Abstract
The Prosigna® assay is a United States Food and Drug Administration (US-FDA) cleared molecular test for prognostic use in hormone receptor-positive stage I/II breast cancer in postmenopausal women. We analyzed histopathologic features of 79 cases with Prosigna® assay results and found a significant correlation between tumor size, grade, and Ki-67 labeling index with Prosigna® score (0–40, 41–60, and 61–100) and Prosigna® risk categories. Since the Prosigna® risk stratification is influenced by lymph node status, we designed an index that included lymph node status and the two most correlated variables (size and Ki-67 labeling index). This was termed the size, nodal, and Ki-67 (SiNK™) index and is calculated as follows: (size in mm) + (pN × 10) + (Ki-67 labeling index). The SiNK™ index was divided into ≤40 and >40 to test its prognostic significance in a well-characterized dataset of 106 ER+/HER2-negative stage I–II invasive breast cancers treated with standard multi-modality therapy with long term follow-up (average 101 months follow-up). Patients with SiNK™ ≤40 showed significantly improved distant recurrence-free survival (96% distant recurrence-free survival in SiNK™ ≤40 compared to 81% in SiNK™ >40; log-rank test p value: 0.0027). SiNK™ provides strong prognostic information in ERo+/HER2-negative breast cancers. SiNK™ index is simple to calculate using data from routine pathology reports. This should be further evaluated in larger datasets.
Similar content being viewed by others
Introduction
Commercial multigene expression assays (such as Oncotype DX®, MammaPrint®, EndoPredict®, Breast Cancer Index®, and Prosigna®) are now routinely performed for breast cancer management. All of these assays were initially devised for prognostic use, but are now routinely used for making therapy decisions in estrogen receptor (ER) positive breast cancers [1,2,3,4,5,6,7]. Low-risk patients are spared chemotherapy, while high-risk patients are offered chemotherapy with the hope of reducing the risk of recurrence and improving survival [8]. This approach seems to have merit, but the principle does not always hold as there are many patients with a high risk of recurrence whose risk cannot be always minimized with chemotherapy (large tumor size, multiple positive nodes, but low tumor grade with strong hormone receptor expression, and low proliferation). With routine usage, it appears that although these tests have both prognostic and predictive value, they are not interchangeable and some tests are more predictive, and while some others are more prognostic [9,10,11,12,13]. Prosigna® is a second-generation multigene expression assay, which comprises of 50 discriminator genes and 8 controls, popularly known as prediction analysis of microarray 50 (PAM50) gene signature [14]. This test has been cleared by the United States Food and Drug Administration (US-FDA) for prognostic use in hormone receptor-positive early-stage breast cancer in postmenopausal women. The Prosigna® score is derived from the correlation of information obtained from the PAM50 gene expression profiling, as well as proliferation and the pathologic tumor size. The risk of recurrence (ROR) at 10 years is classified as low (<5% risk, scores 0–40), intermediate (~10% risk, scores 41–60), and high (>15%, scores 61–100) in node-negative patients. The ROR is low (~5% risk, scores 0–40) and high (~25%, scores 41–100) in node-positive patients [15]. Given the inclusion of tumor size and nodal status in the classification of recurrence risk, Prosigna® test is a strong prognostic assay while the predictive value has not been widely tested. In the past, we have devised and demonstrated simple algorithmic measures using morphologic and immunohistochemical characteristics like Magee Equations™/Magee Decision Algorithm™ to be of significant clinical utility which can be used in lieu of expensive molecular assays like oncotype DX [16,17,18]. To the best of our knowledge, no prior studies have compared the clinical–pathologic and immunohistochemical parameters with the Prosigna® assay. Our current study was undertaken to study histopathologic features that correlate with Prosigna® scores. Subsequently, these features were used to devise a simple-to-use prognostic tool for estimating breast cancer prognosis.
