Key Points
-
Clinical studies suggest that type 2 diabetes mellitus (T2DM) is a risk factor for cognitive decline and dementia, and have found evidence that insulin resistance (IR) occurs in the brain of patients with T2DM and Alzheimer disease (AD).
-
Structural and functional deficits in synaptic plasticity, as well as impairments in a variety of behavioural tests of learning and memory, are observed in the hippocampus in rodent models of T2DM.
-
Evidence for hippocampal IR has also been observed in rodent models of AD.
-
Data from these experimental studies suggest that hippocampal IR is an important mechanistic mediator of the synaptic plasticity and cognitive deficits in T2DM and AD.
-
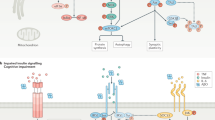
Several pathological features of T2DM and AD may contribute to the development of hippocampal IR, including increases in oxidative stress and in the amount of pro-inflammatory cytokines and amyloid-β peptides, as well as hypothalamic–pituitary–adrenal axis dysfunction.
-
Importantly, both lifestyle (diet and exercise) and pharmacological interventions that are known to alleviate peripheral IR effectively restore hippocampal neuroplasticity in rodent models of T2DM and AD, and this effect may be due to restoration of insulin signalling in the hippocampus.
Abstract
Clinical studies suggest a link between type 2 diabetes mellitus (T2DM) and insulin resistance (IR) and cognitive dysfunction, but there are significant gaps in our knowledge of the mechanisms underlying this relationship. Animal models of IR help to bridge these gaps and point to hippocampal IR as a potential mediator of cognitive dysfunction in T2DM, as well as in Alzheimer disease (AD). This Review highlights these observations and discusses intervention studies which suggest that the restoration of insulin activity in the hippocampus may be an effective strategy to alleviate the cognitive decline associated with T2DM and AD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
International Diabetes Federation. IDF Diabetes Atlas 6th edition. IDF [online], (2013).
Kahn, S. E., Cooper, M. E. & Del Prato, S. Pathophysiology and treatment of type 2 diabetes: perspectives on the past, present, and future. Lancet 383, 1068–1083 (2014).
Koekkoek, P. S., Kappelle, L. J., van den, B. E., Rutten, G. E. & Biessels, G. J. Cognitive function in patients with diabetes mellitus: guidance for daily care. Lancet Neurol. 14, 329–340 (2015).
Luchsinger, J. A. Adiposity, hyperinsulinemia, diabetes and Alzheimer's disease: an epidemiological perspective. Eur. J. Pharmacol. 585, 119–129 (2008).
Gudala, K., Bansal, D., Schifano, F. & Bhansali, A. Diabetes mellitus and risk of dementia: a meta-analysis of prospective observational studies. J. Diabetes Investig. 4, 640–650 (2013).
Biessels, G. J., Strachan, M. W., Visseren, F. L., Kappelle, L. J. & Whitmer, R. A. Dementia and cognitive decline in type 2 diabetes and prediabetic stages: towards targeted interventions. Lancet Diabetes Endocrinol. 2, 246–255 (2014). This review highlights the impact of T2DM and pre-diabetic stages of IR on cognition at different ages.
Norton, S., Matthews, F. E., Barnes, D. E., Yaffe, K. & Brayne, C. Potential for primary prevention of Alzheimer's disease: an analysis of population-based data. Lancet Neurol. 13, 788–794 (2014).
Kiliaan, A. J., Arnoldussen, I. A. & Gustafson, D. R. Adipokines: a link between obesity and dementia? Lancet Neurol. 13, 913–923 (2014).
Palta, P., Schneider, A. L., Biessels, G. J., Touradji, P. & Hill-Briggs, F. Magnitude of cognitive dysfunction in adults with type 2 diabetes: a meta-analysis of six cognitive domains and the most frequently reported neuropsychological tests within domains. J. Int. Neuropsychol. Soc. 20, 278–291 (2014).
Convit, A., Wolf, O. T., Tarshish, C. & de Leon, M. J. Reduced glucose tolerance is associated with poor memory performance and hippocampal atrophy among normal elderly. Proc. Natl Acad. Sci. USA 100, 2019–2022 (2003).
Bruehl, H., Sweat, V., Hassenstab, J., Polyakov, V. & Convit, A. Cognitive impairment in nondiabetic middle-aged and older adults is associated with insulin resistance. J. Clin. Exp. Neuropsychol. 32, 487–493 (2010).
Brundel, M., Kappelle, L. J. & Biessels, G. J. Brain imaging in type 2 diabetes. Eur. Neuropsychopharmacol. 24, 1967–1981 (2014).
Moran, C. et al. Brain atrophy in type 2 diabetes: regional distribution and influence on cognition. Diabetes Care 36, 4036–4042 (2013).
Espeland, M. A. et al. Influence of type 2 diabetes on brain volumes and changes in brain volumes: results from the Women's Health Initiative magnetic resonance imaging studies. Diabetes Care 36, 90–97 (2013).
Luitse, M. J., Biessels, G. J., Rutten, G. E. & Kappelle, L. J. Diabetes, hyperglycaemia, and acute ischaemic stroke. Lancet Neurol. 11, 261–271 (2012).
