Abstract
Background
To assess whether postnatal plasma cholesterol levels are associated with microstructural and macrostructural regional brain development in preterm newborns.
Methods
Sixty preterm newborns (born 24–32 weeks gestational age) were assessed using MRI studies soon after birth and again at term-equivalent age. Blood samples were obtained within 7 days of each MRI scan to analyze for plasma cholesterol and lathosterol (a marker of endogenous cholesterol synthesis) levels. Outcomes were assessed at 3 years using the Bayley Scales of Infant Development, Third Edition.
Results
Early plasma lathosterol levels were associated with increased axial and radial diffusivities and increased volume of the subcortical white matter. Early plasma cholesterol levels were associated with increased volume of the cerebellum. Early plasma lathosterol levels were associated with a 2-point decrease in motor scores at 3 years.
Conclusions
Higher early endogenous cholesterol synthesis is associated with worse microstructural measures and larger volumes in the subcortical white matter that may signify regional edema and worse motor outcomes. Higher early cholesterol is associated with improved cerebellar volumes. Further work is needed to better understand how the balance of cholesterol supply and endogenous synthesis impacts preterm brain development, especially if these may be modifiable factors to improve outcomes.
Similar content being viewed by others
Introduction
Children born preterm are at risk for brain injury, impaired brain development, and adverse neurological and developmental outcomes. As we improve our skills at improving survival of preterm newborns, significant strides have also been made to better understand the mechanisms for injury and impaired development, and such mechanisms have been targeted in the neonatal intensive care unit with dramatic successes. For example, optimization of ventilation settings have decreased the incidence of cystic periventricular leukomalacia.1 With increasing understanding of the range of factors that may impact preterm brain development, the role of perinatal nutrition is becoming more important. While some studies have focused on energy intake or proportions of macronutrients,2,3 others have focused on specific nutritional factors, such as polyunsaturated fatty acids.4
Cholesterol, a necessary component of the plasma cell membrane, plays an important role not only in cell membrane integrity but also in cell signaling, especially in cerebellar development.5 In the mature human body, 25% of total body cholesterol content exists in the brain, particularly within myelin. While studies in adults have looked at the relationship between cholesterol levels and brain hemorrhage outcomes6,7 and preterm newborns have been shown to be at risk for marked elevations in cholesterol levels due to stimulation of endogenous biosynthesis,8 there have been no studies addressing the relationship between cholesterol and neurodevelopmental outcomes after preterm birth. This prospective cohort study of preterm newborns uses serial blood samples and brain magnetic resonance imaging (MRI) studies to address the macrostructural (regional brain volumes) and microstructural (diffusion tensor imaging metrics) associations between postnatal cholesterol levels and brain injury and development after preterm birth. Lathosterol, a precursor in the pathway of cholesterol synthesis and a biochemical marker of endogenous cholesterol synthesis, is also assessed to better understand the relationships found. The hypothesis of this exploratory study is that higher cholesterol levels in the early postnatal period are associated with decreased risk of brain injury and improved development of the white matter and cerebellum in preterm newborns.
Methods
Study subjects
Preterm infants born at ≤32 weeks gestational age and admitted to the neonatal intensive care units at the University of California San Francisco Benioff Children’s Hospital and the British Columbia Children’s and Women’s Hospital between March 2010 and November 2011 were approached for this prospective cohort study, with 60 subjects enrolled. Consent was obtained following a protocol approved by the research ethics boards at both institutions. Infants were excluded if there was a congenital malformation or syndrome, congenital infection, or they were too clinically unstable to transport to the MRI scanner within the study timeframe. Clinical history was collected prospectively from patient charts. Feeding protocols in these infants are as previously described, including parenteral nutrition and intravenous fat emulsion as needed, followed by enteral feeds of expressed breast milk or formula, sometimes supplemented for protein.4
MRI studies
MRI studies were performed soon after birth as soon as clinically stable and at term-equivalent age on 1.5-T MRI scanners (General Electric Sigma, GE Medical Systems, Milwaukee, WI or Siemens Avanto, Siemens Medical Solutions, Malvern, PA). Sequences obtained include volumetric three-dimensional T1-weighted, T2-weighted, and diffusion tensor imaging (DTI) sequences as previously described.4 All imaging analysis was performed blinded to patient clinical history. Scans were visually graded for brain injury, including intraventricular hemorrhage (IVH),9 white matter injury (WMI),10 and cerebellar hemorrhage,11 by a single neuroradiologist at each study site (A.J.B., Kenneth J. Poskitt). The highest injury score from both scans was selected as the injury severity score for each infant.
