Abstract
Resilience in the face of major life stressors is changeable over time and with experience. Accordingly, differing sets of neurobiological factors may contribute to an adaptive stress response before, during, and after the stressor. Longitudinal studies are therefore particularly effective in answering questions about the determinants of resilience. Here we provide an overview of the rapidly-growing body of longitudinal neuroimaging research on stress resilience. Despite lingering gaps and limitations, these studies are beginning to reveal individual differences in neural circuit structure and function that appear protective against the emergence of future psychopathology following a major life stressor. Here we outline a neural circuit model of resilience to trauma. Specifically, pre-trauma biomarkers of resilience show that an ability to modulate activity within threat and salience networks predicts fewer stress-related symptoms. In contrast, early post-trauma biomarkers of subsequent resilience or recovery show a more complex pattern, spanning a number of major circuits including attention and cognitive control networks as well as primary sensory cortices. This novel synthesis suggests stress resilience may be scaffolded by stable individual differences in the processing of threat cues, and further buttressed by post-trauma adaptations to the stressor that encompass multiple mechanisms and circuits. More attention and resources supporting this work will inform the targets and timing of mechanistic resilience-boosting interventions.
Similar content being viewed by others
Introduction
Resilience has been defined as the set of complex and dynamic processes that allow individuals to maintain psychological well-being in the face of adversity [1, 2]. By this conception, resilience is not merely the absence of psychopathology, but involves a number of processes that can change over time. These dynamic processes are nowhere more apparent than in the psychological response to trauma exposure. Cohort studies tracing symptom levels from hours to years post-trauma show that many people initially experience high levels of depression, anxiety, and posttraumatic stress disorder (PTSD) symptoms in the first days after a traumatic event, but the majority recover naturally [3,4,5]. Therefore resilience does not provide a blanket protection against symptoms. Instead it is manifested in the capacity to recover to a state of well-being within a few weeks to months of a major stressful life event. Notably, resilience is the most common response to a traumatic event, as only a minority of individuals go on to maintain persistent impairing symptoms [4].
Neuroimaging data collected from before a traumatic event, during the peri-trauma, and during the post-trauma period can show the full sequence of neural features and adaptations that lead to recovery. Understanding neural circuit contributors to an adaptive response to trauma is critical to our ability to create new interventions that will improve and accelerate recovery. This is in line with current research priorities of the National Institute of Mental Health, which underscores the need for “the development of translatable biomarkers [to] facilitate the study of stress responses, resilience, and vulnerability across both human and animal studies [6]” . To facilitate the development of a longitudinal model of neural contributors to resilience, here we review the emerging body of literature employing neuroimaging in longitudinal cohort studies to predict psychological well-being following trauma exposure. For the purposes of the review, trauma is defined as an event meeting DSM-5 criterion A for trauma- and stressor-related disorders [7] such that participants experienced, witnessed, or were confronted with the threat of death or serious injury or threat to personal integrity. This definition was expanded to consider studies of other major life stressors, such as the death of a family member, job loss, or medical illness. Such stressful life events may elicit similar neurocognitive adaptations as “criterion A” traumas, a topic of active investigation. Finally, because resilience is not merely the absence of a psychiatric diagnosis, where possible, we highlight findings that show neural predictors of multidimensional aspects of well-being following trauma or other major stressor.
We conducted a literature review of neuroimaging studies from January 1995 to May 2021, using the following search terms: trauma OR PTSD OR “posttraumatic stress disorder” AND prospective OR longitudinal OR cohort OR predict AND MRI OR neuroimaging OR “magnetic resonance imaging” OR “neural correlates”. This search was conducted in Pubmed, Web of Science, and Scopus. We included all unique studies that had a longitudinal component such that neuroimaging was conducted either at one or multiple timepoints, with subsequent assessments of trauma-related symptoms including posttraumatic stress disorder (PTSD) or depression, and excluded findings from drug or intervention studies. Table 1 contains a full list of the longitudinal neuroimaging studies of trauma.
We discuss the findings following general principles outlined by Williams et al. [8], in terms of the role of each region within a larger established network. Emerging findings suggest that network-level alterations provide a parsimonious explanation of psychiatric symptoms [9, 10]. For example, neuromodulation therapies for depression, obsessive-compulsive disorder, and Parkinson’s disease show similar symptom-reducing effects across multiple nodes within a target network [11, 12]. Such findings indicate that the network may be the ideal unit of analysis for treatment targeting and characterization of psychiatric disorders. We also theorize that summarizing findings across networks will allow patterns to emerge which may not be clear at the single-region level. We, therefore, discuss the role of regions typically engaged within the default mode, salience, threat, reward, attention, and cognitive control networks, as well as primary and associative sensory cortices. This is further elaborated in Box 1.
Pre-trauma predictors of resilience
Longitudinal studies have proved to be a powerful tool in identifying aspects of neural structure and function that contribute to resilience. One particularly important goal has been to search for traits that exist prior to a trauma, which confer protection from chronic stress-related symptoms following the trauma. Due to challenges in predicting who will be exposed to trauma and when, it has been difficult to study pre-existing neural traits of resilience. However, an emerging literature across both military-related and civilian longitudinal studies has captured pre-trauma structural and functional neural features that predict changes in later brain function and adaptive natural recovery from stress-related symptoms after trauma. These studies provide key insights into neural resilience and how the brain changes and recovers after exposure to psychological stress.
Threat and salience
The majority of research on risk and resilience factors for trauma-related psychopathology links the psychological response to trauma with individual differences in the structure and function of areas associated with threat processing, which has some overlap with the salience network. The areas discussed can be categorized by those involved in threat inhibition (hippocampus, vmPFC), versus those involved in threat response and salience detection (amygdala, insula, dACC, dmPFC).
Pre-trauma protective factors
The amygdala is widely studied in trauma research due to its role in responding to threatening stimuli and the expression of fear (for review, see ref. [13]). It has been hypothesized that individual differences in the structure and function of the amygdala may contribute to stress resilience, such that individuals who show less threat reactivity may be more resilient after trauma [14]. There is now evidence to support this idea across a variety of different cohorts, in which individuals with less amygdala reactivity measured before trauma exposure were more resilient and reported fewer PTSD symptoms post-exposure, in children studied prior to a terrorist event [15] and young adults prior to military deployment [16,17,18]. These effects appear to generalize beyond PTSD to more general stress-related symptoms. For example, healthy young adults who showed less amygdala reactivity to emotional faces at an initial study visit went on to report fewer stress symptoms following stressful life events that occurred 3 months to 4 years later [19]. Lower amygdala reactivity to face stimuli has also been linked with fewer later depressive symptoms [20]. However, individual differences in pre-trauma reactivity may not be accompanied by gross structural differences—van Wingen and colleagues found that amygdala volume pre-trauma does not appear to be predictive of resilience post-trauma [21,22,23].
Individuals with less amygdala reactivity may also show less sympathetic output when new threats are encountered. For example, among medical students scanned prior to a stressful ED internship, less connectivity between the amygdala and a key downstream noradrenergic output region the locus coeruleus (LC) [24] measured before the internship predicted later resilience to anxiety and depression symptoms, as did lower reactivity of the LC during an emotional conflict task [25]. These studies show that pre-existing differences in amygdala and its regulation of sympathetic nervous system activity may predict resilience to stress in some individuals. Resilient individuals may be better able to modulate the activity of the amygdala in concordance with current environmental demands.
Similarly, the ventromedial prefrontal cortex (vmPFC) and its connections with the amygdala or hippocampus may be important contributors to adaptive threat responses. The vmPFC implements top-down control over fear-related regions like the amygdala, and helps to adaptively modulate threat responses [13]. A recent study found that greater vmPFC activation pre-combat training positively predicted resilience to stress symptoms post-training [26]. Structural studies of earthquake survivors who were scanned prior to the event have found that greater grey matter volume (GMV) in the right rACC [27] predicted less anxiety and PTSD symptoms measured 3–4 months after the event. In the same cohort of earthquake survivors, Sekiguchi and colleagues also found that higher pre-trauma fractional anisotropy (FA) of the right anterior cingulum, a tract that is part of the larger cingulum bundle connecting several PFC and temporal lobe regions, was predictive of fewer anxiety and PTSD symptoms evaluated 3–4 months after trauma exposure [28]. Together, these findings link greater vmPFC function, volume, and connectivity with stress resilience.
The hippocampus is another region likely to aid in appropriate regulation of physiological arousal responses to threat, known to be important for contextualizing memories and experiences [29]. Classic findings implicating the hippocampus in resilience came from twin studies of combat veterans and their twins who were not exposed to combat stress. Veterans who were diagnosed with PTSD and their unexposed twins had lower hippocampal volumes compared to veterans with similar exposures but without PTSD and their twins [30]. This suggested that greater hippocampal volume may be a familial PTSD resilience factor. However, this work was conducted many years after the trauma had occurred. A more recent study provided confirmatory observation of hippocampal volume prior to the stressful training experiences. In police recruits, Koch et al. found that greater left hippocampal dentate gyrus volume measured before training predicted fewer PTSD and stress symptoms 16 months post-training [31]. Similarly, in functional studies with a civilian cohort exposed to a terrorist attack, greater bilateral hippocampal activity during an emotional regulation task pre-exposure to trauma was predictive of resilience to PTSD symptoms measured post-exposure [15], suggesting another potential inherent resilience trait. However, another study of military recruits reported no significant difference in hippocampal response to stressful stimuli measured before trauma exposure and the association with post-exposure symptoms [16]. This discrepancy in pre-trauma functional results may be explained by the context in which their traumas occurred and the mental preparedness of the military cohort fulfilling their service versus the civilian cohort. Overall, evidence suggests that greater hippocampal volume before a trauma may be protective, but future longitudinal studies will be needed to determine if pre-trauma function relates to resilience after trauma exposure and how context of the trauma may impact these results.
