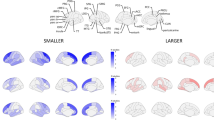
Abstract
Substance use in adolescence is a known risk factor for the development of neuropsychiatric and substance use disorders in adulthood. This is in part due to the fact that critical aspects of brain development occur during adolescence, which can be altered by drug use. Despite concerted efforts to educate youth about the potential negative consequences of substance use, initiation remains common amongst adolescents world-wide. Additionally, though there has been substantial research on the topic, many questions remain about the predictors and the consequences of adolescent drug use. In the following review, we will highlight some of the most recent literature on the neurobiological and behavioral effects of adolescent drug use in rodents, non-human primates, and humans, with a specific focus on alcohol, cannabis, nicotine, and the interactions between these substances. Overall, consumption of these substances during adolescence can produce long-lasting changes across a variety of structures and networks which can have enduring effects on behavior, emotion, and cognition.
Similar content being viewed by others
Adolescence is a period of critical development in the brain and body. Developmental changes in the brain lead to adolescents exhibiting heightened impulsiveness, which can lead to risky behaviors that may have long-term consequences [1, 2]. In particular, the use of both licit and illicit substances in adolescence can produce both acute and enduring effects on brain function and behavior. Of great concern is the fact that the prevalence of substance use disorders as an adult is greater if substance use is initiated during adolescence [3], however, other issues can persist into adulthood both related and unrelated to continued use. Alcohol, cannabis, and nicotine are among the most commonly used substances in adolescents, in part due to their availability, perceived lack of risk, and use in social settings [4].
Importantly, these substances act on receptors widely expressed in the brain (i.e. dopamine, GABAergic, and glutamate receptors), particularly in regions important for reward and cognition. Moreover, these receptor systems and brain regions undergo critical developmental changes during adolescence including a reduction in gray matter volume (GMV) [5] accompanied by an increase in white matter volume [6], changes in connections from subcortical to cortical circuits for emotional control [7], elimination of excess neural connections [8], refinement of the GABAergic system in the neocortex [9, 10], increases in dopamine (DA) receptor expression [11, 12], and development of the mesocorticolimbic system [13], to name a few. The molecular and structural changes in the brain are accompanied by changes in mood, behavior, and cognition, including heightened reward sensitivity [14], reduced inhibitory control [15, 16], and deficits in executive function relative to adults [17]. Furthermore, increases in sex hormones, such as testosterone and estrogen, have been shown to influence the brain’s response to reward [18, 19]. These changes in the brain and behavior make adolescents particularly likely to engage in substance use and susceptible to the long-term negative consequences of drug use. Given the potential societal impact of adolescent drug use, a number of researchers have investigated the long-term consequences of adolescent drug exposure in both clinical and preclinical studies. Indeed, several recent reviews have highlighted much of this research [20,21,22,23,24,25], so the present review is focused on the most recent work investigating the consequences of adolescent use of alcohol [Tables 1 & 5], cannabis [Tables 2 & 5], nicotine [Tables 3 & 5], or polysubstance combinations [Tables 4 & 5] in the human, non-human primate, and rodent literatures.
Alcohol
Alcohol is one of the most commonly used recreational drugs in the world, with adolescents constituting a large group of consumers. However, alcohol has neurotoxic effects and can modify a number of structures and circuits in the brain, including the mesocorticolimbic and striatal systems [26,27,28]. During adolescence, important changes occur in brain circuits that respond to stress and emotional stimuli, which are sensitive to alcohol exposure [29]. Furthermore, there is a well-established relationship between adolescent alcohol exposure (AAE), brain development, and cognitive functioning [20, 30], as well as data indicating that AAE is associated with increased rate and severity of stress-related psychopathologies [31]. AAE also increases future alcohol consumption in rodents [32, 33], as well as humans [34]. Importantly, adolescents are less sensitive than adults to many of the intoxication cues that suppress drinking, such as motor-impairment, sedation, and hangover, and are more sensitive to the reinforcing effects of alcohol, such as social facilitation [35], which may explain why both human and laboratory animal adolescents will consume more alcohol (relative to body weight) per session of drinking than their adult counterparts [36]. Of equal importance is that repeated AAE can cause neuroinflammation via the release of pro-inflammatory cytokines, which can disrupt synaptic plasticity and lead to neuropathology and cell death [37,38,39].
In addition, AAE is known to trigger a series of behavioral effects than can often persist into adulthood, many of which are related to anxiety- and depression- like behavior [40,41,42,43,44]. For example, AAE is associated with increased rates of major depressive disorder [45, 46], particularly in females [47]. Studies in animal models suggest that these effects may have multiple sources, one of which is changes in glucocorticoid receptor density and corticotropin-releasing factor (CRF) expression [48]. For example, AAE via two-bottle choice has been shown to increase glucocorticoid receptor densities in the prelimbic cortex (PL), the paraventricular nucleus (PVN), the central amygdala (CeA,) and the basolateral amygdala (BLA) in both late adolescent and adult mice, as well as lead to higher levels of CRF expression in the PVN and CeA in male mice [42]. In addition to directly contributing to the development of anxiety- and depression-like behaviors, AAE can indirectly contribute to the development of anxiety- and depression-like behavior by altering the effect of stress on the brain, particularly in the nucleus accumbens (NAc). Voluntary AAE in rats has been reported to increase dopamine (D)1 receptor expression while decreasing D2 receptor expression in the NAc and alter postsynaptic excitatory signaling following stressors [49]. Epigenetic modifications have also been linked to AAE’s long-lasting effects on anxiety- and depression-like behavior. For example, AAE can lead to long-lasting histone modifications that alter synaptic function in the amygdala and likely contributes anxiety-like behavior [50]. Recent evidence points to AAE-induced epigenetic repression of the synaptic activity response element (SARE) within the immediate-early gene activity-regulated cytoskeleton-associated protein (Arc) in the central amygdala (CeA) [51, 52] as a critical mediator for anxiety-like behavior. Bohnsack and colleagues [52] found that restoring histone acetylation at the Arc SARE site of the CeA following voluntary AAE in male rats caused a reduction in anxiety-like behavior and excessive drinking to control levels.
AAE also has long-lasting effects on cognitive abilities [53]. Recent studies have reported deficits in recall of an extinguished fear response [54], deficits in reversal learning [43], and impaired working memory [55, 56] following AAE. Unsurprisingly, alcohol’s effect on the medial prefrontal cortex (mPFC) is an important factor in producing these cognitive deficits [57]. Indeed, AAE has been reported to cause a myriad of effects in the mPFC, including greater PL spine density [54], decreased infralimbic cortex (IL) spine density [58], altered PL pyramidal neuron excitability [55, 59], activation of microglia and pro-inflammatory factors [60], decreases in resting state connectivity between PFC subregions [61], and decreases in myelinated fiber density (in males but not females) [62]. AAE also impacts the hippocampus, which is critical in supporting cognitive abilities [63]. Researchers have found that AAE inhibits neurogenesis throughout the hippocampus [64, 65], produces long-lasting reductions in dendritic spine density and alterations in morphology [66], increased levels of astrocytic glial fibrillary acidic protein (GFAP), and decreased levels of brain-derived neurotrophic factor (BDNF) [56].
Potential mechanisms underlying the long-lasting effects of AAE have begun to emerge, one of which is glial functioning and morphology [67, 68]. Astrocyte morphology and astrocyte-neuronal proximity undergo developmental changes, which may make astrocytes vulnerable to AAE [69]. The hippocampus seems to be a critical target for these effects. AAE produces long-lasting alterations to astrocyte activity in the hippocampus [70, 71] and diminishes astrocytic synaptic contact in hippocampal CA1 [72]. Furthermore, AAE elevates levels of astrocytic glutamate transporter (GLT)-1 in the dorsal hippocampus (DH), as well as the ventral hippocampus (VH), in both male and female rats [68]. This finding is particularly relevant given the evidence suggesting AAE alters glutamatergic function [73, 74], and increases in hippocampal glutamate have been linked to schizophrenia [75] and psychosis [76]. However, the effects of AAE are not limited to the hippocampus. Adolescent injections of alcohol in rats also cause changes in PFC subregion astrocyte morphology in the anterior cingulate cortex (ACC) and ventral orbital frontal cortex (vOFC) [77], implying that AAE’s cognitive effects come from changes to multiple brain regions.
Microglia have also been shown to be important mediators of alcohol’s neurotoxic effects [78], in addition to their known role in adolescent brain development [79]. Thus, alcohol’s effects on microglia signaling could lead to a wide array of long-lasting effects. Similar to astrocytes, hippocampal microglia seem to be particularly vulnerable to alcohol. AAE via gavage in adolescent male rats has been shown to produce dramatic increases in microglial activation markers in the entorhinal cortex (EC) and the hippocampus [80]. In another recent example, injections of alcohol in male adolescent mice caused significant loss and dystrophy of microglia in the dentate gyrus (DG) [81] and similar results have been reported in the perirhinal cortex and ECin adult and adolescent male rats [82]. While the long-term consequences of AAE’s effect on microglia are still being determined, it is known that microglia-induced systemic inflammation has many consequences including contributing to long-term neurodegenerative disease [83].
Another consequence of AAE may be long-lasting dysregulation of the endocannabinoid system (ECS). The ECS is widespread throughout the central nervous system and is known to play important roles in many cognitive and behavioral processes [84, 85]. Importantly, the ECS has been implicated in alcohol consumption as well as alcohol addiction [86]. There is some evidence to suggest that cannabinoid (CB)1 receptors may be particularly important in adolescent alcohol consumption. Inhibition of CB1 receptors reduced adolescent alcohol intake in male mice down to adult levels, but did not affect alcohol intake in adults [87]. Moreover, Sánchez-Marín and colleagues have reported several effects of AAE via injections on the ECS. Some of their findings include brain-region dependent changes in mRNA levels of endocannabinoid synthetic enzymes in the PFC and amygdala [44], higher mRNA expression of EC signaling in the mPFC and hippocampus [88], and increases in amygdalar CB1 and CB2 receptor expression (all in male rats) [89]. Furthermore, voluntary AAE in male mice produces long-term deficits in CB1 expression in the hippocampus and interferes with CB1-dependent long-term depression (LTD) [90]. These data suggest that AAE has wide-spread effects on the developing ECS, which could contribute to a wide array of behavioral and cognitive effects.
Research in humans is also producing novel evidence of the harmful effects of AAE. It has been known from some time that AAE disrupts changes in neurodevelopmental trajectories [91]. Specifically, there are accelerated decreases in gray matter and attenuated increases in white matter [92, 93] following AAE. One recent analysis of GMV decline in college students found that, over a two-year period, heavy drinkers had more GMV decline than low drinkers in several brain regions, including the inferior/medial gyrus, parahippocampus, and ACC [94]. Others have recently reported similar results regarding cortical and cingulate GMV [95, 96]. Critically, Sun and colleagues [97] reported that AAE accelerated GMV decline particularly in young adolescents relative to older adolescents. Other recent reports have confirmed that AAE has deleterious effects on white matter microstructural integrity [98, 99]. Furthermore, AAE can alter the overall volume of some brain structures. For example, AAE has recently been linked to smaller whole hippocampal volume and increased volume of the right basal nucleus of the amygdala [100].
Human electroencephalography (EEG) research has also found impairments in overall cognitive functioning following AAE. A recent systematic review reported that AAE increases P3 (an event-related potential related to decision making) amplitude during attention, working memory, and inhibition tasks, suggesting that additional resources were needed to correctly complete these tasks [101]. Together these changes in cortical volume and neural functioning are likely to decrease the overall efficiency of the brain, which could potentially lead to long-lasting cognitive and emotional consequences.
