Abstract
In neuroscience, the term ‘Stress’ has a negative connotation because of its potential to trigger or exacerbate psychopathologies. Yet in the face of exposure to stress, the more common reaction to stress is resilience, indicating that resilience is the rule and stress-related pathology the exception. This is critical because neural mechanisms associated with stress-related psychopathology are expected to differ significantly from those associated with resilience.
Research labels and terminology affect research directions, conclusions drawn from the results, and the way we think about a topic, while choice of labels is often influenced by biases and hidden assumptions. It is therefore important to adopt a terminology that differentiates between stress conditions, leading to different outcomes.
Here, we propose to conceptually associate the term ‘stress’/‘stressful experience’ with ‘stress resilience’, while restricting the use of the term ‘trauma’ only in reference to exposures that lead to pathology. We acknowledge that there are as yet no ideal ways for addressing the murkiness of the border between stressful and traumatic experiences. Yet ignoring these differences hampers our ability to elucidate the mechanisms of trauma-related pathologies on the one hand, and of stress resilience on the other. Accordingly, we discuss how to translate such conceptual terminology into research practice.
Similar content being viewed by others
Introduction
Our choice of labels and metaphors for the information we observe in the world impacts the way we process and understand it [1]. This is true also in science, where choice of terminology is often influenced by hidden assumptions and predispositions, which in turn may affect not only research directions, but also the conclusions drawn from the results [1]. Taking the time, every now and then, to reflect on customary terminology and on the potential benefit of considering alternative terminology is important for any field of research. It is certainly relevant for the field of the neurobiology of stress to reflect and consider hidden biases related with the use of the term Stress [2, 3].
Stress has a negative connotation. Particularly because of its known potential to trigger or exacerbate pathologies [4,5,6,7]. Yet both humans and animals demonstrate surprisingly high resilience to it [8,9,10], indicating that resilience is the rule and stress-related pathology the exception. Indeed, the physiological stress response has been selected throughout evolution as an important survival reaction [11, 12]. Thus, it seems likely that the neural mechanisms associated with trauma-related psychopathology are different from the neural mechanisms that are triggered by stressful experiences that do not lead to trauma-related psychopathology [13]. This likely distinction, emphasizes the importance of differentiating between the terms ‘stress’ and ‘trauma’. In order to differentiate between neural mechanisms that are associated with the pathology and those actually associated with stress resilience, it is critical to be able to differentiate between individuals who were exposed to a significant emotional challenge but did not develop pathology and those who did.
Understanding the stress-response: from simplistic text-book descriptions to a complex integrative view
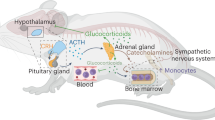
The stress-response system is often described as a combined neuroendocrine response, with a bias toward emphasizing its endocrinological arm: an encounter with a stressful challenge activates simultaneously the sympathetic nervous system (SNS) and the hypothalamus–pituitary–adrenal (HPA) axis. This response, often referred to as ‘the’ stress response system, comprises two stages: first, a fast response, in which the SNS is activated to trigger a myriad of peripheral actions, both through direct action of sympathetic nerves and through the release of adrenaline from the core of the adrenal gland. Second, a slower response, known as HPA axis response, in which the release of corticotropin-releasing hormone (CRH) from the paraventricular nucleus of the hypothalamus induces the release of adrenocorticotropic hormone (ACTH) from the anterior pituitary gland, which in turn triggers the release of cortisol (or corticosterone) from the adrenal cortex.
While these textbook descriptions are correct, they often present a misleading oversimplification of what is taking place and miss the essence of the stress response. Too frequently, cortisol is referred to as the ‘stress hormone’, implying that it is the most significant aspect of the stress response, and reflecting literature’s overemphasis of its role in the stress response [14]. Yet, while cortisol is indeed secreted during stress, it takes over ten-to-twenty minutes for cortisol blood levels to rise to their stress-related peak [15]. This is significant since many highly stressful situations, such as predator–prey encounters, normally last only a few minutes, as any National Geographic documentary fan well knows.
In fact, cortisol has a dual role in the stress response: in addition to mobilizing resources to cope with a current stressor, once the threat is over, cortisol is actually responsible for bringing the stress response to its end, via its inhibitory feedback effect on the hypothalamus and pituitary gland [16, 17]. If the threat is still present, the role of cortisol is to shift the stress response into a longer-period response, balancing between the emergency needs for energy for physical responses and the need to maintain the ability to respond for a longer period of time. Furthermore, cortisol is not secreted only in response to stress and is not triggered only by negative challenges or adverse experiences. Pleasurable activities, such as sports and sex, also lead to increased secretion of cortisol [15, 18].
More recently, there is growing attention to the energy component of the stress response [19, 20]. Cortisol’s role as a metabolic hormone is emphasized in this context, because of its effects on increasing blood glucose levels and its capacity to eventually directly affect mitochondrial function [19,20,21,22]. Indeed, an important part of the stress response is metabolic control, as increases in glucocorticoid levels can lead to increases in glucose and modulate adiponectin levels [22,23,24,25,26]. Furthermore, stress and trauma-related psychopathologies, such as post-traumatic stress disorder (PTSD), or stress-induced mood disorders, are known to have high comorbidity with metabolic dysregulation and metabolic syndromes [19, 27, 28].
Therefore, the stress response emerges nowadays as a complex and integrative process that involves, among others, neuronal–neuroendocrine-immune interactions [19, 23, 27, 29,30,31,32,33,34] and these interactions are also influenced by an individual’s genetic background [35,36,37,38,39].
The challenge of defining a stress response
Attempts to define the concept of Stress frequently fail to encompass its complexity [14, 15, 40, 41]. Hans Selye, often credited as the modern-day father of stress research, defined stress in 1936 as “the nonspecific response of the body to any demand for change” [42]. This definition is clearly too general and circumscribes the term stress response to the body’s response to a stressor. Yet more than eighty years after Selye’s original definition [42], a clear definition of the stress response remains elusive. One important reason for this is that any response to any environmental stimulus involves some physiological reaction. Intuitively, when researchers refer to a response to a stressor, they refer to a response to a challenging event, which would seem to require a significant reaction. Yet at what stage does a response start to fall into the category of a stress response? This is where the lines begin to blur. To make matters worse, many of the physiological reactions that could form part of a stress response could just as well be components of responses to much milder or positive challenges that would not fall into the category of a stressor.
Attempts to address this difficulty in definition brought up the terms Allostasis and Allostatic Load [31]. Allostasis refers to the mechanisms employed to achieve stability through change in response to constantly changing social and physical environments [43]. In many cases, Allostasis involves adaptive plasticity, i.e., lasting neurobiological, endocrine, or immune-response alterations, designed to adjust the body physiology to the new conditions [31]. The term Allostatic Load was later proposed to refer to the long-term consequences of activating allostatic mechanisms [44], and to emphasize that, while a brief activation of allostatic mechanisms in response to environmental challenges may be adaptive, prolonged activation, in response to chronic challenges, is likely to lead to pathological outcomes [45]. Indeed, the term allostatic overload was introduced in order to emphasize that the same stress response, when activated for long periods of time, may come with a cost to the organism and establish the foundation of various metabolic, cardiovascular, neurological and mental diseases [34, 46, 47]. Recently, there have been attempts to create a diagnostic scale of allostatic overload that would help guiding treatment [48].