The salience network has been implicated in the monitoring of affective environmental cues. Structural and functional differences in distinct subregions of the ACC may denote specific PTSD resilience factors. Studies of combat exposed veterans and their unexposed twins have found that less resting state dACC and mid-cingulate activity [32], as well as less responsivity of the dACC during a cognitive interference task [33] were associated with resilience to the development of PTSD. Less functional activity in the dACC pre-trauma may therefore be a protective factor. These studies indicate that less dACC activity and greater ACC volume or connectivity via the anterior cingulum may be protective traits.
Pre- to post-trauma changes
Regardless of symptom presentation, regions involved in threat detection and response, including the amygdala and insula [22, 31, 34] have generally been found to increase in volume and threat reactivity from pre- to post-trauma exposure. Changes in these regions may be dependent on the actual or perceived severity of the trauma experienced. Participant reports of perceived threat during military deployment were correlated with pre- to post-deployment increases in amygdala-insula and decreases in amygdala–dACC functional connectivity (FC) in response to social threat cues 1.5 months after deployment [22]. However, data collected in the same military cohort 1 year later showed that amygdala activity, insula activity, and amygdala-insula FC returned to pre-deployment baseline levels, while only decreased amygdala–dACC coupling remained significantly correlated with perceived stress [23]. Therefore, changes in amygdala and insula as a result of trauma may not necessarily be permanent.
A pattern of conserved hippocampal volume and function from pre- to post-exposure to stress has also been associated with resilience. Hippocampal volume appears to decrease as a function of stress exposure; for example, individuals who experienced a greater number of stressful life events demonstrated larger decreases in left parahippocampal and right hippocampal volumes [35]. Thus, it seems that inherently larger hippocampal volume, which is then sustained post-trauma, may indicate individuals who are less at risk for developing PTSD. However, the individual’s response to the stressful event appears important—soldiers who showed less change in hippocampus reactivity to threat stimuli from pre- to post- deployment reported lower stress symptom severity post-deployment [16]. In this same study, increases in hippocampal-vmPFC functional coupling from pre- to post-deployment predicted resilience, as measured by fewer stress-related symptoms [16]. Therefore, while larger hippocampal volume is protective, individuals with less hippocampal reactivity but improved connectivity from pre- to post-trauma may be more resilient.
The uncinate fasciculus (Uf) is the primary white matter tract connecting the vmPFC and orbitofrontal cortex to the amygdala, and is involved in emotional processing [36]. Standard neurocognitive models of stress resilience predict that greater integrity of the Uf should facilitate regulation of the amygdala by the vmPFC, enhancing resilience. However, detailed investigation of this tract among earthquake survivors suggests that there is a more complex temporal association between Uf integrity and resilience that unfolds over the course of recovery. Sekiguchi and colleagues found that a longitudinal increase in FA of the left Uf from pre- to 3+ months post-trauma was negatively associated with resilience, measured by more anxiety symptoms post-trauma [28]. However, by the time of a 1 year follow up, participants who had initially shown the increase in Uf FA now showed a decrease in both left and right Uf FA, indicating recovery of this tract even if symptoms did not significantly improve [37]. Although pre-trauma results suggested that greater anterior cingulum bundle (Cg) FA is a protective factor, additional analyses of these same earthquake survivors reveal that increased left Cg FA from pre- to 3+ months post-exposure was associated with higher anxiety scores and therefore less resilience [28]. Likewise, PTSD severity was positively correlated with increased FA of the right Cg 1+ year later [37]. These studies imply that individuals who were resilient, showing fewer chronic PTSD or anxiety symptoms in the aftermath of the earthquake, were less likely to show any changes in anterior cingulum white matter diffusivity from pre- to post-exposure. Additionally, changes in GMV of the bilateral ACC were negatively correlated with the number of stressful life events experienced within a 3 month time period [35], suggesting that exposure to stressful experiences could lead to reductions in ACC volume, though this was only demonstrated in one small study. It is possible that more resilient participants with initially greater anterior cingulum FA and ACC volume did not experience as great of changes from pre- to post-exposure due to a ceiling effect. However, similar to results with Uf FA, over the course of a year, participants that initially had increased r Cg FA showed some reductions and recovery of the tract [37]. These studies indicate that greater changes of the anterior cingulum bundle are associated with less resilience to trauma but may not be permanent changes to the brain.
Reward
Very little work has focused on the reward circuit pre- to post- trauma exposure. One study found that differences in nucleus accumbens (NAc) activation pre-deployment did not predict resilience, but that at the post-deployment assessment, more resilient individuals showed greater NAc response to reward compared to individuals experiencing worse post-trauma PTSD symptoms [17]. Additionally, studies of earthquake survivors found that individuals with fewer PTSD symptoms 3 months post-trauma showed less of a decrease in GMV of the left orbitofrontal cortex (OFC) from pre- to post-trauma [27]. Based on these studies, it appears that pre-exposure differences in reward circuitry may not indicate any protective factors, but that resilient individuals retain higher reward circuitry response and structure after trauma exposure. As blunted affect is a symptom of both PTSD and depression, it is imperative that future longitudinal studies further explore the relationship between trauma exposure and changes in reward circuitry.
Cognitive control
The anterior prefrontal cortex (aPFC) and in particular the frontopolar cortex is thought to be involved in action selection and reasoning [38, 39]. Koch and colleagues further suggest that the aPFC and lateral frontal pole may be important for emotional cognitive control and switching between alternative emotional strategies during cognitive decision-making tasks [40]. A recent study of a large cohort of police recruits found that greater pre-trauma anterior PFC, dorsal and medial frontal pole response during an approach-avoidance task was associated with fewer PTSD symptoms measured after exposure to trauma [41]. This suggests that having greater top-down emotional control may be another protective trait against the development of PTSD. Perhaps more resilient individuals with greater aPFC activity are better able to adaptively alternate their emotional response in the face of trauma.
Summary of pre-trauma resilience factors
Potential resilience or protective factors prior to a trauma (Fig. 1) include lower engagement of threat response and salience detection regions, such as the amygdala, insula, LC, and dACC. Likewise, greater hippocampal volume, vmPFC, and anterior prefrontal activation pre-trauma may protect against later effects of trauma. Individuals with greater resilience demonstrate regional conservation of volume, function, and connectivity from pre- to post-trauma across multiple networks, as well as increased hippocampal-vmPFC coupling. However, pre-trauma studies have not yet reported findings in networks including dorsal and ventral attention, default mode, or sensory cortex. Future research evaluating brain-wide, multi-circuit interactions in individuals before and after trauma exposure is necessary to determine the potential involvement of other networks in predicting pre-trauma resilience.
Pre-trauma factors of resilience include larger vmPFC and hippocampus volume, greater activation of emotional regulatory regions like the vmPFC, hippocampus, and aPFC, and less activation in threat response regions such as the amygdala, dACC, and LC. Resilient individuals demonstrated fewer functional and structural changes from pre to post trauma compared to those with symptoms of PTSD. However, greater coupling between the vmPFC and hippocampus from pre to post trauma is associated with resilience. Peri-trauma features that predict later resilience include larger structural features in the hippocampus, parahippocampus, vmPFC, sgACC, dlPFC, temporal lobe, as well as greater rACC surface area volume. Similar to pre-trauma, decreased amygdala and increased hippocampal/parahippocampal functional activity soon after trauma are associated with resilience. Increased dlPFC-amygdala and decreased PCC-amygdala or PCC-PAG connectivity positively correlate with later resilience. Notably, however, many of these findings appear the opposite among those at risk for chronic PTSD with dissociative features. Over the course of recovery, functional reactivity in the amygdala, insula, and dACC decrease or return to pre-trauma levels. Amygdala-insula, -thalamus, and -vmPFC connectivity also return to baseline levels, and connectivity between the PCC and PAG continues to weaken over time. Structural increases in frontal regions such as the OFC or vmPFC as well as increases in the thalamus are related to recovery. There is greater activation of emotional regulatory regions like the vmPFC and hippocampus as well as less activity in the dmPFC and PCC. Yellow/blue fill = positive/negative correlations between resilience and functional activation or reactivity. Orange/blue border = positive/negative correlation between resilience and structural volume. Green/blue arrows = positive/negative correlation between resilience and connectivity between regions.
It is important to note that while being a “resilient” individual with a dampened threat and salience response or greater threat inhibition may be adaptive in times of safety and security, these same traits may be harmful in situations where responding to threat cues is vital for survival, like in the case of a soldier on the battlefield. Trait-like, lower reactivity to threatening cues may not keep a person safe in times of real danger. Resilience may therefore be reflected in the adaptive regulation of circuits that modulate autonomic and emotional arousal, but not defined by any specific level of regional reactivity.
Peri-trauma to long-term predictors of resilience
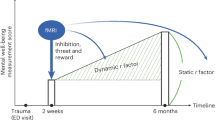
Most civilian trauma neuroimaging research occurs in the aftermath of trauma, during peri-trauma and in the years following. The main focus of this research has been to establish structural and functional differences or changes that predict the future emergence of chronic trauma-related symptoms or resilience. As many trauma-exposed individuals seek acute treatment for injuries or related medical issues, the peri-traumatic period is a key time window for early assessment of stress resilience and for the deployment of early interventions to boost resilience and recovery.
Threat and salience
The majority of peri- and post-trauma neuroimaging findings implicate the structure and function of areas involved in threat inhibition (hippocampus, vmPFC) and those involved in threat response and salience detection (amygdala, insula, dACC, dmPFC).
Consistent with pre-trauma findings, amygdala reactivity to threat cues acutely post-trauma correlates negatively with later resilience. For example, in participants recruited from the emergency department (ED), who were scanned 1-month post-trauma, amygdala reactivity to negative emotional stimuli negatively correlated with resilience, as measured by fewer PTSD symptoms months later [42]. While this occurs in the likely context of a pre- to post-trauma increase in amygdala reactivity [22, 34], relatively lower post-trauma amygdala reactivity may indicate PTSD resilience. However, the relationship with amygdala structure remains unclear. Stein and colleagues showed no association between amygdala volume 2 weeks following a head injury trauma and later PTSD symptoms [43], although there have been few early posttrauma studies of amygdala volume with respect to later symptoms or well-being.
