Abstract
Asthma is an allergic inflammatory lung disease affecting nearly 300 million people worldwide. To better understand asthma, new regulators must be identified. We conducted a study to investigate the effect and mechanisms of action of surfactant protein A (SPA) in OVA-induced asthmatic mice. Treatment with SPA delayed the onset of asthma, decreased its severity, as well as notably suppressed pro-inflammatory cytokine production. Furthermore, SPA-treated mice possessed more leukocytes; more CD4+ T cells infiltrated the spleen in the SPA-treated mice than in the control mice, and there were decreased percentages of Th1 and Th17 cells in vivo. In addition, expression levels of the T-bet (Th1 transcription factor) and RORγt (Th17 transcription factor) genes were significantly downregulated by SPA treatment. Moreover, SPA reduced the production and mRNA expression of pro-inflammatory cytokine mRNAs in activated T cells in vivo. Mechanistically, SPA could inhibit STAT1/4 and STAT3 phosphorylation, resulting in the differentiation of Th1 and suppression of Th17 cells, respectively. In the presence of CD3/CD28 expression, STAT1/4 and STAT3 were activated but suppressed by SPA, which was responsible for the augmentation of Th1 and Th17 differentiation. This result showed that SPA can effectively modulate the JAK/STAT pathway by suppressing Th1 and Th17 differentiation, thus preventing asthma. The present study reveals the novel immunomodulatory activity of SPA and highlights the importance of further investigating the effects of SPA on asthma.
Similar content being viewed by others
Introduction
Asthma is an allergic inflammatory disease characterized by dyspnea and cough or chest tightness accompanied by varying degrees of airway remodeling. There are nearly 300 million asthmatic individuals worldwide. The complex pathophysiology of asthma makes it challenging to understand it at both the cellular and molecular levels. At present, the commonly used beta-2 receptor agonists only have certain beneficial effects on immediate symptoms, while long-term control depends on the inhalation of glucocorticoid therapy [1]. The long-term use of hormones disturbs the immune function of the body and easily leads to drug antagonism [2]. Specific immunotherapy may markedly improve asthma, but the clinical application of this approach is greatly limited because of the numerous inducing factors and the inability to identify allergens. Therefore, there is still a long way to go to further understand the pathogenesis of asthma and identify potential therapeutic targets.
On the surface of the pulmonary mucosa, alveolar surfactant reduces gas-liquid surface tension and participates in maintaining mucosal immune balance. Surfactant protein A (SPA), a type of alveolar surfactant protein, is a natural immune mediator of the lectin family. It exerts a regulatory effect on innate and adaptive immune effector cells [3]. While SPA can inhibit the maturation and antigen presentation function of dendritic cells, it can also directly inhibit the proliferation of T cells by inducing expression of the regulatory cytokine TGFβ [4]. Evidence suggests that the decrease or disappearance of SPA leads to a decrease in its immunoprotective effect [5]. Several clinical and experimental studies have also found that SPA is downregulated or inactivated in asthmatic patients and animal models of asthma [6]. Relevant studies based on SPA-deficient animals further demonstrated the regulatory effect of SPA on lung allergic inflammation and asthma [7]. Therefore, SPA may serve as a potential therapeutic agent against asthma.
Asthma is most likely mediated by the pathogenic responses of lymphocytes, followed by a series of various cytokine secretion processes [8]. A growing number of therapeutic approaches targeting T cells have been considered for the treatment of asthma [9]. T lymphocytes can mainly differentiate into CD4+ and CD8+ T cells according to the capacity of their TCR to interact with MHCII or MHCI, respectively [10]. Activated CD4+ T cells can further differentiate into a variety of effector subtypes, including classical T-helper 1 cells (Th1), T-helper 2 cells (Th2), T-helper 17 cells (Th17) and regulatory T cells (Treg) [11]. Their differentiation is driven by a network of specific cytokines. Th1 cells are characterized by the production of IFN-γ, priming the development of Th1 cells through a cascade of reactions [12]. Th2 cells can release a large quantity of IL-4 and IL-13 by upregulation of IL-4 and phosphorylation of STAT6 [13]. The differentiation of Th17 cells, which produce IL-17A and IL-22, requires transforming growth factor-β and induction of the transcription factor RORγt [14]. Regulatory T cells are important for maintaining self-tolerance and immune homeostasis. Forkhead box protein 3 (Foxp3) and other transcription factors are essential for the differentiation of Treg cells [15]. Aberrant Th17 and Th1 cell responses are pathogenic in several autoimmune diseases. For example, a number of studies have demonstrated that Th17 and Th1 cells play a central role in the development and pathogenesis of asthma [16]. Some other studies have demonstrated that the inhibition of Th1 and Th17 differentiation yields a major effect in the treatment of asthma [17,18,19].