The subjective nature of the experience
When people describe a stressful experience, the intensity of the stress is often related to the characteristics of the stressor: “…it was a terrible car accident…,” “…they experienced an awful earthquake…,” “…she survived a horrible terrorist attack…”. Indeed, a serious car accident, which involves people injured, is more likely to lead to the development of psychopathological symptoms than a mild car accident, in which no one was hurt. The same principle is used for designing animal experiments. For example, using 0.4 mA as a mild-intensity foot-shock experience, and 1 mA as a severe stressful experience [49, 50]. On average, it can be expected that the stress-response measurements of the group that received 1-mA foot shock would be higher than those of the group receiving 0.4 mA.
Yet the characteristics of the stressor by themselves do not define the intensity of the individual stress experience, they only contribute to it [51, 52]. This is a critical aspect of stress, and it makes it impossible to define a stress experience by using only the parameters of the stressor. The impact of exposure to a stressor is a combination of the parameters of the stressor, the characteristics of the individual’s physiological machinery, and the subjective way the stressor is perceived by the individual [53,54,55,56]. The subjective nature and the huge individual variability observed following exposure to a stressor is one of the defining hallmarks of the stress response [13, 54, 57,58,59]. For example, the lifetime prevalence of exposure to severely stressful events like combat, accidents, natural disasters, assault, or rape is as high as 75–80% [60, 61]. If the characteristics of the stressor were the major factor in defining the outcome, we would expect that the prevalence of trauma-related disorders, such as PTSD, would reflect this high percentage of exposure to severely stressful events. Yet only about 10–20% of the population exposed to these types of stressors will suffer from clinically relevant PTSD [60, 62,63,64,65]. In other words, regardless of the characteristics of the trauma, only a minority of the exposed individuals eventually develop pathology. This signifies the importance of both the neurobiological constitution of individuals in producing stress responses and the subjective nature of the impact of stress and trauma, i.e., the same stressful event may lead to very different responses in different individuals. This is true for both humans and animals. It is now becoming clear that stressful experiences cannot be defined solely by the conditions to which an individual was exposed, because the eventual emotional experience of these conditions will also greatly depend on the combination of their physiological constitution and their subjective perception of the event by the individual—the perceived experience.
The realization of the critical role of the subjectivity of stress experiences represents a significant challenge to the neurobiology of stress field. The scientific method, which often relies on statistical analyses of averages of treatment groups, is not very efficient in analyzing subjective experiences. Animal models, which are the main window to understanding physiological and neurobiological processes underlying behavior and psychopathologies, are even less geared to do so.
The need for a stronger conceptual differentiation between Stress and Trauma
Our claim is that the use of the term Stress in a nondistinctive way to describe emotional experiences anywhere on the range from mild to severe, contributes to the difficulty in addressing the subjective nature of the response to emotional, stressful, or traumatic experiences.
Not every challenge should come under the umbrella of the term Stress. We propose to refer to three distinct conceptual levels of emotional experiences, which will be distinguished from one another according to three parameters: the level of emotional reaction associated with the experience, the scale of induced plasticity from the experience, and, most importantly, the experience’s impact on the ability or inability of the individual experiencing it to cope later on with daily challenges.
The following are the three distinct conceptual levels we propose:
Arousing experience
An experience that activates a detectable emotional reaction and may affect the immediate choice of behavioral response, but does not lead to substantial long-term consequences in physiological systems, i.e., it does not lead to physiological plasticity. Following an arousing emotional experience, physiological systems generally return to their former baseline.
Stressful experience
An emotionally significant experience that activates a substantial emotional response—a response that not only acutely changes the choice of an immediate behavioral response, but also induces lasting alterations that have the potential of changing the response of the individual to a variety of future experiences (metaplasticity—plasticity of plasticity) [66, 67]. Those alterations are not pathological, however, as they remain within the coping abilities of the individual [15]. As a result, their functional capacity, understood as the degree to which the individuals can adapt with normative responses to future challenges, is not affected.
Traumatic experience
An experience that activates a robust emotional response, which not only acutely changes the choice of a behavioral response, but also induces lasting alterations that would change the response of the individual to a variety of future experiences. Critically, those alterations are pathological, compromising functional capacity, i.e., the ability of the individual to cope later on with daily challenges. In other words, it can be said that the experience induces pathological metaplasticity.
It should be emphasized that we propose these definitions as conceptual distinctions. They obviously leave much to be more precisely defined: what should be considered as a ‘significant’ or a ‘robust’ emotional response? What should be considered as a ‘substantial level of plasticity’? How do we define healthy functional capacity? What should be considered a pathological outcome? When does an experience transform from ‘stressful’ to ‘traumatic’? etc. For both humans and animals, these are all challenging concepts to define. We will propose how to start translating these concepts into practice below, but regardless of whether and how precisely we can draw the dividing lines, it is critical to conceptually distinguish between these notions (Fig. 1). Particularly, it is important to distinguish between stressful and traumatic experiences. As mentioned above, our choice of terminology shapes our way of thinking about scientific challenges, and the way we address them [1,2,3]. The questions indicated above, such as how to define a ‘substantial level of plasticity’ or ‘healthy functional capacity’, would not have been considered if not for such conceptual distinction.
As of now, there are no ideal ways for effectively addressing the subjective nature of the response to stress, the murkiness of the border between stressful and traumatic experiences, and the complexity of individual variability. Nevertheless, we propose to conceptually associate the term ‘stress’/‘stressful experience’ with ‘stress resilience’ (the ability to be exposed to a stressful experience and cope with it), while restricting the use of the term ‘trauma’ only in reference to exposures that lead to pathology. In accordance with that, additional tools and approaches should be developed, making it more practical to establish the distinction between Stress and Trauma. These would enable to more effectively dissociate neural mechanisms underlying coping with stress from those mechanisms underlying failure to cope, which lead to the development of trauma-related psychopathologies.
In our view, the use of the term Stress for both stressful and traumatic experiences has hampered the field’s ability to dissociate between two distinctive mechanisms. People who have undergone a severe experience, may change their behavior in significant ways and, if examined, will exhibit neuronal alterations at different levels of analysis. Yet many of them will continue with what is considered regular daily life and a satisfactory level of day-to-day functional capacity. What this means is that these people do not suffer from a pathology. Somehow, they have managed to cope with the experience. It is, therefore, important to clearly distinguish these individuals’ experience from that of others, who, as a result of being exposed to a similar experience, have lost a significant part of their previous neurobehavioral functional capacities. Accordingly, the same experience affects neural mechanisms differently and induces a different level of plasticity in the two cases. Similarly, in animal studies, we should be able to distinguish between alterations in behavior and physiology that lead to a significant compromise of the animal’s functional capacity and those alterations that do not [68, 69]. Lumping together the results from both affected and coping individuals and averaging them out would mask any possibility to identify the mechanisms of stress vulnerability, pathology or stress resilience.
The proposed conceptual definitions emphasize the consequences of exposure to an experience. This raises a problem when referring to resilience or vulnerability to traumatic experiences, because resilience and vulnerability refer to characteristics that are assumed to be present prior to the exposure and influence the exposure outcome [70,71,72]. Below, we suggest a way to use the concepts of resilience and vulnerability in the context of the above-proposed dissociation between stress and trauma.
Translating the conceptual terminology into research practice
The conceptual dissociation between stress and trauma suggested above raises practical problems for studies of stress and trauma. It becomes imperative to consider when a stress experience should be defined as traumatic and how to distinguish between a stressful experience and a traumatic one. The main challenge arises from the subjectivity of the response: the very same experience could be experienced as stressful by one individual (i.e., significantly challenging but nevertheless still within the coping abilities of that individual), and as traumatic by another one. Currently, there is no convenient way to define and quantify the ‘perceived experience’. Therefore, in order to address this issue, we propose that animal studies undertaken within this field (and to some extent, also human studies) categorize parameters in the following five complementary aspects:
-
A.