Other areas of overlap between the threat and salience network include the dACC and insula. Within these areas, increased reactivity and structural integrity are associated with resilience. A pattern of larger insula, dACC, and rACC volume assessed shortly after trauma predicts fewer future PTSD symptoms after head injury trauma [43], and motor vehicle accident [44]. Greater structural integrity in this network may lead to adaptive regulation of emotional responses to threat stimuli. However, functional imaging findings have been more mixed. PTSD symptom improvement in military service members over the first few months following the return from deployment was positively correlated with engagement of the anterior insula and dACC during an affective task requiring effortful attention allocation in the face of distractors [45]. In contrast, PTSD symptom improvement was negatively correlated with dmPFC engagement during a less effortful emotional appraisal task 2 weeks after motor vehicle collision and with insular engagement 3 months after. This study also showed an increase in dmPFC activation from 2 weeks to 3 months which was accompanied by an increase in PTSD severity over this time [46]. This network’s contributions to trauma recovery may therefore depend on its role in various task demands, such as attention allocation, and depend on emotional context and nature of the trauma. Further work is needed to disentangle the relative contributions of these effects, employing tasks with varying effort levels or attentional complexity, and comparing across trauma types.
The amygdala is regulated by the vmPFC, and longitudinal findings support that an increase of vmPFC morphometry or activity is important for later resilience. Among PTSD patients scanned at varying times since trauma, a longitudinal increase in sgACC over 6–9 months predicted natural recovery over the same timespan [47]. Similarly, trauma survivors with greater rACC and vmPFC surface area and volume collected peri-trauma were less likely to develop PTSD symptoms three to twelve months later [43, 44, 48]. DTI studies also implicate the vmPFC in resilience, finding larger FA and less mean diffusivity in the vmPFC predicted resilience 2 days following a traumatic event, and larger vmPFC FA predicted lower PTSD severity at 1 or 6 months later [49]. Interestingly, a study of prenatal mothers found that greater pre-delivery personal growth initiative correlated with larger maternal vmPFC grey matter volume after birth, indicating a link between proactive coping and vmPFC structure post-trauma [50]. Concerning functional findings, activation to trauma-related pictures in bilateral vmPFC 2 months post-trauma positively correlated with resilience to acute PTSD [51]. Similarly, persistent activation of the sgACC over repeated threat stimuli presentation was positively associated with PTSD resilience months later [42]. Although there are some mixed findings [46], overall, greater vmPFC volume, FA, and engagement in the context of threat predict less PTSD severity post-trauma, consistent with the role this area plays in inhibiting fear responses.
Hippocampal activation and structural integrity overall appear to be predictive of resilience. Studies of hippocampal structure suggest that greater hippocampal volume correlates with greater resilience to trauma, with larger hippocampal volumes early post-trauma among those who demonstrated the fewest future PTSD symptoms [52,53,54]. However, studies also suggest that the experience of stressful life events correlates with smaller hippocampal volumes weeks and months post trauma, even among resilient individuals who demonstrated few PTSD symptoms after trauma [35, 54]. This may be compounded among individuals who continue to experience high levels of distress following the trauma, for example, high anxiety symptoms 5–12 months following traumatic brain injury (TBI) predicted atrophy of the right hippocampus between 12 and 30 months post-trauma [55]. When individual hippocampal subfield volumes were assessed in individuals two weeks and six months post-trauma, no correlation with PTSD symptoms was found [56], although this study was underpowered to assess group differences in PTSD and had low PTSD severity as a whole. These structural differences appear to be accompanied by differences in function. Greater engagement of the hippocampus during response inhibition 1-month post-trauma predicts PTSD resilience, with individuals who experienced similar types of traumas showing lower symptom severity up to 6 months post-trauma [57]. Hippocampal activation during fear conditioning also positively correlates with trait resilience at two months post-trauma, as measured by the Connor-Davidson Resilience Scale (CD-RISC) [58]. Furthermore, longitudinal increases in hippocampal activity in response to social threat cues and during fear extinction are associated with PTSD symptom improvement several months later [58, 59]. It may be that traumatic events result in smaller hippocampal volume and impaired function, or that smaller hippocampal volume or reduced function is a vulnerability factor. However, these effects may be seen to a lesser degree in resilient individuals who show fewer symptoms of anxiety and PTSD. Larger hippocampal volumes and greater engagement in inhibition and extinction, in turn, appears to facilitate later reductions in PTSD symptoms in the post-trauma recovery period, and correlate with trait resilience. It is thought that hippocampal activation during fear conditioning tasks may lead to better contextual threat processing, which then results in improved contextual behavior modulation [58].
Connectivity within and to the threat network appears highly important to the response to the trauma, with many studies finding amygdala FC to be involved in fear learning and symptom severity following trauma. Amygdala-PFC, -PCC, -dACC, -superior temporal gyrus, and -insula functional connectivity have all been implicated in recovery from trauma-related psychopathology. Amygdala-PFC connectivity, and PFC sub areas, has previously been supported as a resilience factor [60]. The vmPFC is suggested to inhibit fear responses via the amygdala [13]. Concurrently, DTI studies point to greater amygdala-PFC or Uf structural connectivity as a protective factor for resilience, as measured by fewer PTSD symptoms [61]. Increased post-trauma connectivity negatively associated with longitudinal resilience include amygdala-PCC [62], -dACC [23], -left superiortemporal gyrus [63], and with somatosensory areas [64]. As these areas are part of the default mode network and sensory network, higher connectivity in PTSD between these areas may reflect hypervigilance, increased emotional self-reflection, or hypersensitivity to sensory signals. Increased PAG connectivity, which receives output from the amygdala and hippocampus, with the PCC and frontal pole is also shown to negatively correlate with resilience 6 months posttrauma [65]. One study, which monitored shifts in amygdala connections in PTSD patients post-trauma for 5 years using DTI, found that amygdala-insula connectivity initially strengthened and then normalized during recovery from PTSD symptoms, -prefrontal cortex connectivity initially was unchanged, strengthened, and then normalized, -thalamus connectivity normalized during recovery, and -hippocampus connectivity remained low [61]. This highlights the potential complexity in network dynamics during recovery from PTSD, and the utility of assessment over several years.
These findings support proposed models suggesting that hippocampus and vmPFC modulation of amygdala activity early post-trauma supports resilience and recovery. On a basic level, less amygdala reactivity, greater hippocampus, and vmPFC activity and volume, and greater functional and structural connectivity between these regions and the amygdala early post-trauma correlate with better PTSD recovery. However, evidence across the collected studies suggests that functional correlations are task dependent, with vmPFC engagement across repeated presentations of threat correlating with quicker recovery, whereas the vmPFC response to threatening versus neutral social cues was associated with greater future symptom maintenance. Similarly, in the hippocampus, engagement during inhibition is associated with less future PTSD symptom symptoms, but greater response to threat cues during conditioning correlates only with trait resilience. Therefore, a multifaceted approach is necessary to fully understand these three areas of the threat network. Methods for assessing resilience more broadly than PTSD symptoms and recovery are necessary.
Reward
In the peri-trauma period, again, a very limited set of studies have focused on reward circuits and resilience. One longitudinal study in cancer survivors did find that GMV in the right orbitofrontal cortex 3–15 months post-surgery and two years later was significantly larger in resilient survivors and non-trauma controls compared to those with PTSD [66], which in line with findings from a pre- to post-trauma study [27]. Orbitofrontal cortex activation correlates positively with magnitude of presented reward [67] and reward “pleasantness” [68], and dysregulation of this area may play a part in anhedonia symptoms post-trauma. However, reward circuits are also highly overlapping with areas involved in the threat network, including the amygdala and mPFC regions [8], and thus may also play a role in emotional regulation [69, 70]. Considering that a decrease in positive affect is a symptom of PTSD, further research understanding the relationship between resilience and reward circuits is necessary.
Default mode
Default mode network (DMN) activation is observed while at rest and during spontaneous reflection [8, 71, 72]. The role of the DMN in resilience is highly consistent across early post-trauma studies. Structurally, greater FA across nodes of the network [49], greater cortical thickness [73], GMV [21, 44,74,75,76], and surface area [77] within DMN nodes all positively predict resilience weeks and months following trauma. This may extend from the core DMN to the temporal subnetwork as well, as one study showed that individuals with increasing PTSD symptoms over time also demonstrate accelerated temporal lobe atrophy [78]. While these findings suggest that structural integrity of the DMN post-trauma promotes resilience, functional studies found a negative correlation between PCC reactivity and DMN-threat network connectivity and early resilience. Early post-trauma, the PCC response to trauma-related images correlates negatively with resilience [79]. Acute post-trauma connections between DMN nodes and aspects of the threat network appear to additionally reduce resilience, and may be particularly related to re-experiencing symptoms. Post-trauma DMN-amygdala FC predicts PTSD severity at time of scan, and negatively correlates with resilience months later [62]. Early post-trauma connectivity between DMN nodes and rACC, periaqueductal grey, inferior frontal gyrus, and thalamus also are all negatively associated with subsequent resilience [62, 65, 80, 81]. Further, reduced centrality of the PCC/precuneus in sexual assault victims peri-trauma is reported in those who did not develop PTSD, compared to non-trauma exposed controls [82]. Thus, early post-trauma DMN engagement via the PCC and threat network connectivity is suggested to have a negative impact on resilience, mainly within the first year following trauma.