Because of the significant role of activated T cells in asthma, we hypothesized that SPA may affect the treatment of asthma. In the present study, we investigated the effect and mechanism of action of SPA on asthma. The results showed that SPA could effectively inhibit the development of asthma, and this was mainly achieved by inhibiting Th1 and Th17 polarization and JAK/STAT pathway activation.
Materials and methods
Mice
Female C57BL/6J mice (18–20 g) were purchased from Shanghai SLAC Laboratory Animal Co., Ltd. (Shanghai, China). All animals were maintained under pathogen-free conditions, with a light/dark cycle of 12 h, and had access to sterilized water and a standard laboratory diet ad libitum. The experimental protocols were approved by the Ethics Committee of Fujian Medical University, and the study was conducted in accordance with the guidelines published in the NIH Guide for the Care and Use of Laboratory Animals.
Asthma model
The methods used were based on our previously published articles with minor modifications [20]. Briefly, mice were sensitized intranasally with three injections of 200 μL 1.36% aluminum hydroxide suspension of 20 μg OVA on days 0, 7, and 14. On day 15, the mice were stimulated by spray inhalation of 2 mg/mL OVA solution in normal saline. All animals were euthanized on day 21 after a 10 h fast prior to euthanasia, and 1 mL bronchoalveolar lavage fluid (BALF) samples were collected for measurement. The control (naive mice) and model mice were injected with the same proportion of solvent.
Histopathology staining
The lung lobules from 6 mice selected at random from each group were fixed in 4% neutral buffered paraformaldehyde, dehydrated for 12 h, embedded in paraffin, sectioned into 3 µm-thick slices, stained with hematoxylin-eosin and periodic acid-Schiff stains refer to a previous report [21] and then examined under a light microscope (Olympus, Tokyo, Japan) for imaging.
Measurement of cytokine production
The levels of IgA, IgE, IFN-γ, IL-17, IL-4, and IL-10 were assayed according to the manufacturer’s instructions (eBioscience, San Diego, CA). BLAF samples were collected to determine the cytokine levels and were analyzed with the LEGENDplex Mouse Th Cytokine Panel kit (catalog 740005, Biolegend, San Diego, CA) according to the manufacturer’s protocol. Briefly, sample and fluorescence-encoded beads were mixed in each well using a V-bottom plate and then shook at 500 rpm for 2 h at room temperature. After wash twice, the biotinylated detection antibodies were added to each well and the plate was incubated under the same conditions. Streptavidin–phycoerythrin, which bound to the antibodies and provided fluorescent signal intensities in proportion to the number of bound analytes, was subsequently added to each well and incubated for 30 min. Finally, samples were washed twice and analyzed on flow cytometry (FACSCalibur, BD Biosciences).
Cell purification, culture, and proliferation
For purification of infiltrating mononuclear cells from the spleen, single-cell suspensions were prepared and purified using a Percoll (GE Healthcare, Piscataway, NJ) gradient. To isolate mouse CD4+ T cells, splenocytes cells were excluded using a mouse CD4+ negative selection kit (Miltenyi Biotec) according to the manufacturer’s instructions. Cell purity was assessed by flow cytometry and was consistently higher than 90%.
For Th1 and Th17 differentiation, naive CD4+ T cells from mice were cultured in RPMI 1640 medium with 10% fetal bovine serum in the presence of CD3/28 antibodies (2 μg/mL; Peprotech, Rocky Hill, NJ) to stimulate the T cell receptor (TCR). A Th1 mixture (10 ng/mL IL-12 and 10 μg/mL IL-4 antibody; Peprotech) or Th17 mixture (30 ng/mL IL-6, 3 ng/mL TGF-β, 10 μg/mL IL-4 antibody, and 10 μg/mL IFN-γ antibody; all from Peprotech) was added to drive Th1 and Th17 polarization for 4 days in culture, respectively. Fludarabine and HO-3867 (Selleck Chemicals) were added to the cell culture at a concentration of 1 μM.