Materials and methods
A search for cases with Prosigna® testing requests at our institution resulted in 79 cases. All cases were ER-positive and HER2-negative invasive breast carcinomas diagnosed between late 2014 to early 2018. Clinical and pathologic information was obtained from the clinical and laboratory information systems. The clinical–pathologic features of each case were recorded and analyzed for its association with the Prosigna® scores categories, 0–40, 41–60, and 61–100. The variables found to be significant in univariable analysis were taken to create an index with similar score categories as the Prosigna® test. The concordance between Prosigna® score categories and the resulting index categories (≤40, >40–60, >60) was evaluated. The resulting index was then used as a binary classifier (≤40 and >40) and tested on a separate set of 106 well-characterized ER+/HER2-negative American Joint Committee on Cancer stage I and II invasive breast carcinomas treated with routine multi-modality therapy with long term follow-up (validation set). The database used for validation is from a prior study where ER/PR and HER2 data was taken from pathology reports and Ki-67 was performed and assessed using semiautomated image analysis system as previously described [19]. Distant recurrence-free survival (DRFS) and breast cancer-specific survival (BCSS) for the validation set cases was analyzed via Kaplan–Meier curves with respect to the newly developed index. DRFS was defined as the time from diagnosis to first distant recurrence or the date of last contact. BCSS was defined as the time from diagnosis to death due to breast cancer or the date of last contact. For comparison of means, independent sample t tests were performed. Univariable analysis was performed using χ2 and Fisher exact tests to compare the differences in percentages between groups. A P value < 0.05 was considered significant. Log-rank test was used to compare Kaplan–Meier curves. Statistical analysis was performed using Prism GraphPad software, version 8.3.0, San Diego, CA.
Results
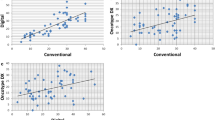
Univariable analysis of the clinical–pathologic features of 79 cases with Prosigna® test results showed a strong association with known clinical–pathologic prognostic factors. Table 1 highlights the significant correlation of Prosigna® score categories with tumor size, grade, and Ki-67 labeling index (p value: <0.05).
Although no association was identified between Prosigna® scores and lymph node status, the final Prosigna® risk categories do take into account the tumor lymph node status. Therefore, lymph node status was taken into consideration along with the two most significantly correlated features (Ki-67 labeling index and tumor size) to create the size, nodal, Ki-67, or the SiNK™ index. SiNK™ index is calculated by adding the tumor size in millimeters to the pathologic nodal status (pN) × 10 and the Ki-67 labeling index. The score can theoretically range from <1 to over 100. Since it is modeled after the Prosigna® test scores, the SiNK™ index was categorized similarly to Prosigna® score categories (≤40, >40–60, >60) and compared to Prosigna® test scores. The comparison is shown in Table 2. The overall concordance between Prosigna® score and SiNK™ index categories was 71% (56 of 79), with 2-step discordance rate of only 2.5% (see Table 2). When both Prosigna® score and SiNK™ index are evaluated using a binary cutoff value (≤40 and >40), the concordance increased to 82% (Table 3). No deaths occurred due to breast cancer in this test development cohort, but four distant recurrences were recorded (average follow-up of 33 months), two in lymph node-positive, and two in lymph node-negative patients. Three patients who recurred had tumors characterized as Prosigna® high risk and one as Prosigna® low risk. However, all recurrences happened in cases with SiNK™ >40 group (4 of 38 or 10.5%) and none in the SiNK™ ≤40 group (0 of 41; p value: 0.0491). Therefore, the binary values (≤40 and >40) were used to test the prognostic significance in the validation set. The patient and tumor characteristics of the 106 cases used for validation are provided in Table 4. SiNK™ indices were calculated, 75 patients (71%) had SiNK™ index ≤40 and 31 (29%) had SiNK™ index >40. The average follow-up for these cases was 101 months. Patients with SiNK™ index ≤40 experienced 4% distant recurrence compared to 19% distant recurrence in the group with SiNK™ index >40. The BCSS was 99% for patients with SiNK index ≤40 compared to 97% for the group with SiNK >40. DRFS and BCSS assessed by Kaplan–Meier curves are shown in Fig. 1. Significantly improved DRFS was noted in patients with SiNK™ ≤40. The BCSS was excellent for all patients.
The significance of the SiNK™ index was also evaluated separately in the lymph node-negative (n = 76) and lymph node-positive (n = 30) groups. Statistically significant improved DRFS for SiNK™ ≤40 was noted but BCSS was not significantly different between SiNK™ ≤40 and >40 in the lymph node-negative group (Fig. 2a, b). Although more distant recurrences were noted for patients with SiNK™ >40 in the lymph node-positive group (3 of 15 or 20% in SiNK™ >40 versus 1 of 15 or 7% in SiNK™ <40), the difference in DRFS was not statistically significant (Fig. 2c). The BCSS was not significantly different based on SiNK™ in lymph node-positive patients (Fig. 2d).