While DMN engagement and threat-network connectivity predict acute trauma-related psychopathology, they may lead to later recovery and long-term resilience. For example, reactivity in the DMN to trauma-related pictures two months post-trauma was negatively correlated with early PTSD resilience, but positively related to resilience 2 years later [51]. Participants in this study showed a general reduction in PTSD symptoms over the two years, and a reduction of PCC and mPFC activation between time points, suggesting that early DMN activation which then decreased within the months following trauma was eventually an indicator of adaptive recovery. Such findings illustrate the major value of longitudinal studies in uncovering unexpected ways that resilience may unfold over time. Further work is necessary to understand how early activation of the PCC post-trauma may play a role in resilience, both peri-trauma, and in the years following, and how FC within and with the DMN may or may not fluctuate over time. These findings also suggest that the DMN may play an important role in treatment and recovery. There is some support for this, with DMN-dlPFC, -ACC, and salience network connectivity increasing pre- to post-treatment in treatment studies [83, 84].
Attention
The attention network is involved in directing and maintaining perceptual resources as a function of task demands [8, 85]. Emerging findings suggest that early post-trauma engagement of this network protects against the development of later trauma-related psychopathology. First, inferior parietal lobule response may change over time following trauma. Resilient mining accident survivors show greater activation of the inferior parietal lobule early post-trauma in response to trauma-related imaged compared to those with PTSD [51], suggesting that resilient individuals may initially maintain engagement of attentional regions to trauma related cues. In turn, sequential increases in inferior parietal responses to emotional stimuli 2–12 months post trauma predicted PTSD symptom improvement over this time in combat deployed soldiers [45], again pointing to an adaptive role of this network in trauma recovery. Increases in the function of this circuit over the post-trauma recovery period appear to have protective effects that extend beyond PTSD, for example, an increase in thalamus-dorsal attention network connectivity from 6 week to four months post-trauma correlated with decreased pain and post-concussive symptoms among mTBI patients [81]. Structural findings also support this possibility. Among soldiers deployed to Iraq/Afghanistan, larger volume and greater FA of the superior longitudinal fasciculus (a major tract connecting the parietal cortex with medial frontal regions) early posttrauma appeared protective against the later development of PTSD after military combat [63] and MVC [86]. Additionally, MVC survivors who developed PTSD showed a decrease in the volume of the left superior frontal gyrus from two weeks to three months post-trauma, whereas resilient participants did not [46]. Given that these findings span multiple types of traumatic experiences and different cohorts, this network may be an important and generalizable target for facilitating recovery.
Cognitive control
The cognitive control network is involved in working memory and selective attention [8, 87, 88]. Within this network, changes in the structure and function of the dorsolateral prefrontal cortex (dlPFC) particularly appear to predict resilience. Adults showed greater dlPFC thickness over a year post-trauma exposure, and this thickness predicted greater recovery from PTSD symptoms over a period of 5 years [89]. These patterns suggest that growth of the dlPFC early after a traumatic event may be a positive adaptation. The dlPFC may engage in the regulation of emotional arousal responses during the early recovery period, as its connectivity with a threat network overlapping the bilateral amygdala and brainstem 2 weeks post-trauma predicts subsequent resilience to PTSD and depressive symptoms months later [74, 80]. This suggests the DLPFC as an interesting target for early intervention, following current neuromodulation treatments for chronic PTSD and MDD which typically target the dlPFC.
Alterations in the cognitive control network may also be related to its reciprocal projections with the thalamus. Traumatic events involving a head injury have been associated with decreased thalamus-frontoparietal control network FC [81], and FA in the thalamus and the superior corona radiata—connecting the thalamus and cortical areas—has been shown to significantly increase from within several months to years post-trauma exposure [37, 90], and this increase is associated with PTSD symptom recovery [90]. These adaptations to trauma may facilitate recovery as trait resilience scores positively correlate with FC between the inferior thalamus and middle frontal gyrus [91]. Cognitive control findings thus suggest that greater connectivity early post-trauma and increases in structural integrity from peri-trauma to years post-trauma promotes later resilience.
Sensory
Sensory areas showing associations with longitudinal resilience include the occipital cortex and fusiform gyrus, involved in vision, and precentral cortex, involved in relaying and regulating sensory input. Vision related activation within the first year post-trauma appears to negatively correlate with resilience. Peritraumatic dissociation, a known risk factor for later chronic PTSD, depression, and chronic pain [92,93,94], positively correlates with activation along the right ventral visual pathway fusiform, lingual, and parahippocampal gyri during a trauma script task 2–4 months later [95], providing evidence for a negative correlation between visual circuit activation and resilience. Similarly, increased occipital centrality during resting-state three weeks following sexual assault predicted PTSD diagnosis six months post-trauma [82]. The occipital lobe is engaged during autobiographical recall of emotional events, and increased activation post-trauma may indicate a propensity for sensory reactivation during trauma-related, intrusive thought [82, 95].
Summary of early and later post-trauma resilience factors
Potential resilience protective factors post-trauma include increased vmPFC structure, reactivity, and connectivity, larger hippocampal volumes and activation, decreased amygdala reactivity, greater salience network activation and structure, and greater reward circuit/orbitofrontal cortex GMV (Fig. 1). A major risk factor appears to be amygdala reactivity and connectivity, which is highly involved in fear learning and symptom severity following trauma. Changes in amygdala connectivity during trauma recovery may be vital to resilience post-trauma. However, the resilient profile extends beyond the canonical circuits involved in threat detection, reward responsivity, and regulation of these functions. The pattern across networks is summarized in Fig. 2. Lower engagement of the DMN and DMN-threat network FC, and greater threat network-cognitive control network FC are also associated with resilience, particularly acutely, in the earlier months following trauma. Interestingly, while hyperactivation of DMN, hyperactivation of cognitive control network, and hypoactivation of the attention network are related to acute trauma-related psychopathology, findings suggest these features may be adaptations to the trauma which only have short-term consequences, and in fact predict longer lasting recovery in the years following trauma.
Across time following trauma, network patterns are noticeable. Pre-trauma findings (A) suggest that structural integrity/activation of inhibition areas in the threat and cognitive control networks positively correlate with resilience, while threat response and salience networks show negative associations with resilience. Post-trauma findings are split into two categories: early, peri-trauma findings that predict early resilience within the first year following trauma (B) and peri-trauma findings that predict later recovery and resilience years following the trauma (C). Correlations for the inhibitory and response areas of the threat network and cognitive control network are consistent over all three timepoints, and across the two post-trauma timepoints, the attention and reward networks both have positive correlations with resilience. However, some networks exhibit changes across time points. The DMN during peri-trauma negatively predicts early resilience, while it positively predicts later recovery and resilience. Changes are also seen across the salience network, with less salience activation pre-trauma predicting resilience, but higher salience activation post-trauma predicting peri-trauma resilience and later recovery. Axis values represent overall negative (purple) or overall positive (blue) correlations. Time points that do not have strong evidence supporting network correlations are indicated by grey dots.
Peri-trauma and post-trauma studies to date are limited by the timing of scans, both in how early individuals are scanned post-trauma and later, longitudinal follow-ups. Neuroimaging studies face logistical constraints, in that most studies are unable to schedule individuals for a scan until weeks after a trauma occurs. In addition, most studies only evaluate trauma-related psychopathology or perform a second scan within the first year following trauma. This prevents evaluation of factors that may predict which individuals display long-term recovery and resilience compared to those with chronic trauma-related psychopathology. Studies that evaluate changes from peri-trauma to years post-trauma within these networks, and how they correlate with resilience over time, are necessary. Further, it is currently difficult to form conclusions regarding the influence of trauma type on findings, as many studies recruit individuals who have been exposed to all different kinds of trauma. While some studies are limited to motor vehicle collisions, a particular natural disaster, or military deployment, for example, the mix of time points that data is collected at and differences in neuroimaging data collected make it difficult to directly compare findings. Finally, while post-trauma longitudinal findings have been made within each of the described networks, the threat network and DMN have had the most findings, and the reward network the least. While the reported studies here reflect the importance of the threat network and DMN in particular in understanding resilience, they also elucidate gaps in the field regarding other network areas.
Seeming contradictions in neurobiological resilience profiles
Several PTSD symptom clusters have a common neural correlate pattern. Specifically, intrusions, alterations in cognition and mood and altered arousal and reactivity symptoms are often associated with deficits in corticolimbic inhibition [96]. The findings outlined in the earlier sections suggest that greater engagement of cortical regions involved in emotion regulation before or shortly after a traumatic experience might then be protective against intrusion, mood, and arousal symptoms associated with posttraumatic stress. However, in a subset of individuals, this may not be the case. Fifteen to thirty percent of individuals with PTSD experience significant, chronic dissociative symptoms of depersonalization and derealization [97, 98]. These experiences of detachment from your sense of self, body and surroundings (e.g., “I feel like I’m looking at my body from outside my body” or “I feel like I’m looking at the world through a fog”) are associated with the opposite neural pattern to that described above. That is, these symptoms occur in conjunction with increased corticolimbic inhibition during emotionally provocative contexts [99]. Specifically, during symptom provocation paradigms, individuals with this subtype show a pattern of hypoactivity in the threat and salience networks in regions involved in salience detection (amygdala) and interoception (insula), and hyperactivity in cortical regions involved in threat inhibition, emotion, and bodily arousal regulation (e.g., vmPFC, dACC). Resting state FC supports this pattern with increased connectivity between subregions of the amygdala and the prefrontal cortex [100], the amygdala and insula [101], and the ventrolateral periaqueductal grey with cortical regions associated with passive responses to threat (e.g., temporoparietal junction) [102]. Furthermore, as discussed previously, peritraumatic dissociative symptoms of depersonalization/derealization are associated with subsequent activity in areas involved in vivid autobiographical memory recall (e.g., fusiform gyrus, lingual gyrus [95]). This suggests peritraumatic dissociation may play a role in the development of intrusive re-experiencing symptoms [95].
Given these findings, it follows that less vmPFC or dACC engagement before or shortly after a traumatic experience might be protective against developing both chronic dissociation and posttraumatic stress symptoms. To our knowledge, this has yet to be directly tested. However, a recent longitudinal treatment study in adolescents supports this conclusion. Specifically, this study demonstrated that improvements in dissociation post treatment were associated with decreased dACC and amygdala activation during an emotionally provocative task [103].
