Abstract
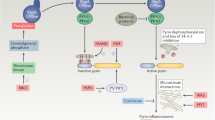
The notion of 'autoinflammatory' disease was introduced at the end of the 1990s, and, since then, this concept has rapidly evolved. As a result, multiple definitions of autoinflammatory disease, and classifications of conditions encompassed by these definitions, have been proposed; this succession highlights advances that have been made in understanding of the innate immune system, and especially the roles of IL-1β and the inflammasome in autoinflammtory conditions. However, the definitions and classifications that have been suggested to date face a number of structure and content issues. We therefore propose another, more clinically-oriented, definition: autoinflammatory diseases are diseases with clinical signs of inflammation, associated with elevated levels of acute-phase reactants, which are attributable to dysfunction of the innate immune system, genetically-determined or triggered by an endogenous factor. From this foundation, we propose a clinically-based classification of autoinflammatory diseases, and go on to discuss how immunological diseases as a whole, including autoimmune diseases, can be appropriately located within a continuum only if the classification process is multidimensional. For this purpose, we appeal to the philosophical concepts of family resemblance and signature.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
McDermott, M. F. et al. Germline mutations in the extracellular domains of the 55 kDa TNF receptor, TNFR1, define a family of dominantly inherited autoinflammatory syndromes. Cell 97, 133–144 (1999).
French FMF Consortium. A candidate gene for familial Mediterranean fever. Nat. Genet. 17, 25–31 (1997).
International FMF Consortium. Ancient missense mutations in a new member of the RoRet gene family are likely to cause familial Mediterranean fever. Cell 90, 797–807 (1997).
McGonagle, D. & McDermott, M. F. A proposed classification of the immunological diseases. PLoS Med. 3, e297 (2006).
Hoffman, H. M., Mueller, J. L., Broide, D. H., Wanderer, A. A. & Kolodner, R. D. Mutation of a new gene encoding a putative pyrin-like protein causes familial cold autoinflammatory syndrome and Muckle–Wells syndrome. Nat. Genet. 29, 301–305 (2001).
Martinon, F., Burns, K. & Tschopp, J. The inflammasome: a molecular platform triggering activation of inflammatory caspases and processing of proIL-β. Mol. Cell 10, 417–426 (2002).
Agostini, L. et al. NALP3 forms an IL-1β-processing inflammasome with increased activity in Muckle–Wells autoinflammatory disorder. Immunity 20, 319–325 (2004).
Hawkins, P. N., Lachmann, H. J. & McDermott, M. F. Interleukin-1-receptor antagonist in the Muckle–Wells syndrome. N. Engl. J. Med. 348, 2583–2584 (2003).
Dinarello, C. A. Mutations in cryopyrin: bypassing roadblocks in the caspase 1 inflammasome for interleukin-1β secretion and disease activity. Arthritis Rheum. 56, 2817–2822 (2007).
de Koning, H. D., Schalkwijk, J., van der Meer, J. W. & Simon, A. Successful canakinumab treatment identifies IL-1β as a pivotal mediator in Schnitzler syndrome. J. Allergy Clin. Immunol. 128, 1352–1354 (2011).
Krause, K., Feist, E., Fiene, M., Kallinich, T. & Maurer, M. Complete remission in 3 of 3 anti-IL-6-treated patients with Schnitzler syndrome. J. Allergy Clin. Immunol. 129, 848–850 (2012).
Arima, K. et al. Proteasome assembly defect due to a proteasome subunit beta type 8 (PSMB8) mutation causes the autoinflammatory disorder, Nakajo–Nishimura syndrome. Proc. Natl Acad. Sci. USA 108, 14914–14919 (2011).
Dinarello, C. A. Blocking interleukin-1β in acute and chronic autoinflammatory diseases. J. Intern. Med. 269, 16–28 (2011).
Murphy, E. A. The Logic of Medicine. 122–126 (Johns Hopkins University Press, Baltimore, 1976).
Masters, S. L., Simon, A., Aksentijevich, I. & Kastner, D. L. Horror autoinflammaticus: the molecular pathophysiology of autoinflammatory disease. Annu. Rev. Immunol. 27, 621–668 (2009).
Kastner, D. L., Aksentijevich, I. & Goldbach-Mansky, R. Autoinflammatory disease reloaded: a clinical perspective. Cell 140, 784–790 (2010).
Wittgenstein, L. Philosophical investigations (Blackwell, Oxford, 1953).
Verbsky, J. W. Monogenic causes of inflammatory disease in rheumatology. Curr. Opin. Rheumatol. 24, 506–514 (2012).
Boisson, B. et al. Immunodeficiency, autoinflammation and amylopectinosis in humans with inherited HOIL-1 and LUBAC deficiency. Nat. Immunol. 13, 1178–1186 (2012).
Ombrello, M. J. et al. Cold urticaria, immunodeficiency, and autoimmunity related to PLCG2 deletions. N. Engl. J. Med. 366, 330–338 (2012).
Lachmann, H. J. et al. Natural history and outcome in systemic AA amyloidosis. N. Engl. J. Med. 356, 2361–2371 (2007).
Foucault, M. Les Mots et les Choses 17–29 (Gallimard, Paris, 1966).
Feramisco, J. D., Sadreyev, R. I., Murray, M. L., Grishin, N. V. & Tsao, H. Phenotypic and genotypic analyses of genetic skin disease through the Online Mendelian Inheritance in Man (OMIM) database. J. Invest. Dermatol. 129, 2628–2636 (2009).
Samarghitean, C., Ortutay, C. & Vihinen, M. Systematic classification of primary immunodeficiencies based on clinical, pathological, and laboratory parameters. J. Immunol. 183, 7569–7575 (2009).
Author information
Authors and Affiliations
Contributions
G. Grateau and O. Steichen researched the data for the article. G. Grateau, S. Amselem and O. Steichen contributed equally to writing the article. All authors provided a substantial contribution to discussion of the content and to review and/or editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Table 1
Proposed categories of autoinflammatory diseases from selected classifications (PDF 94 kb)
Supplementary Table 2
Classification of autoinflammatory diseases according to their main pathological features* (DOC 41 kb)
Supplementary Figure 1
The immune disease continuum. Schematic representation of the continuum of inflammatory diseases proposed by McGonagle and McDermott.4 (PDF 149 kb)
Supplementary Figure 2
The continuum of immune diseases along the gradient of amyloidosis prevalence. (PDF 130 kb)
Rights and permissions
About this article
Cite this article
Grateau, G., Hentgen, V., Stojanovic, K. et al. How should we approach classification of autoinflammatory diseases?. Nat Rev Rheumatol 9, 624–629 (2013). https://doi.org/10.1038/nrrheum.2013.101
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2013.101
This article is cited by
-
Breaking down the fences among registries on autoinflammatory diseases: the E-Merge project
Orphanet Journal of Rare Diseases (2023)
-
Immune cartography of macrophage activation syndrome in the COVID-19 era
Nature Reviews Rheumatology (2021)
-
Suboptimal vaccination coverage of recommended vaccines among French children with recurrent autoinflammatory fever syndromes: a study from the Juvenile Inflammatory Rheumatism cohort
Clinical Rheumatology (2021)
-
Moving towards a systems-based classification of innate immune-mediated diseases
Nature Reviews Rheumatology (2020)
-
Modulation of autoimmune pathogenesis by T cell-triggered inflammatory cell death
Nature Communications (2019)