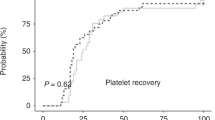
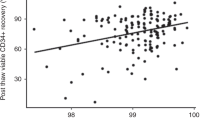
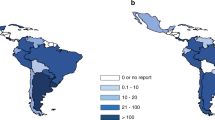
Abstract
The COVID-19 pandemic has had a significant impact on medical practices, including the delivery of allogeneic hematopoietic cell transplantation (HCT). In response, transplant centers have made changes to their procedures, including an increased use of cryopreservation for allogeneic haematopoietic progenitor cell (HPC) grafts. The use of cryopreserved grafts for allogeneic HCT has been reviewed and analysed in terms of potential benefits and drawbacks based on existing data on impact on cell subsets, hematological recovery, and clinical outcomes of approximately 2000 patients from different studies. A survey of European Society for Blood and Marrow Transplantation centers was also conducted to assess changes in practice during the pandemic and any unnecessary burdens on HPC donors. Before the pandemic, only 7.4% of transplant centers were routinely cryopreserving HPC products, but this percentage increased to 90% during the pandemic. The results of this review and survey suggest that cryopreservation of HPC grafts is a viable option for allogeneic HCT in certain situations, but further research is needed to determine long-term effects and ethical discussions are required to balance the needs of donors and patients when using frozen allografts.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article and its supplementary information files.
References
Worel N, Shaw BE, Aljurf M, Koh M, Seber A, Weisdorf D, et al. Changes in Hematopoietic Cell Transplantation Practices in Response to COVID-19: A Survey from the Worldwide Network for Blood & Marrow Transplantation. Transpl Cell Ther. 2021;27:270. https://doi.org/10.1016/j.jtct.2020.11.019.
Faulkner J, O’Brien WJ, McGrane B, Wadsworth D, Batten J, Askew CD, et al. Physical activity, mental health and well-being of adults during initial COVID-19 containment strategies: A multi-country cross-sectional analysis. J Sci Med Sport. 2021;24:320–6. https://doi.org/10.1016/j.jsams.2020.11.016.
Al Hamed R, Bazarbachi AH, Malard F, Harousseau JL, Mohty M. Current status of autologous stem cell transplantation for multiple myeloma. Blood Cancer J. 2019;9:44. https://doi.org/10.1038/s41408-019-0205-9.
Mohty M, Hubel K, Kroger N, Aljurf M, Apperley J, Basak GW, et al. Autologous haematopoietic stem cell mobilisation in multiple myeloma and lymphoma patients: a position statement from the European Group for Blood and Marrow Transplantation. Bone Marrow Transplant. 2014;49:865–72. https://doi.org/10.1038/bmt.2014.39.
Gunn N, Damon L, Varosy P, Navarro W, Martin T, Ries C, et al. High CD34+ cell dose promotes faster platelet recovery after autologous stem cell transplantation for acute myeloid leukemia. Biol Blood Marrow Transpl. 2003;9:643–8. https://doi.org/10.1016/s1083-8791(03)00232-5.
Czerw T, Labopin M, Schmid C, Cornelissen JJ, Chevallier P, Blaise D, et al. High CD3+ and CD34+ peripheral blood stem cell grafts content is associated with increased risk of graft-versus-host disease without beneficial effect on disease control after reduced-intensity conditioning allogeneic transplantation from matched unrelated donors for acute myeloid leukemia - an analysis from the Acute Leukemia Working Party of the European Society for Blood and Marrow Transplantation. Oncotarget. 2016;7:27255–66. https://doi.org/10.18632/oncotarget.8463.
Arcuri LJ, Nabhan SK, Cunha R, Nichele S, Ribeiro AAF, Fernandes JF, et al. Impact of CD34 Cell Dose and Conditioning Regimen on Outcomes after Haploidentical Donor Hematopoietic Stem Cell Transplantation with Post-Transplantation Cyclophosphamide for Relapsed/Refractory Severe Aplastic Anemia. Biol Blood Marrow Transpl. 2020;26:2311–7. https://doi.org/10.1016/j.bbmt.2020.09.007.
Czerw T, Iacobelli S, Malpassuti V, Koster L, Kroger N, Robin M, et al. Impact of donor-derived CD34 + infused cell dose on outcomes of patients undergoing allo-HCT following reduced intensity regimen for myelofibrosis: a study from the Chronic Malignancies Working Party of the EBMT. Bone marrow Transplant. 2022;57:261–70. https://doi.org/10.1038/s41409-021-01540-2.