Prespecified regions of interest were measured on DTI sequences for apparent diffusion coefficient (ADC), fractional anisotropy (FA), axial diffusivity (AD, first eigenvector), and radial diffusivity (RD, average of second and third eigenvector). Regions of interest included subcortical white matter, posterior limb of the internal capsule, optic radiations, and deep gray matter (including basal ganglia and thalamus) (Fig. 1). Data were inadequate for assessment of cerebellar and brainstem regions of interest. Measurements of left and right hemispheres were averaged for each scan.
Diffusion tensor imaging and regions of interest. The anisotropy color maps of an infant born at 26–6/7 weeks gestational age and scanned at 29–3/7 weeks postmenstrual age are presented in two axial planes to demonstrate the regions of interest. Regions of interest in the (1) anterior, (2) central, and (3) posterior cortical white matter were averaged for analysis of cortical white matter. Regions of interest in the (4) basal ganglia and (6) thalamus were averaged for analysis of deep gray matter. Other regions of interest included the (5) posterior limb of the internal capsul and the (7) optic radiations. Colors display the predominant diffusion directions, with red representing right–left, green representing anterior–posterior, and blue presenting superior–inferior directions
Brain tissue volumes were automatically segmented into tissue classes (cortical gray matter, white matter, deep gray matter, cerebellum, and brainstem) as previously described, with Dice Similar Coefficients of 0.883 across tissue classes.12,13
Blood samples
Two blood samples were drawn from each subject, obtained as close in time as possible within a week of each MRI scan, timed with clinically indicated bloodwork. Samples were separated into plasma and red blood cell fractions and stored at −70 °C until analysis. Cholesterol and lathosterol (a marker of de novo cholesterol synthesis14) levels in plasma were analyzed by gas chromatography–mass spectrometry.
Neurodevelopmental outcomes
Infants were reassessed between 30 and 36 months corrected age by a developmental psychologist or physiotherapist blinded to the child’s perinatal course. Standardized assessment was performed using the Bayley Scales of Infant Development, Third Edition, and composite cognitive, language, and motor scores were assigned based on the child’s corrected age.
Statistical analysis
Statistical analysis was performed using R version 3.3.15 All analyses were adjusted for study site to account for variations between sites. Ordinal logistic regression analysis was used to study the association between cholesterol or lathosterol levels and measures of brain injury (including IVH, WMI, and cerebellar hemorrhage). These analyses adjusted for known confounders for brain injury including gestational age at birth, chorioamnionitis (suspected or confirmed), antenatal glucocorticoid exposure, 5-min Apgar score, patent ductus arteriosus (diagnosed by echocardiogram), hypotension requiring medical intervention, intubation, and neonatal sepsis (presumed or confirmed, including necrotizing enterocolitis).
Early plasma levels were compared to neuroimaging measures on both scans. Linear mixed effects analysis was used to study the association between early cholesterol or lathosterol levels and DTI regions of interest, to account for repeated scans in each subject. Linear mixed effects analysis was used to study the association between early cholesterol or lathosterol levels and brain volumes, which were cube-root transformed for normality. Term-equivalent age plasma levels were compared to neuroimaging measures on the term-equivalent age scan only, as it would not be relevant to study the relationship between MRI scans done earlier than the plasma levels. For these analyses, simple linear regression models for DTI and non-linear regression models for brain volumes were used. All analyses were adjusted for postmenstrual age (PMA) at MRI, sex, IVH, WMI, days intubated (log-transformed), sepsis, and patent ductus arteriosus.