CFDA-SE was used to track splenocyte proliferation. CFDA-SE labeled splenocytes (2 × 106 cell/mL) were stimulated by Con A (5 μg/mL; Sigma) in the presence of IL-2 (50 ng/mL; Peprotech) and incubated with SPA for 72 h. The cells were collected and washed with PBS, and their proliferation was analyzed by flow cytometry (FACSCalibur, BD Biosciences).
Flow cytometry analysis
For intracellular staining, splenocytes were stimulated with 100 ng/mL PMA (Sigma) and 750 ng/mL ionomycin (Sigma). Cells were collected and incubated with anti-CD4 antibody and then fixed, permeabilized, and stained with Phenotyping Cocktail (BD Biosciences). For surface staining, splenocytes were collected and washed with PBS once and then incubated for 30 min with the following fluorochrome-conjugated antibodies: PE-CD3, FITC-CD4, and APC-CD8 (eBioscience). Flow cytometry analyses were performed on a FACSCalibur (BD Biosciences).
Purification of SPA
The CHO cell line constitutively expressing SPA was purchased from Cloud Clone Corp. Company and SPA were purified as follows. After thawing, the cells were cultured in a serum-free EXCELL 302 medium. The medium was collected five times (24–48 h interval) and centrifuged. The supernatant was then dialyzed in 20 mM Tris buffer (pH 7.4). A mannose-agarose 6B column was balanced with 5 mM Tris buffer (pH 7.4). Then SPA was eluted with 5 mM Tris buffer containing 5 mM EDTA (pH 7.4), and the EDTA was removed by dialysis against 5 mM Tris buffer (pH 7.4). After purification, the purified protein was analyzed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). After electrophoresis, silver nitrate staining was used to evaluate the purification of SPA.
Quantitative real-time polymerase chain reaction (PCR) analysis
Total RNA was isolated from lungs using RNAiso Plus (Takara) according to the manufacturer’s protocol. One microgram of total RNA was reverse transcribed to yield first-strand cDNA, which was obtained by using Prime ScriptTM RT Master Mix (Takara). The expression levels of mRNA were quantified by real-time PCR using SYBR Premix Ex TaqTM (Takara). The PCR program included 1 cycle at 95 °C for 30 s, 40 cycles at 95 °C for 5 s and 60 °C for 30 s. The specific primers used for amplification are shown in Table 1. The results were expressed by calculating the 2−ΔΔCT values relative to the housekeeping gene GAPDH.
Western blot
To analyze protein expression, Western blot assays were performed using a Phospho-STAT Antibody Sampler Kit (#9914, Cell Signaling Technology), STAT1 antibody (#9172, Cell Signaling Technology), STAT3 antibody (#4904, Cell Signaling Technology), STAT4 antibody (#2653, Cell Signaling Technology), and mouse anti-GAPDH antibody (#5174, Cell Signaling Technology) according to the manufacturer’s instructions. Briefly, 1 × 107 cells were lysed with 100 μL RIPA buffer (50 mM Tris-HCl (pH 7.5), 150 mM NaCl, 1% Triton X-100 and 0.5% Na-deoxycholate) containing protease inhibitors (Complete Mini; Roche). Samples (30 μg) were separated on 8–12% SDS-PAGE gels and transferred to PVDF membranes. The membranes were incubated with the primary antibodies overnight at 4 °C followed by incubation with an HRP-conjugated secondary antibody. The bound antibodies were detected using an ECL kit (Pierce, Rockford, Illinois, USA).
Statistical analysis
The data were analyzed with GraphPad Prism software (Graph Pad Inc., San Diego, CA). All quantitative data are expressed as the means ± SEM as indicated. Comparisons between two groups were analyzed by unpaired Student’s t test, and multiple comparisons were analyzed by one-way analysis of variance followed by Dunnett’s test. The level of statistical significance was established at p < 0.05.
