Abstract
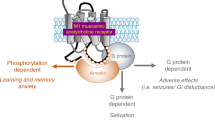
Highly selective positive allosteric modulators (PAMs) of the M1 subtype of muscarinic acetylcholine receptor have emerged as an exciting new approach for improving cognitive function in patients suffering from Alzheimer’s disease and schizophrenia. However, excessive activation of M1 is known to induce seizure activity and have actions in the prefrontal cortex (PFC) that could impair cognitive function. We now report a series of pharmacological, electrophysiological, and behavioral studies in which we find that recently reported M1 PAMs, PF-06764427 and MK-7622, have robust agonist activity in cell lines and agonist effects in the mouse PFC, and have the potential to overactivate the M1 receptor and disrupt PFC function. In contrast, structurally distinct M1 PAMs (VU0453595 and VU0550164) are devoid of agonist activity in cell lines and maintain activity dependence of M1 activation in the PFC. Consistent with the previously reported effect of PF-06764427, the ago-PAM MK-7622 induces severe behavioral convulsions in mice. In contrast, VU0453595 does not induce behavioral convulsions at doses well above those required for maximal efficacy in enhancing cognitive function. Furthermore, in contrast to the robust efficacy of VU0453595, the ago-PAM MK-7622 failed to improve novel object recognition, a rodent assay of cognitive function. These findings suggest that in vivo cognition-enhancing efficacy of M1 PAMs can be observed with PAMs lacking intrinsic agonist activity and that intrinsic agonist activity of M1 PAMs may contribute to adverse effects and reduced efficacy in improving cognitive function.
Similar content being viewed by others
Introduction
The M1 muscarinic acetylcholine receptor (mAChR) has garnered intense interest as a promising therapeutic target for the treatment of the cognitive disruptions in schizophrenia and Alzheimer’s disease (AD). Cholinergic signaling is disrupted in both schizophrenia and AD [1, 2], and a subset of schizophrenia patients display profound decreases in M1 expression in brain regions critical for cognition [3, 4]. Several compounds targeting the M1 receptor have entered clinical trials [5, 6] including a multicenter phase III clinical trial examining the effects of the M1/M4-preferring mAChR partial agonist xanomeline in patients suffering from AD [7] and a subsequent small clinical trial in patients with schizophrenia [8]. Unfortunately, xanomeline and other mAChR partial agonists exhibited dose-limiting adverse effects that are thought to be mediated by nonselective agonist activity and activation of peripheral M2 and M3 receptors [9]. While efforts are underway to reduce peripheral side effects of xanomeline by co-administration of a peripheral muscarinic receptor antagonist (ClinicalTrials.gov; identifier NCT02831231), it may also be possible to develop more selective activators of the M1 receptor.
Despite major investments in medicinal chemistry, previous efforts to develop highly selective orthosteric agonists of the M1 receptor have failed due to the highly conserved orthosteric acetylcholine binding site. To circumvent this problem, multiple research efforts shifted to developing compounds that act via allosteric sites on mAChRs, which are structurally distinct from the orthosteric binding site and may be less highly conserved among receptor subtypes. By targeting allosteric sites on mAChRs, we and others have identified highly subtype-selective positive allosteric modulators (PAMs) of the M1 receptor that avoid activation of other mAChR subtypes [10,11,12,13]. These M1 PAMs induce robust potentiation of M1-mediated responses in animal models and in brain tissue from schizophrenia patients in which the M1 receptor is down regulated [3]. Based on these exciting advances, three highly selective M1 PAMs, MK-7622, VU-319, and TAK-071, have been advanced to clinical testing for potential efficacy in enhancing cognitive function in patients with central nervous system (CNS) disorders (see ClinicalTrials.gov Identifiers NCT01852110, NCT03220295, and NCT02769065).
M1 PAM discovery programs have produced a structurally diverse range of M1 PAMs with distinct pharmacological properties, including differences in stimulus bias [14], and different levels of intrinsic agonist activity [10, 15, 16]. However, little is known about the impact of different modes of efficacy of M1 PAMs on in vivo activity. While M1 activation plays an integral role in regulation of cognitive function [17], overactivation of M1 can have effects that could impair cognition and induce severe adverse effects. For instance, while modest activation of M1 enhances synaptic plasticity in the hippocampus and prefrontal cortex (PFC) [18, 19], excessive M1 activation induces a disordered increase in pyramidal neuron activity [18, 20], and this may impair cognitive function [21, 22]. Additionally, strong activation of M1 may contribute to peripheral cholinergic adverse effects [15, 23] as well as induction of generalized behavioral convulsions [16, 24]. These studies raise the possibility that M1 PAMs that possess direct agonist activity could lead to overactivation of the receptor and have different in vivo effects than M1 PAMs devoid of agonist activity. However, for other receptors, PAMs have been identified that exert allosteric agonist activity in cell lines but not in native systems [25] and it is not known whether M1 PAMs will display allosteric agonist activity in CNS preparations. Lastly, the differences in in vivo effects of compounds lacking M1 agonist vs. possessing intrinsic agonist activity have not been evaluated.
We now report a series of studies in which we characterize two M1 PAMs that exhibit robust M1 agonist activity (MK-7622 and PF-06764427) and two structurally distinct M1 PAMs, VU0453595 [19] and VU0550164, that are devoid of agonist activity in cell lines. We then evaluate the physiological effects of these ago-PAMs and non ago-PAMs in the medial PFC (mPFC), a brain region important in cognition, and the potential cognitive and pro-convulsive effects in awake healthy rodents.
Materials and methods
Cell line and calcium mobilization assay
Chinese hamster ovary (CHO) cells stably expressing rat M1 were used in the calcium mobilization assay to examine the agonist and PAM activities of the M1-positive allosteric modulators. Cell culture and calcium flux assay conditions have been previously described [16]. Detailed methods are described in the Supplementary Methods.
