Abstract
Study design
A non-randomized open-label single-arm clinical trial.
Objectives
To analyze the effect of body weight supported treadmill training (BWSTT) with the voluntary driven exoskeleton (VDE) in persons with differing levels and completeness of spinal cord injury (SCI) and differing walking abilities.
Setting
Keio University Hospital, Tokyo, Japan.
Methods
Twenty individuals with chronic SCI (age, 43 ± 17 years) classified as American Spinal Injury Association Impairment Scale grade A (n = 2), B (n = 4), C (n = 8), or D (n = 6) who had reached a plateau in recovery. Participants underwent twenty 60 min sessions of BWSTT with the hybrid assisted limb. The speed, distance, and duration walked in every 60 min training session were recorded. The Walking Index for SCI Scale II (WISCI-II), 10 meters walk test (10MWT), 2 min walk test, timed up and go (TUG) test, Berg Balance Scale (BBS), lower extremity motor score (LEMS), Barthel Index, and Functional Independence Measure were evaluated at pre and post intervention.
Results
There was a significant improvement in 10MWT, TUG, and BBS after the intervention. Walking ability significantly improved in participants with high walking ability at baseline (WISCI-II score 6–20; n = 12) but not in participants with low walking ability (WISCI-II score 0–3; n = 8). Significant improvement of BBS was also shown in participants with high walking ability at baseline.
Conclusions
Patients with high walking ability at baseline responded better to the training than those with low walking ability.
Similar content being viewed by others
Introduction
Spontaneous recovery after spinal cord injury (SCI) at the chronic stage is limited, and individuals with severe SCI are less likely to recover motor function than individuals with moderate SCI [1]. Over the past 25 years, since the first publication showing that humans possess a central pattern generator [2], body weight-supported treadmill training (BWSTT) has been reported to be effective for restoring gait capacity in SCI patients with varying levels of severity [3]. More recently, robot-assisted gait training (RAGT) devices have been used clinically, including Ekso [4], Lokomat [5], and ReWalk [6] devices. In general, a combination of BWSTT and RAGT offers advantages including reducing the effort required of physical therapists, longer durations of training for patients, and more accurate and reproducible gait patterns. In addition, the sensorimotor cortical regions of the cerebrum can be activated after BWSTT with the Lokomat [7]. It is important to understand who best responds to what type of treatment, and some reports have indicated that individuals with chronic SCI are responsive to each therapy included RAGT [8,9,10].
The hybrid assistive limb (HAL; Cyberdyne Inc., Ibaraki, Japan), one of a voluntary driven exoskeleton (VDE), is also effective in improving gait function for chronic SCI patients when used in conjunction with BWSTT [11,12,13,14,–15]. However, most of these previous studies included participants with some residual lower-limb motor function and walking ability [11, 13] and only showed the effectiveness of VDE for chronic SCI patients with the ability to walk. In the report by Jansen et al., a participant with more severe gait disability at baseline (Walking Index for Spinal Cord Injury II score of one) was included and showed an improvement in gait function after BWSTT with a VDE [12]. Nevertheless, his injury level and zone of partial preservation were enough to walk with orthosis clinically (Th12 level, above L1). Based on this, it is not fully clear if BWSTT with VDE is effective for all severities of SCI.
In order to optimize the use of VDE, it is necessary to understand the treatment effect that can be expected for individuals with severe SCI. The aim of this study was to quantify the effect of BWSTT with VDE in SCI patients according to the severity of SCI, evaluated using baseline walking ability.
Methods
Study participants
Twenty participants with chronic SCI were recruited from the patients of Keio University Hospital by individual referral regardless gait ability between November 2016 and November 2018. The inclusion criteria were as follows: (1) age between 20 and 75 years, (2) more than 6 months post injury, (3) self-reported disturbed gait and plateau in recovery from paralysis symptoms. The exclusion criteria were as follows: (1) disease or skin disorder that would make training impossible or be made worse by training, (2) received interferon-alpha or Botox injection within the past 6 months, (3) participated in other neurorehabilitation training (such as other BWSTT or functional electrical stimulation) within the past 3 months, and (4) underwent walking training with VDE within the past 12 months. This study was registered to UMIN-CTR (UMIN000021907).
Study design
This study was conducted as a non-randomized open-label single-arm study.