Results
SPA protects mice from asthma
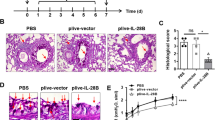
We investigated whether the administration of SPA (Fig. 1A) could affect the inflammation and disease progression of asthma (Fig. 1B). OVA-challenged mice showed marked infiltration of inflammatory cells into the perivascular and connective tissues, lumen narrowing, and mucosa thickening compared with normal lungs (Fig. 1C). These results demonstrated the validity of the OVA-challenge mouse model compared with the normal group. The SPA group showed less infiltration of inflammatory cells than the model group, and SPA significantly decreased abnormal alveolar structure, structural disorder, cell debris, and fibroblasts in the lung tissue sections (Fig. 1C). In addition, SPA treatment significantly increased IgA levels and decreased IgE levels (Fig. 1D). Moreover, expression of the cytokine IFN-γ was significantly decreased after SPA treatment, and IL-17 was significantly decreased after 10 μg/kg SPA treatment compared with the asthma group (Fig. 1E).
A SDS-PAGE (12% w/v) analysis of an affinity-purified recombinant fragment of SPA. Purified SPA appeared as a homogenous band of 36 kDa. B The experimental design. C Tissue histopathology was analyzed by HE and PAS staining (scale bar = 50 μm). D The levels of BLAF IgA and IgE were determined by ELISA. E The expression of cytokines in BLAF was analyzed by ELISA. *p < 0.05 and **p < 0.01 compared with OVA-induced allergic asthma mice (n = 6).
SPA suppresses pro-inflammatory cytokine production and expression in activated T cells in asthmatic mice
We next investigated the effect of SPA on T cell differentiation by multiparameter flow cytometry. SPA administration significantly decreased levels of the pro-inflammatory cytokines TNF-α, IL-6, and IL-1β in the BLAF but slightly increased levels of the anti-inflammatory cytokines IL10 and IL-13 (Fig. 2A). The levels of IFN-γ and IL17A also showed decreasing trends. Because of the noticeably preventive effect of SPA on asthma, we wondered whether SPA could suppress the inflammatory response. Lungs isolated from control, model, and SPA-treated mice were collected and after homogenization, the supernatants were harvested to determine the production of pro-inflammatory cytokines. Compared with the model group, SPA treatment significantly reduced the production of IL-6, IL-1β, and TNF-α but increased levels of the anti-inflammatory cytokine IL10 (Fig. 2B). Subsequently, we investigated the effect of SPA on the mRNA expression of pro-inflammatory cytokines in lungs from different treated mice. The mRNA expression of IL-6, IL-1β, and TNF-α was also decreased, but levels of the anti-inflammatory cytokine IL10 were increased by SPA treatment (Fig. 2C).
A Model and SPA-treated mice were sacrificed to obtain the BLAF, and cytokines were measured by multiparameter flow cytometry. n = 6. B The lungs were collected from different treated mice; after homogenization, the supernatants were harvested to measure cytokine production by ELISA. C The lungs were collected from different treated mice, and the mRNA expression of selected genes was measured by real-time PCR. The data are expressed as the means ± SEM (n = 3). *p < 0.05 and **p < 0.01 vs. model mice. The data presented are representative of three independent experiments.
SPA inhibits Th1 and Th17 differentiation in vivo
We next found that SPA significantly promoted the proliferation of splenocytes (Fig. 3A). Activated T cells can be differentiated into a variety of subtypes, including Th1, Th2, Th17, and Treg cells, and imbalances in subtype differentiation are closely related to the onset of disease. To better understand the above results, in which SPA was shown to significantly increase the number of CD4+ T cells infiltrating into the spleen, we investigated the effect of SPA on the subtypes of CD4+ T cells (Fig. 3B). Splenocytes isolated from naive, asthma, and SPA-treated mice were stimulated with 100 ng/mL IL-2 for 48 h to analyze the content of Th1 (CD4+IFN-γ+), Th2 (CD4+IL-4+), Th17 (CD4+IL-17+), and Treg (CD4+Foxp3+) cell populations by flow cytometry (Fig. 3C). Compared with naive mice, the asthma mice showed significantly increased percentages of all four T cell subtypes, whereas SPA decreased the proportion of Th1 and Th17 cells but increased the Th2 cells in contrast to the model group (Fig. 3C).