Animals
All animal studies were approved by the Vanderbilt University Medical Center Institutional Animal Care and Use Committee and were conducted in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. Male C57BL6/J mice (Jackson laboratories) and M1 receptor knockout (KO) mice (with permission from J. Wess, National Institutes of Health–National Institute of Diabetes and Digestive and Kidney Diseases, Bethesda, MD) were used in electrophysiology and behavioral studies (6–10 weeks old). Mice were group housed 4–5 per cage, maintained on a 12 h light/dark cycle, and food and water were provided ad libitum. Adult male Sprague-Dawley rats weighing between 280 and 350 g (Envigo, Indianapolis, IN) were used in the behavioral studies. Rats were group housed 3 per cage and were maintained on a 12 h light/dark cycle with food and water ad libitum.
Behavioral manifestations of seizure activity
To evaluate induction of behavioral manifestation of seizure activity, C57Bl/6J mice received administration of vehicle or 1, 3, 10, 30, or 100 mg/kg M1 PAM. Compounds were formulated in 10% Tween 80 (pH 7.0) and injected intraperitoneally (n = 3). Animals were monitored continuously and scored for behavioral manifestations of seizure activity at 5, 10, 15, and 30 min, and 1 and 3 h. Behavioral manifestations of seizures were scored using a modified Racine scoring system [16, 26]. Detailed methods are described in the Supplementary Methods.
Extracellular field electrophysiology
Extracellular field potential recordings were performed with 6–10-week-old male C57BL6/J mice (Jackson Laboratories) using 400 µm coronal slices containing the prelimbic prefrontal cortex recorded from layer V and evoked electrically by a concentric bipolar stimulating electrode (200 µs duration, 0.05 Hz; inter-pulse interval of 50 ms) in the superficial layers II–III. Input–output curves were generated to determine the stimulus intensity that produced approximately 70% of the maximum field excitatory postsynaptic potential (fEPSP) slope before each experiment, which was then used as the baseline stimulation. All test compounds, with the exception of carbachol (CCh; Tocris Bioscience, Bristol, UK) which was diluted in H2O, were diluted to the appropriate concentrations in dimethyl sulfoxide (DMSO; <0.1% final) in artificial cerebrospinal fluid (ACSF) and applied to the bath for 20 min. Detailed methods are described in the Supplementary Methods.
Whole-cell electrophysiology
Whole-cell patch-clamp recordings were performed using coronal mPFC slices (300 µm) prepared from 6–10-week-old male C57BL6/J mice (Jackson Laboratories) according to the methods above using pipettes filled with an intracellular solution consisting of the following (in mM): 125 K-gluconate, 4 NaCl, 10 HEPES, 4 MgATP, 0.3 NaGTP, and 10 Tris-phosphocreatine, and had resistances ranging from 3 to 5 MΩ. Pyramidal neurons were visualized based on morphology and were further identified by their regular spiking pattern following depolarizing current injections induced by a series of 500 ms current steps (−150 pA to +100 pA) incremented in +25 pA performed in current clamp mode. Spontaneous EPSCs (sEPSC) were recorded at a holding potential of −70 mV (the reversal potential for GABAA channels). After a stable baseline was recorded for 5–10 min, test compounds were diluted to the appropriate concentrations in DMSO (<0.1% final) in ACSF and applied to the bath for 5 min using a peristaltic pump perfusion system. Cumulative probability plots were constructed using 2 min episodes of baseline and peak affect during drug add interevent interval values. Detailed methods are described in the Supplementary Methods.
Novel object recognition task
Rats were habituated for 10 min for 2 consecutive days in an empty novel object recognition arena consisting of dark-colored plexiglass box (40 × 64 × 33 cm3). On day 3, rats were administered vehicle (0.5% methylcellulose for MK-7622 and 20% b-cyclodextrin for VU0453595) or M1 PAM (0.3−10 mg/kg, per os (p.o.), 3 mL/kg, n = 11–12) and returned to their home cage for 90 min. Rats were then placed in the novel object recognition arena containing two identical objects for 10 min. Following the exposure period, rats were placed back into their home cages for 24 h. The rats were then returned to the arena in which one of the previously exposed (familiar) objects was replaced by a novel object and were video recorded for 5 min while they explored the two objects. Time spent exploring each object was scored by an observer blinded to the experimental conditions and the recognition index was calculated as [(time spent exploring novel object)−(time spent exploring familiar object)]/total time exploring objects.
Statistical analyses
For the novel object recognition task, groups were compared using a one-way analysis of variance (ANOVA), followed by Dunnett’s multiple comparison tests with the vehicle-treated rats as the control group. Changes in sEPSC frequency before and during drug add (peak effect) was compared using a paired t-test after data passed the Kohmogrov–Sminov normality test. For long-term depression (LTD) experiments where only two experimental conditions were compared, a paired t-test was performed to calculate statistical significance after data passed the Kohmogrov–Sminov normality test. For electrophysiological comparisons with more than one group, a one-way ANOVA was performed followed by Dunnett’s multiple comparison test. For all statistical comparisons, the critical p-value was considered to be 0.05. The numbers of animals to be used for each experiment outlined within the study were determined using a power calculation statistical analysis using the Power and Sample Size Calculation software program available at Vanderbilt University (Dupont and Plummer, PS Controlled Clinical Trials. 18:274 1997). Animal numbers are based on a power calculation using standard errors from published studies and previous experience to detect >20% difference for each outlined experiment with an 80% power (alpha = 0.05, power = 80%, delta = 0.2, sigma = 0.18).
Results
MK-7622 and PF-06764427 display robust agonist activity in an in vitro calcium mobilization assay
To assess the in vitro activity of the M1 PAMs used in this study, compounds were tested using CHO cells stably expressing the M1 receptor. The previously published M1 PAM PF-06764427 [10, 16] (Fig. 1a, left) and Merck’s MK-7622 [27] (Fig.1a, right) were evaluated for their ability to mobilize intracellular calcium (Ca2+) in the M1-CHO cells. The raw calcium traces (Fig. 1b) indicate that both PF-06764427 and MK-7622 induce robust increases in Ca2+ mobilization in the absence of an orthosteric mAChR agonist. PF-06764427 alone causes a concentration-dependent increase in the mobilization of intracellular Ca2+ (Ago half-maximal effective concentration (EC50) 610 nM ± 14, Fig. 1c). Interestingly, we report a similar degree of intracellular Ca2+ mobilization with MK-7622 alone (Ago EC50 2930 nM ± 95, Fig. 1d). Both PF-06764427 (PAM EC50 30 nM ± 3, Fig. 1e) and MK-7622 (PAM EC50 16 nM ± 4, Fig. 1f) act as potent and selective [10] (Supplemental Fig. 1) M1 PAMs in the presence of the orthosteric agonist ACh. Therefore, in addition to their PAM activity, both PF-06764427 and MK-7622 have significant intrinsic agonist activity in this cell-based Ca2+ mobilization assay.