Del Bene, A. et al. Is type 2 diabetes related to leukoaraiosis? An updated review. Acta Neurol. Scand. 132, 147–155 (2015).
Qiu, C. et al. Diabetes, markers of brain pathology and cognitive function: the Age, Gene/Environment Susceptibility–Reykjavik Study. Ann. Neurol. 75, 138–146 (2014). This large population-based study identifies structural changes in the brain in individuals with T2DM and finds that these structural changes are associated with impaired cognitive performance.
Musen, G. et al. Resting-state brain functional connectivity is altered in type 2 diabetes. Diabetes 61, 2375–2379 (2012).
Reijmer, Y. D. et al. Microstructural white matter abnormalities and cognitive functioning in type 2 diabetes: a diffusion tensor imaging study. Diabetes Care 36, 137–144 (2013).
Yarchoan, M. & Arnold, S. E. Repurposing diabetes drugs for brain insulin resistance in Alzheimer disease. Diabetes 63, 2253–2261 (2014).
De Felice, F. G. & Ferreira, S. T. Inflammation, defective insulin signaling, and mitochondrial dysfunction as common molecular denominators connecting type 2 diabetes to Alzheimer disease. Diabetes 63, 2262–2272 (2014).
Ahtiluoto, S. et al. Diabetes, Alzheimer disease, and vascular dementia: a population-based neuropathologic study. Neurology 75, 1195–1202 (2010).
Nelson, P. T. et al. Human cerebral neuropathology of type 2 diabetes mellitus. Biochim. Biophys. Acta 1792, 454–469 (2009).
Roberts, R. O. et al. Diabetes and elevated hemoglobin A1c levels are associated with brain hypometabolism but not amyloid accumulation. J. Nucl. Med. 55, 759–764 (2014). This is the first detailed investigation to explore the links between T2DM and brain Aβ accumulation in humans in vivo.
Thambisetty, M. et al. Glucose intolerance, insulin resistance, and pathological features of Alzheimer disease in the Baltimore Longitudinal Study of Aging. JAMA Neurol. 70, 1167–1172 (2013).
Willette, A. A. et al. Insulin resistance predicts brain amyloid deposition in late middle-aged adults. Alzheimers Dement. 11, 504–510 (2015).
Jackson, K. et al. Amylin deposition in the brain: a second amyloid in Alzheimer disease? Ann. Neurol. 74, 517–526 (2013).
Ossenkoppele, R. et al. Prevalence of amyloid PET positivity in dementia syndromes: a meta-analysis. JAMA 313, 1939–1949 (2015).
Schneider, J. A., Arvanitakis, Z., Bang, W. & Bennett, D. A. Mixed brain pathologies account for most dementia cases in community-dwelling older persons. Neurology 69, 2197–2204 (2007).
Marks, J. L., Porte, D. Jr, Stahl, W. L. & Baskin, D. G. Localization of insulin receptor mRNA in rat brain by in situ hybridization. Endocrinology 127, 3234–3236 (1991).
Dore, S., Kar, S., Rowe, W. & Quirion, R. Distribution and levels of [125I]IGF-I, [125I]IGF-II and [125I]insulin receptor binding sites in the hippocampus of aged memory-unimpaired and -impaired rats. Neuroscience 80, 1033–1040 (1997).
Park, C. R., Seely, R. J., Craft, S. & Woods, S. C. Intracerebroventricular insulin enhances memory in a passive-avoidance task. Physiol. Behav. 68, 509–514 (2000).
Benedict, C. et al. Intranasal insulin improves memory in humans. Psychoneuroendocrinology 29, 1326–1334 (2004).
Haj-ali, V., Mohaddes, G. & Babri, S. H. Intracerebroventricular insulin improves spatial learning and memory in male Wistar rats. Behav. Neurosci. 123, 1309–1314 (2009).
Moosavi, M., Naghdi, N. & Choopani, S. Intra CA1 insulin microinjection improves memory consolidation and retrieval. Peptides 28, 1029–1034 (2007).
McNay, E. C. et al. Hippocampal memory processes are modulated by insulin and high-fat-induced insulin resistance. Neurobiol. Learn. Mem. 93, 546–553 (2010).
Stern, S. A., Chen, D. Y. & Alberini, C. M. The effect of insulin and insulin-like growth factors on hippocampus- and amygdala-dependent long-term memory formation. Learn. Mem. 21, 556–563 (2014).
Grillo, C. A., Piroli, G. G., Hendry, R. M. & Reagan, L. P. Insulin-stimulated translocation of GLUT4 to the plasma membrane in rat hippocampus is PI3-kinase dependent. Brain Res. 1296, 35–45 (2009).
Nelson, T. J., Sun, M. K., Hongpaisan, J. & Alkon, D. L. Insulin, PKC signaling pathways and synaptic remodeling during memory storage and neuronal repair. Eur. J. Pharmacol. 585, 76–87 (2008).
Ghasemi, R., Haeri, A., Dargahi, L., Mohamed, Z. & Ahmadiani, A. Insulin in the brain: sources, localization and functions. Mol. Neurobiol. 47, 145–171 (2013).