Before training, neurological classification was determined using the American Spinal Injury Association Impairment Scale (AIS), and all outcome measures were evaluated. Two physical therapists evaluated all outcomes measures throughout the research. In this investigation, every participant underwent 20 sessions of BWSTT with VDE (2–5 sessions per week), at Keio University Hospital referring to previous report [11] and investigator-initiated clinical in Japan [16]. After 20 training sessions, all outcomes were evaluated and feedback was collected from the participants. Written informed consent was obtained from the study participants, including consent to participate and to publish the findings before enrollment. No incentive was given to the study participants.
Treadmill training with VDE
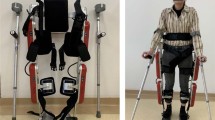
As VDE, we used HAL which was designed for this purpose as it provides voluntary motion assistance to the lower limbs and is controlled by detecting the weak bio-electrical signal generated by active muscle contractions. Electrodes were placed on the skin surface according to a previous report to capture the voluntary bio-electrical signals from extensor and flexor muscles of hip and knee joints [15]. These signals were used to provide motion support.
Training with VDE was performed on a treadmill (Aeromill STM-1250, Nihon Kohden Corporation, Tokyo, Japan) with half of their body weight supported (VDE device weight not included), by a weight-support device (PneuWeight, Pneumex, Idaho, USA). If necessary, participants were allowed to hold the handrails of the treadmill. Assistance for shifting the center of gravity and swinging of the lower limbs was provided by therapists. The velocity of the treadmill was individually set to the participant’s comfortable walking speed (between 0.5 and 2.5 km/h), and there was no incline. The duration of each training session was 60 min, which included time to rest that did not exceed 20 min. Five participants had difficulty walking with VDE and performed weight-shift training or stepping training with VDE, instead of gait training, for the initial one to three sessions.
The treadmill training with VDE was supervised by physical therapists with more than 6 months of experience using VDE, all of whom had attended the safety training courses provided by Cyberdyne, Inc. and were certified to use the device.
Outcome measures
Gait performance on the treadmill with VDE
The speed, distance, and duration the participant walked in one session with VDE was recorded in every training session. On the same days, participants reported the perceived training intensity using the Borg scale, which quantifies the perception of effort during exercise. The scale ranges from 6 to 20 points, where 6 means very very light and 19 means very very hard [17].
Overground walking ability without VDE
Overground walking ability was evaluated without VDE, but with the use of self-selected aids and braces. Walking ability was quantified using the Walking Index for Spinal Cord Injury II Scale (WISCI-II) [18]. Gait speed and number of steps needed to walk 10 m were assessed using the 10 meters walk test (10MWT) [19] performed at maximum speed. The 2 minutes walk test (2MWT) [20] was used to measure the distance walked in 2 min at a self-selected speed. Mobility was evaluated using the timed up and go (TUG) test [19], which is the time needed to stand up from a wheelchair, walk 3 m, return to the chair and sit down.
Balance and performance in activities of daily living (ADL)
Balance was evaluated using the Berg Balance Scale (BBS) [21]. The result of BBS was also evaluated in three categories—sitting balance, standing balance, and dynamic balance (position change) according to the previous study [22]. Lower extremity motor score (LEMS) was evaluated as function of lower limb [23]. Performance of ADL was evaluated using the Barthel Index (BI) [24] and Functional Independence Measure (FIM) [25].
Subjective assessment
The degree of subjective improvement and degree of satisfaction were rated by the participants on the evaluation day set after the last training session. Improvement was rated on a seven-point scale (completely improved, much improved, slightly improved, no change, slightly worse, much worse, very much worse), and satisfaction was rated on a four-point scale (satisfied, slightly satisfied, slightly dissatisfied, dissatisfied).
Statistics
The change in each outcome measure from pre to post training was assessed using a Wilcoxon signed-rank test due to the heterogeneity of the study sample. In addition, participants were categorized into two groups according to the baseline WISCI-II score: low walking ability (low group; unable to walk 10 m or could walk only in parallel bars; WISCI-II score 0–5), and high walking ability (high group; able to walk 10 m with a walker or canes/crutches, braces, or physical assistance, or without any device or assistance; WISCI-II score 6–20). We compare each parameter of participant characteristics between low and high group in using a chi-square test and Mann–Whitney U test. Within each group, the changes in each outcome measure from pre to post training was assessed using a Wilcoxon signed-rank test. Differences were considered statistically significant at p < 0.05. Data were analyzed using IBM SPSS Statistics version 25.0 (IBM Japan, Japan).