PF-06764427 and MK-7622 display robust intrinsic agonist activity in M1-expressing CHO cells. a Structures of the M1 PAMs PF-06764427 (left) and MK-7622 (right). b Representative raw calcium traces following the addition of 10 μM PF-06764427 (black), MK-7622 (light gray), and the subsequent additions of EC20 and EC80 concentrations of acetylcholine (ACh) (dotted line). c Agonist concentration-response curves of rM1-CHO calcium mobilization assay for PF-06764427 and d MK-7622 in the absence of ACh. e PAM concentration-response curves of rM1-CHO calcium mobilization assay for PF-06764427 and f MK-7622 in the presence of an EC20 of ACh. Data represent mean ± S.E.M. from three independent experiments performed in triplicate
VU0453595 and VU0550164 act as highly selective PAMs lacking agonist activity in the M1 in vitro calcium mobilization assay
Previous studies suggest that overactivation of the M1 mAChR can induce cholinergic adverse effects [16, 23]. Based on these studies, we initiated an effort to optimize M1 PAMs that lack intrinsic agonist activity. We have previously reported characterization VU0453595 [19, 28] (Fig. 2a, left) and now disclose the novel M1 PAM VU0550164 (Fig. 2a, right). The raw Ca2+ traces (Fig. 2b) illustrate the lack of agonist activity of both VU0453595 and VU0550164 in the calcium mobilization assay even at high concentrations (>10 µM). As expected, VU0453595 had little to no effect on intracellular Ca2+ mobilization when applied alone (Fig. 2c). Similarly, the optimized M1 PAM VU0550164 lacks agonist activity (Fig. 2d). However, both VU0453595 (PAM EC50 2140 nM ± 436, Fig. 2e) and VU0550164 (PAM EC50 330 nM ± 44, Fig. 2f) can strongly potentiate an EC20 concentration of ACh in a concentration-dependent manner. These data reveal that at concentrations significantly above the PAM EC50, VU0453595 and VU0550164 lack agonist activity in this assay, thereby demonstrating that these compounds are highly selective [19] (Supplemental Fig. 1) PAMs devoid of agonist activity with respect to in vitro Ca2+ mobilization.
VU0453595 and VU0550164 lack significant agonist activity and are potent PAMs in M1-expressing CHO cells. a Structure of M1 PAM VU0453595 (left) and VU0550164 (right). b Representative raw calcium traces following the addition of 10 μM VU0453595 (black) and VU0550164 (light gray) and the subsequent additions of EC20 and EC80 concentrations of acetylcholine (ACh) (dotted line). c Agonist concentration-response curves of rM1-CHO calcium mobilization assay for VU0453595 and d VU0550164 in the absence of ACh. e PAM concentration-response curves of rM1-CHO calcium mobilization assay for VU0453595 and f VU0550164 in the presence of an EC20 of ACh. e Data represent mean ± S.E.M. from three independent experiments performed in triplicate
Ago-PAMs but not PAMs lacking agonist activity increase sEPSC in layer V mPFC pyramidal neurons
To test whether PAMs displaying agonist activity in cell-based assays would have agonist activity in a native system, we evaluated the various M1 PAMs in a series of whole-cell electrophysiology experiments in native brain tissue. Previously, we reported that activation of M1 can dramatically increase the activity of excitatory synaptic inputs onto layer V pyramidal cells in acute mPFC-containing brain slices [20]. We now report that bath application of M1 ago-PAM PF-06764427 (1 µM) induces a marked increase in frequency of spontaneous excitatory postsynaptic currents (sEPSCs) in mPFC layer V pyramidal cells (Fig. 3a, paired t-test, p < 0.05). In addition, the second M1 ago-PAM, MK-7622 (1 µM), also increased sEPSC frequency (Fig. 3b, paired t-test, p < 0.05). However, neither 10 µM VU0453595 (Fig. 3c) nor 10 µM VU0550164 (Fig. 3d) caused any significant change in sEPSC frequency (paired t-test, p > 0.05) at concentrations well above those required for maximal PAM activity. These results show that MK-7622 and PF-06764427 but not VU0453595 or VU0550164 have agonist activity in this native cortical preparation.
Ago-PAMs but not PAMs devoid of agonist activity increase sEPSC frequency in layer V mPFC neurons. a Whole-cell recordings from pyramidal neurons (regular spiking firing) clamped at −70 mV were performed in layer V of the prelimbic prefrontal mouse cortex. A sample trace of baseline (upper-trace) and during drug add (bottom-trace) and the cumulative probability of interevent intervals for a typical cell are shown (middle) during baseline and drug-add. Histogram summarizing the change in frequency of baseline to the drug peak effect (lower). Similar to 1 µM PF-06764427, bath application of b 1 µM MK-7622 produced a statistically significant increase in sEPSC frequency, in contrast to bath application of c 10 µM VU0453595 and d 10 µM VU0550164 which induced no significant change in sEPSC frequency. Scale bars denote 50 pA and 10 s. *p < 0.05, paired t-test
Ago-PAMs but not PAMs lacking agonist activity induce robust depression of fEPSP slopes in the mPFC
We next tested the various M1 PAMs in a more circuit-level brain slice electrophysiology assay by measuring changes in layer V fEPSPs evoked by electrical stimulation of afferents in layer II/III of the mPFC. Previously, we and others found that cholinergic agonists can induce an M1-dependent long-term depression (LTD) of fEPSP slope at this synapse [19, 29, 30]. Consistent with the effects on sEPSCs, bath application of 1 µM PF-06764427 (Fig. 4a) induces a significant LTD of fEPSP slope 46–50 min (shaded area) after drug washout compared to baseline (paired t-test, p < 0.05, Fig. 4b); this effect was absent in the presence of the selective M1 antagonist VU0255035 [31] (Supplemental Fig. 2A) as well as in M1-KO mice (Supplemental Fig. 2B). Quantification of LTD measured at 46–50 min after drug washout (shaded area) indicates a significant difference in the magnitude of LTD observed with application of 1 µM PF-06764427+10 µM VU0255035 as well as 1 µM PF-06764427 in M1-KO mice compared to 1 µM PF-06764427 alone (Supplemental Fig. 2C) (one-way ANOVA, p < 0.05).