Banks, W. A., Owen, J. B. & Erickson, M. A. Insulin in the brain: there and back again. Pharmacol. Ther. 136, 82–93 (2012).
Levin, B. E. & Routh, V. H. Role of the brain in energy balance and obesity. Am. J. Physiol. 271, R491–R500 (1996).
Levin, B. E., Dunn-Meynell, A. A., Balkan, B. & Keesey, R. E. Selective breeding for diet-induced obesity and resistance in Sprague-Dawley rats. Am. J. Physiol. 273, R725–R730 (1997).
Reagan, L. P. Diabetes as a chronic metabolic stressor: causes, consequences and clinical complications. Exp. Neurol. 233, 68–78 (2012).
Greenwood, C. E. & Winocur, G. Learning and memory impairment in rats fed a high saturated fat diet. Behav. Neural Biol. 53, 74–87 (1990). This is the first work to demonstrate that the detrimental effects of an HFD in rodents extend to the CNS and induce deficits in hippocampus-dependent learning.
Greenwood, C. E. & Winocur, G. Cognitive impairment in rats fed high-fat diets: a specific effect of saturated fatty-acid intake. Behav. Neurosci. 110, 451–459 (1996).
Winocur, G. et al. Memory impairment in obese Zucker rats: an investigation of cognitive function in an animal model of insulin resistance and obesity. Behav. Neurosci. 119, 1389–1395 (2005).
Kamal, A., Ramakers, G. M., Gispen, W. H. & Biessels, G. J. Hyperinsulinemia in rats causes impairment of spatial memory and learning with defects in hippocampal synaptic plasticity by involvement of postsynaptic mechanisms. Exp. Brain Res. 226, 45–51 (2013).
Stranahan, A. M. et al. Diet-induced insulin resistance impairs hippocampal synaptic plasticity and cognition in middle-aged rats. Hippocampus 18, 1085–1088 (2008).
Boitard, C. et al. Juvenile, but not adult exposure to high-fat diet impairs relational memory and hippocampal neurogenesis in mice. Hippocampus 22, 2095–2100 (2012).
Li, X. L. et al. Impairment of long-term potentiation and spatial memory in leptin receptor-deficient rodents. Neuroscience 113, 607–615 (2002).
Stranahan, A. M. et al. Diabetes impairs hippocampal function through glucocorticoid-mediated effects on new and mature neurons. Nat. Neurosci. 11, 309–317 (2008).
Grillo, C. A. et al. Obesity/hyperleptinemic phenotype impairs structural and functional plasticity in the rat hippocampus. Physiol. Behav. 105, 138–144 (2011).
Kanoski, S. E., Meisel, R. L., Mullins, A. J. & Davidson, T. L. The effects of energy-rich diets on discrimination reversal learning and on BDNF in the hippocampus and prefrontal cortex of the rat. Behav. Brain Res. 182, 57–66 (2007).
Kanoski, S. E., Zhang, Y., Zheng, W. & Davidson, T. L. The effects of a high-energy diet on hippocampal function and blood–brain barrier integrity in the rat. J. Alzheimers Dis. 21, 207–219 (2010).
Baskin, D. G. et al. Genetically obese Zucker rats have abnormally low brain insulin content. Life Sci. 36, 627–633 (1985).
Stein, L. J. et al. Reduced effect of experimental peripheral hyperinsulinemia to elevate cerebrospinal fluid insulin concentrations of obese Zucker rats. Endocrinology 121, 1611–1615 (1987).
Urayama, A. & Banks, W. A. Starvation and triglycerides reverse the obesity-induced impairment of insulin transport at the blood–brain barrier. Endocrinology 149, 3592–3597 (2008).
Mielke, J. G. et al. A biochemical and functional characterization of diet-induced brain insulin resistance. J. Neurochem. 93, 1568–1578 (2005).
Piroli, G. G. et al. Corticosterone impairs insulin-stimulated translocation of GLUT4 in the rat hippocampus. Neuroendocrinology 85, 71–80 (2007).
Pratchayasakul, W. et al. Effects of high-fat diet on insulin receptor function in rat hippocampus and the level of neuronal corticosterone. Life Sci. 88, 619–627 (2011).
Arnold, S. E. et al. High fat diet produces brain insulin resistance, synaptodendritic abnormalities and altered behavior in mice. Neurobiol. Dis. 67, 79–87 (2014).
Morrison, C. D. et al. High fat diet increases hippocampal oxidative stress and cognitive impairment in aged mice: implications for decreased Nrf2 signaling. J. Neurochem. 114, 1581–1589 (2010).
Agrawal, R. & Gomez-Pinilla, F. 'Metabolic syndrome' in the brain: deficiency in omega-3 fatty acid exacerbates dysfunctions in insulin receptor signalling and cognition. J. Physiol. 590, 2485–2499 (2012).
Plotsky, P. M., Thrivikraman, K. V., Watts, A. G. & Hauger, R. L. Hypothalamic-pituitary-adrenal axis function in the Zucker obese rat. Endocrinology 130, 1931–1941 (1992).
Dinel, A. L. et al. Cognitive and emotional alterations are related to hippocampal inflammation in a mouse model of metabolic syndrome. PLoS ONE 6, e24325 (2011).