Results
Participant characteristics
The characteristics of 20 participants completed the study protocol and were left for the final analysis are shown in Table 1 (see Supplementary Table 1–1 for participant-level data). The mean (SD) age at the time of enrollment was 43.3 (16.6) years. The mean (SD) time since injury was 80.4 (128.8) months. Two participants were categorized as AIS grade A, four as grade B, eight as grade C, and six as grade D. The level of the injury was cervical in ten participants, thoracic in nine, and lumbar in one. The median (range) WISCI-II score was 9 (0–20), including seven participants with a score of zero. Based on baseline WISCI-II score, eight participants were categorized into the low walking ability group, and 12 participants were categorized into the high walking ability group. The mean (SD) frequency of intervention was 2.6 (1.1) days per week. All participants achieved 20 training sessions with VDE. There were no adverse events.
Gait performance on the treadmill with VDE
The speed, distance, and total duration participants walked in one training session increased significantly from the first to the last training session in all participants (Table 2 and also see Supplementary Tables 1–2 for participant-level data). Five participants who initially had difficulty walking with VDE were analyzed afterwards, when they were able to walk with VDE for the first time. Mean ratings of perceived exertion across the 20 sessions were significantly higher in the low group compared with the high group. This provides evidence that the training intensity was adequate in the low group, even though the other three training parameters were significantly lower in this group (see Supplementary Table 2 for training intensity in two groups).
Overground walking ability without VDE
Overground walking ability before and after 20 sessions of VDE training is shown in Table 3 (see Supplementary Tables 1–3 for participant-level data). The WISCI-II score tended to increase. No participants in the low group were able to complete the tests of overground walking ability (10MWT and TUG) at either time point. In the high group, there was a significant improvement in 10MWT time (134.0 to 88.3 s, p = 0.01) and speed (0.26 to 0.34 s/m, p < 0.01), 10MWT number of steps (44.8 to 36.5 steps, p = 0.05), and TUG time (83.5 to 68.5 s, p = 0.01). The decrease in 10MWT number of steps indicates an extension of step length. The WISCI score was not significantly improved in the high group (10.5 to 11.5, p = 0.11). In terms of the relation between relative change in 10MWT time and WISCI-II score at baseline, the relative change in 10MWT time varied among participants (Supplementary Figure).
Balance and performance of ADL
Balance and performance of ADL before and after 20 sessions of VDE training are shown in Table 3 (see Supplementary Tables 1–4 for participant-level data). When the group was considered as a whole, there was a significant improvement in BBS score, but not BI or FIM (Table 3). Although the BBS score improved following training in the high group (p = 0.02, Table 3), there was no change in the low group (p = 0.06, Table 3). When BBS was evaluated by three categories, participants in the low group showed little improvement in dynamic balance and sitting balance but no improvement in standing balance (Supplementary Table 3). Neither the BI nor the FIM changed from pre to post training in high group (p = 0.32, p = 1.00) and low group (p = 1.00, p = 1.00), indicating that there was no effect of training on performance of ADL regardless of baseline walking ability (Table 3).
Subjective assessment
Subjective improvement was reported by 19 participants (two answered “much well” and 17 answered “slight well”), and all participants were either fully or slightly satisfied with the training (eight were satisfied and 12 were slightly satisfied).
Discussion
This study is novel in the point that the performance in balance and gait of chronic SCI patients was improved after as few as 20 sessions of BWSTT with VDE. The degree of improvement depended on baseline walking ability, with patients with a baseline WISCI-II score of less than six hardly benefitting from the training. This result was against previous study that reported the improvement of walking ability even in the patients with severe motor dysfunction with a baseline WISCI-II score of one [12]. Although the overall change in BBS score was not significant for our participants with a baseline WISCI-II score of less than six, some of them did show an improvement in sitting balance.
Jansen et al. showed improvement in gait ability after 60 sessions with VDE in chronic SCI participants who included a participant with severe gait disability at baseline (WISCI-II score of one) [12]. He was classified as AIS grade A, however, his injury was at Th12 and zone of partial preservation was above L1. In addition, his baseline Janda muscle function testing grade was more than two for the hip joint flexors and knee joint extensors, which meant that he could move his hip and knee joints when supported against gravity. In contrast, the low group in the present study had more severe motor dysfunction than the patient reported by Jansen et al. Other reports also showed that gait function was not or poorly improved in the participant without baseline gait ability by locomotion training [26] or RAGT [27] too. Moreover, the above intervention by Jansen et al. included regular physical therapy and exercises in addition to BWSTT with VDE [12]. By contrast, participants in the present study performed only BWSTT with VDE. This may require additional interventions for chronic SCI patient with severe motor dysfunction and gait disability.