Ago-PAMs but not PAMs devoid of agonist activity robustly depress fEPSP slopes recorded in layer V of the prelimbic mPFC evoked by electrical stimulation in layer II/III. a Time course graph showing that bath application of 1 µM PF-06764427 for 20 min leads to a robust long-term depression (LTD) of fEPSP slope. b Quantification of fEPSP slope 46–50 min following drug washout (shaded area) indicates a significant depression of fEPSP slope. / = 9 brain slice experiments. *P < 0.05, paired t-test. c Under similar conditions bath application of 1 µM MK-7622 for 20 min led to an acute depression followed by a LTD of fEPSPs measured 46–50 min following drug washout. d Quantification of fEPSP slope 46–50 min following drug washout (shaded area) indicates a significant depression of fEPSP slope; n = 9 brain slice experiments; ***p < 0.001, paired t-test. e Time course graph showing that bath application of 10 µM VU0453595 for 20 min led to no significant change in fEPSP slopes (f) measured 46–50 min following drug washout; n = 4 brain slice experiments; p > 0.05, paired t-test. g Similar to VU04553595, 10 µM VU0550164 failed to induce any significant change in fEPSP 46–50 min following drug washout (h); n = 7 brain slice recordings, p < 0.05. paired t-test. Time course data are expressed as mean ± S.E.M. Insert contains fEPSP sample traces during baseline (red) and 46–50 min following drug washout (black). Scale bar denotes 0.5 mV and 5 ms
Similarly, bath application of 1 µM MK-7622 for 20 min (Fig. 4c) induces a robust and statistically significant LTD measured at 46–50 min following drug washout (paired t-test, p < 0.001) (Fig. 4d). This LTD was absent in the presence of VU0255035 (Supplemental Fig. 2D) as well as in brain slices obtained from M1-KO mice (Supplemental Fig. 2E). Quantification of LTD (normalized fEPSP slopes) 46–50 min after drug washout (shaded area) indicates a significant difference in the magnitude of LTD observed with application of 1 µM MK-7622 + 10 µM VU0255035 as well as 1 µM MK-7622 in M1-KO mice compared to 1 µM MK-7622 alone (one-way ANOVA, p < 0.01) (Supplemental Fig. 1F).
In contrast to the effects of MK-7622 and PF-06764427, high concentrations of M1 PAMs VU0453595 and VU0550164 induce no change in fEPSP slope. As illustrated in Fig. 4e, f, bath application of 10 µM VU0453595, a concentration known to potentiate the LTD induced by a submaximal concentration of CCh [19], for 20 min did not significantly change fEPSP slope compared to baseline (p > 0.05). Furthermore, the second M1 PAM devoid of agonist activity in vitro, VU0550164 (10 µM), did not significantly change fEPSP slope compared to baseline when bath applied alone (p > 0.05) (Fig. 4g, h), but potentiated the response to 10 μM CCh (Supplemental Fig. 3A) and induced a robust LTD only in the presence of an orthosteric agonist (Supplemental Fig. 3B). Quantification of LTD indicates a significant difference in the magnitude of LTD observed with co-application of 10 µM VU0550164 + 10 µM CCh compared to 10 µM alone (p < 0.05) (Supplemental Fig. 3C). Taken together, these data demonstrate that MK-7622 and PF-06764427 display strong intrinsic M1 agonist activity in this cortical brain slice electrophysiological assay, in contrast to VU0453595 and VU0550164 which lack agonist activity and maintain activity dependence of M1 activation in these native brain tissue assays.
MK-7622, but not VU0453595, induces behavioral convulsions in rodents
In light of our finding that both PF-06764427 and MK-7622 have robust allosteric agonist activity in both cell line and native tissue assays, we hypothesized that MK-7622 would induce behavioral convulsions in a manner similar to those observed with PF-06764427 [16]. Therefore, we performed a dose-escalation study in mice to assess seizure liability of the M1 ago-PAM MK-7622. Consistent with our previous study with PF-06764427, 30 and 100 mg/kg MK-7622 induces robust convulsions that reached stage 5 on the modified Racine scale [16, 26, 32] in wild-type but not M1 KO mice (Fig. 5a). While the M1 PAM VU0550164 does not have suitable pharmacokinetic properties for systemic administration, VU0453595 has excellent properties for use in in vivo studies [19]. Interestingly, VU0453595 induces no gross changes in behavior even at doses up to 100 mg/kg (Fig. 5a), well above doses that enhance cognition in rodents [19]. Collectively, these findings suggest that M1 ago-PAMs, such as MK-7622 and PF-06764427, induce behavioral convulsions that are not observed with PAMs lacking agonist activity such as VU0453595.