Politikos I, Davis E, Nhaissi M, Wagner JE, Brunstein CG, Cohen S, et al. Guidelines for Cord Blood Unit Selection. Biol Blood Marrow Transpl. 2020;26:2190–6. https://doi.org/10.1016/j.bbmt.2020.07.030.
Ruggeri A. Optimizing cord blood selection. Hematol Am Soc Hematol Educ Program. 2019;2019:522–31. https://doi.org/10.1182/hematology.2019000056.
Blazar BR, Hill GR, Murphy WJ. Dissecting the biology of allogeneic HSCT to enhance the GvT effect whilst minimizing GvHD. Nat Rev Clin Oncol. 2020;17:475–92. https://doi.org/10.1038/s41571-020-0356-4.
Kuball J, Boelens JJ. Clinical and Biological Concepts for Mastering Immune Reconstitution After HSCT: Toward Practical Guidelines and Greater Harmonization. In: th, Carreras E, Dufour C, Mohty M, Kroger N (eds). The EBMT Handbook: Hematopoietic Stem Cell Transplantation and Cellular Therapies: Cham (CH), 2019, pp 69–74.
Cox JH, Ferrari G, Kalams SA, Lopaczynski W, Oden N, D’Souza MP, et al. Results of an ELISPOT proficiency panel conducted in 11 laboratories participating in international human immunodeficiency virus type 1 vaccine trials. AIDS Res Hum Retroviruses. 2005;21:68–81. https://doi.org/10.1089/aid.2005.21.68.
Ramachandran H, Laux J, Moldovan I, Caspell R, Lehmann PV, Subbramanian RA. Optimal thawing of cryopreserved peripheral blood mononuclear cells for use in high-throughput human immune monitoring studies. Cells. 2012;1:313–24. https://doi.org/10.3390/cells1030313.
Li Y, Mateu E, Diaz I. Impact of Cryopreservation on Viability, Phenotype, and Functionality of Porcine PBMC. Front Immunol. 2021;12:765667. https://doi.org/10.3389/fimmu.2021.765667.
Panch SR, Srivastava SK, Elavia N, McManus A, Liu S, Jin P, et al. Effect of Cryopreservation on Autologous Chimeric Antigen Receptor T Cell Characteristics. Mol Ther. 2019;27:1275–85. https://doi.org/10.1016/j.ymthe.2019.05.015.
Pi CH, Hornberger K, Dosa P, Hubel A. Understanding the freezing responses of T cells and other subsets of human peripheral blood mononuclear cells using DSMO-free cryoprotectants. Cytotherapy. 2020;22:291–300. https://doi.org/10.1016/j.jcyt.2020.01.013.
Xu H, Cao W, Huang L, Xiao M, Cao Y, Zhao L, et al. Effects of cryopreservation on chimeric antigen receptor T cell functions. Cryobiology. 2018;83:40–47. https://doi.org/10.1016/j.cryobiol.2018.06.007.
Costantini A, Mancini S, Giuliodoro S, Butini L, Regnery CM, Silvestri G, et al. Effects of cryopreservation on lymphocyte immunophenotype and function. J Immunol Methods. 2003;278:145–55. https://doi.org/10.1016/s0022-1759(03)00202-3.
Luo Y, Wang P, Liu H, Zhu Z, Li C, Gao Y. The state of T cells before cryopreservation: Effects on post-thaw proliferation and function. Cryobiology. 2017;79:65–70. https://doi.org/10.1016/j.cryobiol.2017.08.008.
Capelle CM, Cire S, Ammerlaan W, Konstantinou M, Balling R, Betsou F, et al. Standard PBMC cryopreservation selectively decreases detection of nine clinically-relevant T-cell markers. ImmunoHorizons. 2021. https://doi.org/10.1101/2021.05.18.443634.
Svalgaard JD, Talkhoncheh MS, Haastrup EK, Munthe-Fog L, Clausen C, Hansen MB, et al. Pentaisomaltose, an Alternative to DMSO. Engraftment of Cryopreserved Human CD34(+) Cells in Immunodeficient NSG Mice. Cell Transpl. 2018;27:1407–12. https://doi.org/10.1177/0963689718786226.
