRNA-133a in head and neck squamous cell carcinoma. Int J Oncol. 2012;40:1770–8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest
Ethics
The study was approved by the Institutional Review Board of Linyi People’s Hospital and in accordance with the declaration of Helsinki. Written informed consent was obtained from each participant.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article has been retracted. Please see the retraction notice for more detail:https://doi.org/10.1038/s41417-023-00649-4
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, W., Shi, X. & Wang, B. RETRACTED ARTICLE: microRNA-133a exerts tumor suppressive role in oral squamous cell carcinoma through the Notch signaling pathway via downregulation of CTBP2. Cancer Gene Ther 29, 62–72 (2022). https://doi.org/10.1038/s41417-020-00289-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41417-020-00289-y