Plasma cholesterol and lathosterol levels were also compared to Bayley cognitive, language, and motor scores at 30–36 months using linear regression analyses.
Associations were considered significant at P ≤ 0.050.
Results
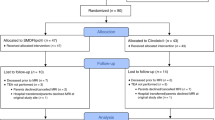
A total of 60 preterm newborns were enrolled from the two study centers (37 males), demographics described in Supplemental Table S1, as previously reported.4 Plasma cholesterol and lathosterol levels, as well as brain MRIs were successfully completed early in 59/60 (98%) subjects (mean 31.44 ± 2.15 weeks PMA), while near-term MRI and blood sampling were completed in 43/60 (72%) of subjects (mean 37.68 ± 3.01 weeks PMA). Mean early plasma cholesterol levels were 2.50 ± 0.76 mmol/L (range 1.3–6.1 mmol/L) and lathosterol levels 5.85 ± 2.96 μmol/L (range 1.5–15.5μmol/L). Mean near-term plasma cholesterol levels were 2.34 ± 0.78 mmol/L (range 1.2–5.1 mmol/L) and lathosterol levels 3.71 ± 1.53 μmol/L (range 1.2–8.5 μmol/L).
Brain injury
Ordinal logistic regression analysis adjusting for confounding factors found no associations between early or near-term cholesterol and lathosterol levels and IVH, WMI, or cerebellar hemorrhage (Table 1).
Diffusion tensor imaging
Mixed effects models adjusting for confounders, including study site, demonstrated that early plasma lathosterol levels were associated with increased AD (0.02, 95% confidence interval (CI) 0.002–0.047, P = 0.035) and RD (0.012, 95% CI 0.00004–0.02, P = 0.049) in the subcortical white matter on both MRI scans but in no other brain regions measured. Early plasma cholesterol and lathosterol associations with DTI measures are summarized in Table 2. Linear regression analyses showed no associations between near-term cholesterol or lathosterol levels and DTI measures in the regions assessed (P > 0.1, data not shown).
Brain volumes
Mixed effects models adjusting for confounders demonstrated that, per 1 mmol/L increase in early plasma cholesterol levels, there is an associated 1.11 cm3 increase in cerebellar volumes on both MRI scans (95% CI 0.37–1.86 cm3, P = 0.009). Meanwhile, per 1 mmol/L increase in early plasma lathosterol levels, there is an associated 1.98 cm3 increase in subcortical white matter volume on both MRI scans (95% CI 0.53–3.46 cm3, P = 0.018). As well, per 1 mmol/L increase in near-term plasma cholesterol levels, there is an associated 8.00 cm3 decrease in near-term cortical gray matter volumes (95% CI 0.52–15.48 cm3, P = 0.048). Associations between plasma levels and brain volumes are summarized in Table 3.
Neurodevelopmental outcome
Mean scores on neurodevelopmental testing were 104 ± 14 on cognitive testing (range 80–145), 101. ± 18 on language testing (range 65–138), and 98 ± 14 on motor testing (range 64–124). Using linear regression analysis, each 1-μmol/L increase in early plasma lathosterol levels was associated with a 2.0-point decrease in Bayley motor scores (95% CI 0.3–3.6, P = 0.018), but other plasma levels were not associated with outcomes (Table 4).
Discussion
Although cholesterol plays an important role in cell signaling through neurodevelopment and is a key component of myelin, there has been little research into its impact on preterm brain injury, growth, and neurodevelopment. This current study demonstrates associations between cholesterol and lathosterol levels and brain development in the subcortical white matter, cortical gray matter, and cerebellum, as well as associations with motor outcomes at 3 years corrected age.