Duchez P, Chevaleyre J, Brunet de la Grange P, Vlaski M, Boiron JM, Wouters G, et al. Cryopreservation of hematopoietic stem and progenitor cells amplified ex vivo from cord blood CD34+ cells. Transfusion. 2013;53:2012–9. https://doi.org/10.1111/trf.12015.
Sartor M, Antonenas V, Garvin F, Webb M, Bradstock KF. Recovery of viable CD34+ cells from cryopreserved hemopoietic progenitor cell products. Bone Marrow Transpl. 2005;36:199–204. https://doi.org/10.1038/sj.bmt.1705009.
Berens C, Heine A, Müller J, Held SAE, Mayer K, Brossart P, et al. Variable resistance to freezing and thawing of CD34-positive stem cells and lymphocyte subpopulations in leukapheresis products. Cytotherapy. 2016;18:1325–31. https://doi.org/10.1016/j.jcyt.2016.06.014.
Kaushal R, Jahan S, McGregor C, Pineault N. Dimethyl sulfoxide-free cryopreservation solutions for hematopoietic stem cell grafts. Cytotherapy. 2022;24:272–81. https://doi.org/10.1016/j.jcyt.2021.09.002.
Pollock K, Yu G, Moller-Trane R, Koran M, Dosa PI, McKenna DH, et al. Combinations of Osmolytes, Including Monosaccharides, Disaccharides, and Sugar Alcohols Act in Concert During Cryopreservation to Improve Mesenchymal Stromal Cell Survival. Tissue Eng Part C Methods. 2016;22:999–1008. https://doi.org/10.1089/ten.TEC.2016.0284.
Matsumura K, Hyon SH. Polyampholytes as low toxic efficient cryoprotective agents with antifreeze protein properties. Biomaterials. 2009;30:4842–9. https://doi.org/10.1016/j.biomaterials.2009.05.025.
Ota A, Matsumura K, Lee JJ, Sumi S, Hyon SH. StemCell Keep Is Effective for Cryopreservation of Human Embryonic Stem Cells by Vitrification. Cell Transpl. 2017;26:773–87. https://doi.org/10.3727/096368916X692654.
Li R, Hornberger K, Dutton JR, Hubel A. Cryopreservation of Human iPS Cell Aggregates in a DMSO-Free Solution-An Optimization and Comparative Study. Front Bioeng Biotechnol. 2020;8:1. https://doi.org/10.3389/fbioe.2020.00001.
Pasley S, Zylberberg C, Matosevic S. Natural killer-92 cells maintain cytotoxic activity after long-term cryopreservation in novel DMSO-free media. Immunol Lett. 2017;192:35–41. https://doi.org/10.1016/j.imlet.2017.09.012.
Lamure S, Paul F, Gagez AL, Delage J, Vincent L, Fegueux N, et al. A Retrospective Comparison of DLI and gDLI for Post-Transplant Treatment. J Clin Med. 2020;9. https://doi.org/10.3390/jcm9072204.
Lasky LC, Van Buren N, Weisdorf DJ, Filipovich A, McGlave P, Kersey JH, et al. Successful allogeneic cryopreserved marrow transplantation. Transfusion. 1989;29:182–4. https://doi.org/10.1046/j.1537-2995.1989.29289146840.x.
Eckardt JR, Roodman GD, Boldt DH, Clark GM, Alvarez R, Page C, et al. Comparison of engraftment and acute GVHD in patients undergoing cryopreserved or fresh allogeneic BMT. Bone Marrow Transpl. 1993;11:125–31.
Stockschlader M, Kruger W, tom Dieck A, Horstmann M, Altnoder M, Loliger C. et al. Use of cryopreserved bone marrow in unrelated allogeneic transplantation. Bone Marrow Transpl. 1996;17:197–9.
Stockschlader M, Hassan HT, Krog C, Kruger W, Loliger C, Horstman M, et al. Long-term follow-up of leukaemia patients after related cryopreserved allogeneic bone marrow transplantation. Br J Haematol. 1997;96:382–6. https://doi.org/10.1046/j.1365-2141.1997.d01-2032.x.
Parody R, Caballero D, Marquez-Malaver FJ, Vazquez L, Saldana R, Madrigal MD, et al. To freeze or not to freeze peripheral blood stem cells prior to allogeneic transplantation from matched related donors. Eur J Haematol. 2013;91:448–55. https://doi.org/10.1111/ejh.12140.