Cannabis
Cannabis is one of the most popular drugs worldwide and initiation of cannabis use commonly occurs in adolescence. Its availability is rapidly increasing with both medical and recreational legality, and its perception as a potentially addictive and harmful substance is decreasing [102]. Despite this, cannabis still has the potential for misuse, which has been shown to have long-term behavioral and biological consequences [103]. While perceived risk has decreased, evidence for a corresponding increase in adolescent use is mixed, with some studies showing increases in adolescent use [104] and others showing no changes [105]. The primary psychoactive effects of cannabis are thought to be mediated by delta-9-tetrahydrocannabinol (THC), while some nonpsychoactive effects may be mediated by cannabidiol (CBD) and other minor cannabinoids and terpenes [106]. Importantly, the concentration of THC in cannabis products has been increasing over the years, leading to a much more potent product than what has previously been available [107], which could also have greater long-term consequences that are yet to be understood. Furthermore, a recent systematic review found increases in both life-time prevalence and past 12-month use of cannabis vaping among adolescents in the United States and Canada [108], emphasizing a need for continued research in this area.
THC primarily acts on the ECS by binding to CB1 and CB2 receptors. As described above, the ECS undergoes important changes during adolescence that are critical to normative development, such that over-use of cannabis during adolescence might interfere with these changes. Adolescent cannabis exposure (ACE) has also been linked to multiple psychiatric disorders. Of great interest to researchers is its role as a risk factor in schizophrenia and psychosis [109], but it has also been linked to depression, anxiety, and addiction [110]. Furthermore, cannabis use has been shown to have stronger acute behavioral and cognitive effects in late adolescent humans (18–20) than in adults (30–40) [111]. In addition, ACE has been noted to have effects on several brain regions, including the PFC, hippocampus, ventral tegmental area (VTA), and striatum (see [22] for a review).
One of the most concerning potential consequences of ACE is its propensity to precipitate first episode psychosis and the development of schizophrenia [112]. A recent report suggests that the risk of psychosis is elevated in adolescents who have consumed cannabis at least five times [113]. ACE may contribute to the development of schizophrenia by interfering with natural changes in GMV that occur throughout adolescence [114]. One recent investigation found that early cannabis use was associated with greater GMV in the cerebellar schizophrenia-related network as well as more severe positive symptoms of recent onset psychosis (ROP) [115]. Interestingly, some argue that microglia could be important contributors to ACE-induced psychosis [116, 117]. Indeed, ACE is known to activate microglia [118, 119], which are responsible for much of the synaptic pruning that reduces GMV during adolescence [120], potentially leading to unhealthy amounts of synaptic pruning and loss of cortical gray matter [117]. Further research is needed in order establish if microglial effects can explain ACE’s relations with schizophrenia. Importantly factors that moderate the relationship between ACE and psychosis are being identified, and include age of onset, frequency of cannabis use, exposure to childhood trauma, concurrent use of other substances, and genetic factors [121]. On the other hand, recent evidence has also emerged suggesting that genetic risk for schizophrenia may be a risk factor for ACE [122], making causal conclusions about the role of ACE in psychosis difficult to determine. More research is needed to clarify how genetic predisposition and ACE interact to increase risk of schizophrenia and psychosis.
One important target of ACE that may influence risk for schizophrenia is GABAergic signaling in the PFC [123], with ACE generally inhibiting GABAergic activity [124]. Renard and colleagues [125] found that adolescent injections of THC reduced levels of the GABA synthesizing enzyme GAD67 in the mPFC in adult male rats, which was coupled with a hyperactive dopaminergic state. Reductions in PFC GABA levels were also observed in female rats following adolescent injections of THC [126], and adolescent cannabis users have been shown to have significantly reduced GABA levels in the anterior cingulate cortex (ACC) [127]. Interestingly, one recent report has shown that while adolescent injections of pure THC in female rats reduced expression of GAD67 in the PFC, injections of THC-CBD combinations actually increased expression [128], suggesting that the interactions between THC and CBD can have complex effects which need further study.
The mesolimbic pathway is also an important target for the effects of cannabis, and the ongoing reorganization of this pathway in adolescence makes it particularly vulnerable to ACE. Mesolimbic activity is critical for reward learning and is involved in the development of substance use disorders [129]. The VTA in particular may be critically important for changes in reward processing induced by ACE. For example, there is evidence that ACE induces a hyperdopaminergic state in the VTA [130]. Conversely, repeated CB1 receptor activation during adolescence has been found to reduce firing of DA neurons in the VTA and DA release in the NAc shell [131]. CB1 receptors in the VTA are located on both GABAergic and glutamatergic synapses [132], meaning both systems have important roles in modulating dopaminergic activity and reward learning. One recent study reported downregulation of VTA CB1 expression in glutamatergic terminals following ACE via THC-containing gelatin consumption in male, but not female adolescent rats, which was affiliated with an increase in the value of reward predictive cues. These authors argued that loss of CB1 receptors on glutamatergic terminals resulted in greater VTA dopamine firing and increased dopamine release in the NAc [133]. Enhanced VTA dopaminergic activity following ACE is also related to inhibition of GABAergic CB1 expressing neurons. Adolescent injections of THC induce synaptic depression of excitatory synapses onto VTA GABAergic neurons, disinhibiting VTA dopamine neurons in male mice. More work needs to be done to understand to complicated balance between dopamine, glutamate, and GABA in the mesolimbic system, particularly after ACE.
In addition, a number of studies report sex differences in the effects of ACE. For example, there are sex-specific disruptions in long-term potentiation (LTP) of the Schaffer-commisural projection to CA1 and in the lateral perforant pathway (LPP) in adolescent mice and rats following injections of THC, with females showing greater LTP impairment than males [134]. The sex differences in the effects of cannabis may be explained by differences in the metabolism of THC [135, 136], and/or by different ACE-induced changes in gene expression networks in the brain. For example, Zuo and colleagues [137] reported that female mice exhibited a larger number of differentially expressed genes (DEGs) across the amygdala and dorsal medial striatum (DMS) compared to males. Differences in DEGs following adolescent THC exposure have also been shown in the NAc [138]. These studies represent just a part of the growing literature on sex differences in outcomes related to cannabis [139].
ACE also has important implications for cognitive function [140]. Many demonstrations of impaired working memory have been observed in rodents. For example, male and female mice show deficits in working memory in adulthood following ACE via experimenter-administered injections [141]. ACE-induced cognitive impairments have also been observed in nonhuman primates. Injections of THC in adolescent male squirrel monkeys impaired performance in a working memory task [142] and injections of THC in adolescent rhesus monkeys impaired spatial working memory [143]. In humans, studies have reported effects of ACE on academic achievement [144], IQ [145], and performance on specific cognitive tasks [146, 147] (but see also [148]). A systematic review of recent papers investigating the cognitive impacts of ACE found significant effects on outcomes such as executive control, working memory, and academic performance [149]. As is often the case in human studies, baseline differences in cognitive performance are also predictive of early cannabis use [150], meaning that trait level cognitive ability and ACE may interact with each other.
In clinical populations, a 5-year longitudinal study found that cannabis use was negatively associated with thickness in the left and right PFC. Furthermore, thinning in the right PFC was associated with attentional impulsiveness [151, 152]. Moreover, a recent systematic review of the neuroimaging literature on adolescent cannabis users noted preliminary evidence for functional and structural alterations in frontoparietal, frontolimbic, frontostriatal, and cerebellar regions [153]. However, other studies have failed to find effects of adolescent cannabis use on brain volume. A recent meta-analysis of 16 studies that examined global brain volume (GBV) of 12–16 year-olds found no evidence of differences in GBV between cannabis users and non-users [154]. Therefore, it will be important for future research to identify underlying causes for these disparate results, such as frequency of use, total years of use, the potency of the cannabis used, and other confounding factors, such as use of other substances.
The mechanisms by which ACE produces long-term effects are not well established, but recent research suggests that epigenetic factors may play a role. For example, ACE in female rats produced age-dependent alterations in histone modifications in several brain regions, including the PFC, amygdala, and hippocampus [155]. The same group has also reported chromatin modifications in the PFC following ACE in female rats, which was associated with cognitive deficits [156]. ACE in male rats causes a disruption in the developmental trajectory of PL pyramidal neuron transcriptomes along with premature pruning of synaptic spines. Furthermore, the alterations in PFC networks observed in the THC-exposed animals were common to subjects with schizophrenia [157]. There have also been reports of age-dependent alterations in the hippocampal transcriptome following acute administration of THC [158]. Evidence of epigenetic changes have also been shown in humans, as Clark and colleagues reported alterations in several DNA methylation sites following ACE [159].
It should be stated that not all of the literature on ACE has produced evidence of deleterious effects. There is emerging evidence that, at least in rodent studies, that detrimental effects of ACE on working memory could be due to the experimenter-administration of high doses of cannabinoids. Studies from our lab were the first to use adolescent self-administration of cannabinoids (THC or the full agonist WIN55,212-2) to investigate long-term effects on adult working memory performance. We found that adolescent THC or WIN55,212-2 self-administration actually led to improved working memory performance in males, while having no effect or a tendency to decrease performance in females [160,161,162]. Importantly, self-administration versus experimenter-administration of drugs can often produce very different effects based on the stress of injections and having volitional control over the dose consumed [22]. Further research should be conducted in order to solidify our understanding of the role of dose and administration methods regarding ACE and working memory. There have been conflicting reports of the effects of ACE in other domains as well. There are failures to find any effects of cannabis smoke or THC vapor exposures in male and female rats during adolescence using assays for cognition and anxiety-like behaviors [163, 164]. In humans, moderate adolescent cannabis users have reported less anhedonia than nonusers [165]. Another study found no effect of moderate adolescent cannabis use on ventral striatum activity in a test of reward anticipation [166], again indicating the importance of considering dose in analyses of the effects of all drugs.
Nicotine
While researchers have been addressing the effects of adolescent nicotine exposure (ANE) for decades, the advent of electronic cigarette products has led to a resurgence of nicotine use among adolescents [167, 168], resulting in the development of a new domain in adolescent nicotine research. In addition to the harmful effects of electronic cigarette products on the body [169, 170], nicotine has a myriad of harmful effects on adolescent development, mood, and behavior. ANE is associated with increased mood and anxiety disorder risk [171,172,173,174], in part through alterations in mescorticolimbic dopaminergic transmission [172]. Furthermore, there are known impairments in cognitive abilities [175,176,177]. ANE also increases the likelihood of continued nicotine use [178], cannabis use [179], and alcohol use in humans [180], as well as other drugs in rodents such as methamphetamine [181], alcohol [182], cocaine [183, 184], and fentanyl [183]. Nicotine also modulates multiple neurotransmitter systems, including dopamine, GABA, glutamate, serotonin, and acetylcholine, all of which require regulation by precise neurodevelopmental mechanisms [185].
Nicotine produces reinforcing effects primarily through its action on the nicotinic acetylcholine receptor (nAchR) system, which is still immature during adolescence [186]. For example, α4β2 and α7 nAchR expression and binding are higher in adolescents than adults [187, 188]. This suggests that adolescents may be particularly vulnerable to the reinforcing properties of nicotine, which are likely mediated through mesocorticolimbic structures. Nicotine injections induce greater DA release in the NAc shell (NAcSh) and NAc core of adolescents relative to adults [189] and adolescents of both sexes show stronger conditioned place preference (CPP) than adults following nicotine injections [190], as well as enhanced locomotor activity following nicotine self-administration [191]. Furthermore, nicotine self-administration has been shown to be greater in male adolescent mice than in adults, which correlated strongly with α4 nAchR expression in the VTA [192]. In humans, adolescent smokers display increased reward-related activity in the NAc, insula, and mPFC [193]. However, studies using self-administration models do not always support the conclusion that nicotine is more reinforcing for adolescents than adults. For example, there is a report of adolescent male and female rats self-administered less nicotine than adults at a low dose [194], and that there are minimal long-term impacts of ANE on reinforcement enhancement [195]. The discrepancy in these results again highlights the importance of considering the method of drug administration and drug dose in interpreting experimental results.
ANE has also been implicated in the development of anxiety- and depression-like symptoms [172, 196,197,198]. Many of ANE’s effects on anxiety and depression-like behavior are thought to be related to changes in mesolimbic function [185]. One important report comes from Jobson and colleagues [172]. These authors injected male rats with nicotine during adolescence or adulthood and found increases in anxiety-like behavior specifically in adolescent-exposed animals. Furthermore, they reported that ANE resulted in a hyperdopaminergic state during adulthood in the VTA and hyperexcitability of PFC pyramidal neurons (PNs). Importantly, disturbances in mesolimbic DA transmission and hyperexcitability of PFC PNs are both associated with mood and anxiety disorder symptoms [199, 200]. In addition, the authors reported that ANE produced a downregulation in D1 receptor expression and upregulation in phosphorylated extracellular-signal-related kinase (pERK) 1–2 molecular signaling pathways in the PFC, both of which have been associated with increases in anxiety-like behavior [201, 202].