On the other hand, our result in high group (WISCI-II score of 6–20) was consistent with previous studies [11, 13]. More specifically, they improved gait performances without VDE at less times of training sessions using VDE than that of them. Considering our result and previous studies that included participants with some residual lower limb motor function and walking ability (i.e., WISCI-II score ≥ 6), BWSTT with VDE is effective for chronic SCI patients with the ability to walk with some aid at baseline. In other word, the patients who are most likely to benefit from BWSTT with VDE have certain stability of the lower limbs, which means enough to move one’s leg under the condition without gravity. Some previous studies reported that showed baseline strength of lower muscle was useful for person with SCI to predict responsiveness in BWSTT and activity-based therapy [8, 26]. Meanwhile, in this study, a small improvement in WISCI-II scores was shown and this was not statistically significant in the high group, which was consistent with two previous reports that enrolled 8 patients [15] and 21 patients [12] with chronic SCI using VDE. These results may indicate that the sample is too small sample to achieve a statistically significant change in WISCI-II scores.
In the present study, there was significant improvement in the BBS score in all patients and high group following BWSTT with VDE, which is novel finding. Although there was no significant change in the low group, some of them showed an improvement in sitting balance or dynamic balance (Supplementary Table 3). This is in accordance with a previous study that demonstrated RAGT improved trunk muscular activity during gait in high-thoracic, motor-completed SCI patients [28]. Thus, similar to previous research [29], RAGT may improve trunk muscular function and sitting balance in SCI patients with severe motor impairment.
The present study has several limitations. First, the duration of the intervention (20 sessions) may have been insufficient to improve the ability of the low group. We chose this duration because we considered it worthwhile to prove information on the minimum training duration required to elicit an effect, regarding many chronic SCI patients have returned to social life and are difficult to spend long time on training. The selection of duration was based on studies that adopted 9 sessions [16] and 30 sessions [11]. While a previous report showed that RAGT was ineffective for re-establishing gait in chronic SCI patients without baseline gait function after 60 sessions [10], longitudinal research utilizing >60 sessions is required. Second, the frequency of the intervention differed across participants, and this could have led to dispersion of the results. However, as there was no significant difference in intervention frequency between the high and low group, it is likely our main finding was not severely affected. Third, this was a heterogeneous study sample that included a wide range of time since injury, a wide range of injury level and type (nontraumatic and traumatic), a mixture of complete and incomplete SCI, and was also no control group. For this reason, we could not confirm significant difference of the treatment effect of our patients by analysis by the level of injury or functional severity, as was shown in previous studies [2, 30]. Although this baseline heterogeneity may lead effect modification, baseline data showed no significant difference between groups except for AIS grade and WISCI score in Table 1, which may be related at baseline gait ability. Fourth, we did not perform a detailed evaluation of spinal circuitry (e.g., electromyography, somatosensory, or motor evoked potentials) after RAGT in our study. Sczesny-Kaiser et al. reported that BWSTT with VDE improved cortical excitability in the primary somatosensory cortex for people with chronic SCI [14]. Accordingly, it is possible that the difference in the recovery of gait function between the low and high group of our study was also influenced by differences in spinal circuitry, especially sensory function. Further research is needed to clarify the sensory functional influence on effectiveness in BWSTT using VDE. Lastly, it was not possible to collect data in a blinded manner.
We found that chronic SCI patients with high walking ability at baseline showed significant improvement in walking function and balance function by 20 sessions of BWSTT with VDE. On the other hand, patients with a baseline WISCI-II score of less than six did not show significant improvement in either of these functions. In the near future, BWSTT with VDE combined with new therapies under development, such as cell transplantation or medication, may further promote functional recovery even in individuals with severe chronic SCI.
Data availability
This can be found in the Supplementary Tables 1–1, 1–2, 1–3, and 1–4.
References
Wilson JR, Jaja BNR, Kwon BK, Guest JD, Harrop JS, Aarabi B, et al. Natural history, predictors of outcome, and effects of treatment in thoracic spinal cord injury: a multi-center cohort study from the North American Clinical Trials Network. J Neurotrauma. 2018;35:2554–60.
Dietz V, Colombo G, Jensen L, Baumgartner L. Locomotor capacity of spinal cord in paraplegic patients. Ann Neurol. 1995;37:574–82.