Bomfim, T. R. et al. An anti-diabetes agent protects the mouse brain from defective insulin signaling caused by Alzheimer's disease- associated Aβ oligomers. J. Clin. Invest. 122, 1339–1353 (2012). This study illustrates that Aβ oligomers induce IR in hippocampal neuronal cultures, APP/PS1 mice and non-human primates, and also shows that these pathologies are inhibited by the GLP1 analogue exendin-4.
Boitard, C. et al. Impairment of hippocampal-dependent memory induced by juvenile high-fat diet intake is associated with enhanced hippocampal inflammation in rats. Brain Behav. Immun. 40, 9–17 (2014).
Pistell, P. J. et al. Cognitive impairment following high fat diet consumption is associated with brain inflammation. J. Neuroimmunol. 219, 25–32 (2010).
Erion, J. R. et al. Obesity elicits interleukin 1-mediated deficits in hippocampal synaptic plasticity. J. Neurosci. 34, 2618–2631 (2014).
Kwon, H. & Pessin, J. E. Adipokines mediate inflammation and insulin resistance. Front. Endocrinol. (Lausanne) 4, 71 (2013).
Henriksen, E. J., Diamond-Stanic, M. K. & Marchionne, E. M. Oxidative stress and the etiology of insulin resistance and type 2 diabetes. Free Radic. Biol. Med. 51, 993–999 (2011).
Park, H. R. et al. A high-fat diet impairs neurogenesis: involvement of lipid peroxidation and brain-derived neurotrophic factor. Neurosci. Lett. 482, 235–239 (2010).
Hwang, I. K. et al. Strain-specific differences in cell proliferation and differentiation in the dentate gyrus of C57BL/6N and C3H/HeN mice fed a high fat diet. Brain Res. 1241, 1–6 (2008).
Lindqvist, A. et al. High-fat diet impairs hippocampal neurogenesis in male rats. Eur. J. Neurol. 13, 1385–1388 (2006).
Granholm, A. C. et al. Effects of a saturated fat and high cholesterol diet on memory and hippocampal morphology in the middle-aged rat. J. Alzheimers Dis. 14, 133–145 (2008).
Freeman, L. R., Haley-Zitlin, V., Stevens, C. & Granholm, A. C. Diet-induced effects on neuronal and glial elements in the middle-aged rat hippocampus. Nutr. Neurosci. 14, 32–44 (2011).
Tomassoni, D. et al. Astrogliosis in the brain of obese Zucker rat: a model of metabolic syndrome. Neurosci. Lett. 543, 136–141 (2013).
Freeman, L. R. & Granholm, A. C. Vascular changes in rat hippocampus following a high saturated fat and cholesterol diet. J. Cereb. Blood Flow Metab. 32, 643–653 (2012).
Davidson, T. L. et al. Inter-relationships among diet, obesity and hippocampal-dependent cognitive function. Neuroscience 253, 110–122 (2013).
Davidson, T. L. et al. The effects of a high-energy diet on hippocampal-dependent discrimination performance and blood–brain barrier integrity differ for diet-induced obese and diet-resistant rats. Physiol. Behav. 107, 26–33 (2012).
Kanoski, S. E. & Davidson, T. L. Different patterns of memory impairments accompany short- and longer-term maintenance on a high-energy diet. J. Exp. Psychol. Anim. Behav. Process. 36, 313–319 (2010).
Gerges, N. Z., Aleisa, A. M. & Alkadhi, K. A. Impaired long-term potentiation in obese Zucker rats: possible involvement of presynaptic mechanism. Neuroscience 120, 535–539 (2003).
Alzoubi, K. H., Aleisa, A. M. & Alkadhi, K. A. Impairment of long-term potentiation in the CA1, but not dentate gyrus, of the hippocampus in obese Zucker rats: role of calcineurin and phosphorylated CaMKII. J. Mol. Neurosci. 27, 337–346 (2005).
Karimi, S. A. et al. Effect of high-fat diet and antioxidants on hippocampal long-term potentiation in rats: an in vivo study. Brain Res. 1539, 1–6 (2013).
Grillo, C. A. et al. Obesity/hyperleptinemic phenotype adversely affects hippocampal plasticity: effects of dietary restriction. Physiol. Behav. 104, 235–241 (2011).
Nisticò, R. et al. Insulin receptor β-subunit haploinsufficiency impairs hippocampal late-phase LTP and recognition memory. Neuromolecular Med. 14, 262–269 (2012).
Costello, D. A. et al. Brain deletion of insulin receptor substrate 2 disrupts hippocampal synaptic plasticity and metaplasticity. PLoS ONE 7, e31124 (2012).
Belanger, A., Lavoie, N., Trudeau, F., Massicotte, G. & Gagnon, S. Preserved LTP and water maze learning in hyperglycaemic-hyperinsulinemic ZDF rats. Physiol. Behav. 83, 483–494 (2004).
Grillo, C. A. et al. Hippocampal insulin resistance impairs spatial learning and synaptic plasticity. Diabetes http://dx.doi.org/10.2337/db15-0596 (2015). This research demonstrates that hippocampal region-specific decreases in insulin receptor activity impair hippocampal synaptic plasticity in the absence of peripheral IR.