MK-7622 induces robust behavioral convulsions, and lacks efficacy in enhancing rodent cognition. a C57Bl6/j mice were administered 3, 10, 30, and 100 mg/kg MK-7622, and behavioral convulsions were measured for 3 h using the modified Racine scale (0−5). M1-KO mice exhibit no behavioral convulsions, suggesting that MK-7622 induces behavioral convulsions in a M1-dependent fashion. Compounds were formulated in 10% Tween 80 and delivered intraperitoneally. Data represent mean ± S.E.M. (n = 3 mice per dose). This is in contrast to VU0453595, which displays no overt adverse effects at concentrations up to 100 mg/kg (n = 3 mice). b Schematic of the rodent Novel Object Recognition paradigm. c Pretreatment with 1, 3, and 10 mg/kg of MK-7622 90 min before the familiar phase failed to significantly enhance object recognition assessed 24 h later (p = 0.9110, n = 11–12 rats per group). d Under similar conditions for MK-7622, VU0453595 dose-dependently enhances object recognition in rats. Pretreatment with 1, 3, and 10 mg/kg VU0453595 90 min before the familiar phase enhanced object recognition memory assessed 24 h later (p = 0.008, n = 11–12 rats per group). Data are expressed as mean ± S.E.M.; *p < 0.05
VU0453595 but not MK-7622 can enhance rodent object recognition
While M1 ago-PAMs such as MK-7622 and PF-06764427 do not induce overt seizures at lower doses, it is possible that overactivation of M1 with ago-PAMs such as MK-7622 and PF-06764427 could disrupt cortical function and reduce efficacy of these compounds in enhancing cognitive function. Therefore, we performed a series of studies to evaluate the effects of the M1 ago-PAM MK-7622 and the PAM VU0453595 in an established model of recognition memory in healthy adult rats (Fig. 5b). Rats were chosen for these studies because they do not display overt behavioral convulsions in response to administration of M1 ago-PAMs and, for these studies, we used doses below those shown to induce behavioral convulsions in mice.
Interestingly, MK-7622 (1, 3, and 10 mg/kg) did not significantly improve performance in the novel object recognition task (p = 0.9110, one-way ANOVA) (Fig. 5c). In contrast, VU0453595, which lacks agonist activity, induces a robust improvement in recognition memory in healthy adult rats assessed using the novel object recognition task. At 1, 3, and 10 mg/kg, VU0453595 enhances object recognition as indicated by a significant increase in the recognition index (Dunnett’s multiple comparison test: p = 0.0008) (Fig. 5d). Taken together, these data suggest that ago-PAMs and PAMs devoid of agonist activity can be differentiated in multiple cell-based, native tissue, and in vivo assays.
Discussion
We report that PF-06764427 and MK-7622 exert considerable agonist activity in inducing calcium mobilization in M1-expressing cell lines, and in two cortical brain slice electrophysiology assays. Conceptually, while prototypical PAMs only potentiate responses to an orthosteric agonist, these “ago-PAMs” potentiate responses to orthosteric agonist but also directly activate M1 when added alone. Importantly, ago-PAMs are also distinct from agonists, in that pure agonists do not potentiate responses to ACh but only act by directly activating the receptor. The current findings suggest that the presence of agonist activity when assessing M1 PAMs in cell lines translates into robust ago-PAM activity in native preparations. In contrast, two structurally distinct M1 PAMs optimized to eliminate agonist activity in cell lines, VU0453595 and VU0550164, have no significant agonist activity in mPFC-containing brain slices. Thus, unlike ago-PAMs, VU0453595 and VU0550164 maintain dependence on ACh for receptor activation and would be expected to maintain activity dependence of M1 activation. Furthermore, similar to PF-06764427 [16], the ago-PAM MK-7622, but not VU0453595, induces robust behavioral convulsions in adult mice that were absent in M1-KO mice, suggesting that overactivation of M1 is responsible for the MK-7622-induced behavioral convulsions. Finally, VU0453595 but not MK-7622, has robust efficacy in improving object recognition memory in rats. Consistent with a more limited therapeutic window of M1 ago-PAMs in this assay, we previously reported that another M1 ago-PAM, PF-06764427, fails to improve performance in this cognition assay [16].
The lack of efficacy of PF-06764427 and MK-7622 in improving recognition memory in unimpaired rodents is interesting in light of previous studies showing that M1 PAMs within these structural classes can reverse scopolamine-induced deficits in rodent and monkey working memory [10, 33,34,35, 44]. Thus, it is possible that the efficacy observed with M1 PAMs will be impacted by the specific assay being used. It is important to note that PF-06764427 and other M1 ago-PAMs in the MK-7622 structural class have been shown to exhibit strong negative cooperativity with scopolamine binding to the orthosteric site, and are therefore able to inhibit [3H]-N-methyl-scopoloamine binding to this site [16, 36,37,38]. This ability of some M1 PAMs to inhibit scopolamine binding may confound studies using scopolamine to impair cognitive function and may reflect the negative cooperativity between M1 ago-PAM and scopolamine binding. In future studies, it will be important to fully evaluate M1 ago-PAMs and PAMs devoid of agonist activity across a broader range of models, especially in relevant disease models that do not rely on disruption of cognitive function with scopolamine or other mAChR orthosteric antagonists in order to determine whether M1 PAMs lacking agonist activity have the superior efficacy and safety across multiple models.
The data reported here suggest that it will be important to optimize M1 PAMs that lack intrinsic agonist activity in order to minimalize risk of adverse effects and to maximize the M1 PAM efficacy in improving cognitive function in diseases such as schizophrenia and AD [19, 20]. This is reminiscent of previous studies demonstrating that the presence of allosteric agonist activity also leads to severe adverse effect liability for select PAMs for the mGlu5 subtype of metabotropic glutamate (mGlu) receptors [26, 32]. This liability is minimized with PAMs lacking agonist activity, and therefore suggests that, for certain receptors and channels [39, 40], it is critical to optimize PAMs that maintain a strict dependence of receptor signaling on activation of receptors by endogenous transmitters. This is directly analogous to the favorable safety profile of positive allosteric modulators of GABAA receptors in patients [41]. The need to optimize compounds that maintain a strict dependence on activity of presynaptic afferents is likely to be especially important for any receptor subtype for which excessive activation is known to lead to severe adverse effects or disruptive effects on CNS function. Even in instances where potential adverse effect liability is not as severe as is the case for M1, mGlu5, and ligand-gated channels, optimizing PAMs lacking agonist activity, as opposed to ago-PAMs, may provide a critical advantage in maintaining the high degree of spatial and temporal signaling that is inherent in the activity of virtually all brain circuits. Thus, the current studies suggest that, by bypassing the need for orthosteric M1 activation by cholinergic afferents, ago-PAMs induce a profound LTD and persistent increase in spontaneous excitatory synaptic activity in the mPFC, which could disrupt mPFC-dependent cognitive function. However, unlike agonists or ago-PAMs, PAMs devoid of agonist activity remain effectively silent until the endogenous neurotransmitter is released and subsequently binds to the receptor’s orthosteric site.