The choice of the stress/trauma protocol
-
B.
Considering modulating factors
-
C.
The choice of tests to assess functional capacity
-
D.
The data analysis approach
-
E.
The discussion of the results
We will now discuss each of these aspects in more detail.
The choice of the stress/trauma protocol
As discussed above, the subjective nature of the stress experience leaves no possibility of clearly defining a protocol as either stressful or traumatic. Yet taking the conceptual distinction between the two into consideration is instrumental for effective progress in understanding the psychological, neurobiological, and pathological aspects of stress and trauma. When planning a study, already at the first step, defining whether the focus will be on stress or trauma in their suggested definitions here, will make a difference in the selection of examined populations and the experimental procedures employed.
Although a clear distinction between a stressful or traumatic experience cannot be made based only on the exposure protocol (i.e., on the characteristics of the stressor/s), it is still true that the milder the protocol, the more likely it is that more individuals will experience it just as stressful, and the more intense it is, the greater proportion of tested individuals will experience it as traumatic.
In fact, several factors are known to make an experience more intense
Physical intensity of the experience
Although the physical intensity of the triggering challenge alone cannot define the individual’s outcome, it is in correlation with the outcome. The stronger the electric shock or the colder the water individuals are exposed to, the more likely the incident to be experienced as traumatic [49, 73]. Another factor that may add to the intensity of the experience is the chronicity of a stressor [5, 18, 45, 47, 69].
Level of predictability
Predictability is known to be a protective factor against demanding challenges [74, 75]. The less prepared the individual is for the experience, the more surprising the experience, the greater the impact it will have [76,77,78].
Level of controllability
Probably the most influential factor to determine the impact on the individual of exposure to challenging experiences is the level of controllability over the exposure. It has been elegantly and convincingly demonstrated that the precise same physical exposure could be stressful under controllable conditions but traumatic under uncontrollable conditions [79,80,81,82,83].
Taking the above into consideration, when the research goal is specifically to study trauma mechanisms, it is important to use a stressor or a combination of conditions that are more likely to be experienced as trauma by a larger percentage of the exposed population.
It is important to note here that an animal model of PTSD presents an ethical challenge: on the one hand, a key ethical principle is to reduce unnecessary suffering of the animals, but on the other hand, another important principle is to perform experiments in a way that would maximize the probability of gaining the searched for understanding. Aiming to understand the mechanisms of trauma and related pathologies requires exposure of animals to what would most likely be perceived as trauma. Exposing them to a mild stressor, which is not likely to be perceived as traumatic, may be more acceptable by the ethical committees, but would compromise the relevance of the experiment to trauma-related psychopathologies. Accordingly, we would like to add an important warning here that choosing a “less stressful” protocol in order to get the ethical committee consensus or being enforced by the committee to choose a “less stressful protocol” for ethical reasons may be counteractive, as this approach often leads to the usage of animal models devoid of any translational value, causing a much greater ethical problem.
Considering predisposition factors
Several modulating or predisposition factors were found to influence both the magnitude of physiological reaction and the subjective experience of an exposure to a stressor. These may either be used in order to aim for an outcome of stress or trauma, when planning an experiment, and when choosing the study population, or should be taken into consideration when discussing the outcome of a study.
Mostly, three predisposing factors are considered
Genetic predisposition
It is widely accepted that there are genetic influences on the development of PTSD [84, 85], although available data suggest that stress-related disorders are highly polygenic and the relations to specific genes are complex [84,85,86]. Several genes, such as glucocorticoid-induced leucine zipper (GILZ), a transcription factor encoded by the gene Tsc22d3 on the X chromosome, the serotonin transporter gene (SLC6A4), or pituitary adenylate cyclase-activating polypeptide (PACAP), are suggested as being more influentially associated with the risk of developing stress-related psychopathologies [86, 87]. More often, gene polymorphisms were found to be of influence only when associated with another predisposing factor—childhood adversities [87, 88].
Previous life experiences
It has been demonstrated that exposure to harsh experiences at critical developmental periods could serve as a risk factor for the reaction to subsequent stressful experiences, transforming a stressful experience into a traumatic one for the individual. Early-life adversities [89,90,91,92,93], childhood adversities [94, 95], and adolescence adversities [96,97,98] have been indicated as risk factors that intensify the impact of exposure to a stressful experience in adulthood. Proximal factors, such as sleep disturbances, appear to act as well as risk factors [99,100,101]. Pre-exposure to such adversities would increase the probability of a stressful experience later in life to be traumatic, i.e., to lead to high levels of pathological symptoms and to significantly compromise the ability of the individual to cope with day-to-day challenges. It should be noted though that some forms of pre-exposure to stressors may actually result in the development of resilience, if that pre-disposing experience leads to the development of coping strategies that support coping with stressors later in life [102, 103]. While this further complicates translating the dissociation between ‘Stress’ and ‘Trauma’ into practicality, conceptually, it actually strengthens the need to dissociate between the two. In order to correctly dissociate and study early-life experiences that lead to vulnerability or to resilience, one needs to define whether they increased or not the likelihood of developing pathology. In order to faithfully answer that, it is critical to be able to dissociate between individuals who eventually did or did not develop pathology.
Level of social support leading to, during and following the exposure
Studies have shown that the lack of social support leading to, during and following the exposure to a stressful experience, could influence to a large extent the response of the individual to the exposure [104,105,106,107,108].
The choice of tests to assess functional capacity
The newly proposed definitions for stress and trauma require typifying each participant and determining whether or not their functional capacity was impaired in a significant way. Functional capacity is not, as such, a well-defined or quantitative term. Further studies are required in order to develop widely accepted tests. Yet one principle has already emerged from the literature: in both human and animal studies, a battery of tests that examines a wide range of behavioral domains is required. A single test can identify a specific alteration but cannot depict the true impact of the experience on an individual. A battery of tests is required also in order to be able to identify sub-populations of individuals who may be affected by exposure to the challenging circumstances in different ways. For example, the exposure to a traumatic experience may lead to a different pathological profile (e.g., more anxious or more depressive) in subgroups of exposed individuals [109,110,111,112] or to different clusters of symptoms, as it has been shown, for example, for males and females [95, 113,114,115,116].
It is too early to propose employing an agreed-upon, unified set of tests that would serve as an accepted standard for functional capacity. Different labs are still at the important exploratory phase, examining the efficacy of different test batteries. Furthermore, it is likely that different standardized batteries of tests would be required for different behavioral predispositions or pathological clustering. However, the eventual goal should probably be to standardize tests, at least in the context of specific pathologies, and we make a call here for a future focus on this endeavor.
Importantly, as indicated above, findings from both humans and animals indicate that males and females are differently sensitive to different stressors and may be affected in different behavioral domains [117,118,119,120,121,122,123]. Thus, when choosing the exposure protocol and the test batteries to be used, it is critical to take sex differences into consideration.
The data analysis approach
Whether an event will be experienced as stressful or traumatic cannot be delineated from the characteristics of the stressor. As indicated above, there is high individual variability in responses to the same experience. This is true even for rodent-inbred lines, which are supposed to be genetically identical, due to experience-induced plasticity and epigenetic processes [123,124,125,126,127,128,129,130,131,132]. Typically, individual differences are concealed by the use of group averages, by increasing the sample size, and even by excluding outliers [133]. Yet with respect to stress and trauma, individual variability and outliers are at the essence of the phenomena. It is crucial to dissociate and differentiate between individual clusters of responses in order to associate specific biological mechanisms to precise behavioral responses. For example, in the case of PTSD, if only about 10–20% of the exposed population is expected to develop the disorder, working with the averaged data of the exposed group would clearly hamper the possibility of identifying biological mechanisms associated with the pathology.