Considering that 25% of the total body cholesterol is found in the brain, concentrated in the myelin, it would be expected that variations in cholesterol supply would have consequences on the developing subcortical white matter. Indeed, preterm newborns have been found to be at risk for marked elevations in plasma cholesterol levels due to upregulation of endogenous cholesterol synthesis.8 Here we show that higher early plasma lathosterol levels are associated with higher AD and RD, without significant change in ADC or FA, and higher regional volumes in the subcortical white matter. Interestingly, other regions of interest in the white matter, including the posterior limb of the internal capsule and the optic radiations, do not seem to be involved. It has been demonstrated that, with increasing gestational age, FA increases while ADC, AD, and RD decrease.16 The constellation of increasing AD and RD and increasing volume could suggest regional tissue swelling and impaired or delayed development of the cortical white matter. This theory is further supported by the corresponding worsened Bayley motor scores at 3 years with higher early lathosterol. Since lathosterol is a marker of endogenous cholesterol synthesis, this may suggest that this may represent an increased endogenous production either in response to inadequate cholesterol supplies or an endogenous drive for higher cholesterol levels. Since higher early plasma cholesterol levels were not found to be associated with worse DTI metrics, this suggests that inadequate cholesterol supply may be more likely. This fits with what has previously been described in preterm newborns.8 With standardized feeding protocols postnatally, cholesterol supply becomes more standard, which may explain the lack of associations found with later lathosterol levels.
The most statistically significant finding in this study was an association between early plasma cholesterol levels and increased cerebellar volumes. Unfortunately, the DTI measures could not be accurately assessed in the posterior fossa using the sequences obtained for this study,17 thus only volumetric data are available for the cerebellum and brainstem. Cholesterol is known to play a critical role in upregulating Sonic hedgehog signaling,5,18,19,20,21 which in turn is a key pathway supporting the development of the cerebellum.22 Cholesterol covalently modifies hedgehog proteins and is a necessary posttranslational modification to initiate the function of Sonic hedgehog protein.5 Inhibition of cholesterol synthesis has been shown to result in abnormalities in cerebellar development.23,24,25 Thus there is mechanistic evidence linking cholesterol and cerebellar development to support our observed association.
We have previously reported from this cohort that larger cortical and deep gray matter, cerebellar, and brainstem volumes were associated with improved language scores at 30–36 months corrected age. Also, larger cerebellar and brainstem volumes were associated with improved motor scores.12 Although we did not find that cholesterol levels were associated with improved language or motor scores, an association between cholesterol levels and cerebellar volumes suggests that these larger volumes may also be contributing to improved outcomes.
The association of higher endogenous synthesis with worse subcortical white matter development and that of higher overall cholesterol levels with better cerebellar development seems superficially contradictory. However, one could postulate that higher endogenous synthesis can be a response to lower supply, creating a need to synthesize more endogenously to meet demand. This endogenous synthesis may be adequate to normalize plasma levels, while remaining inadequate to maintain subcortical white matter development. Thus, in such a way, higher cholesterol levels and lower need to synthesize endogenously are both associated with improved subcortical white matter and cerebellar development.
Another aspect of the relationship between cholesterol and preterm brain development is the issue of the blood–brain barrier. The fetus has been shown to receive cholesterol supply via the placenta.26 However, there is evidence in sheep that cholesterol carried in low-density lipoproteins are not incorporated into the brain.27 In the adult brain, it is known that the blood–brain barrier prevents transport of cholesterol,28 but the exact timing of the closure of the blood–brain barrier to cholesterol is not known in humans. The associations found in this study suggest that there may be either some level of transfer of cholesterol into the brain or at least signaling effects that alter brain development. Indeed, it is unknown what factors result in variability in cholesterol and lathosterol levels in preterm newborns, and these factors may also be important in driving these observed associations.