Takeda, S. et al. Diabetes-accelerated memory dysfunction via cerebrovascular inflammation and Aβ deposition in an Alzheimer mouse model with diabetes. Proc. Natl Acad. Sci. USA 107, 7036–7041 (2010).
Bhat, N. R. & Thirumangalakudi, L. Increased tau phosphorylation and impaired brain insulin/IGF signaling in mice fed a high fat/high cholesterol diet. J. Alzheimers Dis. 36, 781–789 (2013).
Li, J., Deng, J., Sheng, W. & Zuo, Z. Metformin attenuates Alzheimer's disease-like neuropathology in obese, leptin-resistant mice. Pharmacol. Biochem. Behav. 101, 564–574 (2012).
Kim, B., Backus, C., Oh, S., Hayes, J. M. & Feldman, E. L. Increased tau phosphorylation and cleavage in mouse models of type 1 and type 2 diabetes. Endocrinology 150, 5294–5301 (2009).
Wrighten, S. A., Piroli, G. G., Grillo, C. A. & Reagan, L. P. A look inside the diabetic brain: contributors to diabetes-induced brain aging. Biochim. Biophys. Acta 1792, 444–453 (2008).
Schubert, M. et al. Role for neuronal insulin resistance in neurodegenerative diseases. Proc. Natl Acad. Sci. USA 101, 3100–3105 (2004).
Pearson-Leary, J. & McNay, E. C. Intrahippocampal administration of amyloid-β1-42 oligomers acutely impairs spatial working memory, insulin signaling, and hippocampal metabolism. J. Alzheimers Dis. 30, 413–422 (2012).
Zhao, W. Q. et al. Amyloid beta oligomers induce impairment of neuronal insulin receptors. FASEB J. 22, 246–260 (2008).
Talbot, K. et al. Demonstrated brain insulin resistance in Alzheimer's disease patients is associated with IGF-1 resistance, IRS-1 dysregulation, and cognitive decline. J. Clin. Invest. 122, 1316–1338 (2012).
Fadel, J. R., Jolivalt, C. G. & Reagan, L. P. Food for thought: the role of appetitive peptides in age-related cognitive decline. Ageing Res. Rev. 12, 764–776 (2013).
McNay, E. C. & Recknagel, A. K. Brain insulin signaling: a key component of cognitive processes and a potential basis for cognitive impairment in type 2 diabetes. Neurobiol. Learn. Mem. 96, 432–442 (2011).
Marks, D. R., Tucker, K., Cavallin, M. A., Mast, T. G. & Fadool, D. A. Awake intranasal insulin delivery modifies protein complexes and alters memory, anxiety, and olfactory behaviors. J. Neurosci. 29, 6734–6751 (2009).
McNeilly, A. D., Williamson, R., Balfour, D. J., Stewart, C. A. & Sutherland, C. A high-fat-diet-induced cognitive deficit in rats that is not prevented by improving insulin sensitivity with metformin. Diabetologia 55, 3061–3070 (2012).
Wang, J. et al. Metformin activates an atypical PKC-CBP pathway to promote neurogenesis and enhance spatial memory formation. Cell Stem Cell 11, 23–35 (2012).
Pathan, A. R., Gaikwad, A. B., Viswanad, B. & Ramarao, P. Rosiglitazone attenuates the cognitive deficits induced by high fat diet feeding in rats. Eur. J. Pharmacol. 589, 176–179 (2008).
Pipatpiboon, N., Pratchayasakul, W., Chattipakorn, N. & Chattipakorn, S. C. PPARγ agonist improves neuronal insulin receptor function in hippocampus and brain mitochondria function in rats with insulin resistance induced by long term high-fat diets. Endocrinology 153, 329–338 (2012).
Fakhoury, W. K., Lereun, C. & Wright, D. A meta-analysis of placebo-controlled clinical trials assessing the efficacy and safety of incretin-based medications in patients with type 2 diabetes. Pharmacology 86, 44–57 (2010).
Angeli, F. S. & Shannon, R. P. Incretin-based therapies: can we achieve glycemic control and cardioprotection? J. Endocrinol. 221, T17–T30 (2014).
Pintana, H., Apaijai, N., Chattipakorn, N. & Chattipakorn, S. C. DPP-4 inhibitors improve cognition and brain mitochondrial function of insulin-resistant rats. J. Endocrinol. 218, 1–11 (2013).
Pipatpiboon, N., Pintana, H., Pratchayasakul, W., Chattipakorn, N. & Chattipakorn, S. C. DPP4-inhibitor improves neuronal insulin receptor function, brain mitochondrial function and cognitive function in rats with insulin resistance induced by high-fat diet consumption. Eur. J. Neurosci. 37, 839–849 (2013).
Gault, V. A., Porter, W. D., Flatt, P. R. & Holscher, C. Actions of exendin-4 therapy on cognitive function and hippocampal synaptic plasticity in mice fed a high-fat diet. Int. J. Obes. (Lond.) 34, 1341–1344 (2010).