A Splenocytes were isolated from model and SPA-treated mice on day 18 after immunization and cultured in the presence of 100 ng/mL IL-2 for 48 h. The proliferation of cells was measured by CFDA-SE. B The percentages of CD4 and CD8 subsets in splenocytes were determined by flow cytometry. C Splenocytes were analyzed for the percentages of Th1, Th2, Th17, and Treg cells expressing CD4 by flow cytometry. The percentages of cells positive for these antigens are shown. The data are expressed as the means ± SEM (n = 6). *p < 0.05 and **p < 0.01 vs. model mice.
SPA regulates Th cell polarization in part by the JAK/STAT signaling pathway
Because the JAK/STAT signaling pathway is closely associated with the differentiation of T helper cells, we determined whether SPA affects the regulation of Th cell polarization. After treatment with the pan-STAT inhibitor SH-4-54, the OVA-challenged mice showed less infiltration of inflammatory cells into the perivascular and connective tissues, lumen narrowing, and mucosa thickening compared with normal lungs (Fig. 4A). In addition, inhibition of the JAK/STAT signaling pathway significantly increased the IgA level and decreased the IgE level (Fig. 4B). Moreover, expression of the cytokines IFN-γ and IL-17 was significantly decreased after SPA treatment compared with the asthma group (Fig. 4C).
SPA suppresses Th1 differentiation in vitro by inhibiting STAT1
As shown in Fig. 5A, the Th1 differentiation mixture induced the differentiation of approximately 40% of naive CD4+ T cells into Th1 cells. SPA significantly decreased the proportion of Th1 cells without inhibiting the proliferation or vitality of CD4+ T cells. We next examined the lineage markers of Th1, IFN-γ and T-bet. As expected, the mRNA levels of both genes were downregulated by SPA (Fig. 5B). To study the mechanisms underlying the suppressive effect of SPA on Th1 differentiation, we next examined the STAT signaling pathway. Interestingly, we found that fludarabine (a STAT1 inhibitor) could inhibit Th1 differentiation, which was consistent with earlier studies (Fig. 5C). We also found that 20 μg/mL SPA treatment downregulated the phosphorylation of STAT1 (Fig. 5C), suggesting that SPA inhibits Th1 differentiation by decreasing STAT1 activation.
A Naive CD4+ T cells from mice were cultured under Th1 conditions in the absence or presence of SPA at the indicated concentration for 4 days. Intracellular staining for IFN-γ is shown. **p < 0.05 vs. control. B Naive CD4+ T cells were isolated from mice and cultured with CD3/28 to stimulate the TCR in the absence or presence of a Th1 differentiation mixture. SPA was then added at the indicated concentration. Three days later, RNA was extracted, reverse transcribed into cDNA and subjected to real-time PCR. Gene expression is expressed relative to that of GAPDH. **p < 0.05 vs. control. C Naive CD4+ T cells from mice were exposed to different treatments. Cell lysates were subjected to Western blot analysis for phosphorylated STAT1 and STAT4. Quantification of the optical density of the bands is shown in the right panel. **p < 0.05 vs. CD3/28.
SPA suppresses Th17 differentiation in vitro by inhibiting STAT3 phosphorylation
Next, we investigated the mechanism by which SPA affects cytokine-driven Th17 differentiation. After 4 days of culture under Th17 conditions, approximately 30% of the naive CD4+ T cells were polarized into IL-17-secreting Th17 cells. The addition of SPA significantly inhibited this polarization (Fig. 6A). As shown in Fig. 6B, the expression of IL-17A, IL-17F, and Rorγt associated with Th17 differentiation was suppressed by SPA. To characterize the mechanisms underlying the suppression of Th17 by SPA, we used a specific STAT3 inhibitor HO-3867 as a positive control. We found that 20 μg/mL SPA treatment downregulated the phosphorylation of STAT3, suggesting that SPA inhibits Th17 differentiation by decreasing STAT3 activation (Fig. 6C).
A Naive CD4+ T cells from mice were cultured under Th17 conditions in the absence or presence of SPA at the indicated concentration for 4 days. Intracellular staining for IL-17 is shown. **p < 0.05 vs. control. B Naive CD4+ T cells were isolated from mice and cultured with CD3/28 to stimulate the TCR in the absence or presence of a Th17 differentiation mixture. SPA was added at the indicated concentration. Three days later, RNA was extracted, reverse transcribed into cDNA, and subjected to real-time PCR. The gene expression is expressed relative to that of actin. **p < 0.05 vs. control. C Naive CD4+ T cells from mice were exposed to different treatments. The cell lysates were subjected to Western blot analysis for phosphorylated STAT3. Quantification of the optical density of the bands is shown in the right panel. **p < 0.05 vs. CD3/28.