Discussion
In this genomic/molecular era, multigene commercial assays are increasingly used for breast cancer management. The five commonly available assays include the 12 gene assay (EndoPredict®), 21 gene (OncotypeDX®), 70 gene (MammaPrint®), 2–5 gene (HOXB13/IL-17BR or H/I index® and Breast Cancer Index®) or 58 gene assay (Prosigna®) [1,2,3,4,5,6,7,8]. The intended use is prognostic (in fact the FDA approval for MammaPrint® and Prosigna® is for prognostic use only, others are not FDA cleared), but these tests are frequently used for predictive/therapeutic purposes. Generally, such use may be acceptable as tumors that are at high ROR have more biologically aggressive features and high proliferation that can be targeted with drugs that inhibit the cell cycle (i.e., chemotherapy). In fact, the majority of these assays are influenced by tumor cell proliferation [20]. However, some tumors that present at an anatomically advanced stage but lack increased proliferation and such tumors are at increased ROR but this risk cannot always be minimized by the use of chemotherapy. Therefore, these multigene assays are now coupled with clinical parameters (such as using EndoPredict® clinical score or EPclin over just the EP score) to provide a more accurate assessment of prognosis [21]. Prosigna® assay is a bit different from the rest as it accounts for the tumor size (≤2 cm or more) in Prosigna® score calculation and then the Prosigna® risk is defined based on the lymph node status. The Prosigna® test is based on the 50 gene classifier algorithm previously named as PAM50 [14, 22]. The Prosigna® score is calculated using coefficients from a Cox model that includes the Pearson correlation of a 46-gene subset of the 50 genes to each PAM50 centroid (prototypical gene expression profiles of each of the four PAM50 molecular subtypes), a proliferation score, and tumor size [7]. The score is adjusted to a 0–100 scale and risk categories are defined based on lymph node status (low, intermediate, and high risk in lymph node-negative; low and high risk in lymph node-positive). Although the Prosigna® test result is also heavily influenced by the proliferation gene score, the inclusion of tumor size, and lymph node status in defining risk makes it a robust prognostic assay. However, Prosigna® and other molecular tests are expensive with the cost of over $4000 per test. We also believe that similar prognostic and predictive information can be derived from routine histopathology and immunohistochemistry testing of breast cancer but similar to multigene assays, the pathologic variables also need to be combined into one index to provide more succinct information to the oncologist [16, 17, 23, 24].
Our primary goal of this study was to compare the histopathologic features of the tumors that were tested by the Prosigna® assay (n = 79) and identify the pathologic variables that correlate best with the Prosigna® score. The secondary goal was to attempt combining these significant pathologic variables into one index that could provide an alternative to Prosigna® score. We found that a high Prosigna® score correlated with larger tumor size, higher tumor grade, and higher Ki-67 labeling index. These histopathologic features are known to have strong prognostic value [25,26,27,28]. Although the high Prosigna® score cases showed slightly lower ER and PR H-scores, the differences were not statistically significant in this cohort—this is in contrast to the histopathologic correlation that is usually seen with other molecular assays. This may be due to only a small number of cases with high Prosigna® score or reflects the difference in genes that comprise Prosigna® and other molecular assays. In fact, a few studies that have directly compared different molecular tests have shown significant variability in risk classification on an individual case and it is likely that each molecular assay correlates with different morpho-immunohistologic features [10, 12].
Due to the significant correlation of Prosigna® test results with the known prognostic features, we created an index modeled on Prosigna® scores and tested its prognostic significance on a set of well-characterized ER+/HER2-negative stage I and II invasive breast cancers (n = 106). This SiNK™ index is easy to calculate and provides useful prognostic information that can be obtained using only tumor size, lymph node status, and Ki-67 labeling index. However, it is difficult to make an exact comparison of the SiNK™ index in this study tested on heterogeneously treated patients with distant recurrence risk defined by Prosigna® score derived from clinical trial data. Prosigna® test is currently intended for use on stage I and II postmenopausal breast cancer patients with up to three positive lymph nodes. The clinical trial data has shown that the risk of distant recurrence at 10 years based on Prosigna® score is <10% with low-risk and >20% for high-risk assuming patients receive 5 years of endocrine therapy [15, 22]. In comparison, the distant recurrence rate in the current study was 4% in patients with SiNK™ ≤40 and 19% in patients with SiNK™ >40 with an average follow-up of 101 months. This suggests a comparable prognostic value of the SiNK™ index. SiNK™ is particularly useful in estimating prognosis for stage I and II, lymph node-negative patients. Because the SiNK index includes tumor size and lymph node status, it correlates with tumor stage, such that almost all stage III patients generally have SiNK™ index >40. Therefore, we did not include stage III patients in the validation cohort. In contrast to individual parameters that are hard to factor in for estimating prognosis, the SiNK™ index can provide one cohesive variable for breast cancer management. Moreover, it is simple to calculate based on the pathology report and can be used for counseling patients during office visits without waiting for additional testing.