Hudson and colleagues [198] expanded on this by providing evidence for upregulation of NAcSh biomarkers of ANE-induced anxiety-like behavior. The authors gave male rats injections of nicotine during adolescence or adulthood and found several consequences that were only present in adolescent-exposed animals. In addition to increases in anxiety-like behavior, the adolescent-exposed animals displayed several anxiety-related biomarkers, including upregulation of the ERK 1–2 and the protein-kinase B (Akt) glycogen-synthase-kinase-3 (GSK-3) signaling pathways in the NAcSh, as well as a large decrease in D1 receptor expression. Critically, these authors found that selectively targeting the Akt-GSK-3 and ERK 1–2 signaling pathways in the NAc was sufficient to ablate the effects of ANE on anxiety-like behavior, implying that these signaling pathways could be important targets for therapeutic interventions.
ANE has also been shown to produce cognitive deficits, including effects in humans on working memory, attention, and PFC activation [176, 203, 204]. Many of the cognitive effects of ANE are likely mediated by its effects on the mPFC. ANE has been reported to cause short-term increases and long-term decreases in synaptic metabotropic glutamate receptor (mGluR)2 in the rat mPFC, which was associated with deficits in attention [205]. These changes in mGluR2 expression are also thought to mediate changes in pre and postsynaptic activity in the mPFC [206]. Other research points to a role for the hippocampus in ANE-induced cognitive deficits [196, 207, 208]. For example, subcutaneous nicotine delivery in male rats via osmotic pump reduced dendritic length and complexity in CA1 branches of pyramidal cells at adulthood [196]. Moreover, in male mice ANE was shown to alter hippocampal gene methylation and expression in adulthood. Interestingly, administration of the essential nutrient choline ameliorated the ANE-induced decrements in fear conditioning as well as the epigenetic changes in the hippocampus [209], suggesting that choline may have potential as a ANE therapy.
Research in humans generally corroborates the findings from the preclinical literature. ANE is associated with long-lasting effects on attention [210], risky sexual behavior [211], and progression to traditional cigarettes [212]. However, as is the case with much human research, directionality of the effect is subject to debate and more research is needed. In terms of the effects of ANE on the brain, initiation of tobacco use in late childhood has been associated with inferior cognitive performance and smaller cortical volume in frontal, parietal, and temporal lobes at a 2-year follow-up [213]. Moreover, even exposure to low doses of nicotine have been linked to volume reductions in the ventral medial PFC (vmPFC) and altered connectivity in the corpus callosum [214]. There are also reductions in thalamus volume following ANE [215]. In addition, there are alterations in both right dorsal and left ventral frontostriatal tracts in young adult male smokers, which corresponded with poorer performance in cognitive tasks [187, 188]. All of these alterations in brain structure and volume are likely to contribute to the long-lasting effect of ANE on cognition and mood disorders.
In addition, microglial signaling is emerging as an important participant in the long-term effects of ANE, and there are important differences in the effects of ANE on adults versus adolescents. For example, while nicotine inhibits microglia activation in adults, the opposite effect has been found in adolescents [186]. Linker and colleagues [216] reported a myriad of differences in how microglia in adult and adolescent rats respond to injections of nicotine, including differences in IBA1 expression in the NAc and BLA and microglial morphology. Importantly, the authors reported that NAc D2 receptors were responsible for nicotine-induced increases in microglial activation in adolescent rats, as well as the increased cocaine self-administration that was also observed. Furthermore, the effects of ANE on microglia may not be limited to reward-related circuitry. Exposure to nicotine and e-cigarette aerosols during pregnancy have been shown to upregulate hippocampal microglia [217, 218], which is likely to have long-lasting impacts on learning and memory. However, whether these results will hold with ANE is still unknown.
Finally, sex differences surrounding adolescent nicotine have been a subject of interest for some time, with results generally suggesting that females are more susceptible to nicotine’s effects than males [219]. One recent study reported that adolescent nicotine injections reduced brain reward threshold (via intracranial self-stimulation) more in adolescent female rats than males [220]. Another study found that exposure to high-nicotine tobacco smoke increased later nicotine self-administration in adolescent female rats but not males [221]. In humans, adolescent females that smoke are more likely to develop nicotine dependence than males [222]. However, another study reported that male e-cigarette users were more likely than female e-cigarette users to report past 30-day cigarette use at a one-year follow up [223]. More research is needed to make sense of these conflicting reports.
Poly use
Researchers are increasingly studying the effects of drugs in combination to better model the way humans consume drugs [224,225,226,227]. However, this approach is still novel and there is much that we do not know. Alcohol, cannabis, and nicotine use are often directly related to each other [226, 228] with all three often used in combination. Even use of one substance during adolescence can be predictive of use of other substances later in life [229]. Moreover, these drugs can serve to augment the reinforcing properties of each other and continued use of one drug is predictive of relapse of another [230]. When taken in combination, drugs can often have unexpected effects on the brain and body, making it critical to examine the effects of these drugs when they are taken in combination. However, the complexity and novelty of the field has led to some contradictory and unexpected results. Continued work is necessary to gain a full understanding of the effects of polysubstance use.
One recent review of studies investigating individuals that use both cannabis and alcohol reported mixed results, with some findings suggesting that cannabis use may be protective of the effects of alcohol use, while other studies find more negative outcomes such as low academic performance [227]. As human studies in this domain can be difficult to interpret due to factors such as the ratio of alcohol to cannabis use and additional drug use, rodent research has proven highly valuable given the increased experiment control. One study exposed adolescent rats to vaporized cannabis on alternating days while also allowing them to self-administer both alcohol and water in a two-bottle choice paradigm daily. Interestingly, on days when the rats were exposed to cannabis, there was no preference between alcohol and water. However, on days when cannabis was not administered, the rats preferred the alcohol over water [231]. One interpretation is that the rats were increasing their alcohol consumption to compensate for the loss of cannabis, which might replicate substitution effects observed in humans [232]. Other research has examined a possible role for alcohol-cannabis poly use in the development of fear responding. While the combination of THC and alcohol did produce heightened fear responding in male rats as well as greater glutamatergic activity in the PFC, there were not any additive effects compared to cannabis and alcohol alone [233].
There is mixed evidence in humans suggesting additive effects of ACE and AAE. One investigation reported decreases in GMV following poly use of alcohol and cannabis starting in early adolescence, but did not observe this in participants who had exclusively used alcohol or cannabis [234]. However, there is also evidence that the two do not have additive effects in some domains. A 14-year longitudinal study on youth who used both alcohol and cannabis found that there were cognitive performance decrements affiliated with both substances individually, but did not observe additive effects of the two [235]. Furthermore, one recent analysis of GMV and white matter integrity found that alcohol use led to reduced GMV, but that cannabis use in the past 30-days did not have any additional impact on GMV or white matter integrity [236]. Further studies are needed to determine what additive effects AAE and ACE may or may not have.
Examinations of the interactions between alcohol and nicotine are also emerging. Recent analyses of high school e-cigarette users found that a large proportion of their samples reported poly substance use, with alcohol being the most frequently reported drug used in combination with e-cigarettes [237, 238]. There is evidence from rodent studies suggesting that the combination of nicotine and alcohol is more reinforcing in adolescents than adults. One study has found that previous exposure to alcohol and nicotine increased responding for alcohol and alcohol consumption relative to only alcohol exposure, however, this effect was limited to male rats [239]. Furthermore, administration of a low dose of alcohol and nicotine to male adolescent and adult rats produced an increase in locomotor behavior and a decrease in anxiety-like behavior only in the adolescents [240]. Importantly, this experiment did not compare alcohol and nicotine together to the drugs separately, so it is difficult to determine if any additive effects were present. Another study exposed male and female adolescent rats to vaporized nicotine/vehicle vapor and voluntary alcohol/water, which produced long-term changes in reward- and cognition-based behaviors in males but not females, however, they did not find any evidence for additive effects of the drugs [241].
While research examining cannabis-nicotine poly use is limited, recent evidence suggests that e-cigarette use and cannabis use develop in close parallel from middle to late adolescence [242] Studies in rodents have produced interesting sex differences. For example, Dukes and colleagues [243] reported that injections of WIN55,212-2 and nicotine in adolescence increased nicotine self-administration in adult males, but that it decreased nicotine self-administration in females. Courtney and colleagues [244] recently evaluated the effects of cannabis and nicotine poly use on white matter microstructure in a cohort of late adolescents (ages 16–22) and reported that cannabis was associated with white matter reductions, but only when nicotine was also being used. Another study in adults found that the use of both nicotine and cannabis enhanced nAChR availability in the PFC and thalamus to a higher degree than single substance-use alone [245]. More research is needed to corroborate this finding in adolescents, as well as more research on cannabis-nicotine poly use in general.
Conclusions
Adolescence is a period of both great developmental and behavioral change, making it a particularly vulnerable period for long-term effects of drug exposure. Over consumption of alcohol, cannabis and nicotine, and combinations of these drugs can have long-lasting effects on the brain and body. Continued research is needed to isolate the predictors, mechanisms, and consequences of adolescent drug use, as well as efficacious treatments and pre-use interventions. In particular, many questions still exist around polysubstance use. Further research is needed to establish how these substances pharmacodynamically interact, and how acute and long-term administration of one substance effects consumption of another substance. An understanding of this will enable the use of better models to understand the way that humans consume these substances. Furthermore, we still lack a complete understanding of how adolescent substance use promotes sustained substance use in adulthood. Many theories have been proposed, including dysregulation of the mesolimbic reward system [129], loss of goal-directed control [246], and hyperkaitefia [247], but other mechanisms are likely at work.
Going forward, it will be important to solidify our understanding of the similarities, and the differences of different drug types on adolescents to create therapies that work on the widest possible range of substance use disorders. For example, it has become clear that many drugs can cause proinflammatory central immune signaling, including the acute activation of microglia [248]. This neuroinflammation can have critical long-term effects [67, 249,250,251] which may contribute to the development of substance-use disorders (see [251] for a review). Targeting neuroinflammation may prove to be an important target for therapies addressing adolescent drug use [252,253,254]. The EC system also seems to be a common factor related to all three substances. The EC system contributes to the reinforcing effects of all three substances, and undergoes changes following adolescent exposure to them [255]. EC- and inflammation-modulating therapies such as CBD [49, 256, 257] and exercise [258,259,260] show promise, but more work is needed to determine their efficacy in clinical populations as well as optimal therapeutic protocols. Furthermore, researchers should continue to be aware of potential differences between preclinical studies that use self-administration models of drug taking and studies that use experiment-administered drugs, as the differences in these methods can lead to discrepancies in the dose of drug the animal consumes and produce conflicting results [22, 161]. Furthermore, experimenter-administered methods of drug administration, such as injection or gavage, could be a stressful experience with the ability to induce immune effects in rodents [261], which must be considered in the interpretation of results. Finally, while great progress has been made in including both males and females in research samples, many of the studies mentioned above exclusively studied males (particularly the preclinical studies). Furthermore, an often ignored factor in experimental designs is that male and female rodents (particularly rats) can reach sexual maturity at different ages [262], which can complicate the interpretation of results when male and female adolescent rodents of the same age are compared. Continued emphasis should be placed on the study of both sexes, which has already yielded interesting and important sex differences, and will help to develop more targeted therapeutic interventions.
References
Steinberg L, Icenogle G, Shulman EP, Breiner K, Chein J, Bacchini D. Around the world, adolescence is a time of heightened sensation seeking and immature self-regulation. Dev Sci. 2018;21:e12532.
Shulman EP, Smith AR, Silva K, Icenogle G, Duell N, Chein J, et al. The dual systems model: review, reappraisal, and reaffirmation. Develop Cogn Neurosci. 2016;17:103–17.
DuPont RL, Han B, Shea CL, Madras BK. Drug use among youth: National survey data support a common liability of all drug use. Prev Med 2018. 2018;113:68–73.