Discussion
In this work, we hypothesized that asthmatic conditions would be reflected in alterations in the BALF of individuals suffering from asthma. SPA was administered to mice to investigate the effects of interfering with the disordered BALF metabolism induced by asthma. In the present study, SPA was found to ameliorate asthma and we demonstrated that SPA protected against asthma by inhibiting the differentiation of Th1 and Th17 cells. Treatment with SPA effectively inhibited the development of asthma, reduced the levels of BALF pro-inflammatory cytokine, and notably decreased inflammatory cell infiltration. SPA-treated mice exhibited less aggregation of leukocytes and CD4+ T cells in the spleen. With regard to CD4+ T cells, we demonstrated that SPA decreased the percentages of Th1 cells and increased the Th2 and Treg cells in the spleen. Expression of the T-bet and RORγt genes was also significantly downregulated by SPA treatment. Furthermore, SPA reduced the production and mRNA expression of pro-inflammatory cytokines in vivo.
Inflammatory and autoimmune diseases are the most common illnesses and are a major challenge for our healthcare system. Autoimmune diseases are characterized by autologous tissue damage triggered by dysfunctional immune responses to cellular- or organ-specific self-antigens. In this process, innate immune cells play a significant role. Thus, we investigated the effect of SPA on these immune cells. In this study, the percentages of total leukocytes, CD4+ T cells, and CD8+ T cells were investigated in asthma model and SPA-treated mice. SPA was found to significantly decrease the percentages of total leukocytes and CD4+ T cells infiltrating into the spleen.
CD4+ T cells play a crucial role in both the occurrence and prevention of asthma. The diverse functions of CD4+ T cells depend on their multiple subtypes. Activated CD4+ T cells can be differentiated into at least 4 subtypes: Th1, Th2, Th17, and Treg cells. Among them, Th1 and Th17 cells always play a pathogenic role in asthma. Th2 cells display an antagonistic function toward Th1 and Treg cells to regulate the immune response [22, 23]. CD4+ T cells play an important role in the development of asthma. Effector cells promote the differentiation of activated CD4+ T cells more into Th1 and Th17 subtypes than into Th2 and Treg subtypes. As demonstrated above, we observed that the effect of SPA may target CD4+ T cells. Therefore, we isolated the splenocytes from the naive, model, and SPA-treated mice to further investigate the effect of SPA on the differentiation of CD4+ T cells. The results showed that SPA-treated mice had decreasing trends in the percentages of Th1 and Th17 cells compared to the model mice, but no obvious changes were found for Th2 and Treg cells. We demonstrated that SPA ameliorated asthma by reducing the Th1 and Th17 cells. Furthermore, we investigated the related cytokines that participate in controlling the differentiation of CD4+ T cells. We found that SPA inhibits the expression of IFN-γ and IL-17 in the spleen, suggesting that SPA inhibits the differentiation of Th1 and Th17 cells in peripheral lymphoid organs and further suppresses the follow-up immune responses to ameliorate asthma.
Asthma is a chronic inflammatory process, with a number of pro-inflammatory cytokines released by activated immune cells in peripheral immune organs. Previous studies have shown that inflammatory cytokines, such as TNF-α, IL-6, and IL-1β, are predominantly detected in asthma mice [24]. The high level of IL-6 mRNA expression in peripheral immune organs correlates with the progression of asthma. IL-6 and TNF-α are also important cytokines secreted by Th17 and Th1 cells [25, 26]. We observed that SPA suppresses the production of IL-6, IL-1β, and TNF-α along with their expression at the mRNA level in splenocytes, indicating that SPA also prevents the onset of asthma through inhibition of pro-inflammatory cytokines in peripheral immune organs.