Lam T, Eng JJ, Wolfe DL, Hsieh JT, Whittaker M. A systematic review of the efficacy of gait rehabilitation strategies for spinal cord injury. Top Spinal Cord Inj Rehabil. 2007;13:32–57.
Bach Baunsgaard C, Vig Nissen U, Katrin Brust A, Frotzler A, Ribeill C, Kalke YB, et al. Gait training after spinal cord injury: safety, feasibility and gait function following 8 weeks of training with the exoskeletons from Ekso Bionics. Spinal Cord. 2018;56:106–16.
Colombo G, Wirz M, Dietz V. Driven gait orthosis for improvement of locomotor training in paraplegic patients. Spinal Cord. 2001;39:252–5.
Esquenazi A, Talaty M, Packel A, Saulino M. The ReWalk powered exoskeleton to restore ambulatory function to individuals with thoracic-level motor-complete spinal cord injury. Am J Phys Med Rehabil. 2012;91:911–21.
Winchester P, McColl R, Querry R, Foreman N, Mosby J, Tansey K, et al. Changes in supraspinal activation patterns following robotic locomotor therapy in motor-incomplete spinal cord injury. Neurorehabilit Neural Repair. 2005;19:313–24.
Yang JF, Norton J, Nevett-Duchcherer J, Roy FD, Gross DP, Gorassini MA. Volitional muscle strength in the legs predicts changes in walking speed following locomotor training in people with chronic spinal cord injury. Phys Ther. 2011;91:931–43.
Piira A, Lannem AM, Sorensen M, Glott T, Knutsen R, Jorgensen L, et al. Manually assisted body-weight supported locomotor training does not re-establish walking in non-walking subjects with chronic incomplete spinal cord injury: a randomized clinical trial. J Rehabil Med. 2019;51:113–9.
Piira A, Lannem AM, Sorensen M, Glott T, Knutsen R, Jorgensen L, et al. Robot-assisted locomotor training did not improve walking function in patients with chronic incomplete spinal cord injured: a randomized clinical trial. J Rehabil Med. 2019;51:385–9.
Jansen O, Schildhauer TA, Meindl RC, Tegenthoff M, Schwenkreis P, Sczesny-Kaiser M, et al. Functional outcome of neurologic-controlled HAL-exoskeletal neurorehabilitation in chronic spinal cord. Injury: a pilot one year treat variable treat frequency. Glob Spine J. 2017;7:735–43.
Jansen O, Grasmuecke D, Meindl RC, Tegenthoff M, Schwenkreis P, Sczesny-Kaiser M, et al. Hybrid Assistive Limb exoskeleton HAL in the rehabilitation of chronic spinal cord injury: proof of concept; the results in 21 patients. World Neurosurg. 2018;110:e73–8.
Grasmucke D, Zieriacks A, Jansen O, Fisahn C, Sczesny-Kaiser M, Wessling M, et al. Against the odds: what to expect in rehabilitation of chronic spinal cord injury with a neurologically controlled Hybrid Assistive Limb exoskeleton. A subgroup analysis of 55 patients according to age and lesion level. Neurosurg Focus. 2017;42:E15.
Sczesny-Kaiser M, Hoffken O, Aach M, Cruciger O, Grasmucke D, Meindl R, et al. HAL(R) exoskeleton training improves walking parameters and normalizes cortical excitability in primary somatosensory cortex in spinal cord injury patients. J Neuroeng Rehabil. 2015;12:68.
Aach M, Cruciger O, Sczesny-Kaiser M, Hoffken O, Meindl R, Tegenthoff M, et al. Voluntary driven exoskeleton as a new tool for rehabilitation in chronic spinal cord injury: a pilot study. Spine J. 2014;14:2847–53.
Nakajima T. Cybernic neurorehabilitation using robot suit HAL. Neurol Ther. 2016;33:396–8.
Borg GA. Psychophysical bases of perceived exertion. Med Sci Sports Exerc. 1982;14:377–81.
Dittuno PL, Ditunno JF Jr. Walking index for spinal cord injury (WISCI II): scale revision. Spinal Cord. 2001;39:654–6.
Van Hedel HJ, Wirz M, Dietz V. Assessing walking ability in subjects with spinal cord injury: validity and reliability of 3 walking tests. Arch Phys Med Rehabil. 2005;86:190–6.
Rossier P, Wade DT. Validity and reliability comparison of 4 mobility measures in patients presenting with neurologic impairment. Arch Phys Med Rehabil. 2001;82:9–13.