Johnston L, Miech R, O’Malley P, Bachman J, Schulenberg J, Patrick M. Monitoring the future national survey results on drug use, 1975–2019: overview, key findings on adolescent drug use. 2020.
Tamnes CK, Herting MM, Goddings AL, Meuwese R, Blakemore SJ, Dahl RE, et al. Development of the cerebral cortex across adolescence: a multisample study of inter-related longitudinal changes in cortical volume, surface area, and thickness. J Neurosci. 2017;37:3402–12.
Gogtay N, Giedd JN, Lusk L, Hayashi KM, Greenstein D, Vaituzis AC, et al. Dynamic mapping of human cortical development during childhood through early adulthood. Proc Natl Acad Sci USA. 2004;101:8174–9.
Casey BJ, Heller AS, Gee DG, Cohen AO. Development of the emotional brain. Neurosci Lett. 2019;693:29–34.
Giedd JN. Structural magnetic resonance imaging of the adolescent brain. Ann N. Y Acad Sci. 2004;1021:77–85.
Kilb W. Development of the GABAergic system from birth to adolescence. Neuroscientist. 2012;18:613–30.
Caballero A, Orozco A, Tseng KY. Developmental regulation of excitatory-inhibitory synaptic balance in the prefrontal cortex during adolescence. Semin Cell Develop Biol. 2021;118:60–3.
Rosenberg DR, Lewis DA. Changes in the dopaminergic innervation of monkey prefrontal cortex during late postnatal development: a tyrosine hydroxylase immunohistochemical study. Biol Psychiatry. 1994;36:272–7.
Weickert CS, Webster MJ, Gondipalli P, Rothmond D, Fatula RJ, Herman MM, et al. Postnatal alterations in dopaminergic markers in the human prefrontal cortex. Neuroscience. 2007;144:1109–19.
Reynolds LM, Flores C. Mesocorticolimbic dopamine pathways across adolescence: diversity in development. Front Neural Circuits. 2021;15:735625.
van Duijvenvoorde ACK, van Hoorn J, Blankenstein NE. Risks and rewards in adolescent decision-making. Curr Opin Psychol. 2022;48:101457.
Casey BJ. Beyond simple models of self-control to circuit-based accounts of adolescent behavior. Annu Rev Psychol. 2015;66:295–19.
Romer D, Reyna VF, Satterthwaite TD. Beyond stereotypes of adolescent risk taking: placing the adolescent brain in developmental context. Dev Cogn Neurosci. 2017;27:19–34.
Kundu P, Benson BE, Rosen D, Frangou S, Leibenluft E, Luh WM, et al. The integration of functional brain activity from adolescence to adulthood. J Neurosci. 2018;38:3559–70.
Poon JA, Niehaus CE, Thompson JC, Chaplin TM. Adolescents’ pubertal development: Links between testosterone, estradiol, and neural reward processing. Horm Behav. 2019;114:104504.
Calipari ES, Juarez B, Morel C, Walker DM, Cahill ME, Ribeiro E, et al. Dopaminergic dynamics underlying sex-specific cocaine reward. Nat Commun. 2017;8:13877.
Lees B, Meredith LR, Kirkland AE, Bryant BE, Squeglia LM. Effect of alcohol use on the adolescent brain and behavior. Pharmacol Biochem Behav. 2020;192:172906.
Salmanzadeh H, Ahmadi-Soleimani SM, Pachenari N, Azadi M, Halliwell RF, Rubino T, et al. Adolescent drug exposure: a review of evidence for the development of persistent changes in brain function. Brain Res Bull. 2020;156:105–17.
Stringfield SJ, Torregrossa MM. Disentangling the lasting effects of adolescent cannabinoid exposure. Prog Neuro-Psychopharmacol Biol Psychiatry. 2021;104:110067.
Thorpe HHA, Hamidullah S, Jenkins BW, Khokhar JY. Adolescent neurodevelopment and substance use: receptor expression and behavioral consequences. Pharm Ther. 2020;206:107431.
Mooney-Leber SM, Gould TJ. The long-term cognitive consequences of adolescent exposure to recreational drugs of abuse. Learn Mem. 2018;25:481–91.
Kwan LY, Eaton DL, Andersen SL, Dow-Edwards D, Levin ED, Talpos J, et al. This is your teen brain on drugs: in search of biological factors unique to dependence toxicity in adolescence. Neurotoxicol Teratol. 2020;81:106916.
Alasmari F, Goodwani S, McCullumsmith RE, Sari Y. Role of glutamatergic system and mesocorticolimbic circuits in alcohol dependence. Prog Neurobiol. 2018;171:32–49.
McCool BA. Ethanol modulation of cortico-basolateral amygdala circuits: neurophysiology and behavior. Neuropharmacology. 2021;197:108750.
Ron D, Barak S. Molecular mechanisms underlying alcohol-drinking behaviours. Nat Rev Neurosci. 2016;17:576–91.
Vilpoux C, Warnault V, Pierrefiche O, Daoust M, Naassila M. Ethanol-sensitive brain regions in rat and mouse: a cartographic review, using immediate early gene expression. Alcohol Clin Exp Res. 2009;33:945–69.
Lees B, Mewton L, Stapinski LA, Squeglia LM, Rae CD, Teesson M, et al. Neurobiological and cognitive profile of young Binge drinkers: a systematic review and meta-analysis. Neuropsychol Rev. 2019;29:357–85.
Fortier CB, Whitworth JW, Fonda JR, Currao A, Beck BM, Levin L, et al. Early adolescent binge drinking increases risk of psychopathology in post-9/11 veterans and mild traumatic brain injury exacerbates symptom severity. Alcohol Alcohol. 2021;56:116–24.
Maldonado-Devincci AM, Kirstein CL. Chronic ethanol exposure during adolescence increases voluntary ethanol consumption in adulthood in female Sprague Dawley Rats. Brain Sci. 2020;10:900.
Maldonado‐Devincci AM, Makdisi JG, Hill AM, Waters RC, Hall NI, Shobande MJ, et al. Adolescent intermittent ethanol exposure induces sex-dependent divergent changes in ethanol drinking and motor activity in adulthood in C57BL/6J mice. J Neurosci Res. 2022;100:1560–72.
Tomek S, Bolland KA, Bolland JM, Hooper LM, Church WT, Bolland AC. Age of alcohol initiation matters: examining gender differences in the recency and frequency of alcohol use across adolescence using a sample of impoverished minority adolescents. Youth Soc. 2019;51:120–45.
Spear L. The teenage brain: adolescents and alcohol. Curr Dir Psychol Sci. 2013;22:152–7.
Spear LP. Effects of adolescent alcohol consumption on the brain and behaviour. Nat Rev Neurosci. 2018;19:197–214.
Crews FT, Vetreno RP. Mechanisms of neuroimmune gene induction in alcoholism. Psychopharmacology. 2016;233:1543–57.
Crews FT, Vetreno RP. Addiction, adolescence, and innate immune gene induction. Front Psychiatry. 2011;2:1–11.
Crews FT, Lawrimore CJ, Walter TJ, Coleman LG. The role of neuroimmune signaling in alcoholism. Neuropharmacology. 2017;122:56–73.
Lee KM, Coehlo MA, Solton NR, Szumlinski KK. Negative affect and excessive alcohol intake incubate during protracted withdrawal from binge-drinking in adolescent, but not adult, mice. Front Psychol. 2017;8:1128.
Lee KM, Coelho MA, Class MA, Sern KR, Bocz MD, Szumlinski KK. mGlu5 receptor blockade within the nucleus accumbens shell reduces behavioral indices of alcohol withdrawal-induced anxiety in mice. Front Pharmacol. 2018;9:1306.
Sampedro‐Piquero P, Moreno‐Fernández RD, Begega A, López M, Santín LJ. Long‐term consequences of alcohol use in early adolescent mice: focus on neuroadaptations in GR, CRF and BDNF. Addict Biol. 2022;27:1–13.
Van Hees L, Didone V, Charlet‐Briart M, Van Ingelgom T, Alexandre A, Quertemont E, et al. Voluntary alcohol binge‐drinking in adolescent C57Bl6 mice induces delayed appearance of behavioural defects in both males and females. Addict Biol. 2022;27:1–15.
Sanchez-Marin L, Pavon FJ, Decara J, Suarez J, Gavito A, Castilla-Ortega E, et al. Effects of intermittent alcohol exposure on emotion and cognition: a potential role for the endogenous cannabinoid system and neuroinflammation. Front Behav Neurosci. 2017;11:15.
Pedrelli P, Shapero B, Archibald A, Dale C. Alcohol use and depression during adolescence and young adulthood: a summary and interpretation of mixed findings. Curr Addict Rep. 2016;3:91–7.
Edwards AC, Heron J, Dick DM, Hickman M, Lewis G, Macleod J, et al. Adolescent alcohol use is positively associated with later depression in a population-based UK cohort. J Stud Alcohol Drugs. 2014;75:758–65.
Danzo S, Connell AM, Stormshak EA. Associations between alcohol‐use and depression symptoms in adolescence: examining gender differences and pathways over time. J Adolescence. 2017;56:64–74.
Whitaker AM, Priddy BM, Edwards S, Vendruscolo LF. The role of brain glucocorticoid systems in alcohol dependence. in Neuropathology of Drug Addictions and Substance Misuse 610–617 (Elsevier, 2016). https://doi.org/10.1016/B978-0-12-800213-1.00056-0.
Brancato A, Castelli V, Lavanco G, Tringali G, Micale V, Kuchar M, et al. Binge-like alcohol exposure in adolescence: behavioural, neuroendocrine and molecular evidence of abnormal neuroplasticity… and return. Biomedicines. 2021;9:1161.
Pandey SC, Sakharkar AJ, Tang L, Zhang H. Potential role of adolescent alcohol exposure-induced amygdaloid histone modifications in anxiety and alcohol intake during adulthood. Neurobiol Dis. 2015;82:607–19.
Kyzar EJ, Zhang H, Pandey SC. Adolescent alcohol exposure epigenetically suppresses amygdala arc enhancer RNA expression to confer adult anxiety susceptibility. Biol Psychiatry. 2019;85:904–14.
Bohnsack JP, Zhang H, Wandling GM, He D, Kyzar EJ, Lasek AW, et al. Targeted epigenomic editing ameliorates adult anxiety and excessive drinking after adolescent alcohol exposure. Sci Adv. 2022;8:eabn2748.
Seemiller LR, Gould TJ. The effects of adolescent alcohol exposure on learning and related neurobiology in humans and rodents. Neurobiol Learn Mem. 2020;172:107234.
Lawson K, Scarlata MJ, Cho WC, Mangan C, Petersen D, Thompson HM, et al. Adolescence alcohol exposure impairs fear extinction and alters medial prefrontal cortex plasticity. Neuropharmacology. 2022;211:109048.
Salling MC, Skelly MJ, Avegno E, Regan S, Zeric T, Nichols E, et al. Alcohol consumption during adolescence in a mouse model of binge drinking alters the intrinsic excitability and function of the prefrontal cortex through a reduction in the hyperpolarization-activated cation current. J Neurosci. 2018;38:6207–22.
Fernandes LM, Cartágenes SC, Barros MA, Carvalheiro TC, Castro NC, Schamne MG, et al. Repeated cycles of binge-like ethanol exposure induce immediate and delayed neurobehavioral changes and hippocampal dysfunction in adolescent female rats. Behavioural Brain Res. 2018;350:99–108.
Wei G, Sirohi S, Walker BM. Dysregulated kappa‐opioid receptors in the medial prefrontal cortex contribute to working memory deficits in alcohol dependence. Addict Biol. 2022;27:1–11.
Jury NJ, Pollack GA, Ward MJ, Bezek JL, Ng AJ, Pinard CR, et al. Chronic ethanol during adolescence impacts corticolimbic dendritic spines and behavior. Alcohol Clin Exp Res. 2017;41:1298–308.
Galaj E, Guo C, Huang D, Ranaldi R, Ma Y-Y. Contrasting effects of adolescent and early-adult ethanol exposure on prelimbic cortical pyramidal neurons. Drug Alcohol Depend. 2020;216:108309.