The current study is the first to correlate histopathologic findings with Prosigna® test and reports on a novel index, it does have some limitations. First, the number of events was low despite long term follow-up, which is partly due to successful breast cancer treatments and partly due to small sample size. Secondly, SiNK™ index contains a variable (Ki-67) for which standardization is lacking and many pathologists and clinicians still do not routinely use it in clinical practice [29]. Several pre-analytical and analytical factors (cold ischemic time, time in formalin, different clones, detection systems, etc.), intra-observer, and interlaboratory variability can alter Ki-67 measurement which can impact the SiNK™ index. In routine practice, Ki-67 labeling index should be used as a continuous score rather than a hard cut off and should be combined along with other pathology variables for clinical management. With most laboratories in the United States adhering to proper fixation guidelines for breast biomarkers [30, 31], and with limited number of clones used for clinical testing, interobserver variability remains the most important source of variability in Ki-67 measurement [32]. For Ki-67 evaluation, the pathologists at our institution have often used a more pragmatic approach rather than actual counting of 500 or 1000 tumor cells. We first estimate the Ki-67 labeling index on the entire tumor. If the estimate falls below 10 or above 50, then estimate stands as the final Ki-67 labeling index. If the estimate is between 10 and 50, then 50–100 cells are counted in representative areas based on the pathologist’s discretion to arrive at the labeling index. This approach seems to have worked, with Ki-67 being an important component of a multivariable model (i.e., Magee Equations™) that can estimate Oncotype DX® score and predict benefit from chemotherapy in the neoadjuvant setting [16, 18]. It is possible that signal counting software (free online software expected to be made available by Ki-67 international working group) may make the task easier and more accurate when the estimated Ki-67 index is between 10 and 50%. Although some variability is expected with the SiNK™ index based on Ki-67 score, the two other components in the SiNK™ index (tumor size and nodal status) are more consistently measured and the combined index can be a strong prognostic factor provided a more consistent approach is taken for Ki-67 measurement [33,34,35].
Based on our findings in this study, we believe that SiNK™ provides long term prognostic information in stage I or II ER+/HER2-negative breast cancers. The patients with higher SiNK™ index may need more intensive treatment to improve recurrence-free survival. However, it is to be noted that worse prognosis does not always mandate the use of chemotherapy, and certainly, our current study did not evaluate if chemotherapy use will benefit patients with a higher SiNK™ index. Depending on individual tumor features, higher SiNK™ index patients may be treated upfront with chemotherapy, extended endocrine therapy, or a combination of endocrine therapy with cyclin-dependent kinase 4/6 inhibitors in the future [36, 37]. It is, however, important to note that the patients in the validation cohort of the current study were heterogeneously treated, but SiNK™ index was still able to predict prognosis, suggesting that SiNK™ identifies inherent tumor features present at diagnosis, that does not appear to be impacted with standard therapy.
In summary, Prosigna® test scores correlate with known prognostic factors. The SiNK™ index derived from this correlation provides a cost-effective alternative that can provide useful prognostic information in ER+/HER2-negative tumors. However, this study needs to be further verified in a larger cohort of patients for potential wider applicability.
References
Filipits M, Rudas M, Jakesz R, Dubsky P, Fitzal F, Singer CF, et al. A new molecular predictor of distant recurrence in ER-positive, HER2-negative breast cancer adds independent information to conventional clinical risk factors. Clin Cancer Res. 2011;17:6012–20.
Jerevall PL, Ma XJ, Li H, Salunga R, Kesty NC, Erlander MG, et al. Prognostic utility of HOXB13:IL17BR and molecular grade index in early-stage breast cancer patients from the Stockholm trial. Br J Cancer. 2011;104:1762–9.
Ma XJ, Hilsenbeck SG, Wang W, Ding L, Sgroi DC, Bender RA, et al. The HOXB13:IL17BR expression index is a prognostic factor in early-stage breast cancer. J Clin Oncol. 2006;24:4611–9.