Kim DH, Jamal N, Saragosa R, Loach D, Wright J, Gupta V, et al. Similar outcomes of cryopreserved allogeneic peripheral stem cell transplants (PBSCT) compared to fresh allografts. Biol Blood Marrow Transpl. 2007;13:1233–43. https://doi.org/10.1016/j.bbmt.2007.07.003.
Alotaibi AS, Prem S, Chen S, Lipton JH, Kim DD, Viswabandya A, et al. Fresh vs. frozen allogeneic peripheral blood stem cell grafts: A successful timely option. Am J Hematol. 2021;96:179–87. https://doi.org/10.1002/ajh.26033.
Medd P, Nagra S, Hollyman D, Craddock C, Malladi R. Cryopreservation of allogeneic PBSC from related and unrelated donors is associated with delayed platelet engraftment but has no impact on survival. Bone marrow Transplant. 2013;48:243–8. https://doi.org/10.1038/bmt.2012.118.
Dagdas S, Ucar MA, Ceran F, Gunes AK, Falay M, Ozet G. Comparison of allogenic stem cell transplantations performed with frozen or fresh stem cell products with regard to GVHD and mortality. Transfus Apher Sci. 2020;59:102742. https://doi.org/10.1016/j.transci.2020.102742.
Ghobadi A, Fiala MA, Ramsingh G, Gao F, Abboud CN, Stockerl-Goldstein K, et al. Fresh or Cryopreserved CD34(+)-Selected Mobilized Peripheral Blood Stem and Progenitor Cells for the Treatment of Poor Graft Function after Allogeneic Hematopoietic Cell Transplantation. Biol Blood Marrow Transpl. 2017;23:1072–7. https://doi.org/10.1016/j.bbmt.2017.03.019.
Jacob RP, Flynn J, Devlin SM, Maloy M, Giralt SA, Maslak P, et al. Universal Engraftment after Allogeneic Hematopoietic Cell Transplantation Using Cryopreserved CD34-Selected Grafts. Transpl Cell Ther. 2021;27:697.e691–5. https://doi.org/10.1016/j.jtct.2021.04.026.
Hamadani M, Zhang MJ, Tang XY, Fei M, Brunstein C, Chhabra S, et al. Graft Cryopreservation Does Not Impact Overall Survival after Allogeneic Hematopoietic Cell Transplantation Using Post-Transplantation Cyclophosphamide for Graft-versus-Host Disease Prophylaxis. Biol Blood Marrow Transpl. 2020;26:1312–7. https://doi.org/10.1016/j.bbmt.2020.04.001.
Hsu JW, Farhadfar N, Murthy H, Logan BR, Bo-Subait S, Frey N, et al. The Effect of Donor Graft Cryopreservation on Allogeneic Hematopoietic Cell Transplantation Outcomes: A Center for International Blood and Marrow Transplant Research Analysis. Implications during the COVID-19 Pandemic. Transpl Cell Ther. 2021;27:507–16. https://doi.org/10.1016/j.jtct.2021.03.015.
Eapen M, Zhang MJ, Tang XY, Lee SJ, Fei MW, Wang HL, et al. Hematopoietic Cell Transplantation with Cryopreserved Grafts for Severe Aplastic Anemia. Biol Blood Marrow Transpl. 2020;26:e161–6. https://doi.org/10.1016/j.bbmt.2020.04.027.
Fadeyi E, Mamo YT, Saha AK, Wilson E, Pomper G. The Viability of Hematopoietic Progenitor Cell Grafts after Cryopreservation Does Not Predict Delayed Engraftment in Allogeneic Hematopoietic Stem Cell Transplantation. Lab Med. 2022;53:509–13. https://doi.org/10.1093/labmed/lmac042.
Novitzky-Basso I, Remberger M, Chen C, Pasic I, Lam W, Law A, et al. Anti-thymocyte globulin and post-transplant cyclophosphamide predisposes to inferior outcome when using cryopreserved stem cell grafts. Eur J Haematol. 2022;108:61–72. https://doi.org/10.1111/ejh.13714.
Maurer K, Kim HT, Kuczmarski TM, Garrity HM, Weber A, Reynolds CG, et al. Impact of cryopreservation and transit times of allogeneic grafts on hematopoietic and immune reconstitution. Blood Adv. 2021;5:5140–9. https://doi.org/10.1182/bloodadvances.2021005139.