Silva-Gotay A, Davis J, Tavares ER, Richardson HN. Alcohol drinking during early adolescence activates microglial cells and increases frontolimbic Interleukin-1 beta and Toll-like receptor 4 gene expression, with heightened sensitivity in male rats compared to females. Neuropharmacology. 2021;197:108698.
Broadwater MA, Lee SH, Yu Y, Zhu H, Crews FT, Robinson DL, et al. Adolescent alcohol exposure decreases frontostriatal resting-state functional connectivity in adulthood: AIE alters adult functional connectivity. Addiction Biol. 2018;23:810–23.
Tavares ER, Silva-Gotay A, Vargas Riad W, Bengston L, Richardson NH. Sex differences in the effect of alcohol drinking on myelinated axons in the anterior cingulate cortex of adolescent rats. Brain Sci. 2019;9:167.
Lisman J, Buzsáki G, Eichenbaum H, Nadel L, Ranganath C, Redish AD. Viewpoints: how the hippocampus contributes to memory, navigation and cognition. Nat Neurosci. 2017;20:1434–47.
Vetreno RP, Crews FT. Binge ethanol exposure during adolescence leads to a persistent loss of neurogenesis in the dorsal and ventral hippocampus that is associated with impaired adult cognitive functioning. Front. Neurosci. 2015;9.
Wooden JI, Thompson KR, Guerin SP, Nawarawong NN, Nixon K. Consequences of adolescent alcohol use on adult hippocampal neurogenesis and hippocampal integrity. in International Review of Neurobiology vol. 160, 281–304 (Elsevier, 2021).
Mulholland PJ, Teppen TL, Miller KM, Sexton HG, Pandey SC, Swartzwelder HS. Donepezil reverses dendritic spine morphology adaptations and Fmr1 epigenetic modifications in hippocampus of adult rats after adolescent alcohol exposure. Alcohol Clin Exp Res. 2018;42:706–17.
Melbourne JK, Chandler CM, Van Doorn CE, Bardo MT, Pauly JR, Peng H, et al. Primed for addiction: a critical review of the role of microglia in the neurodevelopmental consequences of adolescent alcohol drinking. Alcohol Clin Exp Res. 2021;45:1908–26.
Healey KL, Kibble S, Bell A, Hodges S, Swartzwelder HS. Effects of adolescent intermittent ethanol on hippocampal expression of glutamate homeostasis and astrocyte-neuronal tethering proteins in male and female rats. J Neurosci Res. 2021;99:1908–21.
Testen A, Ali M, Sexton HG, Hodges S, Dubester K, Reissner KJ, et al. Region-specific differences in morphometric features and synaptic colocalization of astrocytes during development. Neuroscience. 2019;400:98–109.
Risher ML, Fleming RL, Risher WC, Miller KM, Klein RC, Wills T, et al. Adolescent intermittent alcohol exposure: persistence of structural and functional hippocampal abnormalities into adulthood. Alcohol Clin Exp Res. 2015;39:989–97.
Nwachukwu KN, King DM, Healey KL, Swartzwelder HS, Marshall SA. Sex-specific effects of adolescent intermittent ethanol exposure-induced dysregulation of hippocampal glial cells in adulthood. Alcohol. 2022;100:31–39.
Healey KL, Kibble S, Hodges S, Reissner KJ, Testen A, Wills TA, et al. Enduring alterations in hippocampal astrocyte-synaptic proximity following adolescent alcohol exposure: reversal by gabapentin. Neural Regen Res. 2020;15:1496.
Swartzwelder HS, Risher ML, Miller KM, Colbran RJ, Winder DG, Wills TA. Changes in the adult GluN2B associated proteome following adolescent intermittent ethanol exposure. PLoS ONE. 2016;11:e0155951.
Swartzwelder HS, Park MH, Acheson S. Adolescent ethanol exposure enhances NMDA receptor-mediated currents in hippocampal neurons: reversal by Gabapentin. Sci Rep. 2017;7:13133.
Kraguljac NV, White DM, Reid MA, Lahti AC. Increased Hippocampal glutamate and volumetric deficits in unmedicated patients with Schizophrenia. JAMA Psychiatry. 2013;70:1294–302.
Bossong MG, Antoniades M, Azis M, Samson C, Quinn B, Bonoldi I, et al. Association of Hippocampal glutamate levels with adverse outcomes in individuals at clinical high risk for psychosis. JAMA Psychiatry. 2019;76:199–207.
Walker CD, Sexton HG, Hyde J, Greene B, Risher ML. Diverging effects of adolescent ethanol exposure on tripartite synaptic development across prefrontal cortex subregions. Cells. 2022;11:3111.
Henriques JF, Portugal CC, Canedo T, Relvas JB, Summavielle T, Socodato R. Microglia and alcohol meet at the crossroads: Microglia as critical modulators of alcohol neurotoxicity. Toxicol Lett. 2018;283:21–31.
Dziabis JE, Bilbo SD. Microglia and sensitive periods in brain development. in Sensitive Periods of Brain Development and Preventive Interventions (ed. Andersen SL) 55–78 (Springer International Publishing, 2022). https://doi.org/10.1007/7854_2021_242.
Peng H, Nixon K. Microglia phenotypes following the induction of alcohol dependence in adolescent rats. Alcohol: Clin Exp Res. 2021;45:105–16.
Hu P, Wang D, Zhang Y, Cai Z, Ye T, Tong L, et al. Apoptosis-triggered decline in hippocampal microglia mediates adolescent intermittent alcohol exposure-induced depression-like behaviors in mice. Neuropharmacology. 2020;170:108054.
Marshall SA, McClain JA, Wooden JI, Nixon K. Microglia dystrophy following binge-like alcohol exposure in adolescent and adult male rats. Front Neuroanatomy. 2020;14:1–11.
Perry VH, Nicoll JAR, Holmes C. Microglia in neurodegenerative disease. Nat Rev Neurol. 2010;6:193–201.
Lu HC, Mackie K. Review of the endocannabinoid system. Biol Psychiatry: Cogn Neurosci Neuroimaging. 2021;6:607–15.
Cristino L, Bisogno T, Di Marzo V. Cannabinoids and the expanded endocannabinoid system in neurological disorders. Nat Rev Neurol. 2020;16:9–29.
Basavarajappa BS, Joshi V, Shivakumar M, Subbanna S. Distinct functions of endogenous cannabinoid system in alcohol abuse disorders. Br J Pharmacol. 2019;176:3085–109.
Agoglia AE, Holstein SE, Eastman VR, Hodge CW. Cannabinoid CB1 receptor inhibition blunts adolescent-typical increased binge alcohol and sucrose consumption in male C57BL/6J mice. Pharmacol Biochem Behav. 2016;143:11–17.
Sánchez-Marín L, Gavito AL, Decara J, Pastor A, Castilla-Ortega E, Suarez J, et al. Impact of intermittent voluntary ethanol consumption during adolescence on the expression of endocannabinoid system and neuroinflammatory mediators. Eur Neuropsychopharmacol. 2020;33:126–38.
Sánchez-Marín L, Flores-López M, Pastor A, Gavito AL, Suárez J, de la Torre R, et al. Acute stress and alcohol exposure during adolescence result in an anxious phenotype in adulthood: role of altered glutamate/endocannabinoid transmission mechanisms. Prog Neuro-Psychopharmacol Biol Psychiatry. 2022;113:110460.
Peñasco S, Rico-Barrio I, Puente N, Fontaine CJ, Ramos A, Reguero L, et al. Intermittent ethanol exposure during adolescence impairs cannabinoid type 1 receptor-dependent long-term depression and recognition memory in adult mice. Neuropsychopharmacol 2020;45:309–18.
Brown SA, Brumback TY, Tomlinson K, Cummins K, Thompson WK, Nagel BJ, et al. The national consortium on alcohol and neuro development in adolescence (NCANDA): a multisite study of adolescent development and substance use. J Stud Alcohol Drugs. 2015;76:895–908.
Squeglia LM, Tapert SF, Sullivan EV, Jacobus J, Meloy MJ, Rohlfing T, et al. Brain development in heavy-drinking adolescents. AJP. 2015;172:531–42.
Luciana M, Collins PF, Muetzel RL, Lim KO. Effects of alcohol use initiation on brain structure in typically developing adolescents. Am J Drug Alcohol Abus. 2013;39:345–55.
Meda SA, Dager AD, Hawkins KA, Tennen H, Raskin S, Wood RM, et al. Heavy drinking in college students is associated with accelerated gray matter volumetric decline over a 2 year period. Front Behav Neurosci. 2017;11:176.
Infante MA, Eberson SC, Zhang Y, Brumback T, Brown SA, Colrain IM, et al. Adolescent binge drinking is associated with accelerated decline of gray matter volume. Cereb Cortex. 2022;32:2611–20.
El Marroun H, Klapwijk ET, Koevoets M, Brouwer RM, Peters S, Van’t Ent D, et al. Alcohol use and brain morphology in adolescence: a longitudinal study in three different cohorts. Eur J Neurosci. 2021;54:6012–26.
Sun D, Adduru VR, Phillips RD, Bouchard HC, Sotiras A, Michael AM, et al. Adolescent alcohol use is linked to disruptions in age-appropriate cortical thinning: an unsupervised machine learning approach. Neuropsychopharmacology. 2022. https://doi.org/10.1038/s41386-022-01457-4.
Zhao Q, Sullivan EV, Honnorat N, Adeli E, Podhajsky S, De Bellis MD, et al. Association of heavy drinking with deviant fiber tract development in frontal brain systems in adolescents. JAMA Psychiatry. 2021;78:407.
Shen Q, Heikkinen N, Kärkkäinen O, Gröhn H, Könönen M, Liu Y, et al. Effects of long-term adolescent alcohol consumption on white matter integrity and their correlations with metabolic alterations. Psychiatry Res: Neuroimaging. 2019;294:111003.
Phillips RD, De Bellis MD, Brumback T, Clausen AN, Clarke-Rubright EK, Haswell CC, et al. Volumetric trajectories of hippocampal subfields and amygdala nuclei influenced by adolescent alcohol use and lifetime trauma. Transl Psychiatry. 2021;11:154.
Almeida-Antunes N, Crego A, Carbia C, Sousa SS, Rodrigues R, Sampaio A, et al. Electroencephalographic signatures of the binge drinking pattern during adolescence and young adulthood: a PRISMA-driven systematic review. NeuroImage: Clin. 2021;29:102537.
Wen H, Hockenberry JM, Druss BG. The effect of medical marijuana laws on marijuana-related attitude and perception among US adolescents and young adults. Prev Sci. 2019;20:215–23.
Hurd YL, Manzoni OJ, Pletnikov MV, Lee FS, Bhattacharyya S, Melis M. Cannabis and the developing brain: insights into its long-lasting effects. J Neurosci. 2019;39:8250–8.
Paschall MJ, García-Ramírez G, Grube JW. Recreational marijuana legalization and use among California adolescents: findings from a statewide survey. J Stud Alcohol Drugs. 2021;82:103–11.
Sarvet AL, Wall MM, Keyes KM, Cerdá M, Schulenberg JE, O’Malley PM, et al. Recent rapid decrease in adolescents’ perception that marijuana is harmful, but no concurrent increase in use. Drug Alcohol Depend. 2018;186:68–74.
Lucas CJ, Galettis P, Schneider J. The pharmacokinetics and the pharmacodynamics of cannabinoids. Br J Clin Pharm. 2018;84:2477–82.
Wilson J, Freeman TP, Mackie CJ. Effects of increasing cannabis potency on adolescent health. Lancet Child Adolesc Health. 2019;3:121–8.
Lim CC, Sun T, Leung J, Chung JY, Gartner C, Connor J, et al. Prevalence of adolescent cannabis vaping: a systematic review and meta-analysis of US and Canadian studies. JAMA Pediatr. 2022;176:42–51.
Bossong MG, Niesink RJM. Adolescent brain maturation, the endogenous cannabinoid system and the neurobiology of cannabis-induced schizophrenia. Prog Neurobiol. 2010;92:370–85.
Levine A, Clemenza K, Rynn M, Lieberman J. Evidence for the risks and consequences of adolescent cannabis exposure. J Am Acad Child Adolesc Psychiatry. 2017;56:214–25.