Agrawal, R. et al. Deterioration of plasticity and metabolic homeostasis in the brain of the UCD-T2DM rat model of naturally occurring type-2 diabetes. Biochim. Biophys. Acta 1842, 1313–1323 (2014).
Porter, W. D., Flatt, P. R., Holscher, C. & Gault, V. A. Liraglutide improves hippocampal synaptic plasticity associated with increased expression of Mash1 in ob/ob mice. Int. J. Obes. (Lond.) 37, 678–684 (2013).
Porter, D. W., Kerr, B. D., Flatt, P. R., Holscher, C. & Gault, V. A. Four weeks administration of Liraglutide improves memory and learning as well as glycaemic control in mice with high fat dietary-induced obesity and insulin resistance. Diabetes Obes. Metab. 12, 891–899 (2010).
Porter, D. W., Irwin, N., Flatt, P. R., Holscher, C. & Gault, V. A. Prolonged GIP receptor activation improves cognitive function, hippocampal synaptic plasticity and glucose homeostasis in high-fat fed mice. Eur. J. Pharmacol. 650, 688–693 (2011).
Molteni, R. et al. Exercise reverses the harmful effects of consumption of a high-fat diet on synaptic and behavioral plasticity associated to the action of brain-derived neurotrophic factor. Neuroscience 123, 429–440 (2004).
Noble, E. E. et al. Exercise reduces diet-induced cognitive decline and increases hippocampal brain-derived neurotrophic factor in CA3 neurons. Neurobiol. Learn. Mem. 114, 40–50 (2014).
Grayson, B. E. et al. Improvements in hippocampal-dependent memory and microglial infiltration with calorie restriction and gastric bypass surgery, but not with vertical sleeve gastrectomy. Int. J. Obes. (Lond.) 38, 349–356 (2014).
Stranahan, A. M. et al. Voluntary exercise and caloric restriction enhance hippocampal dendritic spine density and BDNF levels in diabetic mice. Hippocampus 19, 951–961 (2009).
Mattson, M. P. Energy intake and exercise as determinants of brain health and vulnerability to injury and disease. Cell Metab. 16, 706–722 (2012).
Lautenschlager, N. T. et al. Effect of physical activity on cognitive function in older adults at risk for Alzheimer disease: a randomized trial. JAMA 300, 1027–1037 (2008).
Baker, L. D. et al. Effects of aerobic exercise on mild cognitive impairment: a controlled trial. Arch. Neurol. 67, 71–79 (2010).
Hillman, C. H., Erickson, K. I. & Kramer, A. F. Be smart, exercise your heart: exercise effects on brain and cognition. Nat. Rev. Neurosci. 9, 58–65 (2008).
Fotuhi, M., Do, D. & Jack, C. Modifiable factors that alter the size of the hippocampus with ageing. Nat. Rev. Neurol. 8, 189–202 (2012).
Baker, L. D. et al. Aerobic exercise improves cognition for older adults with glucose intolerance, a risk factor for Alzheimer's disease. J. Alzheimers Dis. 22, 569–579 (2010).
Alosco, M. L. et al. Cognitive function after bariatric surgery: evidence for improvement 3 years after surgery. Am. J. Surg. 207, 870–876 (2014).
Yang, Y. et al. Intranasal insulin ameliorates tau hyperphosphorylation in a rat model of type 2 diabetes. J. Alzheimers Dis. 33, 329–338 (2013).
Han, W. N. et al. Liraglutide protects against amyloid-β protein-induced impairment of spatial learning and memory in rats. Neurobiol. Aging 34, 576–588 (2013).
Cai, H. Y. et al. Lixisenatide rescues spatial memory and synaptic plasticity from amyloid β protein-induced impairments in rats. Neuroscience 277, 6–13 (2014).
Gengler, S., McClean, P. L., McCurtin, R., Gault, V. A. & Hölscher, C. Val(8)GLP-1 rescues synaptic plasticity and reduces dense core plaques in APP/PS1 mice. Neurobiol. Aging 33, 265–276 (2012).
McClean, P. L., Parthsarathy, V., Faivre, E. & Hölscher, C. The diabetes drug liraglutide prevents degenerative processes in a mouse model of Alzheimer's disease. J. Neurosci. 31, 6587–6594 (2011).
McClean, P. L. & Hölscher, C. Liraglutide can reverse memory impairment, synaptic loss and reduce plaque load in aged APP/PS1 mice, a model of Alzheimer's disease. Neuropharmacology 76, 57–67 (2014).
Cholerton, B., Baker, L. D. & Craft, S. Insulin, cognition, and dementia. Eur. J. Pharmacol. 719, 170–179 (2013).
Kern, W., Born, J., Schreiber, H. & Fehm, H. L. Central nervous system effects of intranasally administered insulin during euglycemia in men. Diabetes 48, 557–563 (1999).
Craft, S. et al. Intranasal insulin therapy for Alzheimer disease and amnestic mild cognitive impairment: a pilot clinical trial. Arch. Neurol. 69, 29–38 (2012).
Novak, V. et al. Enhancement of vasoreactivity and cognition by intranasal insulin in type 2 diabetes. Diabetes Care 37, 751–759 (2014).