Taken together, some studies point toward more threat inhibition network activity and some point toward less as the profile of resilience. How do we begin to reconcile these seemingly opposite patterns of resilience? First, this evidence suggests there are different neural features at early timepoints that appear protective against different potential outcomes. More corticolimbic inhibition associated with threat inhibition and emotion regulation may protect against intrusions, alterations in mood and hyperarousal. However, too much corticolimbic inhibition may interfere with emotional learning [104, 105] and promote chronic dissociation and intrusive re-experiencing [95]. Second, it may be that each individual is on a continuum of possible over- vs. under-modulation in corticolimbic inhibition post-trauma (see the defense cascade model of dissociation [106, 107]). There may be a “goldilocks” level of just the right amount of inhibition that is associated with resilience toward intrusion, mood, hyperarousal symptoms and dissociative symptoms after trauma. Furthermore, this provides a contraindication for a one-size-fits-all approach to early interventions; different therapeutic approaches may be necessary depending on where an individual falls on this continuum.
Special considerations related to development
Exposure to stress and traumatic events is thought to be particularly impactful during childhood, because of rapid brain changes that occur as part of brain maturation [108]. Neuroimaging studies on the longitudinal effects of trauma exposure suggest that early life stress exposure may be an important determinant of individual differences in the structure and function of key circuits involved in resilience. These early experiences may thus influence the way that these circuits adapt and respond in the face of new stressors or traumatic experiences.
Early stress and trauma seem to negatively impact threat and salience network structure and function. For example, early life stress predicted smaller bilateral hippocampal and amygdala volumes [109], with some suggestion of differences in the impact for boys versus girls [110], as well as sequential post-stress reductions in cortical thickness of medial and lateral prefrontal and temporal regions [73]. Also, more violence exposure was related to lower resting state FC between the right hippocampus and bilateral frontotemporal regions 3 to 5 years later [109]. Similarly, studies relating threat and salience brain findings to PTSD in children or adolescents demonstrate its importance for PTSD development versus recovery. The only pre-trauma prospective study showed that greater left amygdala reactivity and lower bilateral hippocampal activity in adolescents who were later exposed to a terrorist attack predicted development of PTSD symptoms [9]. Structural studies showed an association between PTSD severity and a reduction in hippocampal volume 12 to 18 months later was observed in maltreated children [111]. In a sample of maltreated girls but not boys, early onset of psychopathology was associated with the development of the right presubiculum, and late onset psychopathology with right CA1 volumes [42]. However, some studies report no significant difference in hippocampal volumes between those with or without PTSD across multiple timepoints, which could be due to sample size and younger age [112] or older age and lack of chronic PTSD [111]. A sustained right vmPFC GMV reduction and decreases in amygdala-PFC functional connectivity from baseline to a 1-year follow up were observed in children with PTSD, and volume reductions were found to be predictive of PTSD severity [74]. In terms of PTSD recovery, increases in vmPFC and sgACC thickness over time predict natural recovery from PTSD [73, 77]. These studies show profound effects of stress and different forms of trauma exposure reported during late childhood or early adolescence within regions of the threat and salience network that appear to influence later mental health following trauma exposure. Taken together, the findings in children are in line with the adult findings summarized in Fig. 2, with greater peri-trauma salience network structure and function positively predicting resilience or PTSD recovery.
While there is more developmental trauma research needed in networks other than threat and salience, a few findings have been published. For example, one observed that in a 10-year longitudinal study following children into adulthood, early stress exposure at the beginning of the study predicted less dlPFC engagement in error monitoring in adulthood [113], which is in line with observations among adults that more cognitive control is related to greater resilience. In contrast, a decrease in dlPFC GMV has been observed in sequential observations of youth with PTSD who remitted naturally, whereas those who maintained high PTSD symptoms showed dlPFC growth [74]. For the default mode network, reduced vlPFC-hippocampus connectivity is associated with PTSD diagnosis in adolescents [74]. This differs from adult studies of DMN connectivity, and may relate to unique effects of the temporal sub-circuit of the DMN with respect to the psychological adaptation to trauma, or because of the influence of developmental factors.
One important consideration for longitudinal studies in children or adolescents is the timing of traumatic events and its interaction with developmental period. The brain does not develop linearly, instead, there are specific sensitive periods for maturation of different cortical and subcortical structures [114]. The interaction with developmental periods significantly complicates the ability to determine the effect of a traumatic event on the brain, as this depends on the timing of the event and the stage of brain development, and both can vary widely within a study sample. Collecting measures of biological age as well as developmental (pubertal) stage, specific assessment of timing of the traumatic event, and prospective designs could help mitigate this issue.
A second challenge for developmental research aiming to define predictors for PTSD risk is the need for large cohort studies which prospectively assess neural structure and function and trauma exposures over development. Only one prospective study to date has collected MRI data prior to trauma exposure and onset of PTSD when the cohort happened to be exposed to the Boston marathon terrorist attack [15]. Longitudinal MRI studies in at-risk cohorts of children growing up in inner city communities with high levels of violence exposure are underway and will be needed to better understand the relation between MRI abnormalities and PTSD risk. Additionally, large developmental MRI studies that include regular trauma assessments are needed to understand the role of brain structure and function in development of post-trauma psychopathology.
A final consideration for longitudinal developmental studies is related to the long-term consequences of stress and trauma and related brain alterations. It is postulated that traumatic events during development could shape brain structure and function to be adaptive in the current situation [115], but it is unclear whether these changes will be predictive of later PTSD or, alternatively, promote resilience. Furthermore, over time neural compensation could lead to previously overactive regions now showing decreased activation or possibly atrophy, which could be related to different psychiatric risk profiles. Therefore, continuing to monitor developmental cohorts and assessments of psychopathology in adulthood will be instrumental to elucidate the long-term effects of stress and trauma on the brain, which will provide potential targets to promote resilience early on during development.
Summary and conclusions
Longitudinal neuroimaging studies of trauma and related mental health consequences are beginning to outline a consistent set of factors contributing to trauma resilience. The overall pattern across networks and timepoints is illustrated in Fig. 2. Engagement of response areas of the threat network has a consistent negative correlation with resilience, while threat inhibition-related areas, the reward network, and the cognitive control network have a consistent positive correlation. The picture is more complex after the trauma has occurred. The attention network exhibits a positive correlation with resilience post-trauma. However, changes across certain networks appear to support recovery over time. While the DMN engagement peri-trauma at first may be maladaptive, this early engagement predicts later recovery years post-trauma. In addition, pre-trauma salience activity has a negative association with resilience, but higher salience activation post-trauma predicts both peri-trauma resilience and later recovery. Further complicating the picture, there appear to be subgroups of people in which threat inhibition activity is negatively associated with resilience (e.g., the dissociative subtype of PTSD). Together, the emerging literature on longitudinal studies of trauma suggests that stable individual differences in core affective regions predict resilience, whereas higher-order associative networks flexibly adapt following trauma, with an initial tradeoff for early DMN engagement followed by long-term benefit.
These conclusions, however, must be tempered by the fact that this is a relatively new literature, with a number of limitations. Studies of the pre-trauma period are particularly sparse, with very small samples and power-related limits on more exploratory analyses. Future work in larger samples needs to address neural circuit function across the brain, looking at relationships between pre-trauma circuit-level connectivity and psychological outcomes post-trauma, as well as changes in these circuits following trauma. Such studies may very well reveal pre-trauma resilience factors outside the threat and salience networks. Additionally, biomarkers of resilience will only become useful in prognosis and precision medicine approaches with further work to establish measurement variability, reliability, and expected normative values. This has been a major stumbling block for efforts to integrate neuroscience and psychiatry, and must become a key priority area for further research.
With these limitations in mind, we can leverage current findings to guide resilience-boosting interventions targeting different time windows and subgroups. For example, a recent study took a random selection of first-year college students to receive an intervention designed to increase their resilience through training in mindfulness, goal-building, and resilience skills [116]. For this type of intervention, in which the approach is broad, without respect to particular trauma or single stressful event, neurobiological targets might include improving modulation of the threat and salience network. In contrast, for studies in which the goal is to identify trauma exposed individuals shortly after the trauma, such as during an ED visit or in the battlefield, interventions may take an alternative approach of increasing the engagement of the dlPFC, inhibitory threat areas, or the attention networks. Moreover, in studies targeting individuals with chronic dissociative symptoms, interventions may instead attempt to decrease engagement of threat-inhibition regions. Due to changes across the DMN post-trauma and the limited data in respect to the reward network areas, further work is necessary to form new interventions regarding these areas. Future longitudinal work, taking into account potential network changes across pre-, peri-, and post-trauma, will provide critical data that may inform improved interventions that promote resilience.
References
Ioannidis K, Askelund AD, Kievit RA, van Harmelen A-L. The complex neurobiology of resilient functioning after childhood maltreatment. BMC Med. 2020;18:32.
Kalisch R, Cramer A, Binder H, Fritz J, Leertouwer I, Lunansky G, et al. Deconstructing and reconstructing resilience: a dynamic network approach. Perspect Psychol Sci. 2019;14:765–77.
Bonanno GA, Kennedy P, Galatzer-Levy IR, Lude P, Elfström ML. Trajectories of resilience, depression, and anxiety following spinal cord injury. Rehabil Psychol. 2012;57:236–47.
Galatzer-Levy IR, Huang SH, Bonanno GA. Trajectories of resilience and dysfunction following potential trauma: A review and statistical evaluation. Clin Psychol Rev. 2018;63:41–55.
Galatzer-Levy IR, Ankri Y, Freedman S, Israeli-Shalev Y, Roitman P, Gilad M, et al. Early PTSD symptom trajectories: persistence, recovery, and response to treatment: results from the Jerusalem Trauma Outreach and Prevention Study (J-TOPS). PLoS One. 2013;8:e70084.
NOT-MH-18-058: Notice of information: NIMHs Interest in Areas of Stress Biology Research. https://grants.nih.gov/grants/guide/notice-files/NOT-MH-18-058.html (accessed 12 Jul 2021).