Murray CH, Huang Z, Lee R, de Wit H. Adolescents are more sensitive than adults to acute behavioral and cognitive effects of THC. Neuropsychopharmacology. 2022;47:1331–8.
Malone DT, Hill MN, Rubino T. Adolescent cannabis use and psychosis: epidemiology and neurodevelopmental models. Br J Pharmacol. 2010;160:511–22.
Mustonen A, Niemelä S, Nordström T, Murray GK, Mäki P, Jääskeläinen E, et al. Adolescent cannabis use, baseline prodromal symptoms and the risk of psychosis. Br J Psychiatry. 2018;212:227–33.
Patel PK, Leathem LD, Currin DL, Karlsgodt KH. Adolescent neurodevelopment and vulnerability to psychosis. Biol Psychiatry. 2021;89:184–93.
Penzel N, Antonucci LA, Betz LT, Sanfelici R, Weiske J, Pogarell O, et al. Association between age of cannabis initiation and gray matter covariance networks in recent onset psychosis. Neuropsychopharmacology. 2021;46:1484–93.
Lisboa SF, Gomes FV, Guimaraes FS., Campos AC. Microglial cells as a link between cannabinoids and the immune hypothesis of psychiatric disorders. Front Neurol. 2016;7:1–8.
Howes OD, McCutcheon R. Inflammation and the neural diathesis-stress hypothesis of schizophrenia: a reconceptualization. Transl Psychiatry. 2017;7:e1024–4.
Lee HL, Jung KM, Fotio Y, Squire E, Palese F, Lin L, et al. Frequent low-dose Δ9-Tetrahydrocannabinol in adolescence disrupts microglia homeostasis and disables responses to microbial infection and social stress in young adulthood. Biol Psychiatry. 2022;92:845–60.
Wan X, Eguchi A, Qu Y, Yang Y, Chang L, Shan J, et al. Gut–microbiota–brain axis in the vulnerability to psychosis in adulthood after repeated cannabis exposure during adolescence. Eur Arch Psychiatry Clin Neurosci. 2022;272:1297–309.
Schafer DP, Lehrman EK, Kautzman AG, Koyama R, Mardinly AR, Yamasaki R, et al. Microglia sculpt postnatal neural circuits in an activity and complement-dependent manner. Neuron. 2012;74:691–705.
Kiburi SK, Molebatsi K, Ntlantsana V, Lynskey MT. Cannabis use in adolescence and risk of psychosis: are there factors that moderate this relationship? A systematic review and meta-analysis. Subst Abus. 2021;42:527–42.
Jones HJ, Hammerton G, McCloud T, Hines LA, Wright C, Gage SH, et al. Examining pathways between genetic liability for schizophrenia and patterns of tobacco and cannabis use in adolescence. Psychol Med. 2022;52:132–9.
Renard J, Rushlow WJ, Laviolette SR. Effects of adolescent THC exposure on the prefrontal GABAergic system: implications for schizophrenia-related psychopathology. Front Psychiatry. 2018;9:281.
Lewis DA. GABAergic local circuit neurons and prefrontal cortical dysfunction in schizophrenia. Brain Res Rev. 2000;31:270–6.
Renard J, Szkudlarek HJ, Kramar CP, Jobson CE, Moura K, Rushlow WJ, et al. Adolescent THC exposure causes enduring prefrontal cortical disruption of GABAergic inhibition and dysregulation of sub-cortical dopamine function. Sci Rep. 2017;7:11420.
Zamberletti E, Beggiato S, Steardo L Jr, Prini P, Antonelli T, Ferraro L, et al. Alterations of prefrontal cortex GABAergic transmission in the complex psychotic-like phenotype induced by adolescent delta-9-tetrahydrocannabinol exposure in rats. Neurobiol Dis. 2014;63:35–47.
Subramaniam P, Prescot A, McGlade E, Renshaw P, Yurgelun-Todd D. Examination of gamma-aminobutyric acid and glutamate-glutamine levels in association with impulsive behavior in adolescent marijuana users. Drug Alcohol Depend. 2022;233:109326.
Gabaglio M, Zamberletti E, Manenti C, Parolaro D, Rubino T. Long-term consequences of adolescent exposure to THC-Rich/CBD-Poor and CBD-Rich/THC-Poor combinations: a comparison with pure THC treatment in female rats. Int J Mol Sci. 2021;22:8899.
Volkow ND, Morales M. The brain on drugs: from reward to addiction. Cell. 2015;162:712–25.
Renard J, Rosen LG, Loureiro M, De Oliveira C, Schmid S, Rushlow WJ, et al. Adolescent cannabinoid exposure induces a persistent sub-cortical hyper-dopaminergic state and associated molecular adaptations in the prefrontal cortex. Cereb Cortex. bhv335; 2016. https://doi.org/10.1093/cercor/bhv335.
Scherma M, Dessì C, Muntoni AL, Lecca S, Satta V, Luchicchi A, et al. Adolescent Δ9-Tetrahydrocannabinol exposure alters WIN55,212-2 self-administration in adult rats. Neuropsychopharmacol. 2016;41:1416–26.
Mátyás F, Urbán GM, Watanabe M, Mackie K, Zimmer A, Freund TF, et al. Identification of the sites of 2-arachidonoylglycerol synthesis and action imply retrograde endocannabinoid signaling at both GABAergic and glutamatergic synapses in the ventral tegmental area. Neuropharmacology. 2008;54:95–107.
Kruse LC, Cao JK, Viray K, Stella N, Clark JJ. Voluntary oral consumption of Δ9-tetrahydrocannabinol by adolescent rats impairs reward-predictive cue behaviors in adulthood. Neuropsychopharmacol. 2019;44:1406–14.
Le AA, Quintanilla J, Amani M, Piomelli D, Lynch G, Gall CM. Persistent sexually dimorphic effects of adolescent THC exposure on hippocampal synaptic plasticity and episodic memory in rodents. Neurobiol Dis. 2022;162:105565.
Ruiz CM, Torrens A, Lallai V, Castillo E, Manca L, Martinez MX, et al. Pharmacokinetic and pharmacodynamic properties of aerosolized (‘vaped’) THC in adolescent male and female rats. Psychopharmacol (Berl). 2021;238:3595–605.
Torrens A, Vozella V, Huff H, McNeil B, Ahmed F, Ghidini A, et al. Comparative pharmacokinetics of Δ9-Tetrahydrocannabinol in adolescent and adult male mice. J Pharm Exp Ther. 2020;374:151–60.
Zuo Y, Iemolo A, Montilla-Perez P, Li HR, Yang X, Telese F. Chronic adolescent exposure to cannabis in mice leads to sex-biased changes in gene expression networks across brain regions. Neuropsychopharmacology. 2022;47:2071–80.
Orihuel J, Capellán R, Roura-Martínez D, Ucha M, Ambrosio E, Higuera-Matas A. Δ 9-Tetrahydrocannabinol during adolescence reprograms the nucleus accumbens transcriptome, affecting reward processing, impulsivity, and specific aspects of cocaine addiction-like behavior in a sex-dependent manner. Int J Neuropsychopharmacol. 2021;24:920–33.
Ginder DE, Wright HR, McLaughlin RJ. The stoned age: sex differences in the effects of adolescent cannabinoid exposure on prefrontal cortex structure and function in animal models. Int Rev Neurobiol. 2021;161:121–45.
Lorenzetti V, Hoch E, Hall W. Adolescent cannabis use, cognition, brain health and educational outcomes: a review of the evidence. Eur Neuropsychopharmacol. 2020;36:169–80.
Chen H-T, Mackie K. Adolescent Δ9-Tetrahydrocannabinol exposure selectively impairs working memory but not several other mPFC-mediated behaviors. Front Psychiatry. 2020;11:576214.
Withey SL, et al. Effects of daily Δ9-Tetrahydrocannabinol (THC) alone or combined with cannabidiol (CBD) on cognition-based behavior and activity in adolescent nonhuman primates. Drug Alcohol Depend. 2021;221:108629.
Verrico CD, Gu H, Peterson ML, Sampson AR, Lewis DA. Repeated Δ9-tetrahydrocannabinol exposure in adolescent monkeys: persistent effects selective for spatial working memory. Am J Psychiatry. 2014;171:416–25.
Schaefer JD, Hamdi NR, Malone SM, Vrieze S, Wilson S, McGue M, et al. Associations between adolescent cannabis use and young-adult functioning in three longitudinal twin studies. Proc Natl Acad Sci USA. 2021;118:e2013180118.
Meier MH, Caspi A, Ambler A, Harrington H, Houts R, Keefe RS, et al. Persistent cannabis users show neuropsychological decline from childhood to midlife. Proc Natl Acad Sci USA. 2012;109.
Scott JC, Slomiak ST, Jones JD, Rosen AF, Moore TM, Gur RC. Association of cannabis with cognitive functioning in adolescents and young adults: a systematic review and meta-analysis. JAMA Psychiatry. 2018;75:585–95.
Broyd SJ, van Hell HH, Beale C, Yücel M, Solowij N. Acute and chronic effects of cannabinoids on human cognition—a systematic review. Biol Psychiatry. 2016;79:557–67.
Mokrysz C, Landy R, Gage SH, Munafo MR, Roiser JP, Curran HV. Are IQ and educational outcomes in teenagers related to their cannabis use? A prospective cohort study. J Psychopharmacol. 2016;30:159–68.
Cyrus E, Coudray MS, Kiplagat S, Mariano Y, Noel I, Galea JT. A review investigating the relationship between cannabis use and adolescent cognitive functioning. Curr Opin Psychol. 2021;38:38–48.
Castellanos-Ryan N, Pingault JB, Parent S, Vitaro F, Tremblay RE, Seguin JR. Adolescent cannabis use, change in neurocognitive function, and high-school graduation: a longitudinal study from early adolescence to young adulthood. Dev Psychopathol. 2017;29:1253–66.
Albaugh MD, Ottino-Gonzalez J, Sidwell A, Lepage C, Juliano A, Owens MM. Association of cannabis use during adolescence with neurodevelopment. JAMA Psychiatry. 2021;78:1031.
Owens MM, Albaugh MD, Allgaier N, Yuan D, Robert G, Cupertino RB, et al. Bayesian causal network modeling suggests adolescent cannabis use accelerates prefrontal cortical thinning. Transl Psychiatry. 2022;12:188.
Lichenstein SD, Manco N, Cope LM, Egbo L, Garrison KA, Hardee J, et al. Systematic review of structural and functional neuroimaging studies of cannabis use in adolescence and emerging adulthood: evidence from 90 studies and 9441 participants. Neuropsychopharmacology. 2022;47:1000–28.
Lorenzetti V, Kowalczyk M, Duehlmeyer L, Greenwood LM, Chye Y, Yücel M, et al. Brain anatomical alterations in young cannabis users: is it all hype? A meta-analysis of structural neuroimaging studies. Cannabis Cannabinoid Res. 2022. https://doi.org/10.1089/can.2021.0099.
Prini P, Penna F, Sciuccati E, Alberio T, Rubino T. Chronic Δ8-THC exposure differently affects histone modifications in the adolescent and adult rat brain. Int J Mol Sci. 2017;18:2094.
Prini P, Rusconi F, Zamberletti E, Gabaglio M, Penna F, Fasano M, et al. Adolescent THC exposure in female rats leads to cognitive deficits through a mechanism involving chromatin modifications in the prefrontal cortex. J Psychiatry Neurosci. 2018;43:87–101.
Miller ML, Chadwick B, Dickstein DL, Purushothaman I, Egervari G, Rahman T, et al. Adolescent exposure to Δ9-tetrahydrocannabinol alters the transcriptional trajectory and dendritic architecture of prefrontal pyramidal neurons. Mol Psychiatry. 2019;24:588–600.
Leishman E, Murphy M, Mackie K, Bradshaw HB. Δ9-Tetrahydrocannabinol changes the brain lipidome and transcriptome differentially in the adolescent and the adult. Biochim Biophys Acta (BBA) - Mol Cell Biol Lipids. 2018;1863:479–92.
Clark SL, et al. Methylomic investigation of problematic adolescent cannabis use and its negative mental health consequences. J Am Acad Child Adolesc Psychiatry. 2021;60:1524–32.