Ma XJ, Salunga R, Dahiya S, Wang W, Carney E, Durbecq V, et al. A five-gene molecular grade index and HOXB13:IL17BR are complementary prognostic factors in early stage breast cancer. Clin Cancer Res. 2008;14:2601–8.
Paik S, Shak S, Tang G, Kim C, Baker J, Cronin M, et al. A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. N Engl J Med. 2004;351:2817–26.
van de Vijver MJ, He YD, van’t Veer LJ, Dai H, Hart AA, Voskuil DW, et al. A gene-expression signature as a predictor of survival in breast cancer. N Engl J Med. 2002;347:1999–2009.
Wallden B, Storhoff J, Nielsen T, Dowidar N, Schaper C, Ferree S, et al. Development and verification of the PAM50-based Prosigna breast cancer gene signature assay. BMC Med Genom. 2015;8:54.
Paik S, Tang G, Shak S, Kim C, Baker J, Kim W, et al. Gene expression and benefit of chemotherapy in women with node-negative, estrogen receptor-positive breast cancer. J Clin Oncol. 2006;24:3726–34.
Alvarado MD, Prasad C, Rothney M, Cherbavaz DB, Sing AP, Baehner FL, et al. A prospective comparison of the 21-gene recurrence score and the PAM50-based Prosigna in estrogen receptor-positive early-stage breast cancer. Adv Ther. 2015;32:1237–47.
Bartlett JM, Bayani J, Marshall A, Dunn JA, Campbell A, Cunningham C, et al. Comparing breast cancer multiparameter tests in the OPTIMA prelim trial: no test is more equal than the others. J Natl Cancer Inst. 2016;108:djw050.
Dowsett M, Sestak I, Lopez-Knowles E, Sidhu K, Dunbier AK, Cowens JW, et al. Comparison of PAM50 risk of recurrence score with oncotype DX and IHC4 for predicting risk of distant recurrence after endocrine therapy. J Clin Oncol. 2013;31:2783–90.
Sestak I, Buus R, Cuzick J, Dubsky P, Kronenwett R, Denkert C, et al. Comparison of the performance of 6 prognostic signatures for estrogen receptor-positive breast cancer: a secondary analysis of a randomized clinical trial. JAMA Oncol. 2018;4:545–53.
Varga Z, Sinn P, Fritzsche F, von Hochstetter A, Noske A, Schraml P, et al. Comparison of endopredict and oncotype DX test results in hormone receptor positive invasive breast cancer. PLoS ONE. 2013;8:e58483.
Parker JS, Mullins M, Cheang MC, Leung S, Voduc D, Vickery T, et al. Supervised risk predictor of breast cancer based on intrinsic subtypes. J Clin Oncol. 2009;27:1160–7.
Gnant M, Filipits M, Greil R, Stoeger H, Rudas M, Bago-Horvath Z, et al. Predicting distant recurrence in receptor-positive breast cancer patients with limited clinicopathological risk: using the PAM50 risk of recurrence score in 1478 postmenopausal patients of the ABCSG-8 trial treated with adjuvant endocrine therapy alone. Ann Oncol. 2014;25:339–45.
Bhargava R, Clark BZ, Carter GJ, Brufsky AM, Dabbs DJ. The healthcare value of the Magee Decision Algorithm: use of Magee Equations and mitosis score to safely forgo molecular testing in breast cancer. Mod Pathol. 2020;33:1563–70.
Bhargava R, Clark BZ, Dabbs DJ. Breast cancers with magee equation score of less than 18, or 18-25 and mitosis score of 1, do not require oncotype DX testing: a value study. Am J Clin Pathol. 2019;151:316–23.
Farrugia DJ, Landmann A, Zhu L, Diego EJ, Johnson RR, Bonaventura M, et al. Magee Equation 3 predicts pathologic response to neoadjuvant systemic chemotherapy in estrogen receptor positive, HER2 negative/equivocal breast tumors. Mod Pathol. 2017;30:1078–85.
Bhargava R, Striebel J, Beriwal S, Flickinger JC, Onisko A, Ahrendt G, et al. Prevalence, morphologic features and proliferation indices of breast carcinoma molecular classes using immunohistochemical surrogate markers. Int J Clin Exp Pathol. 2009;2:444–55.
Fan C, Oh DS, Wessels L, Weigelt B, Nuyten DS, Nobel AB, et al. Concordance among gene-expression-based predictors for breast cancer. N Engl J Med. 2006;355:560–9.