Finally, we also found in this study that higher term cholesterol levels were associated with decreased cortical gray matter volumes at term age. The biochemical basis of this association is unclear, as there is no mechanistic link previously demonstrated between cholesterol and this brain region. It is reassuring, however, that term cholesterol levels were not associated with neurodevelopmental outcomes. However, cognitive impacts may be more evident on longer developmental follow-up.
This study is limited by its exploratory nature and small sample size. However, consistent findings associating early lathosterol levels and macrostructural and microstructural measures of brain development and cholesterol’s key role in myelin formation suggest that a true relationship exists between early lathosterol levels and subcortical white matter development. Also, strong mechanistic ties between cholesterol and cerebellar development support the plausibility of these reported associations.
It has been well established that cholesterol is a key component of myelin and that cholesterol is necessary for the Sonic hedgehog pathway, which is key to cerebellar development. However, there has been very little research into the implications of cholesterol exposure antenatally and postnatally on brain injury and development in the preterm newborn. This study brings forth new questions regarding the relationship between endogenous cholesterol synthesis and impaired development of the subcortical white matter, as well as cholesterol levels and cerebellar growth. Indeed more work is needed to understand the relationship between plasma and brain cholesterol levels. This study sheds light on an important and poorly understood topic, suggesting that more research is needed to better understand the complex role of cholesterol supply and endogenous synthesis on preterm neurodevelopment.
References
Fujimoto, S. et al. Hypocarbia and cystic periventricular leukomalacia in premature infants. Arch. Dis. Child. 71, F107–F110 (1994).
Beauport, L. et al. Impact of early nutritional intake on preterm brain: a magnetic resonance imaging study. J. Pediatr. 181, 29–36 e21 (2017).
Coviello, C. et al. Effects of early nutrition and growth on brain volumes, white matter microstructure, and neurodevelopmental outcome in preterm newborns. Pediatr. Res. 83, 102–110 (2018).
Tam, E. W. et al. Early postnatal docosahexaenoic acid levels and improved preterm brain development. Pediatr. Res. 79, 723–730 (2016).
Porter, J. A., Young, K. E. & Beachy, P. A. Cholesterol modification of hedgehog signaling proteins in animal development. Science 274, 255–259 (1996).
Roquer, J. et al. Serum lipid levels and in-hospital mortality in patients with intracerebral hemorrhage. Neurology 65, 1198–1202 (2005).
Lindbohm, J. V., Kaprio, J. & Korja, M. Cholesterol as a risk factor for subarachnoid hemorrhage: a systematic review. PLoS ONE 11, e0152568 (2016).
Innis, S. M. & Hamilton, J. J. Effects of developmental changes and early nutrition on cholesterol metabolism in infancy: a review. J. Am. Coll. Nutr. 11(Suppl), 63S–68S (1992).
Papile, L. A., Burstein, J., Burstein, R. & Koffler, H. Incidence and evolution of subependymal and intraventricular hemorrhage: a study of infants with birth weights less than 1,500 gm. J. Pediatr. 92, 529–534 (1978).
Miller, S. P. et al. Early brain injury in premature newborns detected with magnetic resonance imaging is associated with adverse early neurodevelopmental outcome. J. Pediatr. 147, 609–616 (2005).
Tam, E. W. et al. Cerebellar hemorrhage on magnetic resonance imaging in preterm newborns associated with abnormal neurologic outcome. J. Pediatr. 158, 245–250 (2011).
Kamino, D. et al. Postnatal polyunsaturated fatty acids associated with larger preterm brain tissue volumes and better outcomes. Pediatr. Res. 83, 93–101 (2018).
Liu, M. et al. Patch-based augmentation of expectation-maximization for brain MRI tissue segmentation at arbitrary age after premature birth. Neuroimage 127, 387–408 (2016).
Mercer, E. I. & Glover, J. Sterol metabolism. 6. The interconversion of cholesterol, 7-dehydrocholesterol and lathosterol in the rat. Biochem. J. 80, 552–556 (1961).