In conclusion, these findings suggest that the in vivo cognition-enhancing efficacy of M1 PAMs can be observed with PAMs lacking agonist activity and that intrinsic agonist activity of M1 PAMs may contribute to adverse effects and result in reduced efficacy in improving cognitive function. These findings also suggest that it is prudent to use a combination of physiology (e.g., brain slice electrophysiology) and cognition models in addition to cell-based assays (e.g., intracellular Ca2+ mobilization) to fully characterize the pharmacological properties of an M1 allosteric ligand. While in vitro assays are ideal to screen compounds and achieve a first pass look at a ligand’s pharmacological properties, cell-based assays often fail to recapitulate the exact pathways found in native tissue. Lastly, discovery and characterization of functional M1 receptors expressed intracellularly [42] as well as allosteric ligands that display “signal bias” [14] further highlights the need to fully assess activities of allosteric modulators in native preparations. While none of the four compounds examined display known M1 signal bias (data not shown), full understanding of the pharmacological properties responsible for in vivo efficacy of other G protein–coupled receptor allosteric ligands, such as mGlu5 PAMs that display stimulus bias, have provided proof of concept that adverse effect liability can be avoided and in vivo efficacy remain intact [43]. However, little is known about the impact of M1 PAM signal bias on in vivo activity. Future studies are necessary to develop a better understanding of the mechanisms by which M1 PAMs enhance cognitive function and whether allosteric modulators that display stimulus bias can be used to selectively modulate specific aspects of M1 signaling and in vivo activity. Lastly, while these studies shed light on the different properties of structurally distinct M1 PAMs, recent literature has shown that sub-efficacious doses of M1 PAMs co-administered with sub-efficacious concentrations of cholinesterase inhibitors [44, 45] or antipsychotics [46] can yield efficacy in animal models. However, future studies are necessary to fully evaluate the clinical implications M1 PAMs used in conjunction with currently approved neuropsychiatric medications.
References
Davies P, Maloney AJ. Selective loss of central cholinergic neurons in Alzheimer’s disease. Lancet. 1976;2:1403.
Sarter M, Lustig C, Taylor SF. Cholinergic contributions to the cognitive symptoms of schizophrenia and the viability of cholinergic treatments. Neuropharmacology. 2012;62:1544–53.
Dean B, Hopper S, Conn PJ, Scarr E. Changes in BQCA allosteric modulation of [(3)H]NMS binding to human cortex within schizophrenia and by divalent cations. Neuropsychopharmacology. 2016;41:1620–8.
Scarr E, Udawela M, Thomas EA, Dean B.Changed gene expression in subjects with schizophrenia and low cortical muscarinic M1 receptors predicts disrupted upstream pathways interacting with that receptor. Mol Psychiatry. 2016;23:295–303.
Foster DJ, Conn PJ. Allosteric modulation of GPCRs: new insights and potential utility for treatment of schizophrenia and other CNS disorders. Neuron. 2017;94:431–46.
Langmead CJ, Watson J, Reavill C. Muscarinic acetylcholine receptors as CNS drug targets. Pharmacol Ther. 2008;117:232–43.
Bodick NC, Offen WW, Levey AI, Cutler NR, Gauthier SG, Satlin A, et al. Effects of xanomeline, a selective muscarinic receptor agonist, on cognitive function and behavioral symptoms in Alzheimer disease. Arch Neurol. 1997;54:465–73.
Shekhar A, Potter WZ, Lightfoot J, Lienemann J, Dubé S, Mallinckrodt C, et al. Selective muscarinic receptor agonist xanomeline as a novel treatment approach for schizophrenia. Am J Psychiatry. 2008;165:1033–9.
Bymaster FP, Carter PA, Yamada M, Gomeza J, Wess J, Hamilton SE, et al. Role of specific muscarinic receptor subtypes in cholinergic parasympathomimetic responses, in vivo phosphoinositide hydrolysis, and pilocarpine-induced seizure activity. Eur J Neurosci. 2003;17:1403–10.
Davoren JE, O’Neil SV, Anderson DP, Brodney MA, Chenard L, Dlugolenski K, et al. Design and optimization of selective azaindole amide M1 positive allosteric modulators. Bioorg Med Chem Lett. 2016;26:650–5.
Ma L, Seager MA, Wittmann M, Jacobson M, Bickel D, Burno M, et al. Selective activation of the M1 muscarinic acetylcholine receptor achieved by allosteric potentiation. Proc Natl Acad Sci USA. 2009;106:15950–5.
Mistry SN, Jörg M, Lim H, Vinh NB, Sexton PM, Capuano B, et al. 4-Phenylpyridin-2-one derivatives: a novel class of positive allosteric modulator of the M1 muscarinic acetylcholine receptor. J Med Chem. 2016;59:388–409.
Panarese JD, Cho HP, Adams JJ, Nance KD, Garcia-Barrantes PM, Chang S, et al. Further optimization of the M1 PAM VU0453595: Discovery of novel heterobicyclic core motifs with improved CNS penetration. Bioorg Med Chem Lett. 2016;26:3822–5.
Marlo JE, Niswender CM, Days EL, Bridges TM, Xiang Y, Rodriguez AL, et al. Discovery and characterization of novel allosteric potentiators of M1 muscarinic receptors reveals multiple modes of activity. Mol Pharmacol. 2009;75:577–88.
Davoren JE, Lee C-W, Garnsey M, Brodney MA, Cordes J, Dlugolenski K, et al. Discovery of the potent and selective M1 PAM-agonist N-[(3R,4S)-3-Hydroxytetrahydro-2H-pyran-4-yl]-5-methyl-4-[4-(1,3-thiazol-4-yl)benzyl]pyridine-2-carboxamide (PF-06767832): evaluation of efficacy and cholinergic side effects. J Med Chem. 2016;59:6313–28.