It is therefore critical to shift away from working with group averages and move toward more individualized analysis approaches. Some years ago, Cohen et al [134, 135] presented the cutoff behavioral criteria (CBC) analysis approach to differentiate between maladaptive and well-adaptive individuals. While the CBC analysis approach was found to be very productive, yielding a series of highly relevant findings [136], which could not have been identified otherwise, it is different from the way diagnosis is done in humans. While human studies take into consideration responses in nonchallenged individuals, the CBC analysis focuses only on exposed individuals, comparing the performance only among them. More recently, we have developed a variation of the CBC in rodents, termed behavioral profiling, which is an analysis approach based on referring to the performance of a control, nonexposed group as defining normal behavior, and assessing deviation from that “norm” [13, 137, 138]. Importantly, control and exposed animals are examined on a battery of tests, aiming to test their functional capacity and to assess their coping abilities. Employing this analysis approach enabled us to demonstrate key neurobiological differences between animals that were exposed to the traumatic experience and developed symptoms (exposed-affected) and those that were exposed to the same experience but did not develop significant symptoms (exposed–nonaffected) [137,138,139], further verifying the functional significance of such a distinction. This approach is in good alignment with the human population in which there are people who develop PTSD (susceptible) and others who do not (resilient) after trauma exposure. In addition, differentiating between exposed–affected and exposed–nonaffected individuals enables differentiating between neural mechanisms that are associated with the pathology and those actually associated with stress resilience [137, 139]. Moreover, other studies in the literature have succeeded to predict susceptibility to develop a PTSD-like phenotype on the long term, by characterizing animals’ exploratory responses in a novel environment [140] or measuring their changes in startle reactivity [141] shortly after trauma exposure. This line of work aligns well with evidence in humans indicating that a Clinician-Administered PTSD Scale for DSM-IV (CAPS) given within 60 days of trauma exposure is highly predictive of the degree and persistence of PTSD symptoms at later stages [142].
Furthermore, other approaches go beyond the postexposure characterization of animals (i.e., ‘sequalae factors’) to identify ‘susceptibility factors’ pre-existing before trauma [143]. For example, the ‘Revealing Individual Susceptibility to a PTSD-like Phenotype’ model [144] assesses susceptibility to trauma (segregating susceptible, resilient, and intermediate animals) according to rats’ responsiveness (i.e., startle and anxiety-like responses) as assessed a few days after the first encounter of a mild stressor. These approaches may help identifying susceptible individuals a priori and guide neurobiological studies with the potential of paving the way toward the development of preventive treatments.
The discussion of the results
Even before changing anything in the experimental protocols, we believe that the proposed definitions of Stress and Trauma are important for a more productive discussion over existing findings. The confusion arising from the use of the term stress for describing a wide range of different experiences and the responses to them has introduced great difficulties in comparing the findings, discussing them and drawing conclusions from them in a comprehensive way.
Adopting the proposed terminology and aiming to plan and conduct experiments along the line of those definitions will enhance discussion and promote reaching more well-based conclusions in the future.
Vulnerability and resilience versus pathology and no pathology
The proposed conceptual definitions of stress and trauma are based on the eventual outcome of the exposure, namely, whether the individual could maintain its functional capacity or failed to keep effective coping when facing future life events. This terminology is effective when examining the outcome of an exposure. Yet, difficulty arises when attempting to associate them with questions regarding vulnerability versus resilience (and we are back to the importance of semantic definitions). Often, the terms vulnerability and resilience are used in a predictive manner, to describe qualities that would only be manifested if and when individuals will encounter a challenging experience [145]. The assumption behind the use of these terms is that vulnerable or resilient individuals are already different prior to the potential encounter, in a way that would enable the resilient, but not the vulnerable individual, to cope with the challenge if and when it arises [146, 147].
It is important to note that even when referring to vulnerability and resilience as qualities to be measured prior to or without exposure to any challenge, the concept is a predictive concept, referring to currently present mechanisms that would come to action, or fail to come to action in the face of future exposure. In fact, vulnerability and resilience require identifying biomarkers that can predict the coping abilities of the individual. In that sense, the conceptual definitions proposed here for the dissociation between stress and trauma still hold, and they help to identify the need for predictive biomarkers.
As of now, there are no actual biomarkers that could predict vulnerability or resilience in an effective way. However, employing the Behavioral Profiling analysis approach, which enables to differentiate between exposed–affected and exposed–nonaffected individuals, it is possible to design studies that would collect samples prior to the exposure and analyze them in light of the eventual outcome, thus associating them to vulnerability or resilience. In any case, planning such experiments requires dissociating between stressful or traumatic outcome, if to reflect any functional significance to identified biomarkers.
Recovery
A final point to discuss is the fact that PTSD in humans is considered as a failure to follow the normative trajectory of recovery (e.g., in terms of physiological, cognitive, or behavioral symptoms) after exposure to a traumatic event [148]. Recent evidence indicates that some individuals may take long periods of time to recover from PTSD [149] and, therefore, animal studies may need to define the range of testing that would allow characterizing deficiencies in remote symptoms, e.g., [150].
Summary
We acknowledge that there are as yet no ideal ways for effectively addressing the subjective nature of response, the murkiness of the border between stressful and traumatic experiences, and the complexity of individual variability. However, ignoring these challenges hampers our ability to elucidate the mechanism of trauma-related pathologies, and of resilience. There is already sufficient evidence to indicate that pathological metaplasticity, which is expected to take place in the brains of individuals who develop behavioral pathologies following exposure to a traumatic event, would be very different from the type of meta-plasticity that could be found in the brains of those individuals who do not experience the event as traumatic, and do not develop pathology. We have proposed here to start focusing on differentiating between ‘stress’ and ‘trauma’ and conceptualizing the semantic difference between them. Adopting the conception, terminology and differentiation between the concepts of Stress and Trauma, is a first step to inspire planning and conduct of both human and animal studies in accordance with this conception. Likewise, the evaluation of the results and discussion of their most likely interpretation will benefit from the clarity provided by these distinctions. Furthermore, such semantics will encourage the search for more suitable research and analysis tools.
Recently, analysis approaches that put more emphasis on the behavioral profiling triggered by exposure to challenging events by focusing on individuals rather than on group averages have been developed and prove to be very effective and relevant (e.g., [13]). In the future, additional tools and approaches should be developed to allow making the distinction between Stress and Trauma easier to establish. Consequently, neural mechanisms underlying coping with stress could be more effectively dissociated from those mechanisms underlying failure to cope and the development of trauma-related psychopathologies. Ultimately, these dissociations should allow refining our findings and, thus, facilitating the development of more effective treatments for trauma-related psychopathologies.
References
Taylor C, Dewsbury BM. On the problem and promise of metaphor use in science and science communication. J Microbiol Biol Educ. 2018; 19. https://doi.org/10.1128/jmbe.v19i1.1538.
Kagan J. Why stress remains an ambiguous concept: reply to McEwen & McEwen (2016) and Cohen et al. (2016). Perspect Psychol Sci. 2016;11:464–5.
Kagan J. An overly permissive extension. Perspect Psychol Sci. 2016;11:442–50.
VanItallie TB. Stress: a risk factor for serious illness. Metabolism. 2002;51:40–45.
Coccurello R, D’Amato FR, Moles A. Chronic social stress, hedonism and vulnerability to obesity: lessons from Rodents. Neurosci. Biobehav Rev. 2009;33:537–50.
Cirulli F, Laviola G, Ricceri L. Risk factors for mental health: translational models from behavioural neuroscience. Neurosci Biobehav Rev. 2009;33:493–7.