Matsushima M, Yabe I, Uwatoko H, Shirai S, Hirotani M, Sasaki H. Reliability of the Japanese Version of the Berg Balance Scale. Intern Med. 2014;53:1621–4.
Han JY, Kim JM, Kim SK, Chung JS, Lee HC, Lim JK, et al. Therapeutic effects of mechanical horseback riding on gait and balance ability in stroke patients. Ann Rehabil Med. 2012;36:762–9.
Maynard FM, Jr., Bracken MB, Creasey G, Ditunno JF, Jr., Donovan WH, Ducker TB. et al. International standards for neurological and functional classification of spinal cord injury. Spinal Cord. 1997;35:266–74.American Spinal Injury Association.
Collin C, Wade DT, Davies S, Horne V. The Barthel ADL Index: a reliability study. Int Disabil Stud. 2009;10:61–3.
Hamilton BB, Laughlin Ja Fau-Fiedler RC, Fiedler Rc Fau-Granger CV, Granger CV. Interrater reliability of the 7-level functional independence measure (FIM). Scand J Rehab Med. 1994;26:115–9. 0036-5505 (Print).
Jones ML, Evans N, Tefertiller C, Backus D, Sweatman M, Tansey K, et al. Activity-based therapy for recovery of walking in chronic spinal cord injury: results from a secondary analysis to determine responsiveness to therapy. Arch Phys Med Rehabil. 2014;95:2247–52.
Niu X, Varoqui D, Kindig M, Mirbagheri MM. Prediction of gait recovery in spinal cord injured individuals trained with robotic gait orthosis. J Neuroeng Rehabil. 2014;11:42.
Alamro RA, Chisholm AE, Williams AMM, Carpenter MG, Lam T. Overground walking with a robotic exoskeleton elicits trunk muscle activity in people with high-thoracic motor-complete spinal cord injury. J Neuroeng Rehabil. 2018;15:109.
Kim HS, Jeong HJ, Kim MO. Changes of functional outcomes according to the degree of completeness of spinal cord injury. Ann Rehabil Med. 2014;38:335–41.
Wilcox JT, Satkunendrarajah K, Nasirzadeh Y, Laliberte AM, Lip A, Cadotte DW, et al. Generating level-dependent models of cervical and thoracic spinal cord injury: exploring the interplay of neuroanatomy, physiology, and function. Neurobiol Dis. 2017;105:194–212.
Acknowledgements
We thank Cyberdyne Inc. for their technical support in the use of HAL.
Funding
Our institution received grant from Japan Agency for Medical Research and Development (Grant number: 19bk0104017h0002), Japan Science and Technology Agency (Grant number: 05-001-0002), and the Uehara Memorial Foundation for this study, but all author received no specific funding for this work. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
HO and TS conducted evaluation and intervention for all participants, analysis and interpreting the result, and drafting the manuscript. KM was responsible for designing the study protocol, recruiting participants. KS played an important role in interpreting the result and revised the manuscript mainly. OT, NN, and MM played an important role in interpreting the result and revised the manuscript. MN was responsible for all working related to this submission as corresponding author. Also, all authors approved the final version manuscript and agreed to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Statement of ethics
We certify that all applicable institutional and governmental regulations concerning the ethical use of human volunteers were followed during the course of this research. The study protocol was conducted in compliance with ethical guidelines for medical and health research involving human subjects and approved by the Ethics Committees of Keio University School of Medicine (Approval no. 20150355).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Okawara, H., Sawada, T., Matsubayashi, K. et al. Gait ability required to achieve therapeutic effect in gait and balance function with the voluntary driven exoskeleton in patients with chronic spinal cord injury: a clinical study. Spinal Cord 58, 520–527 (2020). https://doi.org/10.1038/s41393-019-0403-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41393-019-0403-0
This article is cited by
-
Exoskeleton-based exercises for overground gait and balance rehabilitation in spinal cord injury: a systematic review of dose and dosage parameters
Journal of NeuroEngineering and Rehabilitation (2024)
-
Wearable powered exoskeletons for gait training in tetraplegia: a systematic review on feasibility, safety and potential health benefits
Acta Neurologica Belgica (2022)
-
Cybernic treatment with wearable cyborg Hybrid Assistive Limb (HAL) improves ambulatory function in patients with slowly progressive rare neuromuscular diseases: a multicentre, randomised, controlled crossover trial for efficacy and safety (NCY-3001)
Orphanet Journal of Rare Diseases (2021)