Rook JM, Abe M, Cho HP, Nance KD, Luscombe VB, Adams JJ, et al. Diverse effects on M1 signaling and adverse effect liability within a series of M1 Ago-PAMs. ACS Chem Neurosci. 2017;8:866–83.
Gould RW, Dencker D, Grannan M, Bubser M, Zhan X, Wess J, et al. Role for the M1 muscarinic acetylcholine receptor in top-down cognitive processing using a touchscreen visual discrimination task in mice. ACS Chem Neurosci. 2015;6:1683–95.
Digby GJ, Noetzel MJ, Bubser M, Utley TJ, Walker AG, Byun NE, et al. Novel allosteric agonists of M1 muscarinic acetylcholine receptors induce brain region-specific responses that correspond with behavioral effects in animal models. J Neurosci. 2012;32:8532–44.
Ghoshal A, Rook JM, Dickerson JW, Roop GN, Morrison RD, Jalan-Sakrikar N, et al. Potentiation of M1 muscarinic receptor reverses plasticity deficits and negative and cognitive symptoms in a schizophrenia mouse model. Neuropsychopharmacology. 2016;41:598–610.
Shirey JK, Brady AE, Jones PJ, Davis AA, Bridges TM, Kennedy JP, et al. A selective allosteric potentiator of the M1 muscarinic acetylcholine receptor increases activity of medial prefrontal cortical neurons and restores impairments in reversal learning. J Neurosci. 2009;29:14271–86.
Dong Y, Nasif FJ, Tsui JJ, Ju WY, Cooper DC, Hu X-T, et al. Cocaine-induced plasticity of intrinsic membrane properties in prefrontal cortex pyramidal neurons: adaptations in potassium currents. J Neurosci. 2005;25:936–40.
Klenowski PM, Fogarty MJ, Shariff M, Belmer A, Bellingham MC, Bartlett SE. Increased synaptic excitation and abnormal dendritic structure of prefrontal cortex layer V pyramidal neurons following prolonged binge-like consumption of ethanol. eNeuro. 2016;3:pii: ENEURO.0248-16.2016
Alt A, Pendri A, Bertekap RL, Li G, Benitex Y, Nophsker M, et al. Evidence for classical cholinergic toxicity associated with selective activation of M1 muscarinic receptors. J Pharmacol Exp Ther. 2016;356:293–304.
Cruickshank JW, Brudzynski SM, McLachlan RS. Involvement of M1 muscarinic receptors in the initiation of cholinergically induced epileptic seizures in the rat brain. Brain Res. 1994;643:125–9.
Noetzel MJ, Rook JM, Vinson PN, Cho HP, Days E, Zhou Y, et al. Functional impact of allosteric agonist activity of selective positive allosteric modulators of metabotropic glutamate receptor subtype 5 in regulating central nervous system function. Mol Pharmacol. 2012;81:120–33.
Rook JM, Noetzel MJ, Pouliot WA, Bridges TM, Vinson PN, Cho HP, et al. Unique signaling profiles of positive allosteric modulators of metabotropic glutamate receptor subtype 5 determine differences in in vivo activity. Biol Psychiatry. 2013;73:501–9.
Beshore D (2017). Targeting positive allosteric modulators of the M1 muscarinic receptor: identification of MK-7622. MEDI: Division of Medicinal Chemistry.
Lv X, Dickerson JW, Rook JM, Lindsley CW, Conn PJ, Xiang Z. M1 muscarinic activation induces long-lasting increase in intrinsic excitability of striatal projection neurons. Neuropharmacology. 2017;118:209–22.
Grannan MD, Mielnik CA, Moran SP, Gould RW, Ball J, Lu Z, et al. Prefrontal cortex-mediated impairments in a genetic model of NMDA receptor hypofunction are reversed by the novel M1 PAM VU6004256. ACS Chem Neurosci. 2016;7:1706–16.
Martin HGS, Bernabeu A, Lassalle O, Bouille C, Beurrier C, Pelissier-Alicot A-L, et al. Endocannabinoids mediate muscarinic acetylcholine receptor-dependent long-term depression in the adult medial prefrontal cortex. Front Cell Neurosci. 2015;9:457.
Sheffler DJ, Williams R, Bridges TM, Xiang Z, Kane AS, Byun NE, et al. A novel selective muscarinic acetylcholine receptor subtype 1 antagonist reduces seizures without impairing hippocampus-dependent learning. Mol Pharmacol. 2009;76:356–68.
Bridges TM, Rook JM, Noetzel MJ, Morrison RD, Zhou Y, Gogliotti RD, et al. Biotransformation of a novel positive allosteric modulator of metabotropic glutamate receptor subtype 5 contributes to seizure-like adverse events in rats involving a receptor agonism-dependent mechanism. Drug Metab Dispos. 2013;41:1703–14.
Uslaner JM, Eddins D, Puri V, Cannon CE, Sutcliffe J, Chew CS, et al. The muscarinic M1 receptor positive allosteric modulator PQCA improves cognitive measures in rat, cynomolgus macaque, and rhesus macaque. Psychopharmacology (Berl). 2013;225:21–30.
Chambon C, Jatzke C, Wegener N, Gravius A, Danysz W. Using cholinergic M1 receptor positive allosteric modulators to improve memory via enhancement of brain cholinergic communication. Eur J Pharmacol. 2012;697:73–80.
Chambon C, Wegener N, Gravius A, Danysz W. A new automated method to assess the rat recognition memory: validation of the method. Behav Brain Res. 2011;222:151–7.
Daval SB, Kellenberger E, Bonnet D, Utard V, Galzi J-L, Ilien B. Exploration of the orthosteric/allosteric interface in human M1 muscarinic receptors by bitopic fluorescent ligands. Mol Pharmacol. 2013;84:71–85.
Keov P, López L, Devine SM, Valant C, Lane JR, Scammells PJ, et al. Molecular mechanisms of bitopic ligand engagement with the M1 muscarinic acetylcholine receptor. J Biol Chem. 2014;289:23817–37.