The JAK/STAT signaling pathway is a common pathway in human physiological and pathological reactions and is closely associated with the differentiation of T helper cells [27]. The STAT family, which includes STAT 1–6, is expressed in T cells [28]. After activation, STAT can transduce the cytokine signal directly from receptors to the nucleus and regulate gene expression [29]. The JAK/STAT pathway has been well-studied in inflammation and immunity [27]. STAT3 directly binds to many of the key genes required for Th17 differentiation, including the IL-17 gene itself [30]. STAT6 is required for mediating the responses to IL-4 and for the development of Th2 cells [13]. STAT1 deletion impaired the responses to type 1 interferon, and STAT4 knockout mice exhibited inhibition of Th1 differentiation [28]. We found that SPA led to the downregulation of STAT3 phosphorylation in Th17 cells and STAT1 and STAT4 phosphorylation in Th1 cells, suggesting that SPA inhibits T cell differentiation by regulating the JAK/STAT pathway. We also examined other members of the Phospho-STAT family in the Sampler Kit (CST), including STAT2, STAT5, and STAT6. However, these latter STAT members were not found to have a regulatory role in the SPA-mediated differentiation of Th cells.
In summary, our study demonstrates that SPA improves asthma by inducing appropriate tolerance. SPA decreases the total leukocytes and CD4+ T cells in vivo, further reduces Th1 cells and increases Th2 and Treg cells, and suppresses the mRNA expression of T-bet and RORγt in asthma mice. Furthermore, SPA also reduces the production of related cytokines in splenocytes, indicating that it ameliorates asthma mainly through suppressing the differentiation of Th1 and Th17 cells and weakening the immune response. Moreover, SPA can effectively modulate the Notch pathway to suppress Th1 and Th17 differentiation. However, this study has only begun to explore the effect of SPA, and further investigations should be performed to elucidate the underlying mechanisms of SPA to evaluate its potential effects on other autoimmune diseases.
Data availability
The data that support the present study are available from the corresponding author upon reasonable request.
References
Edwards MR, Bartlett NW, Hussell T, Openshaw P, Johnston SL. The microbiology of asthma. Nat Rev Microbiol. 2012;10:459–71.
Ferreira LN, Brito U, Ferreira PL. Quality of life in asthma patients. Rev Port Pneumol. 2010;16:23–55.
Ledford JG, Pastva AM, Wright JR. Review: collectins link innate and adaptive immunity in allergic airway disease. Innate Immun. 2010;16:183–90.
Mukherjee S, Giamberardino C, Thomas JM, Gowdy K, Pastva AM, Wright JR. Surfactant protein A modulates induction of regulatory T cells via TGF-β. J Immunol. 2012;188:4376–84.
Deb R, Shakib F, Reid K, Clark H. Major house dust mite allergens Dermatophagoides pteronyssinus 1 and Dermatophagoides farinae 1 degrade and inactivate lung surfactant proteins A and D. J Biol Chem. 2007;282:36808–19.
Wang JY, Shieh CC, Yu CK, Lei HY. Allergen-induced bronchial inflammation is associated with decreased levels of surfactant proteins A and D in a murine model of asthma. Clin Exp Allergy. 2001;31:652–62.
Pastva AM, Mukherjee S, Giamberardino C, Hsia B, Lo B, Sempowski GD, et al. Lung effector memory and activated CD4+ T cells display enhanced proliferation in SP-A-deficient mice during allergen-mediated inflammation. J Immunol. 2011;186:2842–9.
Ayakannu R, Abdullah NA, Radhakrishnan AK, Lechimi Raj, V. Liam CK. Relationship between various cytokines implicated in asthma. Hum Immunol. 2019;80:755–63.
Lloyd CM, Saglani S. T cells in asthma: influences of genetics, environment, and T-cell plasticity. J Allergy Clin Immunol. 2013;131:1267–74.
Wilson CB, Rowell E, Sekimata M. Epigenetic control of T-helper-cell differentiation. Nat Rev Immunol. 2009;9:91–105.
Zhu J, Paul WE. CD4 T cells: fates, functions, and faults. Blood. 2008;112:1557–69.
Aune TM, Collins PL, Chang S. Epigenetics and T helper 1 differentiation. Immunology. 2009;126:299–305.
Kaplan MH, Schindler U, Smiley ST, Grusby MJ. Stat6 is required for mediating responses to IL-4 and for the development of Th2 cells. Immunity. 1996;4:313–9.
Weaver CT, Hatton RD, Mangan PR, Harrington LE. IL-17 family cytokines and the expanding diversity of effector T cell lineages. Annu Rev Immunol. 2007;25:821–52.