Plieger T, Melchers M, Montag C, Meermann R, Reuter M. Life stress as potential risk factor for depression and burnout. Burn Res. 2015;2:19–24.
Franklin TB, Saab BJ, Mansuy IM. Neural mechanisms of stress resilience and vulnerability. Neuron. 2012;75:747–61.
Faye C, Mcgowan JC, Denny CA, David DJ. Neurobiological mechanisms of stress resilience and implications for the aged population. Curr Neuropharmacol. 2018;16:234–70.
Dutcher JM, Creswell JD. The role of brain reward pathways in stress resilience and health. Neurosci Biobehav Rev. 2018;95:559–67.
Bijlsma R, Loeschcke V. Environmental stress, adaptation and evolution: an overview. J Evol Biol. 2005;18:744–9.
Arnoldini M, Mostowy R, Bonhoeffer S, Ackermann M. Evolution of stress response in the face of unreliable environmental signals. PLoS Comput Biol. 2012; 8. https://doi.org/10.1371/journal.pcbi.1002627.
Richter-Levin G, Stork O, Schmidt MV. Animal models of PTSD: a challenge to be met. Mol. Psychiatry. 2019;24:1135–56.
McEwen BS. Physiology and neurobiology of stress and adaptation: central role of the brain. Physiol Rev. 2007;87:873–904.
Koolhaas JM, Bartolomucci A, Buwalda B, de Boer SF, Flügge G, Korte SM, et al. Stress revisited: a critical evaluation of the stress concept. Neurosci Biobehav Rev. 2011;35:1291–301.
de Kloet ER, Karst H, Joëls M. Corticosteroid hormones in the central stress response: quick-and-slow. Front Neuroendocrinol. 2008;29:268–72.
Herman JP, McKlveen JM, Ghosal S, Kopp B, Wulsin A, Makinson R, et al. Regulation of the hypothalamic-pituitary- adrenocortical stress response. Compr Physiol. 2016;6:603–21.
McEwen BS. What is the confusion with cortisol? Chronic Stress. 2019;3:247054701983364.
Picard M, McEwen BS, Epel ES, Sandi C. An energetic view of stress: focus on mitochondria. Front Neuroendocrinol. 2018;49:72–85.
Picard M, McEwen BS. Psychological stress and mitochondria: a systematic review. Psychosom Med. 2018;80:141–53.
Filiou MD, Sandi C. Anxiety and brain mitochondria: a bidirectional crosstalk. Trends Neurosci. 2019;42:573–88.
De Oliveira C, de Mattos AB, Biz C, Oyama LM, Ribeiro EB, do Nascimento CM. High-fat diet and glucocorticoid treatment cause hyperglycemia associated with adiponectin receptor alterations. Lipids Health Dis. 2011;10:10.
Balsevich G, Abizaid A, Chen A, Karatsoreos IN, Schmidt MV. Stress and glucocorticoid modulation of feeding and metabolism. Neurobiol Stress. 2019;11:11.
Papargyri P, Zapanti E, Salakos N, Papargyris L, Bargiota A, Mastorakos G. Links between HPA axis and adipokines: clinical implications in paradigms of stress-related disorders. Expert Rev Endocrinol Metab. 2018;13:317–32.
Sukumaran S, DuBois DC, Jusko WJ, Almon RR. Glucocorticoid effects on adiponectin expression. Vitam Horm. 2012;90:163–86.
Sukumaran S, Jusko WJ, DuBois DC, Almon RR. Mechanistic modeling of the effects of glucocorticoids and circadian rhythms on adipokine expression. J Pharm Exp Ther. 2011;337:734–46.
Mellon SH, Gautam A, Hammamieh R, Jett M, Wolkowitz OM. Metabolism metabolomics, and inflammation in posttraumatic stress disorder. Biol Psychiatry. 2018;83:866–75.
Chan DC. Mitochondrial dynamics and its involvement in disease. Annu Rev Pathol Mech Dis. 2020;15:235–59.
De Souza EB, Grigoriadis DE, Webster EL. Role of brain, pituitary and spleen corticotropin-releasing factor receptors in the stress response. Methods Achiev Exp Pathol. 1991;14:23–44.
Verburg-van Kemenade BML, Cohen N, Chadzinska M. Neuroendocrine-immune interaction: evolutionarily conserved mechanisms that maintain allostasis in an ever-changing environment. Dev Comp Immunol. 2017;66:2–23.
Karatsoreos IN, McEwen BS. Psychobiological allostasis: resistance, resilience and vulnerability. Trends Cogn Sci. 2011;15:576–84.
Hirotsu C, Tufik S, Andersen ML. Interactions between sleep, stress, and metabolism: from physiological to pathological conditions. Sleep Sci. 2015;8:143–52.
Somvanshi PR, Mellon SH, Yehuda R, Flory JD, Makotkine I, Bierer L, et al. Role of enhanced glucocorticoid receptor sensitivity in inflammation in PTSD: insights from computational model for circadian-neuroendocrineimmune interactions. Am J Physiol Endocrinol Metab. 2020;319:E48–66.
Bottaccioli AG, Bottaccioli F, Minelli A. Stress and the psyche–brain–immune network in psychiatric diseases based on psychoneuroendocrineimmunology: a concise review. Ann N Y Acad Sci. 2019;1437:31–42.
Ising M, Holsboer F. Genetics of stress response and stress-related disorders. Dialogues Clin Neurosci. 2006;8:433–44.
Gillespie CF, Phifer J, Bradley B, Ressler KJ. Risk and resilience: genetic and environmental influences on development of the stress response. Depress Anxiety. 2009;26:984–92.
Almli LM, Fani N, Smith AK, Ressler KJ. Genetic approaches to understanding post-traumatic stress disorder. Int J Neuropsychopharmacol. 2014;17:355–70.
Tafet GE, Nemeroff CB. The links between stress and depression: psychoneuroendocrinological, genetic, and environmental interactions. J Neuropsychiatry Clin Neurosci. 2016;28:77–88.
Bogdan R, Pagliaccio D, Baranger DAA, Hariri AR. Genetic moderation of stress effects on corticolimbic circuitry. Neuropsychopharmacology. 2016;41:275–96.
Le Moal M. Historical approach and evolution of the stress concept: a personal account. Psychoneuroendocrinology. 2007; 32. https://doi.org/10.1016/j.psyneuen.2007.03.019.
Cohen S, Gianaros PJ, Manuck SB. A stage model of stress and disease. Perspect Psychol Sci. 2016;11:456–63.
Selye H. A syndrome produced by diverse nocuous agents [13]. Nature. 1936;138:32.
Sterling P, Eyer J. Allostasis: a new paradigm to explain arousal pathology. In: Fisher S & Reason J, editors. Handbook of life stress, cognition and health. John Wiley & Sons; 1988. pp 629-49.
McEwen BS, Stellar E. Stress and the individual: mechanisms leading to disease. Arch Intern Med. 1993;153:2093–101.
McEwen BS. Neurobiological and systemic effects of chronic stress. Chronic Stress. 2017;1:247054701769232.
Juster RP, Russell JJ, Almeida D, Picard M. Allostatic load and comorbidities: a mitochondrial, epigenetic, and evolutionary perspective. Dev Psychopathol. 2016;28:1117–46.
Ullmann E, et al. From allostatic load to allostatic state—an endogenous sympathetic strategy to deal with chronic anxiety and stress? Front Behav Neurosci. 2019; 13. https://doi.org/10.3389/fnbeh.2019.00047.
Fava GA, McEwen BS, Guidi J, Gostoli S, Offidani E, Sonino N. Clinical characterization of allostatic overload. Psychoneuroendocrinology. 2019;108:94–101.