American Psychiatric Association AP, Association AP, Others. Diagnostic and statistical manual of mental disorders: DSM-5. Washington, DC: American Psychiatric Association, 2013.
Williams LM. Defining biotypes for depression and anxiety based on large-scale circuit dysfunction: a theoretical review of the evidence and future directions for clinical translation. Depress Anxiety. 2017;34:9–24.
Williams LM. Precision psychiatry: a neural circuit taxonomy for depression and anxiety. Lancet Psychiatry. 2016;3:472–80.
Ressler KJ. Translating across circuits and genetics toward progress in fear- and anxiety-related disorders. Am J Psychiatry. 2020;177:214–22.
Siddiqi SH, Schaper FLWVJ, Horn A, Hsu J, Padmanabhan JL, Brodtmann A, et al. Brain stimulation and brain lesions converge on common causal circuits in neuropsychiatric disease. Nature Human Behavior. 2021; https://doi.org/10.1038/s41562-021-01161-1.
Li N, Hollunder B, Baldermann JC, Kibleur A, Treu S, Akram H, et al. A Unified Functional Network Target for Deep Brain Stimulation in Obsessive-Compulsive Disorder. Biol Psychiatry 2021; https://doi.org/10.1016/j.biopsych.2021.04.006.
Fitzgerald JM, DiGangi JA, Phan KL. Functional neuroanatomy of emotion and its regulation in PTSD. Harv Rev Psychiatry. 2018;26:116–28.
Admon R, Milad MR, Hendler T. A causal model of post-traumatic stress disorder: disentangling predisposed from acquired neural abnormalities. Trends Cogn Sci. 2013;17:337–47.
McLaughlin KA, Busso DS, Duys A, Green JG, Alves S, Way M, et al. Amygdala response to negative stimuli predicts PTSD symptom onset following a terrorist attack. Depress Anxiety. 2014;31:834–42.
Admon R, Lubin G, Stern O, Rosenberg K, Sela L, Ben-Ami H, et al. Human vulnerability to stress depends on amygdala’s predisposition and hippocampal plasticity. Proc Natl Acad Sci USA. 2009;106:14120–5.
Admon R, Lubin G, Rosenblatt JD, Stern O, Kahn I, Assaf M, et al. Imbalanced neural responsivity to risk and reward indicates stress vulnerability in humans. Cereb Cortex. 2013;23:28–35.
Lin T, Gilam G, Raz G, Or-Borichev A, Bar-Haim Y, Fruchter E, et al. Accessible neurobehavioral anger-related markers for vulnerability to post-traumatic stress symptoms in a population of male soldiers. Front Behav Neurosci. 2017;11:38.
Swartz JR, Knodt AR, Radtke SR, Hariri AR. A neural biomarker of psychological vulnerability to future life stress. Neuron. 2015;85:505–11.
Mattson WI, Hyde LW, Shaw DS, Forbes EE, Monk CS. Clinical neuroprediction: amygdala reactivity predicts depressive symptoms 2 years later. Soc Cogn Affect Neurosci. 2016;11:892–8.
Cwik JC, Vahle N, Woud ML, Potthoff D, Kessler H, Sartory G, et al. Reduced gray matter volume in the left prefrontal, occipital, and temporal regions as predictors for posttraumatic stress disorder: a voxel-based morphometric study. Eur Arch Psychiatry Clin Neurosci. 2020;270:577–88.
van Wingen GA, Geuze E, Vermetten E, Fernández G. Perceived threat predicts the neural sequelae of combat stress. Mol Psychiatry. 2011;16:664–71.
van Wingen GA, Geuze E, Vermetten E, Fernández G. The neural consequences of combat stress: long-term follow-up. Mol Psychiatry. 2012;17:116–8.
Samuels E, Szabadi E. Functional neuroanatomy of the noradrenergic locus coeruleus: its roles in the regulation of arousal and autonomic function part I: principles of functional organisation. Curr Neuropharmacol. 2008;6:235–53.
Grueschow M, Stenz N, Thörn H, Ehlert U, Breckwoldt J, Brodmann Maeder M, et al. Real-world stress resilience is associated with the responsivity of the locus coeruleus. Nat Commun. 2021;12:2275.
Gilam G, Lin T, Fruchter E, Hendler T. Neural indicators of interpersonal anger as cause and consequence of combat training stress symptoms. Psychol Med. 2017;47:1561–72.
Sekiguchi A, Sugiura M, Taki Y, Kotozaki Y, Nouchi R, Takeuchi H, et al. Brain structural changes as vulnerability factors and acquired signs of post-earthquake stress. Mol Psychiatry. 2013;18:618–23.
Sekiguchi A, Sugiura M, Taki Y, Kotozaki Y, Nouchi R, Takeuchi H, et al. White matter microstructural changes as vulnerability factors and acquired signs of post-earthquake distress. PLoS ONE. 2014;9:e83967.
Anand KS, Dhikav V. Hippocampus in health and disease: an overview. Ann Indian Acad Neurol. 2012;15:239–46.
Pitman RK, Gilbertson MW, Gurvits TV, May FS, Lasko NB, Metzger LJ, et al. Clarifying the origin of biological abnormalities in PTSD through the study of identical twins discordant for combat exposure. Ann N Y Acad Sci. 2006;1071:242–54.
Koch SBJ, van Ast VA, Kaldewaij R, Hashemi MM, Zhang W, Klumpers F, et al. Larger dentate gyrus volume as predisposing resilience factor for the development of trauma-related symptoms. Neuropsychopharmacology. 2021;46:1283–92. https://doi.org/10.1038/s41386-020-00947-7
Shin LM, Lasko NB, Macklin ML, Karpf RD, Milad MR, Orr SP, et al. Resting metabolic activity in the cingulate cortex and vulnerability to posttraumatic stress disorder. Arch Gen Psychiatry. 2009;66:1099–107.
Shin LM, Bush G, Milad MR, Lasko NB, Brohawn KH, Hughes KC, et al. Exaggerated activation of dorsal anterior cingulate cortex during cognitive interference: a monozygotic twin study of posttraumatic stress disorder. Am J Psychiatry. 2011;168:979–85.
Reynaud E, Guedj E, Trousselard M, El Khoury-Malhame M, Zendjidjian X, Fakra E, et al. Acute stress disorder modifies cerebral activity of amygdala and prefrontal cortex. Cogn Neurosci. 2015;6:39–43.
Papagni SA, Benetti S, Arulanantham S, McCrory E, McGuire P, Mechelli A. Effects of stressful life events on human brain structure: A longitudinal voxel-based morphometry study. Stress. 2011;14:227–32.
Coad BM, Postans M, Hodgetts CJ, Muhlert N, Graham KS, Lawrence AD. Structural connections support emotional connections: Uncinate fasciculus microstructure is related to the ability to decode facial emotion expressions. Neuropsychologia. 2020;145:106562.
Sekiguchi A, Kotozaki Y, Sugiura M, Nouchi R, Takeuchi H, Hanawa S, et al. Long-term effects of postearthquake distress on brain microstructural changes. Biomed Res Int. 2014;2014:180468.
Koechlin E, Basso G, Pietrini P, Panzer S, Grafman J. The role of the anterior prefrontal cortex in human cognition. Nature. 1999;399:148–51.
Kovach CK, Daw ND, Rudrauf D, Tranel D, O'Doherty JP, Adolphs R. Anterior prefrontal cortex contributes to action selection through tracking of recent reward trends. J Neurosci. 2012;32:8434–42.
Koch SBJ, Mars RB, Toni I, Roelofs K. Emotional control, reappraised. Neurosci Biobehav Rev. 2018;95:528–34.
Kaldewaij R, Koch S, Hashemi MM, Zhang W, Klumpers F, Roelofs K. Anterior prefrontal brain activity during emotion control predicts resilience to post-traumatic stress symptoms. Nat Hum Behav. 2021;5:1055–64. https://doi.org/10.1038/s41562-021-01055-2.
Stevens JS, Kim YJ, Galatzer-Levy IR, Reddy R, Ely TD, Nemeroff CB, et al. Amygdala reactivity and anterior cingulate habituation predict posttraumatic stress disorder symptom maintenance after acute civilian trauma. Biol Psychiatry. 2017;81:1023–9.
Stein MB, Yuh E, Jain S, Okonkwo DO, Mac Donald CL, Levin H, et al. Smaller regional brain volumes predict posttraumatic stress disorder at 3 months after mild traumatic brain injury. Biol Psychiatry Cogn Neurosci Neuroimaging. 2021;6:352–9.
Hu H, Sun Y, Su S, Wang Y, Qiu Y, Yang X, et al. Cortical surface area reduction in identification of subjects at high risk for post-traumatic stress disorder: A pilot study. Aust N Z J Psychiatry. 2018;52:1084–91.
White SF, Costanzo ME, Thornton LC, Mobley AM, Blair JR, Roy MJ. Increased cognitive control and reduced emotional interference is associated with reduced PTSD symptom severity in a trauma-exposed sample: A preliminary longitudinal study. Psychiatry Res Neuroimaging. 2018;278:7–12.
Wang X, Xie H, Cotton AS, Duval ER, Tamburrino MB, Brickman KR, et al. Preliminary study of acute changes in emotion processing in trauma survivors with PTSD symptoms. PLoS ONE. 2016;11:e0159065.
Dickie EW, Brunet A, Akerib V, Armony JL. Anterior cingulate cortical thickness is a stable predictor of recovery from post-traumatic stress disorder. Psychol Med. 2013;43:645–53.
Harnett NG, Stevens JS, van Rooij S, Ely TD, Michopoulos V, Hudak L, et al. Multimodal structural neuroimaging markers of risk and recovery from posttrauma anhedonia: A prospective investigation. Depress Anxiety. 2021;38:79–88.
Sun Y, Wang Z, Ding W, Wan J, Zhuang Z, Zhang Y, et al. Alterations in white matter microstructure as vulnerability factors and acquired signs of traffic accident-induced PTSD. PLoS ONE. 2013;8:e83473.