Valant C, Gregory KJ, Hall NE, Scammells PJ, Lew MJ, Sexton PM, et al. A novel mechanism of G protein-coupled receptor functional selectivity. Muscarinic partial agonist McN-A-343 as a bitopic orthosteric/allosteric ligand. J Biol Chem. 2008;283:29312–21.
Hackos DH, Lupardus PJ, Grand T, Chen Y, Wang T-M, Reynen P, et al. Positive allosteric modulators of GluN2A-containing NMDARs with distinct modes of action and impacts on circuit function. Neuron. 2016;89:983–99.
Watterson LR, Olive MF. Are AMPA receptor positive allosteric modulators potential pharmacotherapeutics for addiction? Pharmceuticals (Basel). 2013;7:29–45.
Möhler H, Fritschy JM, Rudolph U. A new benzodiazepine pharmacology. J Pharmacol Exp Ther. 2002;300:2–8.
Anisuzzaman ASM, Uwada J, Masuoka T, Yoshiki H, Nishio M, Ikegaya Y, et al. Novel contribution of cell surface and intracellular M1-muscarinic acetylcholine receptors to synaptic plasticity in hippocampus. J Neurochem. 2013;126:360–71.
Rook JM, Xiang Z, Lv X, Ghoshal A, Dickerson JW, Bridges TM, et al. Biased mGlu5-positive allosteric modulators provide in vivo efficacy without potentiating mGlu5 modulation of NMDAR currents. Neuron. 2015;86:1029–40.
Lange HS, Cannon CE, Drott JT, Kuduk SD, Uslaner JM. The M1 muscarinic positive allosteric modulator PQCA improves performance on translatable tests of memory and attention in rhesus monkeys. J Pharmacol Exp Ther. 2015;355:442–50.
Vardigan JD, Cannon CE, Puri V, Dancho M, Koser A, Wittmann M, et al. Improved cognition without adverse effects: novel M1 muscarinic potentiator compares favorably to donepezil and xanomeline in rhesus monkey. Psychopharmacology (Berl). 2015;232:1859–66.
Choy KHC, Shackleford DM, Malone DT, Mistry SN, Patil RT, Scammells PJ, et al. Positive allosteric modulation of the muscarinic M1 receptor improves efficacy of antipsychotics in mouse glutamatergic deficit models of behavior. J Pharmacol Exp Ther. 2016;359:354–65.
Acknowledgements
We would like to thank the Dan Marino foundation for their generous support of the Clinical Neuroscience Scholars program at Vanderbilt University and to thank Weimin Peng for excellent assistance in maintaining colonies and genotyping of transgenic mice. Behavioral experiments were performed in part through the use of the Murine Neurobehavior Core lab at the Vanderbilt University Medical Center. We would also like to thank J Wess for generously sharing the M1 KO mice. This work was supported by NIH F31MH114368 (Moran), NIH T32 MH 64913-14 (Moran), U01 MH087965 (Conn), R01 MH062646 (Conn), R01 MH073676 (Conn) and R01 MH082867 (Lindsley), and CIHR DFS146189 (Maksymetz). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. The work of JMR has been funded by the NIH, Alzheimer’s Drug Discovery Foundation, Harrington Discovery Institute, Thome Alzheimer’s Disease Drug Discovery Foundation, and Brain & Behavior Research Foundation. The work of CWL has been funded by the NIH, Johnson and Johnson, Bristol-Myers Squibb, AstraZeneca, Michael J. Fox Foundation, as well as Seaside Therapeutics. PJC has been funded by NIH, Michael J. Fox Foundation, Dystonia Medical Research Foundation, CHDI Foundation and others. The work of CMN has been funded by the NIH, rettsyndrome.org, Autism Speaks, Johnson and Johnson, Bristol-Myers Squibb, AstraZeneca, Michael J. Fox Foundation, as well as Seaside Therapeutics.
Author contributions
SPM, JMR, ZX, CWL, CMN, and PJC designed experiments; SPM, HPC, DHR, JM, XL, JWD, CAD, and DWE performed experiments; SPM, JM, JWD, JMR, and PJC wrote the manuscript with input from all the authors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
JMR is an inventor on patents that protect different classes of muscarinic receptor allosteric modulators. CWL has consulted for AbbVie and received compensation. He is an inventor on patents that protect different classes of metabotropic glutamate and muscarinic receptor allosteric modulators. Over the past three years, PJC has served on the Scientific Advisory Boards for Michael J. Fox Foundation, Stanley Center for Psychiatric Research Broad Institute (MIT/Harvard), Karuna Pharmaceuticals, Lieber Institute for Brain Development, Clinical Mechanism (POCM) and Proof of Concept (POC) Consortium, and Neurobiology Foundation for Schizophrenia and Bipolar Disorder. He is an inventor on patents that protect different classes of metabotropic glutamate and muscarinic receptor allosteric modulators. CMN is an inventor on patents that protect different classes of metabotropic glutamate and muscarinic receptor allosteric modulators. DWE is an inventor on patents that protect different classes of metabotropic glutamate and muscarinic allosteric modulators. The remaining authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Moran, S.P., Dickerson, J.W., Cho, H.P. et al. M1-positive allosteric modulators lacking agonist activity provide the optimal profile for enhancing cognition. Neuropsychopharmacol 43, 1763–1771 (2018). https://doi.org/10.1038/s41386-018-0033-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41386-018-0033-9
This article is cited by
-
Acetylcholine deficiency disrupts extratelencephalic projection neurons in the prefrontal cortex in a mouse model of Alzheimer’s disease
Nature Communications (2022)
-
Clinical and Preclinical Evidence for M1 Muscarinic Acetylcholine Receptor Potentiation as a Therapeutic Approach for Rett Syndrome
Neurotherapeutics (2022)
-
Beneficial effects of levetiracetam in streptozotocin-induced rat model of Alzheimer’s disease
Metabolic Brain Disease (2022)
-
G-protein coupled receptor, PI3K and Rho signaling pathways regulate the cascades of Tau and amyloid-β in Alzheimer’s disease
Molecular Biomedicine (2021)
-
Leveraging bias to your advantage
Nature Chemical Biology (2020)