Cordero MI, Merino JJ, Sandi C. Correlational relationship between shock intensity and corticosterone secretion on the establishment and subsequent expression of contextual fear conditioning. Behav Neurosci. 1998;112:885–91.
Merino JJ, Cordero MI, Sandi C. Regulation of hippocampal cell adhesion molecules NCAM and L1 by contextual fear conditioning is dependent upon time and stressor intensity. Eur J Neurosci. 2000;12:3283–90.
Salehi B, Cordero MI, Sandi C. Learning under stress: The inverted-U-shape function revisited. Learn Mem. 2010;17:522–30.
Goette L, Bendahan S, Thoresen J, Hollis F, Sandi C. Stress pulls us apart: anxiety leads to differences in competitive confidence under stress. Psychoneuroendocrinology. 2015;54:115–23.
Lieberman PB. ‘objective’ methods and ‘subjective’ experiences. Schizophr Bull. 1989;15:267–75.
Creamer M, McFarlane AC, Burgess P. Psychopathology following trauma: the role of subjective experience. J. Affect Disord. 2005;86:175–82.
Huzard D, Ghosal S, Grosse J, Carnevali L, Sgoifo A, Sandi C. Low vagal tone in two rat models of psychopathology involving high or low corticosterone stress responses. Psychoneuroendocrinology. 2019;101:101–10.
Owens SA, Helms SW, Rudolph KD, Hastings PD, Nock MK, Prinstein MJ. Interpersonal stress severity longitudinally predicts adolescent girls’ depressive symptoms: the moderating role of subjective and HPA axis stress responses. J Abnorm Child Psychol. 2019;47:895–905.
Sapolsky RM. Stress and the brain: individual variability and the inverted-U. Nat Neurosci. 2015;18:1344–6.
Walker SE, Zanoletti O, Guillot de Suduiraut I, Sandi C. Constitutive differences in glucocorticoid responsiveness to stress are related to variation in aggression and anxiety-related behaviors. Psychoneuroendocrinology. 2017;84:1–10.
Ebner K, Singewald N. Individual differences in stress susceptibility and stress inhibitory mechanisms. Curr Opin Behav Sci. 2017;14:54–64.
Kessler RC, Berglund P, Demler O, Jin R, Merikangas KR, Walters EE. Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the national comorbidity survey replication. Arch Gen Psychiatry. 2005;62:593–602.
Kilpatrick DG, Resnick HS, Milanak ME, Miller MW, Keyes KM, Friedman MJ. National estimates of exposure to traumatic events and PTSD prevalence using DSM-IV and DSM-5 criteria. J Trauma Stress. 2013;26:537–47.
Richardson LK, Frueh BC, Acierno R. Prevalence estimates of combat-related post-traumatic stress disorder: critical review. Aust N Z J Psychiatry. 2010;44:4–19.
Olaya B, Alonso J, Atwoli L, Kessler RC, Vilagut G, Haro JM. Association between traumatic events and post-traumatic stress disorder: results from the ESEMeD-Spain study. Epidemiol Psychiatr Sci. 2015;24:172–83.
Charlson FJ, et al. Post-traumatic stress disorder and major depression in conflict-affected populations: an epidemiological model and predictor analysis. Glob Ment Health. 2016; 3. https://doi.org/10.1017/gmh.2015.26.
Bromet EJ, Atwoli L, Kawakami N, Navarro-Mateu F, Piotrowski P, King AJ, et al. Post-traumatic stress disorder associated with natural and human-made disasters in the World Mental Health Surveys. Psychol Med. 2017;47:227–41.
Abraham WC, Bear MF. Metaplasticity: the plasticity of synaptic plasticity. Trends Neurosci. 1996;19:126–30.
Schmidt MV, Abraham WC, Maroun M, Stork O, Richter-Levin G. Stress-induced metaplasticity: from synapses to behavior. Neuroscience. 2013;250:112–20.
Krishnan V, Han MH, Graham DL, Berton O, Renthal W, Russo SJ, et al. Molecular adaptations underlying susceptibility and resistance to social defeat in brain reward regions. Cell. 2007;131:391–404.
Larrieu T, Cherix A, Duque A, Rodrigues J, Lei H, Gruetter R, et al. Hierarchical status predicts behavioral vulnerability and nucleus accumbens metabolic profile following chronic social defeat stress. Curr Biol. 2017;27:2202–.e4.
Lorsch ZS, et al. Estrogen receptor α drives pro-resilient transcription in mouse models of depression. Nat Commun 2018; 9. https://doi.org/10.1038/s41467-018-03567-4.
Walker SE, Sandi C. Long-term programing of psychopathology-like behaviors in male rats by peripubertal stress depends on individual’s glucocorticoid responsiveness to stress. Stress. 2018;21:433–42.
Cherix A, et al. Metabolic signature in nucleus accumbens for anti-depressant-like effects of acetyl-l-carnitine. Elife. 2020; 9. https://doi.org/10.7554/eLife.50631.
Siegmund A, Wotjak CT. A mouse model of posttraumatic stress disorder that distinguishes between conditioned and sensitised fear. J Psychiatr Res. 2007;41:848–60.
Zucchi FC, Kirkland SW, Jadavji NM, van Waes LT, Klein A, Supina RD, Metz GA. Predictable stress versus unpredictable stress: a comparison in a rodent model of stroke. Behav Brain Res. 2009;205:67–75.
Trow JE, Jones AM, McDonald RJ. Comparison of the effects of repeated exposures to predictable or unpredictable stress on the behavioural expression of fear in a discriminative fear conditioning to context task. Physiol Behav. 2019; 208. https://doi.org/10.1016/j.physbeh.2019.05.017.
Keyes KM, Pratt C, Galea S, McLaughlin KA, Koenen KC, Shear MK. The burden of loss: unexpected death of a loved one and psychiatric disorders across the life course in a national study. Am J Psychiatry. 2014;171:864–71.
Ursano RJ, Wang J, Fullerton CS, Ramsawh H, Gifford RK, Russell D, et al. Post-deployment mental health in reserve and national guard service members: Deploying with or without one’s unit and deployment preparedness. Mil Med. 2018;183:e51–58.
Cucciare MA, Mengeling MA, Han X, Torner J, Sadler AG. Associations between augmentee status, deployment stress preparedness and depression, post-traumatic stress disorder, and binge drinking in US servicewomen. Womens Health Issues. 2020;30:207–13.
Maier SF. Learned helplessness and animal models of depression. Prog Neuro-Psychopharmacol Biol Psychiatry. 1984;8:435–46.
Harris JC. Experimental animal modeling of depression and anxiety. Psychiatr Clin North Am. 1989;12:815–36.
Dohrenwend BP. The role of adversity and stress in psychopathology: some evidence and its implications for theory and research. J Health Soc Behav. 2000;41:1–19.
Lucas M, Ilin Y, Anunu R, Kehat O, Xu L, Desmedt A, et al. Long-term effects of controllability or the lack of it on coping abilities and stress resilience in the rat. Stress. 2014;17:423–30.
Maier SF, Seligman MEP. Learned helplessness at fifty: Insights from neuroscience. Psychol Rev. 2016;123:1–19.
Duncan LE, Ratanatharathorn A, Aiello AE, Almli LM, Amstadter AB, Ashley-Koch AE, et al. Largest GWAS of PTSD (N=20 070) yields genetic overlap with schizophrenia and sex differences in heritability. Mol Psychiatry. 2018;23:666–73.
Nievergelt CM, et al. International meta-analysis of PTSD genome-wide association studies identifies sex- and ancestry-specific genetic risk loci. Nat Commun. 2019; 10. https://doi.org/10.1038/s41467-019-12576-w.