Mangelsdorf J. Coping with childbirth: Brain structural associations of personal growth initiative. Front Psychol. 2017;8:1829.
Ke J, Zhang L, Qi R, Li W, Hou C, Zhong Y, et al. A longitudinal fMRI investigation in acute post-traumatic stress disorder (PTSD). Acta Radio. 2016;57:1387–95.
Xie H, Claycomb Erwin M, Elhai JD, Wall JT, Tamburrino MB, Brickman KR, et al. Relationship of hippocampal volumes and posttraumatic stress disorder symptoms over early posttrauma periods. Biol Psychiatry Cogn Neurosci Neuroimaging. 2018;3:968–75.
Ben-Zion Z, Artzi M, Niry D, Keynan NJ, Zeevi Y, Admon R, et al. Neuroanatomical risk factors for posttraumatic stress disorder in Recent Trauma Survivors. Biol Psychiatry Cogn Neurosci Neuroimaging. 2020;5:311–9.
Quidé Y, Andersson F, Dufour-Rainfray D, Descriaud C, Brizard B, Gissot V, et al. Smaller hippocampal volume following sexual assault in women is associated with post-traumatic stress disorder. Acta Psychiatr Scand. 2018;138:312–24.
Terpstra AR, Girard TA, Colella B, Green REA. Higher anxiety symptoms predict progressive hippocampal atrophy in the chronic stages of moderate to severe traumatic brain injury. Neurorehabil Neural Repair. 2017;31:1063–71.
Weis CN, Webb EK, Huggins AA, Kallenbach M, Miskovich TA, Fitzgerald JM, et al. Stability of hippocampal subfield volumes after trauma and relationship to development of PTSD symptoms. Neuroimage. 2021;236:118076.
van Rooij SJH, Stevens JS, Ely TD, Hinrichs R, Michopoulos V, Winters SJ, et al. The role of the hippocampus in predicting future posttraumatic stress disorder symptoms in recently traumatized civilians. Biol Psychiatry. 2018;84:106–15.
van Rooij SJH, Ravi M, Ely TD, Michopoulos V, Winters SJ, Shin J, et al. Hippocampal activation during contextual fear inhibition related to resilience in the early aftermath of trauma. Behav Brain Res. 2021;408:113282.
Dickie EW, Brunet A, Akerib V, Armony JL. Neural correlates of recovery from post-traumatic stress disorder: a longitudinal fMRI investigation of memory encoding. Neuropsychologia. 2011;49:1771–8.
Stevens JS, Jovanovic T, Fani N, Ely TD, Glover EM, Bradley B, et al. Disrupted amygdala-prefrontal functional connectivity in civilian women with posttraumatic stress disorder. J Psychiatr Res. 2013;47:1469–78.
Yoon S, Kim JE, Hwang J, Kang I, Jeon S, Im JJ, et al. Recovery from posttraumatic stress requires dynamic and sequential shifts in amygdalar connectivities. Neuropsychopharmacology. 2017;42:454–61.
Lanius RA, Bluhm RL, Coupland NJ, Hegadoren KM, Rowe B, Théberge J, et al. Default mode network connectivity as a predictor of post-traumatic stress disorder symptom severity in acutely traumatized subjects. Acta Psychiatr Scand. 2010;121:33–40.
Roy MJ, Costanzo M, Gill J, Leaman S, Law W, Ndiongue R, et al. Predictors of neurocognitive syndromes in combat veterans. Cureus. 2015;7:e293.
Nilsen AS, Blix I, Leknes S, Ekeberg Ø, Skogstad L, Endestad T, et al. Brain activity in response to trauma-specific, negative, and neutral stimuli. A fMRI study of recent road traffic accident survivors. Front Psychol. 2016;7:1173.
Webb EK, Huggins AA, Belleau EL, Taubitz LE, Hanson JL, deRoon-Cassini TA, et al. Acute posttrauma resting-state functional connectivity of periaqueductal gray prospectively predicts posttraumatic stress disorder symptoms. Biol Psychiatry Cogn Neurosci Neuroimaging. 2020;5:891–900.
Hakamata Y, Matsuoka Y, Inagaki M, Nagamine M, Hara E, Imoto S, et al. Structure of orbitofrontal cortex and its longitudinal course in cancer-related post-traumatic stress disorder. Neurosci Res. 2007;59:383–9.
O’Doherty J, Kringelbach ML, Rolls ET, Hornak J, Andrews C. Abstract reward and punishment representations in the human orbitofrontal cortex. Nat Neurosci. 2001;4:95–102.
Kringelbach ML, O’Doherty J, Rolls ET, Andrews C. Activation of the human orbitofrontal cortex to a liquid food stimulus is correlated with its subjective pleasantness. Cereb Cortex. 2003;13:1064–71.
Gottfried JA, Dolan RJ. Human orbitofrontal cortex mediates extinction learning while accessing conditioned representations of value. Nat Neurosci. 2004;7:1144–52.
Ochsner KN, Gross JJ. The cognitive control of emotion. Trends Cogn Sci. 2005;9:242–9.
Greicius MD, Supekar K, Menon V, Dougherty RF. Resting-state functional connectivity reflects structural connectivity in the default mode network. Cereb Cortex. 2008;19:72–78.
Greicius MD, Krasnow B, Reiss AL, Menon V. Functional connectivity in the resting brain: a network analysis of the default mode hypothesis. Proc Natl Acad Sci. 2003;100:253–8.
Busso DS, McLaughlin KA, Brueck S, Peverill M, Gold AL, Sheridan MA. Child abuse, neural structure, and adolescent psychopathology: a longitudinal study. J Am Acad Child Adolesc Psychiatry. 2017;56:321–328.e1.
Heyn SA, Keding TJ, Ross MC, Cisler JM, Mumford JA, Herringa RJ. Abnormal prefrontal development in pediatric posttraumatic stress disorder: a longitudinal structural and functional magnetic resonance imaging study. Biol Psychiatry Cogn Neurosci Neuroimaging. 2019;4:171–9.
Gong Q, Li L, Tognin S, Wu Q, Pettersson-Yeo W, Lui S, et al. Using structural neuroanatomy to identify trauma survivors with and without post-traumatic stress disorder at the individual level. Psychol Med. 2014;44:195–203.
Zsoldos E, Filippini N, Mahmood A, Mackay CE, Singh-Manoux A, Kivimäki M, et al. Allostatic load as a predictor of grey matter volume and white matter integrity in old age: the Whitehall II MRI study. Sci Rep. 2018;8:6411.
Heyn SA, Herringa RJ. Longitudinal cortical markers of persistence and remission of pediatric PTSD. Neuroimage Clin. 2019;24:102028.
Cardenas VA, Samuelson K, Lenoci M, Studholme C, Neylan TC, Marmar CR, et al. Changes in brain anatomy during the course of posttraumatic stress disorder. Psychiatry Res. 2011;193:93–100.
Cwik JC, Sartory G, Nuyken M, Schürholt B, Seitz RJ. Posterior and prefrontal contributions to the development posttraumatic stress disorder symptom severity: an fMRI study of symptom provocation in acute stress disorder. Eur Arch Psychiatry Clin Neurosci. 2017;267:495–505.
Harnett NG, van Rooij SJH, Ely TD, Lebois LAM, Murty VP, Jovanovic T, et al. Prognostic neuroimaging biomarkers of trauma-related psychopathology: resting-state fMRI shortly after trauma predicts future PTSD and depression symptoms in the AURORA study. Neuropsychopharmacology. 2021;46:1263–71. https://doi.org/10.1038/s41386-020-00946-8.
Banks SD, Coronado RA, Clemons LR, Abraham CM, Pruthi S, Conrad BN, et al. Thalamic functional connectivity in mild traumatic brain injury: Longitudinal associations with patient-reported outcomes and neuropsychological tests. Arch Phys Med Rehabil. 2016;97:1254–61.
Quidé Y, Zine A, Descriaud C, Saint-Martin P, Andersson F, El-Hage W. Aberrant intrinsic connectivity in women victims of sexual assault. Brain Imaging Behav 2021; https://doi.org/10.1007/s11682-020-00431-2.
King AP, Block SR, Sripada RK, Rauch S, Giardino N, Favorite T, et al. Altered default mode network resting state functional connectivity following a mindfulness-based exposure therapy for posttraumatic stress disorder in combat veterans of Afghanistan and Iraq. Depress Anxiety. 2016;33:289–99.
Sheynin J, Duval ER, King AP, Angstadt M, Phan KL, Simon NM, et al. Associations between resting-state functional connectivity and treatment response in a randomized clinical trial for posttraumatic stress disorder. Depress Anxiety. 2020;37:1037–46.
Fornito A, Harrison BJ, Zalesky A, Simons JS. Competitive and cooperative dynamics of large-scale brain functional networks supporting recollection. Proc Natl Acad Sci USA. 2012;109:12788–93.
Hu H, Zhou Y, Wang Q, Su S, Qiu Y, Ge J, et al. Association of abnormal white matter integrity in the acute phase of motor vehicle accidents with post-traumatic stress disorder. J Affect Disord. 2016;190:714–22.
Chen AC, Oathes DJ, Chang C, Bradley T, Zhou ZW, Williams LM, et al. Causal interactions between fronto-parietal central executive and default-mode networks in humans. Proc Natl Acad Sci USA. 2013;110:19944–9.
Niendam TA, Laird AR, Ray KL, Dean YM, Glahn DC, Carter CS. Meta-analytic evidence for a superordinate cognitive control network subserving diverse executive functions. Cogn Affect Behav Neurosci. 2012;12:241–68.
Lyoo IK, Kim JE, Yoon SJ, Hwang J, Bae S, Kim DJ. The neurobiological role of the dorsolateral prefrontal cortex in recovery from trauma. Longitudinal brain imaging study among survivors of the South Korean subway disaster. Arch Gen Psychiatry. 2011;68:701–13.