R Core Team. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, Vienna, 2017). https://www.r-project.org/.
Partridge, S. C. et al. Diffusion tensor imaging: serial quantitation of white matter tract maturity in premature newborns. Neuroimage 22, 1302–1314 (2004).
Tam, E. W. Y. et al. Cerebellar development in the preterm neonate: effect of supratentorial brain injury. Pediatr. Res. 66, 102–106 (2009).
Cooper, M. K. et al. A defective response to Hedgehog signaling in disorders of cholesterol biosynthesis. Nat. Genet. 33, 508–513 (2003).
Beachy, P. A. et al. Multiple roles of cholesterol in hedgehog protein biogenesis and signaling. Cold Spring Harb. Symp. Quant. Biol. 62, 191–204 (1997).
Gruchy, N. et al. Involvement and alteration of the Sonic Hedgehog pathway is associated with decreased cholesterol level in trisomy 18 and SLO amniocytes. Mol. Genet. Metab. 112, 177–182 (2014).
Hennekam, R. C. Congenital brain anomalies in distal cholesterol biosynthesis defects. J. Inherit. Metab. Dis. 28, 385–392 (2005).
Dahmane, N. & Ruiz i Altaba, A. Sonic hedgehog regulates the growth and patterning of the cerebellum. Development 126, 3089–3100 (1999).
Repetto, M. et al. Teratogenic effect of the cholesterol synthesis inhibitor AY 9944 on rat embryos in vitro. Teratology 42, 611–618 (1990).
Lanoue, L. et al. Limb, genital, CNS, and facial malformations result from gene/environment-induced cholesterol deficiency: further evidence for a link to sonic hedgehog. Am. J. Med. Genet. 73, 24–31 (1997).
Dehart, D. B., Lanoue, L., Tint, G. S. & Sulik, K. K. Pathogenesis of malformations in a rodent model for Smith-Lemli-Opitz syndrome. Am. J. Med. Genet. 68, 328–337 (1997).
Spellacy, W. N., Ashbacher, L. V., Harris, G. K. & Buhi, W. C. Total cholesterol content in maternal and umbilical vessels in term pregnancies. Obstet. Gynecol. 44, 661–665 (1974).
Turley, S. D., Burns, D. K., Rosenfeld, C. R. & Dietschy, J. M. Brain does not utilize low density lipoprotein-cholesterol during fetal and neonatal development in the sheep. J. Lipid Res. 37, 1953–1961 (1996).
Dietschy, J. M., Turley, S. D. & Spady, D. K. Role of liver in the maintenance of cholesterol and low density lipoprotein homeostasis in different animal species, including humans. J. Lipid Res. 34, 1637–1659 (1993).
Acknowledgments
The authors would like to dedicate this manuscript to late Sheila M. Innis, RD, PhD and thank her for her guidance and mentorship. The authors also thank Roger Dyer for his assistance with analyzing blood samples for cholesterol and lathosterol levels for this study after Dr. Innis’ passing. The authors also thank Kenneth J. Poskitt, MD, FRCPC for his assistance in reviewing MRI scans at the University of British Columbia study site. The authors also thank the families and children who have participated in this study. The study was supported by the Gerber Foundation (Fremont, MI) [EWYT], Canadian Institutes for Health Research (CIHR) CHI 151135 [to S.P.M.], National Institutes of Health (NIH) NIH R01 NS046432 [to A.J.B.], and NIH P01 NS082330 [to D.X.]. The funders had no role in study design, data collection, analysis or interpretation, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
All authors made substantial contributions to the conception and design of the study, acquisition of data, or analysis and interpretation of data, drafted the article or revised it critically for important intellectual content, and approved the final version of the article to be published.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Kamino, D., Chau, V., Studholme, C. et al. Plasma cholesterol levels and brain development in preterm newborns. Pediatr Res 85, 299–304 (2019). https://doi.org/10.1038/s41390-018-0260-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-018-0260-0