Smoller JW. The genetics of stress-related disorders: PTSD, depression, and anxiety disorders. Neuropsychopharmacology. 2016;41:297–319.
Lebow MA, Schroeder M, Tsoory M, Holzman-Karniel D, Mehta D, Ben-Dor S, et al. Glucocorticoid-induced leucine zipper “quantifies” stressors and increases male susceptibility to PTSD. Transl Psychiatry. 2019;9:9.
Binder EB, Bradley RG, Liu W, Epstein MP, Deveau TC, Mercer KB, et al. Association of FKBP5 polymorphisms and childhood abuse with risk of posttraumatic stress disorder symptoms in adults. JAMA. 2008;299:1291–305.
Faturi CB, Tiba PA, Kawakami SE, Catallani B, Kerstens M, Suchecki D. Disruptions of the mother-infant relationship and stress-related behaviours: altered corticosterone secretion does not explain everything. Neurosci Biobehav Rev. 2010;34:821–34.
Carr CP, Martins CMS, Stingel AM, Lemgruber VB, Juruena MF. The role of early life stress in adult psychiatric disorders: a systematic review according to childhood trauma subtypes. J Nerv Ment Dis. 2013;201:1007–20.
Haller J, Harold G, Sandi C, Neumann ID. Effects of adverse early-life events on aggression and anti-social behaviours in animals and humans. J Neuroendocrinol. 2014;26:724–38.
Nettis MA, Pariante CM, Mondelli V. Early-life adversity, systemic inflammation and comorbid physical and psychiatric illnesses of adult life. Curr Top Behav Neurosci. 2020;44:207–25.
Meyer HC, Lee FS. Translating developmental neuroscience to understand risk for psychiatric disorders. Am J Psychiatry. 2019;176:179–85.
Molnar BE, Berkman LF, Buka SLPsychopathology. childhood sexual abuse and other childhood adversities: relative links to subsequent suicidal behaviour in the US. Psychol Med. 2001;31:965–77.
Horovitz O, Tsoory MM, Yovell Y, Richter-Levin G. A rat model of pre-puberty (Juvenile) stress-induced predisposition to stress-related disorders: sex similarities and sex differences in effects and symptoms. World J Biol Psychiatry. 2014;15:36–48.
McCormick CM, Mathews IZ. HPA function in adolescence: role of sex hormones in its regulation and the enduring consequences of exposure to stressors. Pharm Biochem Behav. 2007;86:220–33.
Smith SS. The influence of stress at puberty on mood and learning: role of the α4βδ GABAA receptor. Neuroscience. 2013;249:192–213.
Tzanoulinou S, Riccio O, De Boer MW, Sandi C. Peripubertal stress-induced behavioral changes are associated with altered expression of genes involved in excitation and inhibition in the amygdale. Transl Psychiatry 2014; 4. https://doi.org/10.1038/tp.2014.54.
Spiegelhalder K, Regen W, Nanovska S, Baglioni C, Riemann D. Comorbid sleep disorders in neuropsychiatric disorders across the life cycle. Curr Psychiatry Rep 2013; 15. https://doi.org/10.1007/s11920-013-0364-5.
Schmidt RE, Van Der Linden M. The relations between sleep, personality, behavioral problems, and school performance in adolescents. Sleep Med Clin. 2015;10:117–23.
Palagini L, Domschke K, Benedetti F, Foster RG, Wulff K, Riemann D. Developmental pathways towards mood disorders in adult life: Is there a role for sleep disturbances? J Affect Disord. 2019;243:121–32.
Daskalakis NP, Bagot RC, Parker KJ, Vinkers CH, de Kloet ER. The three-hit concept of vulnerability and resilience: toward understanding adaptation to early-life adversity outcome. Psychoneuroendocrinology. 2013;38:1858–73.
Santarelli S, Lesuis SL, Wang XD, Wagner KV, Hartmann J, Labermaier C, et al. Evidence supporting the match/mismatch hypothesis of psychiatric disorders. Eur Neuropsychopharmacol. 2014;24(Jun):907–18. Epub 2014 Feb 15. PMID: 24589292
Kessler RC, Price RH, Wortman CB. Social factors in psychopathology: stress, social support, and coping processes. Annu Rev Psychol. 1985;36:531–72.
Charuvastra A, Cloitre M. Social bonds and posttraumatic stress disorder. Annu Rev Psychol. 2008;59:301–28.
Guay S, Billette V, Marchand A. Exploring the links between posttraumatic stress disorder and social support: processes and potential research avenues. J Trauma Stress. 2006;19:327–38.
Ditzen B, Heinrichs M. Psychobiology of social support: The social dimension of stress buffering. Restor Neurol Neurosci. 2014;32:149–62.
Zoladz PR, Diamond DM. Predator-based psychosocial stress animal model of PTSD: preclinical assessment of traumatic stress at cognitive, hormonal, pharmacological, cardiovascular and epigenetic levels of analysis. Exp Neurol. 2016;284:211–9.
Tsoory M, Richter-Levin G. Learning under stress in the adult rat is differentially affected by ‘juvenile’ or ‘adolescent’ stress. Int J Neuropsychopharmacol. 2006;9:713–28.
Tsoory M, Cohen H, Richter-Levin G. Juvenile stress induces a predisposition to either anxiety or depressive-like symptoms following stress in adulthood. Eur Neuropsychopharmacol. 2007;17:245–56.
Lanius RA, Brand B, Vermetten E, Frewen PA, Spiegel D. The dissociative subtype of posttraumatic stress disorder: rationale, clinical and neurobiological evidence, and implications. Depress Anxiety. 2012;29:701–8.
Yehuda R, et al. Post-traumatic stress disorder. Nat Rev Dis Prim 2015; 1. https://doi.org/10.1038/nrdp.2015.57.
Jacobson-Pick S, Richter-Levin G. Differential impact of juvenile stress and corticosterone in juvenility and in adulthood, in male and female rats. Behav Brain Res. 2010;214:268–76.
Hillerer KM, Slattery DA, Pletzer B. Neurobiological mechanisms underlying sex-related differences in stress-related disorders: effects of neuroactive steroids on the hippocampus. Front Neuroendocrinol 2019; 55. https://doi.org/10.1016/j.yfrne.2019.100796.
Christiansen DM, Berke ET. Gender- and sex-based contributors to sex differences in PTSD. Curr Psychiatry Rep 2020; 22. https://doi.org/10.1007/s11920-020-1140-y.
Ford JD, Schneeberger AR, Komarovskaya I, Muenzenmaier K, Castille D, Opler LA, et al. The symptoms of trauma scale (SOTS): psychometric evaluation and gender differences with adults diagnosed with serious mental illness. J Trauma Dissociation. 2017;18:559–74.
Breslau N, Davis GC, Andreski P, Peterson EL, Schultz LR. Sex differences in posttraumatic stress disorder. Arch Gen Psychiatry. 1997;54:1044–8.
Peters L, Issakidis C, Slade T, Andrews G. Gender differences in the prevalence of DSM-IV and ICD-10 PTSD. Psychol Med. 2006;36:81–89.
Tolin DF, Foa EB. Sex differences in trauma and posttraumatic stress disorder: a quantitative review of 25 years of research. Psychol Bull. 2006;132:959–92.
Olff M, Langeland W, Draijer N, Gersons BPR. Gender differences in posttraumatic stress disorder. Psychol Bull. 2007;133:183–204.
Cohen H, Yehuda R. Gender differences in animal models of posttraumatic stress disorder. Dis Markers. 2011;30:141–50.