Voss, M. W., Vivar, C., Kramer, A. F. & van Praag, H. Bridging animal and human models of exercise-induced brain plasticity. Trends Cogn. Sci. 17, 525–544 (2013).
Hunter, K. & Hölscher, C. Drugs developed to treat diabetes, liraglutide and lixisenatide, cross the blood brain barrier and enhance neurogenesis. BMC Neurosci. 13, 33 (2012).
Kastin, A. J., Akerstrom, V. & Pan, W. Interactions of glucagon-like peptide-1 (GLP-1) with the blood–brain barrier. J. Mol. Neurosci. 18, 7–14 (2002).
Dunphy, J. L., Taylor, R. G. & Fuller, P. J. Tissue distribution of rat glucagon receptor and GLP-1 receptor gene expression. Mol. Cell. Endocrinol. 141, 179–186 (1998).
Ballantyne, G. H., Wasielewski, A. & Saunders, J. K. The surgical treatment of type II diabetes mellitus: changes in HOMA insulin resistance in the first year following laparoscopic Roux-en-Y gastric bypass (LRYGB) and laparoscopic adjustable gastric banding (LAGB). Obes. Surg. 19, 1297–1303 (2009).
Mavros, Y. et al. Changes in insulin resistance and HbA1c are related to exercise-mediated changes in body composition in older adults with type 2 diabetes: interim outcomes from the GREAT2DO trial. Diabetes Care 36, 2372–2379 (2013).
Winblad, B. et al. Mild cognitive impairment — beyond controversies, towards a consensus: report of the International Working Group on Mild Cognitive Impairment. J. Intern. Med. 256, 240–246 (2004).
Petersen, R. C. Mild cognitive impairment as a diagnostic entity. J. Intern. Med. 256, 183–194 (2004).
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders 4th edn (American Psychiatric Association, 1994).
McKhann, G. M. et al. The diagnosis of dementia due to Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 7, 263–269 (2011).
Tucsek, Z. et al. Obesity in aging exacerbates blood–brain barrier disruption, neuroinflammation, and oxidative stress in the mouse hippocampus: effects on expression of genes involved in beta-amyloid generation and Alzheimer's disease. J. Gerontol. A Biol. Sci. Med. Sci. 69, 1212–1226 (2014).
Mattson, M. P. Roles of the lipid peroxidation product 4-hydroxynonenal in obesity, the metabolic syndrome, and associated vascular and neurodegenerative disorders. Exp. Gerontol. 44, 625–633 (2009).
Fu, S., Watkins, S. M. & Hotamisligil, G. S. The role of endoplasmic reticulum in hepatic lipid homeostasis and stress signaling. Cell Metab. 15, 623–634 (2012).
Lu, J. et al. Ursolic acid improves high fat diet-induced cognitive impairments by blocking endoplasmic reticulum stress and IκB kinase β/nuclear factor-κB-mediated inflammatory pathways in mice. Brain Behav. Immun. 25, 1658–1667 (2011).
Lourenco, M. V. et al. TNF-α mediates PKR-dependent memory impairment and brain IRS-1 inhibition induced by Alzheimer's β-amyloid oligomers in mice and monkeys. Cell Metab. 18, 831–843 (2013).
Ma, T. et al. Suppression of eIF2α kinases alleviates Alzheimer's disease-related plasticity and memory deficits. Nat. Neurosci. 16, 1299–1305 (2013).
Steen, E. et al. Impaired insulin and insulin-like growth factor expression and signaling mechanisms in Alzheimer's disease — is this type 3 diabetes? J. Alzheimers Dis. 7, 63–80 (2005).
Moloney, A. M. et al. Defects in IGF-1 receptor, insulin receptor and IRS-1/2 in Alzheimer's disease indicate possible resistance to IGF-1 and insulin signalling. Neurobiol. Aging 31, 224–243 (2010).
Hotting, K. & Roder, B. Beneficial effects of physical exercise on neuroplasticity and cognition. Neurosci. Biobehav. Rev. 37, 2243–2257 (2013).
Okonkwo, O. C. et al. Physical activity attenuates age-related biomarker alterations in preclinical AD. Neurology 83, 1753–1760 (2014).
Anderson, R. J., Freedland, K. E., Clouse, R. E. & Lustman, P. J. The prevalence of comorbid depression in adults with diabetes: a meta-analysis. Diabetes Care 24, 1069–1078 (2001).
Ali, S., Stone, M. A., Peters, J. L., Davies, M. J. & Khunti, K. The prevalence of co-morbid depression in adults with Type 2 diabetes: a systematic review and meta-analysis. Diabet. Med. 23, 1165–1173 (2006).
Lustman, P. J. & Clouse, R. E. Depression in diabetic patients: the relationship between mood and glycemic control. J. Diabetes Complications 19, 113–122 (2005).
Testa, M. A. & Simonson, D. C. Health economic benefits and quality of life during improved glycemic control in patients with type 2 diabetes mellitus: a randomized, controlled, double-blind trial. JAMA 280, 1490–1496 (1998).