Stringfield SJ, Torregrossa MM. Intravenous self-administration of delta-9-THC in adolescent rats produces long-lasting alterations in behavior and receptor protein expression. Psychopharmacol (Berl). 2021;238:305–19.
Kirschmann EK, Pollock MW, Nagarajan V, Torregrossa MM. Effects of adolescent cannabinoid self-administration in rats on addiction-related behaviors and working memory. Neuropsychopharmacol. 2017a;42:989–1000.
Kirschmann EK, McCalley DM, Edwards CM, Torregrossa MM. Consequences of adolescent exposure to the cannabinoid receptor agonist WIN55,212-2 on working memory in female rats. Front. Behavioral Neurosci. 2017b;11.
Bruijnzeel AW, Knight P, Panunzio S, Xue S, Bruner MM, Wall SC, et al. Effects in rats of adolescent exposure to cannabis smoke or THC on emotional behavior and cognitive function in adulthood. Psychopharmacol (Berl). 2019;236:2773–84.
Hernandez CM, Orsini CA, Blaes SL, Bizon JL, Febo M, Bruijnzeel AW, et al. Effects of repeated adolescent exposure to cannabis smoke on cognitive outcomes in adulthood. J Psychopharmacol. 2021;35:848–63.
Skumlien M, Mokrysz C, Freeman TP, Valton V, Wall MB, Bloomfield M, et al. Anhedonia, apathy, pleasure, and effort-based decision-making in adult and adolescent cannabis users and controls. Int J Neuropsychopharmacol. 2022a;pyac056. https://doi.org/10.1093/ijnp/pyac056.
Skumlien M, Mokrysz C, Freeman TP, Wall MB, Bloomfield M, Lees R, et al. Neural responses to reward anticipation and feedback in adult and adolescent cannabis users and controls. Neuropsychopharmacology. 2022b;47:1976–83.
Cullen KA, et al. Flavored tobacco product use among middle and high school students - United States, 2014–2018. MMWR Morb Mortal Wkly Rep. 2019;68:839–44.
Miech R, Johnston L, O’Malley PM, Bachman JG, Patrick ME. Adolescent vaping and nicotine use in 2017–2018 - U.S. National Estimates. N. Engl J Med. 2019;380:192–3.
Carroll BJ, et al. Impaired lung function following e‐cigarette or vaping product use associated lung injury in the first cohort of hospitalized adolescents. Pediatr Pulmonol. 2020;55:1712–8.
Tang MS, Wu XR, Lee HW, Xia Y, Deng FM, Moreira AL, et al. Electronic-cigarette smoke induces lung adenocarcinoma and bladder urothelial hyperplasia in mice. Proc Natl Acad Sci USA. 2019;116:21727–31.
Boden JM, Fergusson DM, Horwood LJ. Cigarette smoking and depression: tests of causal linkages using a longitudinal birth cohort. Br J Psychiatry. 2010;196:440–6.
Jobson CL, Renard J, Szkudlarek H, Rosen LG, Pereira B, Wright DJ, et al. Adolescent nicotine exposure induces dysregulation of mesocorticolimbic activity states and depressive and anxiety-like prefrontal cortical molecular phenotypes persisting into adulthood. Cereb Cortex. 2019;29:3140–53.
Moylan S, Jacka FN, Pasco JA, Berk M. How cigarette smoking may increase the risk of anxiety symptoms and anxiety disorders: a critical review of biological pathways. Brain Behav. 2013;3:302–26.
Counotte DS, Spijker S, Van de Burgwal LH, Hogenboom F, Schoffelmeer AN, De Vries TJ, et al. Long-lasting cognitive deficits resulting from adolescent nicotine exposure in rats. Neuropsychopharmacology. 2009;34:299–306.
Spaeth AM, Barnet RC, Hunt PS, Burk JA. Adolescent nicotine exposure disrupts context conditioning in adulthood in rats. Pharmacol Biochem Behav. 2010;96:501–6.
Jacobsen LK, Mencl WE, Constable RT, Westerveld M, Pugh KR. Impact of smoking abstinence on working memory neurocircuitry in adolescent daily tobacco smokers. Psychopharmacol (Berl). 2007;193:557–66.
Falcone M, Wileyto EP, Ruparel K, Gerraty RT, LaPrate L, Detre JA, et al. Age-related differences in working memory deficits during nicotine withdrawal: Nicotine and working memory. Addiction Biol. 2014;19:907–17.
Kinnunen JM, Ollila H, Minkkinen J, Lindfors PL, Timberlake DS, Rimpelä AH, et al. Nicotine matters in predicting subsequent smoking after e-cigarette experimentation: a longitudinal study among Finnish adolescents. Drug Alcohol Depend. 2019;201:182–7.
Dierker L, Braymiller J, Rose J, Goodwin R, Selya A. Nicotine dependence predicts cannabis use disorder symptoms among adolescents and young adults. Drug Alcohol Depend. 2018;187:212–20.
Kristjansson AL, Mann MJ, Sigfusdottir ID. Licit and illicit substance use by adolescent e-cigarette users compared with conventional cigarette smokers, dual users, and nonusers. J Adolesc Health. 2015;57:562–4.
Cardenas A, Lotfipour S. Age- and sex-dependent nicotine pretreatment effects on the enhancement of methamphetamine self-administration in Sprague-Dawley Rats. Nicotine Tob Res. 2022;24:1186–92.
Locker AR, Marks MJ, Kamens HM, Klein LC. Exposure to nicotine increases nicotinic acetylcholine receptor density in the reward pathway and binge ethanol consumption in C57BL/6J adolescent female mice. Brain Res Bull. 2016;123:13–22.
Cardenas A, Martinez M, Saenz Mejia A, Lotfipour S. Early adolescent subchronic low-dose nicotine exposure increases subsequent cocaine and fentanyl self-administration in Sprague-Dawley rats. Behav Pharm. 2021;32:86–91.
Reed SC, Izenwasser S. Nicotine produces long-term increases in cocaine reinforcement in adolescent but not adult rats. Brain Res. 2017;1654:165–70.
Laviolette SR. Molecular and neuronal mechanisms underlying the effects of adolescent nicotine exposure on anxiety and mood disorders. Neuropharmacology. 2021;184:108411.
Leslie FM. Unique, long-term effects of nicotine on adolescent brain. Pharm Biochem Behav. 2020;197:173010.
Adriani W, et al. Evidence for enhanced neurobehavioral vulnerability to nicotine during periadolescence in rats. J Neurosci 2003;23:4712–6.
Doura MB, Gold AB, Keller AB, Perry DC. Adult and periadolescent rats differ in expression of nicotinic cholinergic receptor subtypes and in the response of these subtypes to chronic nicotine exposure. Brain Res. 2008;1215:40–52.
Corongiu S, Dessì C, Cadoni C. Adolescence versus adulthood: Differences in basal mesolimbic and nigrostriatal dopamine transmission and response to drugs of abuse. Addict Biol. 2020;25:e12721.
Lenoir M, Starosciak AK, Ledon J, Booth C, Zakharova E, Wade D, et al. Sex differences in conditioned nicotine reward are age-specific. Pharmacol Biochem Behav. 2015;132:56–62.
Cao J, Belluzzi JD, Loughlin SE, Dao JM, Chen Y, Leslie FM. Locomotor and stress responses to nicotine differ in adolescent and adult rats. Pharmacol Biochem Behav. 2010;96:82–90.
Renda A, Penty N, Komal P, Nashmi R. Vulnerability to nicotine self-administration in adolescent mice correlates with age-specific expression of α4* nicotinic receptors. Neuropharmacology. 2016;108:49–59.
Garrison KA, Yip SW, Balodis IM, Carroll KM, Potenza MN, Krishnan-Sarin S. Reward-related frontostriatal activity and smoking behavior among adolescents in treatment for smoking cessation. Drug Alcohol Depend. 2017;177:268–76.
Schassburger RL, Pitzer EM, Smith TT, Rupprecht LE, Thiels E, Donny EC, et al. Adolescent rats self-administer less nicotine than adults at low doses. Nicotine Tob Res. 2016;18:1861–8.
Weaver MT, Geier CF, Levin ME, Caggiula AR, Sved AF, Donny EC. Adolescent exposure to nicotine results in reinforcement enhancement but does not affect adult responding in rats. Drug Alcohol Depend. 2012;125:307–12.
Holliday ED, Nucero P, Kutlu MG, Oliver C, Connelly KL, Gould TJ, et al. Long-term effects of chronic nicotine on emotional and cognitive behaviors and hippocampus cell morphology in mice: comparisons of adult and adolescent nicotine exposure. Eur J Neurosci. 2016;44:2818–28.
de la Peña JB, Ahsan HM, Botanas CJ, Dela Pena IJ, Woo T, Kim HJ, et al. Cigarette smoke exposure during adolescence but not adulthood induces anxiety-like behavior and locomotor stimulation in rats during withdrawal. Int J Dev Neurosci. 2016;55:49–55.
Hudson R, Green M, Wright DJ, Renard J, Jobson CE, Jung T, et al. Adolescent nicotine induces depressive and anxiogenic effects through ERK 1‐2 and Akt‐GSK‐3 pathways and neuronal dysregulation in the nucleus accumbens. Addict Biol. 2021;26.
Nikolaus S, Mamlins E, Hautzel H, Müller H-W. Acute anxiety disorder, major depressive disorder, bipolar disorder and schizophrenia are related to different patterns of nigrostriatal and mesolimbic dopamine dysfunction. Rev Neurosci. 2019;30:381–26.
Berg L, Eckardt J, Masseck OA. Enhanced activity of pyramidal neurons in the infralimbic cortex drives anxiety behavior. PLOS ONE. 2019;14:1–19.
Ailing F, Fan L, Li S, Manji S. Role of extracellular signal-regulated kinase signal transduction pathway in anxiety. J Psychiatr Res. 2008;43:55–63.
Beyer DKE, Mattukat A, Freund N. Prefrontal dopamine D1 receptor manipulation influences anxiety behavior and induces neuroinflammation within the hippocampus. Int J Bipolar Disord. 2021;9:9.
Jacobsen LK, Krystal JH, Mencl WE, Westerveld M, Frost SJ, Pugh KR. Effects of smoking and smoking abstinence on cognition in adolescent tobacco smokers. Biol Psychiatry. 2005;57:56–66.
Musso F, Bettermann F, Vucurevic G, Stoeter P, Konrad A, Winterer G, et al. Smoking impacts on prefrontal attentional network function in young adult brains. Psychopharmacology. 2007;191:159–69.
Counotte DS, Goriounova NA, Li KW, Loos M, van Der Schors RC, Schetters D, et al. Lasting synaptic changes underlie attention deficits caused by nicotine exposure during adolescence. Nat Neurosci. 2011;14:417–9.
Goriounova NA, Mansvelder HD. Nicotine exposure during adolescence leads to short- and long-term changes in spike timing-dependent plasticity in rat prefrontal cortex. J Neurosci. 2012;32:10484–93.
Connor DA, Gould TJ. Chronic fluoxetine ameliorates adolescent chronic nicotine exposure-induced long-term adult deficits in trace conditioning. Neuropharmacology. 2017;125:272–83.
Portugal GS, Wilkinson DS, Turner JR, Blendy JA, Gould TJ. Developmental effects of acute, chronic, and withdrawal from chronic nicotine on fear conditioning. Neurobiol Learn Mem. 2012;97:482–94.
Gitik M, Holliday ED, Leung M, Yuan Q, Logue SF, Tikkanen R, et al. Choline ameliorates adult learning deficits and reverses epigenetic modification of chromatin remodeling factors related to adolescent nicotine exposure. Neurobiol Learn Mem. 2018;155:239–48.
Treur JL, Willemsen G, Bartels M, Geels LM, van Beek JH, Huppertz C, et al. Smoking during adolescence as a risk factor for attention problems. Biol Psychiatry. 2015;78:656–63.
Rigsby DC, Keim SA, Milanaik R, Adesman A. Electronic vapor product use and sexual risk behaviors in US adolescents. Pediatrics. 2021;147:e2020022533.