Chen Z, Lin F, Gao Y, Li Z, Zhang J, Xing Y, et al. FOXP3 and RORγt: transcriptional regulation of Treg and Th17. Int Immunopharmacol. 2011;11:536–42.
Zhou L, Chong MM, Littman DR. Plasticity of CD4+ T cell lineage differentiation. Immunity. 2009;30:646–55.
Liang, P, Peng, S, Zhang, M, Ma, Y, Zhen, X Li, H. Huai Qi Huang corrects the balance of Th1/Th2 and Treg/Th17 in an ovalbumin-induced asthma mouse model. Biosci Rep. 2017;37:BSR20171071.
Ding F, Fu Z, Liu B. Lipopolysaccharide exposure alleviates asthma in mice by regulating Th1/Th2 and Treg/Th17 balance. Med Sci Monit. 2018;24:3220–29.
Zhu M, Liang Z, Wang T, Chen R, Wang G, Ji Y. Th1/Th2/Th17 cells imbalance in patients with asthma with and without psychological symptoms. Allergy Asthma Proc. 2016;37:148–56.
Peng X, Cui Z, Gu W, Xu W, Guo X. Low level of LAT-PLC-γ1 interaction is associated with Th2 polarized differentiation: a contributing factor to the etiology of asthma. Cell Immunol. 2014;290:131–7.
Zeng Z, Wang L, Ma W, Zheng R, Zhang H, Zeng X, et al. Inhibiting the Notch signaling pathway suppresses Th17-associated airway hyperresponsiveness in obese asthmatic mice. Lab Investig. 2019;99:1784–94.
Zhang H-L, Zheng X-Y, Zhu J. Th1/Th2/Th17/Treg cytokines in Guillain–Barre syndrome and experimental autoimmune neuritis. Cytokine Growth Factor Rev. 2013;24:443–53.
Shevach EM. Mechanisms of foxp3+ T regulatory cell-mediated suppression. Immunity. 2009;30:636–45.
Lambrecht BN, Hammad H, Fahy JV. The cytokines of asthma. Immunity. 2019;50:975–91.
Traves SL, Donnelly LE. Th17 cells in airway diseases. Curr Mol Med. 2008;8:416–26.
Munroe ME, Businga TR, Kline JN, Bishop GA. Anti-inflammatory effects of the neurotransmitter agonist Honokiol in a mouse model of allergic asthma. J Immunol. 2010;185:5586–97.
Villarino AV, Kanno Y, Ferdinand JR, O’Shea JJ. Mechanisms of Jak/STAT signaling in immunity and disease. J Immunol. 2015;194:21–27.
O’Shea JJ, Schwartz DM, Villarino AV, Gadina M, Mcinnes IB, Laurence A. The JAK-STAT pathway: impact on human disease and therapeutic intervention. Annu Rev Med. 2015;66:311–28.
Heim MH. The Jak-STAT pathway: specific signal transduction from the cell membrane to the nucleus. Eur J Clin Investig. 2015;26:1–12.
Durant L, Watford WT, Ramos HL, Laurence A, Vahedi G, Wei L, et al. Diverse targets of the transcription factor STAT3 contribute to T cell pathogenicity and homeostasis. Immunity. 2010;32:605.
Funding
The work was supported by the Youth Program of Natural Science Foundation of China (No. 81900017), the Natural Science Foundation of China (Nos. 81770023 and 81770022), the Natural Science Foundation of Fujian Province (2019J01303), and the Joint Funds for the Innovation of Science and Technology, Fujian Province (2018Y9075).
Author information
Authors and Affiliations
Contributions
The study was conceived and designed by WG and XG. XC and RY conducted experiments. XC, RY, XL, WY, WG, and XG analyzed the data and interpreted the results. XC, RY, and XG wrote the paper. We confirm that all the listed authors have participated actively in the study, and have reviewed and approved the submitted paper.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethics approval
The experimental protocols were approved by the Ethics Committee of Fujian Medical University, and the study was conducted in accordance with the guidelines published in the NIH Guide for the Care and Use of Laboratory Animals.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen, X., Yue, R., Li, X. et al. Surfactant protein A modulates the activities of the JAK/STAT pathway in suppressing Th1 and Th17 polarization in murine OVA-induced allergic asthma. Lab Invest 101, 1176–1185 (2021). https://doi.org/10.1038/s41374-021-00618-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41374-021-00618-1