Collin, M., Hakansson-Ovesjo, M. L., Misane, I., Ogren, S. O. & Meister, B. Decreased 5-HT transporter mRNA in neurons of the dorsal raphe nucleus and behavioral depression in the obese leptin-deficient ob/ob mouse. Brain Res. Mol. Brain Res. 81, 51–61 (2000).
Yamada, N. et al. Impaired CNS leptin action is implicated in depression associated with obesity. Endocrinology 152, 2634–2643 (2011).
Sharma, A. N., Elased, K. M., Garrett, T. L. & Lucot, J. B. Neurobehavioral deficits in db/db diabetic mice. Physiol. Behav. 101, 381–388 (2010).
Sharma, S. & Fulton, S. Diet-induced obesity promotes depressive-like behaviour that is associated with neural adaptations in brain reward circuitry. Int. J. Obes. (Lond.) 37, 382–389 (2013).
Grillo, C. A. et al. Downregulation of hypothalamic insulin receptor expression elicits depressive-like behaviors in rats. Behav. Brain Res. 222, 230–235 (2011).
Grillo, C. A. et al. Dietary restriction reverses obesity-induced anhedonia. Physiol. Behav. 128, 126–132 (2014).
Sharma, A. N., Elased, K. M. & Lucot, J. B. Rosiglitazone treatment reversed depression- but not psychosis-like behavior of db/db diabetic mice. J. Psychopharmacol. 26, 724–732 (2012).
Lustman, P. J., Griffith, L. S., Freedland, K. E., Kissel, S. S. & Clouse, R. E. Cognitive behavior therapy for depression in type 2 diabetes mellitus. A randomized, controlled trial. Ann. Intern. Med. 129, 613–621 (1998).
Lustman, P. J., Freedland, K. E., Griffith, L. S. & Clouse, R. E. Fluoxetine for depression in diabetes: a randomized double-blind placebo-controlled trial. Diabetes Care 23, 618–623 (2000).
Renn, B. N., Feliciano, L. & Segal, D. L. The bidirectional relationship of depression and diabetes: a systematic review. Clin. Psychol. Rev. 31, 1239–1246 (2011).
Schwartz, M. W. et al. Central nervous system control of food intake. Nature 404, 661–671 (2000).
Acknowledgements
The authors thank V. Macht for assistance with the figures. The work of G.J.B. is supported by a Vidi grant from The Netherlands Organisation for Health Research and Development (ZonMw; grant 91711384), and a clinical established investigator grant from The Netherlands Heart Foundation (grant 2010 T073). The work of L.P.R. is supported by the US Department of Veterans Affairs (grants I21 BX002085 and IO1 BX001804) and the University of South Carolina Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
L.P.R. serves as a consultant for I.R.I.S. (Servier) and has received research support for studies involving animal models of depression. G.J.B. consults for and receives research support from Boehringer Ingelheim, consults for Takeda Pharmaceuticals and has received speaker's fees from Eli Lily. Financial compensation for these services is transferred to the University Medical Center Utrecht and not to G.J.B. personally.
Related links
FURTHER INFORMATION
Glossary
- Insulin resistance
-
(IR). A state in which an increase in release of insulin from the pancreas is required to maintain normal plasma glucose levels. In the CNS, IR may be characterized by the decreased ability of insulin to promote structural and functional plasticity.
- Obesity
-
The condition in which an individual has a body mass index of >30 kg per m2, which is normally the result of an increase in fat mass.
- Lacunar infarcts
-
Strokes that, in most cases, are caused by the occlusion of a small perforating artery, visible on MRI as a round or ovoid, subcortical, fluid-filled cavity (with a signal similar to that of cerebrospinal fluid) of 3–15 mm in diameter.
- White matter hyperintensities
-
Signal abnormalities observed on MRI, consisting of white matter that is hyperintense on T2-weighted images (such as fluid-attenuated inversion recovery images) and is without cavitation (it has a signal different to that of cerebrospinal fluid). White matter hyperintensities, presumed to be of vascular origin, reflect tissue abnormalities that range from slight disentanglement of the white matter structure to varying degrees of myelin and axonal loss.
- Amyloid plaques
-
Extracellular deposits of amyloid-β surrounded by dystrophic neurites, reactive astrocytes and microglia. These plaques are a core pathological hallmark of Alzheimer disease.
- Neurofibrillary tangles
-
Intracellular aggregates composed of a hyperphosphorylated form of the microtubule-associated protein tau. These aggregates are a core pathological hallmark of Alzheimer disease.
- Pittsburgh compound B
-
(PiB). A positive emission tomography (PET) tracer that is well retained in amyloid-containing areas of the brain.
- Leptin
-
A hormone that is synthesized and released by adipocytes in direct proportion to body fat mass. Like insulin, leptin is proposed to facilitate neuroplasticity in the hippocampus.
- Peroxisome proliferator-activated receptor-γ
-
(PPARγ). A nuclear receptor that regulates the expression of genes which control glucose metabolism and homeostasis, among other functions.
Rights and permissions
About this article
Cite this article
Biessels, G., Reagan, L. Hippocampal insulin resistance and cognitive dysfunction. Nat Rev Neurosci 16, 660–671 (2015). https://doi.org/10.1038/nrn4019
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrn4019