Primack BA, Soneji S, Stoolmiller M, Fine MJ, Sargent JD. Progression to traditional cigarette smoking after electronic cigarette use among US adolescents and young adults. JAMA Pediatr. 2015;169:1018–23.
Dai HD, Doucet GE, Wang Y, Puga T, Samson K, Xiao P, et al. Longitudinal assessments of neurocognitive performance and brain structure associated with initiation of tobacco use in children, 2016 to 2021. JAMA Netw Open. 2022;5:e2225991.
Chaarani B, Kan KJ, Mackey S, Spechler PA, Potter A, Orr C, et al. Low smoking exposure, the adolescent brain, and the modulating role of CHRNA5 polymorphisms. Biol Psychiatry: Cogn Neurosci Neuroimaging. 2019;4:672–9.
Yu D, Yuan K, Cheng J, Guan Y, Li Y, Bi Y, et al. Reduced thalamus volume may reflect nicotine severity in young male smokers. Nicotine Tob Res. 2018;20:434–9.
Linker KE, Gad M, Tawadrous P, Cano M, Green KN, Wood MA, et al. Microglial activation increases cocaine self-administration following adolescent nicotine exposure. Nat Commun. 2020;11:306.
Liu F, Tao X, Pang G, Wu D, Hu Y, Xue S, et al. Maternal nicotine exposure during gestation and lactation period affects behavior and hippocampal neurogenesis in mouse offspring. Front Pharmacol. 2020;10.
Zelikoff JT, Parmalee NL, Corbett K, Gordon T, Klein CB, Aschner M. Microglia activation and gene expression alteration of neurotrophins in the hippocampus following early-life exposure to E-cigarette aerosols in a murine model. Toxicolog Sci. 2018;162:276–86.
Cross SJ, Linker KE, Leslie FM. Sex-dependent effects of nicotine on the developing brain. J Neurosci Res. 2017;95:422–36.
Xue S, Behnood-Rod A, Wilson R, Wilks I, Tan S, Bruijnzeel AW. Rewarding effects of nicotine in adolescent and adult male and female rats as measured using intracranial self-stimulation. Nicotine Tob Res. 2020;22:172–9.
Chellian R, Behnood-Rod A, Wilson R, Kamble SH, Sharma A, McCurdy CR, et al. Adolescent nicotine and tobacco smoke exposure enhances nicotine self-administration in female rats. Neuropharmacology. 2020;176:108243.
Sylvestre MP, Chagnon M, Wellman RJ, Dugas EN, O’Loughlin J. Sex differences in attaining cigarette smoking and nicotine dependence milestones among novice smokers. Am J Epidemiol. 2018;187:1670–77.
Duan Z, Wang Y, Huang J. Sex difference in the association between electronic cigarette use and subsequent cigarette smoking among U.S. adolescents: findings from the PATH study waves 1–4. Int J Environ Res Public Health. 2021;18:1695.
Banks DE, Rowe AT, Mpofu P, Zapolski TCB. Trends in typologies of concurrent alcohol, marijuana, and cigarette use among US adolescents: an ecological examination by sex and race/ethnicity. Drug Alcohol Depend. 2017;179:71–7.
Crummy EA, O’Neal TJ, Baskin BM, Ferguson SM. One is not enough: understanding and modeling polysubstance use. Front Neurosci. 2020;14:569.
Roche DJO, Bujarski S, Green R, Hartwell EE, Leventhal AM, Ray LA. Alcohol, tobacco, and marijuana consumption is associated with increased odds of same-day substance co- and tri-use. Drug Alcohol Depend. 2019;200:40–9.
Karoly HC, Ross JM, Ellingson JM, Feldstein Ewing SW. Exploring cannabis and alcohol co-use in adolescents: a narrative review of the evidence. J Dual Diagnosis. 2020;16:58–74.
Hershberger A, Argyriou E, Cyders M. Electronic nicotine delivery system use is related to higher odds of alcohol and marijuana use in adolescents: meta-analytic evidence. Addict Behav. 2020;105:106325.
Staff J, Kelly BC, Maggs JL, Vuolo M. Adolescent electronic cigarette use and tobacco smoking in the Millennium Cohort Study. Addiction. 2022;117:484–94.
Frie JA, Nolan CJ, Murray JE, Khokhar JY. Addiction-related outcomes of nicotine and alcohol co-use: new insights following the rise in vaping. Nicotine Tob Res. 2022;24:1141–9.
Hamidullah S, Lutelmowski CD, Creighton SD, Luciani KR, Frie JA, Winters BD, et al. Effects of vapourized THC and voluntary alcohol drinking during adolescence on cognition, reward, and anxiety-like behaviours in rats. Prog Neuropsychopharmacol Biol Psychiatry. 2021;106:110141.
Risso C, Boniface S, Subbaraman MS, Englund A. Does cannabis complement or substitute alcohol consumption? A systematic review of human and animal studies. J Psychopharmacol. 2020;34:938–54.
Smiley CE, Saleh HK, Nimchuk KE, Garcia-Keller C, Gass JT. Adolescent exposure to delta-9-tetrahydrocannabinol and ethanol heightens sensitivity to fear stimuli. Behavioural Brain Res. 2021;415:113517.
Luo X, Yang JJ, Buu A, Trucco EM, Li CR. Alcohol and cannabis co‐use and longitudinal gray matter volumetric changes in early and late adolescence. Addict Biol. 2022;27.
Infante MA, Nguyen-Louie TT, Worley M, Courtney KE, Coronado C, Jacobus J. Neuropsychological trajectories associated with adolescent alcohol and cannabis use: a prospective 14-year study. J Int Neuropsychol Soc. 2020;26:480–91.
Thayer RE, YorkWilliams S, Karoly HC, Sabbineni A, Ewing SF, Bryan AD, et al. Structural neuroimaging correlates of alcohol and cannabis use in adolescents and adults: neuroimaging of alcohol and cannabis use. Addiction. 2017;112:2144–54.
Gilbert PA, Kava CM, Afifi R. High-school students rarely use e-cigarettes alone: a sociodemographic analysis of polysubstance use among adolescents in the United States. Nicotine Tob Res. 2021;23,505–10.
Zuckermann AM, Williams G, Battista K, de Groh M, Jiang Y, Leatherdale ST, et al. Trends of poly-substance use among Canadian youth. Addictive Behav Rep. 2019;10:100189.
Lárraga A, Belluzzi JD, Leslie FM. Nicotine increases alcohol intake in adolescent male rats. Front. Behav. Neurosci. 2017;11.
Cross SJ, Leslie FM. Combined nicotine and ethanol age-dependently alter neural and behavioral responses in male rats. Behavioural Pharmacol. 2021;32:321–34.
Ruffolo J, Frie JA, Thorpe HHA, Talhat MA, Khokhar JY. Alcohol and vaporized nicotine co-exposure during adolescence contribute differentially to sex-specific behavioral effects in adulthood. Nicotine Tob Res. 2022;24:1177–85.
Moustafa AF, Rodriguez D, Pianin SH, Testa SM, Audrain-McGovern JE. Dual use of nicotine and cannabis through vaping among adolescents. Am J Preventive Med. 2022;63:60–7.
Dukes AJ, Fowler JP, Lallai V, Pushkin AN, Fowler CD. Adolescent cannabinoid and nicotine exposure differentially alters adult nicotine self-administration in males and females. Nicotine Tob Res. 2020;22:1364–73.
Courtney KE, Sorg S, Baca R, Doran N, Jacobson A, Liu TT, et al. The effects of nicotine and cannabis co-use during late adolescence on white matter fiber tract microstructure. J Stud Alcohol Drugs. 2022;83:287–95.
Brody AL, Hubert R, Mamoun MS, Enoki R, Garcia LY, Abraham P, et al. Nicotinic acetylcholine receptor availability in cigarette smokers: effect of heavy caffeine or marijuana use. Psychopharmacol (Berl). 2016;233:3249–57.
Barker JM, Taylor JR. Habitual alcohol seeking: modeling the transition from casual drinking to addiction. Neurosci Biobehav Rev. 2014;47:281–94.
Koob GF. Drug addiction: hyperkatifeia/negative reinforcement as a framework for medications development. Pharm Rev. 2021;73:163–201.
Coller JK, Hutchinson MR. Implications of central immune signaling caused by drugs of abuse: mechanisms, mediators and new therapeutic approaches for prediction and treatment of drug dependence. Pharmacol Therapeutics. 2012;134:219–45.
Guerri C, Pascual M. Impact of neuroimmune activation induced by alcohol or drug abuse on adolescent brain development. Int J Dev Neurosci. 2019;77:89–98.
Pascual M, Montesinos J, Guerri C. Role of the innate immune system in the neuropathological consequences induced by adolescent binge drinking. J Neurosci Res. 2018;96:765–80.
Linker KE, Cross SJ, Leslie FM. Glial mechanisms underlying substance use disorders. Eur J Neurosci. 2019;50:2574–89.
Namba MD, Leyrer-Jackson JM, Nagy EK, Olive MF, Neisewander JL. Neuroimmune mechanisms as novel treatment targets for substance use disorders and associated comorbidities. Front Neurosci. 2021;15:650785.
Deschamps C, Uyttersprot F, Debris M, Marié C, Fouquet G, Marcq I, et al. Anti-inflammatory drugs prevent memory and hippocampal plasticity deficits following initial binge-like alcohol exposure in adolescent male rats. Psychopharmacology. 2022;239:2245–62.
Monleón S, Gómez J, Duque A, Vinader-Caerols C. Effects of binge drinking and the anti-inflammatory drug indomethacin on spatial memory and cognitive flexibility in mice. Behavioural Brain Res. 2022;417:113619.
Spanagel R. Cannabinoids and the endocannabinoid system in reward processing and addiction: from mechanisms to interventions. Dialogues Clin Neurosci. 2020;22:241–50.
Brancato A, Castelli V, Lavanco G, D’Amico C, Feo S, Pizzolanti G, et al. Social stress under binge-like alcohol withdrawal in adolescence: evidence of cannabidiol effect on maladaptive plasticity in rats. Psychol Med. 2022:1–13.
Capano A, Weaver R, Burkman E. Evaluation of the effects of CBD hemp extract on opioid use and quality of life indicators in chronic pain patients: a prospective cohort study. Postgrad Med. 2020;132:56–61.
Nock NL, Minnes S, Alberts JL. Neurobiology of substance use in adolescents and potential therapeutic effects of exercise for prevention and treatment of substance use disorders. Birth Defects Res. 2017;109:1711–29.
Vetreno RP, Lawrimore CJ, Rowsey PJ, Crews FT. Persistent adult neuroimmune activation and loss of hippocampal neurogenesis following adolescent ethanol exposure: blockade by exercise and the anti-inflammatory drug indomethacin. Front Neurosci 2018;12:200.
Vetreno RP, Bohnsack JP, Kusumo H, Liu W, Pandey SC, Crews FT. Neuroimmune and epigenetic involvement in adolescent binge ethanol‐induced loss of basal forebrain cholinergic neurons: restoration with voluntary exercise. Addict Biol. 2020;25.
Brown AP, Dinger N, Levine BS. Stress produced by gavage administration in the rat. J Am Assoc Lab Anim Sci. 2000;39:17–21.
Sengupta P. The laboratory rat: relating its age with human’s. Int J Prev Med. 2013;4:624–30.
Friend L, Weed J, Sandoval P, Nufer T, Ostlund I, Edwards JG. CB1-dependent long-term depression in ventral tegmental area GABA neurons: a novel target for marijuana. J Neurosci. 2017;37:10943–54.
Zamberletti E, Gabaglio M, Grilli M, Prini P, Catanese A, Pittaluga A, et al. Long-term hippocampal glutamate synapse and astrocyte dysfunctions underlying the altered phenotype induced by adolescent THC treatment in male rats. Pharmacol Res. 2016;111:459–70.
Author information
Authors and Affiliations
Contributions
MS conceptualized the manuscript and wrote the initial copies. MT edited the manuscript. Both authors approved the finals manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Steinfeld, M.R., Torregrossa, M.M. Consequences of adolescent drug use. Transl Psychiatry 13, 313 (2023). https://doi.org/10.1038/s41398-023-02590-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41398-023-02590-4