Buus R, Sestak I, Kronenwett R, Denkert C, Dubsky P, Krappmann K, et al. Comparison of EndoPredict and EPclin With oncotype DX recurrence score for prediction of risk of distant recurrence after endocrine therapy. J Natl Cancer Inst. 2016;108:jw149.
Ohnstad HO, Borgen E, Falk RS, Lien TG, Aaserud M, Sveli MAT, et al. Prognostic value of PAM50 and risk of recurrence score in patients with early-stage breast cancer with long-term follow-up. Breast Cancer Res. 2017;19:120.
Flanagan MB, Dabbs DJ, Brufsky AM, Beriwal S, Bhargava R. Histopathologic variables predict Oncotype DX recurrence score. Mod Pathol. 2008;21:1255–61.
Klein ME, Dabbs DJ, Shuai Y, Brufsky AM, Jankowitz R, Puhalla SL, et al. Prediction of the oncotype DX recurrence score: use of pathology-generated equations derived by linear regression analysis. Mod Pathol. 2013;26:658–64.
Michaelson JS, Silverstein M, Wyatt J, Weber G, Moore R, Halpern E, et al. Predicting the survival of patients with breast carcinoma using tumor size. Cancer. 2002;95:713–23.
Petrelli F, Viale G, Cabiddu M, Barni S. Prognostic value of different cut-off levels of Ki-67 in breast cancer: a systematic review and meta-analysis of 64,196 patients. Breast Cancer Res Treat. 2015;153:477–91.
Rakha EA, Reis-Filho JS, Baehner F, Dabbs DJ, Decker T, Eusebi V, et al. Breast cancer prognostic classification in the molecular era: the role of histological grade. Breast Cancer Res. 2010;12:207.
Sopik V, Narod SA. The relationship between tumour size, nodal status and distant metastases: on the origins of breast cancer. Breast Cancer Res Treat. 2018;170:647–56.
Dowsett M, Nielsen TO, A’Hern R, Bartlett J, Coombes RC, Cuzick J, et al. Assessment of Ki67 in breast cancer: recommendations from the international Ki67 in breast cancer working group. J Natl Cancer Inst. 2011;103:1656–64.
Allison KH, Hammond MEH, Dowsett M, McKernin SE, Carey LA, Fitzgibbons PL, et al. Estrogen and progesterone receptor testing in breast cancer: ASCO/CAP guideline update. J Clin Oncol. 2020;38:1346–66.
Wolff AC, Hammond MEH, Allison KH, Harvey BE, Mangu PB, Bartlett JMS, et al. Human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists Clinical Practice Guideline Focused Update. J Clin Oncol. 2018;36:2105–22.
Polley MY, Leung SC, McShane LM, Gao D, Hugh JC, Mastropasqua MG, et al. An international Ki67 reproducibility study. J Natl Cancer Inst. 2013;105:1897–906.
Leung SCY, Nielsen TO, Zabaglo L, Arun I, Badve SS, Bane AL, et al. Analytical validation of a standardized scoring protocol for Ki67: phase 3 of an international multicenter collaboration. NPJ Breast Cancer. 2016;2:16014.
Leung SCY, Nielsen TO, Zabaglo LA, Arun I, Badve SS, Bane AL, et al. Analytical validation of a standardised scoring protocol for Ki67 immunohistochemistry on breast cancer excision whole sections: an international multicentre collaboration. Histopathology. 2019;75:225–35.
Rimm DL, Leung SCY, McShane LM, Bai Y, Bane AL, Bartlett JMS, et al. An international multicenter study to evaluate reproducibility of automated scoring for assessment of Ki67 in breast cancer. Mod Pathol. 2019;32:59–69.
Lynce F, Shajahan-Haq AN, Swain SM. CDK4/6 inhibitors in breast cancer therapy: current practice and future opportunities. Pharmacol Ther. 2018;191:65–73.
Shah M, Nunes MR, Stearns V. CDK4/6 inhibitors: game changers in the management of hormone receptor-positive advanced breast cancer? Oncology. 2018;32:216–22.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
DJD is an independent contractor breast pathologist at PreludeDx (Laguna Hills, CA). RB is an ad hoc advisor to Eli Lilly & Company, Indianapolis, IN.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Baskota, S.U., Dabbs, D.J., Clark, B.Z. et al. Prosigna® breast cancer assay: histopathologic correlation, development, and assessment of size, nodal status, Ki-67 (SiNK™) index for breast cancer prognosis. Mod Pathol 34, 70–76 (2021). https://doi.org/10.1038/s41379-020-0643-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41379-020-0643-8