Meng L, Chen Y, Xu X, Chen T, Lui S, Huang X, et al. The neurobiology of brain recovery from traumatic stress: A longitudinal DTI study. J Affect Disord. 2018;225:577–84.
Daniels JK, Hegadoren KM, Coupland NJ, Rowe BH, Densmore M, Neufeld RWJ, et al. Neural correlates and predictive power of trait resilience in an acutely traumatized sample: a pilot investigation. J Clin Psychiatry. 2012;73:327–32.
Beaudoin FL, Kessler RC, Hwang I, Lee S, Sampson NA, An X, et al. Pain after a motor vehicle crash: the role of socio-demographics, crash characteristics, and peri-traumatic stress symptoms. Eur J Pain. 2021;25:1119–36. https://doi.org/10.1002/ejp.1733.
Douglas Bremner J, Marmar CR Trauma, Memory, and Dissociation. American Psychiatric Pub, 2002.
Kessler RC, Ressler KJ, House SL, Beaudoin FL, An X, Stevens JS, et al. Socio-demographic and trauma-related predictors of PTSD within 8 weeks of a motor vehicle collision in the AURORA study. Mol Psychiatry. 2020; https://doi.org/10.1038/s41380-020-00911-3.
Daniels JK, Coupland NJ, Hegadoren KM, Rowe BH, Densmore M, Neufeld RWJ, et al. Neural and behavioral correlates of peritraumatic dissociation in an acutely traumatized sample. J Clin Psychiatry. 2012;73:420–6.
Fenster RJ, Lebois LAM, Ressler KJ, Suh J. Brain circuit dysfunction in post-traumatic stress disorder: from mouse to man. Nat Rev Neurosci. 2018;19:535–51.
Wolf EJ, Lunney CA, Miller MW, Resick PA, Friedman MJ, Schnurr PP. The dissociative subtype of PTSD: a replication and extension. Depress Anxiety. 2012;29:679–88.
Stein DJ, Koenen KC, Friedman MJ, Hill E, McLaughlin KA, Petukhova M, et al. Dissociation in posttraumatic stress disorder: Evidence from the world mental health surveys. Biol Psychiatry. 2013;73:302–12.
Lanius RA, Vermetten E, Loewenstein RJ, Brand B, Schmahl C, Bremner JD, et al. Emotion modulation in PTSD: Clinical and neurobiological evidence for a dissociative subtype. Am J Psychiatry. 2010;167:640–7.
Nicholson AA, Densmore M, Frewen PA, Théberge J, Neufeld RW, McKinnon MC, et al. The dissociative subtype of posttraumatic stress disorder: Unique resting-state functional connectivity of basolateral and centromedial amygdala Complexes. Neuropsychopharmacology. 2015;40:2317–26.
Nicholson AA, Sapru I, Densmore M, Frewen PA, Neufeld RW, Théberge J, et al. Unique insula subregion resting-state functional connectivity with amygdala complexes in posttraumatic stress disorder and its dissociative subtype. Psychiatry Res. 2016;250:61–72.
Harricharan S, Rabellino D, Frewen PA, Densmore M, Théberge J, McKinnon MC, et al. fMRI functional connectivity of the periaqueductal gray in PTSD and its dissociative subtype. Brain Behav. 2016;6:e00579.
Garrett A, Cohen JA, Zack S, Carrion V, Jo B, Blader J, et al. Longitudinal changes in brain function associated with symptom improvement in youth with PTSD. J Psychiatr Res. 2019;114:161–9.
Ebner-Priemer UW, Mauchnik J, Kleindienst N, Schmahl C, Peper M, Rosenthal MZ, et al. Emotional learning during dissociative states in borderline personality disorder. J Psychiatry Neurosci. 2009;34:214–22.
Kleindienst N, Priebe K, Görg N, Dyer A, Steil R, Lyssenko L, et al. State dissociation moderates response to dialectical behavior therapy for posttraumatic stress disorder in women with and without borderline personality disorder. Eur J Psychotraumatol. 2016;7:30375.
Kozlowska K, Walker P, McLean L, Carrive P. Fear and the defense cascade: Clinical implications and management. Harv Rev Psychiatry. 2015;23:263–87.
Lanius RA, Boyd JE, McKinnon MC, Nicholson AA, Frewen P, Vermetten E, et al. A review of the neurobiological basis of trauma-related dissociation and its relation to cannabinoid-and opioid-mediated stress response: a transdiagnostic, translational approach. Curr Psychiatry Rep. 2018;20:118.
De Bellis MD, Zisk A. The biological effects of childhood trauma. Child Adolesc Psychiatr Clin N Am. 2014;23:185–222.
Saxbe D, Khoddam H, Piero LD, Stoycos SA, Gimbel SI, Margolin G, et al. Community violence exposure in early adolescence: Longitudinal associations with hippocampal and amygdala volume and resting state connectivity. Dev Sci. 2018;21:e12686.
Whittle S, Simmons JG, Hendriksma S, Vijayakumar N, Byrne ML, Dennison M, et al. Childhood maltreatment, psychopathology, and the development of hippocampal subregions during adolescence. Brain Behav. 2017;7:e00607.
Bonne O, Brandes D, Gilboa A, Gomori JM, Shenton ME, Pitman RK, et al. Longitudinal MRI study of hippocampal volume in trauma survivors with PTSD. Am J Psychiatry. 2001;158:1248–51.
De Bellis MD, Hall J, Boring AM, Frustaci K, Moritz G. A pilot longitudinal study of hippocampal volumes in pediatric maltreatment-related posttraumatic stress disorder. Biol Psychiatry. 2001;50:305–9.
Harms MB, Birn R, Provencal N, Wiechmann T, Binder EB, Giakas SW, et al. Early life stress, FK506 binding protein 5 gene (FKBP5) methylation, and inhibition-related prefrontal function: a prospective longitudinal study. Dev Psychopathol. 2017;29:1895–903.
Hartley CA, Lee FS. Sensitive periods in affective development: nonlinear maturation of fear learning. Neuropsychopharmacology. 2015;40:50–60.
van Rooij SJH, Smith RD, Stenson AF, Ely TD, Yang X, Tottenham N, et al. Increased activation of the fear neurocircuitry in children exposed to violence. Depress Anxiety. 2020;37:303–12.
Akeman E, Kirlic N, Clausen AN, Cosgrove KT, McDermott TJ, Cromer LD, et al. A pragmatic clinical trial examining the impact of a resilience program on college student mental health. Depress Anxiety. 2020;37:202–13.
Daniels JK, Hegadoren K, Coupland NJ, Rowe BH, Neufeld RW, Lanius RA. Lanius Cognitive distortions in an acutely traumatized sample: an investigation of predictive power and neural correlates. Psychol Med. 2011;41:2149–2157. https://doi.org/10.1017/S0033291711000237.
Weems CF, Scott BG, Russell JD, Reiss AL, Carrión VG. Developmental variation in amygdala volumes among children with posttraumatic stress. Dev Neuropsychol. 2013;38:481–95.
Du MY, Liao W, Lui S, Huang X-Q, Li F, Kuang W-H, et al. Altered functional connectivity in the brain default-mode network of earthquake survivors persists after 2 years despite recovery from anxiety symptoms. Soc Cogn Affect Neurosci. 2015;10:1487–1505.
Li L, Sun G, Liu K, Li M, Li B, Qian SW, et al. White matter changes in posttraumatic stress disorder following mild traumatic brain injury. Chin Med J. 2016;129:1091–1099. https://doi.org/10.4103/0366-6999.180518.
Belleau EL, Ehret LE, Hanson JL, Brasel KJ, Larson CL, deRoon-Cassini TA. Amygdala functional connectivity in the acute aftermath of trauma prospectively predicts severity of posttraumatic stress symptoms. Neurobiol. Stress. 2020;12:100217.
Fani N, Michopoulos V, van Rooij S, Clendinen C, Hardy RA, Jovanovic T, et al. Structural connectivity and risk for anhedonia after trauma: A prospective study and replication. J Psychiatr Res. 2019;116:34–41.
Cai W, Ryali S, Chen T, Li CSR. Dissociable roles of right inferior frontal cortex and anterior insula in inhibitory control: Evidence from intrinsic and task-related functional parcellation, connectivity, and response profile analysis across multiple datasets. J Neurosci. 2014;34:14652–67.
Yeo BT, Krienen FM, Sepulcre J, Sabuncu MR, Lashkari D, Hollinshead M, et al. The organization of the human cerebral cortex estimated by intrinsic functional connectivity. J Neurophysiol. 2011;106:1125–65.
Acknowledgements
This work was supported by the National Institute of Mental Health (NIMH), including U01MH110925, K01MH121653 (to SVR), K01MH118467 (to LAML), and T32NS096050 (to KIO), and the Brain and Behavior Research Foundation, NARSAD Young Investigator Grant to SVR.
Author information
Authors and Affiliations
Contributions
ARR, KIO, LAML, SJHVR, and JSS conceptualized the paper and its organization, conducted the structured literature search, wrote sections of the first draft, and contributed to subsequent edits. KIO created the first draft of Fig. 1 and ARR created Fig. 2.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Roeckner, A.R., Oliver, K.I., Lebois, L.A.M. et al. Neural contributors to trauma resilience: a review of longitudinal neuroimaging studies. Transl Psychiatry 11, 508 (2021). https://doi.org/10.1038/s41398-021-01633-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41398-021-01633-y
This article is cited by
-
Effects of lockdowns on neurobiological and psychometric parameters in unipolar depression during the COVID-19 pandemic
Translational Psychiatry (2024)
-
Multimodal neural correlates of dispositional resilience among healthy individuals
Scientific Reports (2024)
-
Broadening the Scope of Resilience in Chronic Pain: Methods, Social Context, and Development
Current Rheumatology Reports (2024)
-
Defining the r factor for post-trauma resilience and its neural predictors
Nature Mental Health (2024)
-
Unexpected circuit-level tradeoffs in human stress resilience
Neuropsychopharmacology (2023)