Susukida R, Mojtabai R, Mendelson T. Sex differences in help seeking for mood and anxiety disorders in the national comorbidity survey-replication. Depress Anxiety. 2015;32:853–60.
Sajti E, van Meeteren N, Kavelaars A, van der Net J, Gispen WH, Heijnen C. Individual differences in behavior of inbred Lewis rats are associated with severity of joint destruction in adjuvant-induced arthritis. Brain Behav Immun. 2004;18:505–14.
Cohen H, Geva AB, Matar MA, Zohar J, Kaplan Z. Post-traumatic stress behavioural responses in inbred mouse strains: can genetic predisposition explain phenotypic vulnerability? Int J Neuropsychopharmacol. 2008;11:331–49.
Claessens SE, Daskalakis NP, van der Veen R, Oitzl MS, de Kloet ER, Champagne DL. Development of individual differences in stress responsiveness: an overview of factors mediating the outcome of early life experiences. Psychopharmacology. 2011;214:141–54.
Hager T, et al. Display of individuality in avoidance behavior and risk assessment of inbred mice. Front Behav Neurosci 2014; 8. https://doi.org/10.3389/fnbeh.2014.00314.
Wellman LL, Fitzpatrick ME, Hallum OY, Sutton AM, Williams BL, Sanford LD. Individual differences in animal stress models: considering resilience, vulnerability, and the amygdala in mediating the effects of stress and conditioned fear on sleep. Sleep. 2016;39:1293–303.
Feyissa DD, Aher YD, Engidawork E, Höger H, Lubec G, Korz V. Individual differences in male rats in a behavioral test battery: a multivariate statistical approach. Front Behav Neurosci. 2017;11:11 https://doi.org/10.3389/fnbeh.2017.00026
King G, Scott E, Graham BM, Richardson R. Individual differences in fear extinction and anxiety-like behavior. Learn Mem. 2017;24:182–90.
Anyan J, Verwey M, Amir S. Individual differences in circadian locomotor parameters correlate with anxiety- and depression-like behavior. PLoS One. 2017; 12. https://doi.org/10.1371/journal.pone.0181375.
Zhang TY, Shahrokh D, Hellstrom IC, Wen X, Diorio J, Breuillaud L, et al. Brain-derived neurotrophic factor in the nucleus accumbens mediates individual differences in behavioral responses to a natural, social reward. Mol Neurobiol. 2020;57:290–301.
de Boer SF, Buwalda B, Koolhaas JM. Untangling the neurobiology of coping styles in rodents: towards neural mechanisms underlying individual differences in disease susceptibility. Neurosci Biobehav Rev. 2017;74:401–22.
Rousseeuw PJ, Hubert M. Robust statistics for outlier detection. Wiley Interdiscip Rev Data Min Knowl Disco. 2011;1:73–79.
Cohen H, Zohar J, Matar M. The relevance of differential response to trauma in an animal model of posttraumatic stress disorder. Biol Psychiatry. 2003;53:463–73.
Cohen H, Zohar J, Matar MA, Zeev K, Loewenthal U, Richter-Levin G. Setting apart the affected: The use of behavioral criteria in animal models of post traumatic stress disorder. Neuropsychopharmacology. 2004;29:1962–70.
Cohen H, Kozlovsky N, Alona C, Matar MA, Joseph Z. Animal model for PTSD: from clinical concept to translational research. Neuropharmacology. 2012;62:715–24.
Ardi Z, Albrecht A, Richter-Levin A, Saha R, Richter-Levin G. Behavioral profiling as a translational approach in an animal model of posttraumatic stress disorder. Neurobiol Dis. 2016;88:139–47.
Ritov G, Boltyansky B, Richter-Levin G. PTSD modeling in rodents shows alternating patterns of limbic activity in various types of reactions to stress. Mol Psychiatry. 2016;21:587.
Ardi Z, Richter-Levin A, Xu L, Cao X, Volkmer H, Stork O, et al. The role of the GABAA receptor Alpha 1 subunit in the ventral hippocampus in stress resilience. Sci Rep. 2019;9:13513.
Colucci P, Marchetta E, Mancini GF, Alva P, Chiarotti F, Hasan MT, et al. Predicting susceptibility and resilience in an animal model of post-traumatic stress disorder (PTSD). Transl Psychiatry. 2020;10:243.
Torrisi SA, Lavanco G, Maurel OM, Gulisano W, Laudani S, Geraci F, et al. A novel arousal-based individual screening reveals susceptibility and resilience to PTSD-like phenotypes in mice. Neurobiol Stress. 2020;14:100286.
Shalev AY, Gevonden M, Ratanatharathorn A, Laska E, van der Mei WF, Qi W, et al. International consortium to predict PTSD. Estimating the risk of PTSD in recent trauma survivors: results of the International Consortium to Predict PTSD (ICPP). World Psychiatry. 2019;18:77–87.
Alexander KS, Nalloor R, Bunting KM, Vazdarjanova A. Investigating individual pre-trauma susceptibility to a PTSD-like phenotype in animals. Front Syst Neurosci. 2020;13:85.
Nalloor R, Bunting K, Vazdarjanova A. Predicting impaired extinction of traumatic memory and elevated startle. PLoS ONE. 2011;6:e19760.
Larrieu T, Sandi C. Stress-induced depression: is social rank a predictive risk factor? BioEssays. 2018; 40. https://doi.org/10.1002/bies.201800012.
Sandi C, Richter-Levin G. From high anxiety trait to depression: a neurocognitive hypothesis. Trends Neurosci. 2009;32:312–20.
Weger M, Sandi C. High anxiety trait: a vulnerable phenotype for stress-induced depression. Neurosci Biobehav Rev. 2018;87:27–37.
Lancaster CL, Teeters JB, Gros DF, Back SE. Posttraumatic stress disorder: overview of evidence-based assessment and treatment. J Clin Med. 2016;5:105.
Jacoby VM, Hale W, Dillon K, Dondanville KA, Wachen JS, Yarvis JS, et al. STRONG STAR Consortium. Depression suppresses treatment response for traumatic loss-related posttraumatic stress disorder in active duty military personnel. J Trauma Stress. 2019;32:774–83.
Silva BA, et al. A thalamo-amygdalar circuit underlying the extinction of remote fear memories. Nat Neurosci. 2021; https://doi.org/10.1038/s41593-021-00856-y.
Acknowledgements
The authors would like to thank Netta Richter-Levin for English editing the paper. This work was supported by the Israel Science Foundation [grant number 1517/16]; by the State of Israel Ministry of Science, Technology, & Space [grant number 3-13563]; by the State of Israel Ministry of Science, Technology, & Space [grant number 3-14356]; by intramural finding from the EPFL.
Author information
Authors and Affiliations
Contributions
GR-L. Conceptualization, methodology, and project administration, Writing—original draft, Writing—review and editing, Visualization. CS. Conceptualization, Writing—review and editing, Visualization.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Richter-Levin, G., Sandi, C. Title: “Labels Matter: Is it stress or is it Trauma?”. Transl Psychiatry 11, 385 (2021). https://doi.org/10.1038/s41398-021-01514-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41398-021-01514-4
This article is cited by
-
Updating a social cognitive model of PTSD: Implications for cross-group PTSD research
Current Psychology (2024)
-
Metabolomics reveals that chronic restraint stress alleviates carbon tetrachloride-induced hepatic fibrosis through the INSR/PI3K/AKT/AMPK pathway
Journal of Molecular Medicine (2024)
-
In medio stat virtus? A reply to Dupras
Epigenetics Communications (2023)
-
Sex-specific roles of hippocampal microRNAs in stress vulnerability and resilience
Translational Psychiatry (2022)