Bankova AK, Caveney J, Yao B, Ramos TL, Bogeholz J, Heydari K, et al. Real-World Experience of Cryopreserved Allogeneic Hematopoietic Grafts during the COVID-19 Pandemic: A Single-Center Report. Transpl Cell Ther. 2022;28:215.e211–e210. https://doi.org/10.1016/j.jtct.2022.01.010.
Guo M, Liu J, Clark P, Ahmad S, Patel R, Varela JC, et al. Cryopreserved versus fresh peripheral blood allogeneic stem cell transplantation outcomes in patients receiving post-transplant cyclophosphamide for graft-versus-host prophylaxis during the COVID-19 pandemic: a single center experience. Int J Hematol. 2022:1–10. https://doi.org/10.1007/s12185-022-03493-8.
Wiercinska E, Schlipfenbacher V, Bug G, Bader P, Verbeek M, Seifried E, et al. Allogeneic transplant procurement in the times of COVID-19: Quality report from the central European cryopreservation site. J Transl Med. 2021;19:145. https://doi.org/10.1186/s12967-021-02810-9.
Nijssen K, Westinga K, Stuut A, Janssen A, van Rhenen A, van der Wagen L, et al. Graft engineering: how long can you wait, how low can you go, and pandemic readiness. Bone Marrow Transpl. 2023: 1–3. https://doi.org/10.1038/s41409-023-01976-8.
Facchin G, Savignano C, Battista ML, Isola M, De Martino M, Petruzzellis G, et al. Impact of Cryopreservation of Peripheral Blood Stem Cells (PBSC) in Transplantation from Matched Unrelated Donor (MUD). J Clin Med. 2022; 11. https://doi.org/10.3390/jcm11144114.
de Witte M, Daenen LGM, van der Wagen L, van Rhenen A, Raymakers R, Westinga K, et al. Allogeneic Stem Cell Transplantation Platforms With Ex Vivo and In Vivo Immune Manipulations: Count and Adjust. Hemasphere. 2021;5:e580. https://doi.org/10.1097/HS9.0000000000000580.
de Witte MA, Mooyaart JE, Hoogenboom JD, Chabannon C, Malard F, Ruggeri A, et al. Activity of ex vivo graft and DLI Engineering within the last decade increases, a survey from the EBMT Cellular Therapy & Immunobiology Working Party. Bone Marrow Transpl. 2023. https://doi.org/10.1038/s41409-023-01953-1.
Purtill D, Antonenas V, Chiappini P, Tong D, O’Flaherty E, Bajel A, et al. Variable CD34+ recovery of cryopreserved allogeneic HPC products: transplant implications during the COVID-19 pandemic. Blood Adv. 2020;4:4147–50. https://doi.org/10.1182/bloodadvances.2020002431.
Wolf J, Lee J, Pearce R, Wilson M, Snowden JA, Orchard K. The impact of COVID-19 on related-donor allogeneic stem cell harvest processes: A British Society of Blood and Marrow Transplantation and Cellular Therapy survey. Br J Haematol. 2022;198:e51–53. https://doi.org/10.1111/bjh.18299.
Schmidt AH, Buk D, Bernas SN, Mengling T, Neujahr E, van den Brink MRM. A DKMS (German Bone Marrow Donor Center) view on cryopreservation of unrelated donor stem cell products during the Covid-19 pandemic. Am J Hematol. 2021;96:E91–92. https://doi.org/10.1002/ajh.26081.
Acknowledgements
We thank the COVID-19 task force, IDWP and CTIWP for their cooperation in the preparation of the survey and EBMT centers for their participation in the survey (Supplementary Table 2).
Funding
The authors declare no funding was received for this study. All authors declare that there are no competing financial interests in relation to the work described.
Author information
Authors and Affiliations
Contributions
NW, PL, DA, CC, RC, and AR designed the survey; NW, PL, ICMV, JDH, NSK, DJE, ISO, DA, CC, RC and AR analyzed the data. CR, IC, and JK prepared the tables. NW, ICMV, JK, and AR wrote the manuscript. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Worel, N., Ljungman, P., Verheggen, I.C.M. et al. Fresh or frozen grafts for allogeneic stem cell transplantation: conceptual considerations and a survey on the practice during the COVID-19 pandemic from the EBMT Infectious Diseases Working Party (IDWP) and Cellular Therapy & Immunobiology Working Party (CTIWP). Bone Marrow Transplant 58, 1348–1356 (2023). https://doi.org/10.1038/s41409-023-02099-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-023-02099-w