Abstract
This article reviews evidence that causally links hormonal disorders with hepatobiliary disease, and gives particular focus to nonalcoholic steatohepatitis (NASH). The downstream mechanisms by which endocrine disturbances cause liver disease might be similar to those involved in the development of primary liver disease. Hypothyroidism, for example, might lead to NASH, cirrhosis and potentially liver cancer via the development of hyperlipidemia and obesity. Patients with growth hormone deficiency have a metabolic-syndrome-like phenotype that is also associated with the development of NASH. Polycystic ovary syndrome is a common endocrine disorder that is often associated with insulin resistance, the metabolic syndrome, altered levels of liver enzymes and the development of NASH. Recent findings support a role of dehydroepiandrosterone sulfate deficiency in the development of advanced NASH. In addition, adrenal failure is increasingly reported in patients with end stage liver disease and in patients who have received a liver transplant, which suggests a bidirectional relationship between liver and endocrine functions. Clinicians should, therefore, be aware of the potential role of endocrine disorders in patients with cryptogenic liver disease and of the effects of liver function on the endocrine system.
Key Points
-
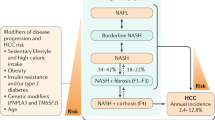
A proportion of patients who have cryptogenic cirrhosis without identifiable etiology of liver disease are expected to have cirrhosis as a complication of nonalcoholic steatohepatitis (NASH)
-
Endocrine hormones control cell metabolism and the distribution of body fat; therefore, endocrine dysfunction might lead to metabolic liver disease in some patients who will eventually be diagnosed with cryptogenic cirrhosis
-
Hypothyroidism is associated with NASH, hepatocellular carcinoma and biliary stones, and dehydroepiandrosterone sulfate (DHEA-S) might have a role in fibrosing NASH
-
Growth hormone deficiency can cause a metabolic-syndrome-like phenotype, the metabolic derangements of which might induce the development of NASH
-
Polycystic ovary syndrome is associated with the development of nonalcoholic liver disease
-
Specific endocrine disorders might promote metabolic liver disease and, given that treatment is available for such endocrine disorders, endocrine etiologies should be ruled out in patients with cryptogenic cirrhosis
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Browning, J. D. et al. Prevalence of hepatic steatosis in an urban population in the United States: impact of ethnicity. Hepatology 40, 1387–1395 (2004).
Byron, D. & Minuk, G. Y. Clinical hepatology: profile of an urban, hospital-based practice. Hepatology 24, 813–815 (1996).
Loria, P. et al. Should nonalcoholic fatty liver disease be renamed? Dig. Dis. 23, 72–82 (2005).
Caldwell, S. H. & Crespo, D. M. The spectrum expanded: cryptogenic cirrhosis and the natural history of non-alcoholic fatty liver disease. J. Hepatol. 40, 578–584 (2004).
Lonardo, A. et al. 'Endocrine NAFLD' a hormonocentric perspective of nonalcoholic fatty liver disease pathogenesis. J. Hepatol. 44, 1196–1207 (2006).
Duntas, L. H. Thyroid disease and lipids. Thyroid 12, 287–293 (2002).
Chidakel, A. et al. Peripheral metabolism of thyroid hormone and glucose homeostasis. Thyroid 15, 899–903 (2005).
Harper, M. E. & Seifert, E. L. Thyroid hormone effects on mitochondrial energetics. Thyroid 18, 145–156 (2008).
Hashimoto, K. et al. Cross-talk between thyroid hormone receptor and liver X receptor regulatory pathways is revealed in a thyroid hormone resistance mouse model. J. Biol. Chem. 281, 295–302 (2006).
Thomas, C. et al. Bile acids and the membrane bile acid receptor TGR5—connecting nutrition and metabolism. Thyroid 18, 167–174 (2008).
Hashimoto, K. et al. Mouse sterol response element binding protein-1c gene expression is negatively regulated by thyroid hormone. Endocrinology 147, 4292–4302 (2006).
Malik, R. & Hodgson, H. The relationship between the thyroid gland and the liver. QJM 95, 559–569 (2002).
Aydemir, S. et al. Effect of hyperthyroidism and propylthiouracil treatment on liver biochemical tests. Int. J. Clin. Pract. 59, 1304–1308 (2005).
Hull, K. et al. Two cases of thyroid storm-associated cholestatic jaundice. Endocr. Pract. 13, 476–480 (2007).
Soylu, A. et al. Intrahepatic cholestasis in subclinical and overt hyperthyroidism: two case reports. J. Med. Case Reports 2, 116 (2008).
Upadhyay, G. et al. Severe hyperthyroidism induces mitochondria-mediated apoptosis in rat liver. Hepatology 39, 1120–1130 (2004).
Carulli, N. et al. Review article: effect of bile salt pool composition on hepatic and biliary functions. Aliment Pharmacol. Ther. 14 (Suppl. 2), 14–18 (2000).
Venditti, P. & Di Meo, S. Thyroid hormone-induced oxidative stress. Cell. Mol. Life Sci. 63, 414–434 (2006).
Sokol, R. J. et al. “Let there be bile”—understanding hepatic injury in cholestasis. J. Pediatr. Gastroenterol. Nutr. 43 (Suppl. 1), S4–S9 (2006).
Sola, J. et al. Liver changes in patients with hyperthyroidism. Liver 11, 193–197 (1991).
Nakamura, H. et al. Comparison of methimazole and propylthiouracil in patients with hyperthyroidism caused by Graves' disease. J. Clin. Endocrinol. Metab. 92, 2157–2162 (2007).
Woeber, K. A. Methimazole-induced hepatotoxicity. Endocr. Pract. 8, 222–224 (2002).
Russo, M. W. et al. Liver transplantation for acute liver failure from drug-induced liver injury in the United States. Liver Transpl. 10, 1018–1023 (2004).
Laukkarinen, J. et al. Increased prevalence of subclinical hypothyroidism in common bile duct stone patients. J. Clin. Endocrinol. Metab. 92, 4260–4264 (2007).
Völzke, H. et al. Association between thyroid function and gallstone disease. World J. Gastroenterol. 11, 5530–5534 (2005).
Bellini, M. A. et al. Hypotonia of the gall bladder, of myxedematous origin. J. Clin. Endocrinol. Metab. 17, 133–142 (1957).
Carulli, N. et al. Thyroid function and sterol metabolism in man. In The Endocrines and the Liver 195–204 (Eds Langer, M. et al.) London and New York: Academic Press (1982).
Ponz de Leon, M. et al. Influence of small-bowel transit time on dietary cholesterol absorption in human beings. N. Engl. J. Med. 307, 102–103 (1982).
Laukkarinen, J. et al. Bile flow to the duodenum is reduced in hypothyreosis and enhanced in hyperthyreosis. Neurogastroenterol. Motil. 14, 183–188 (2002).
Gälman, C. et al. Dramatically increased intestinal absorption of cholesterol following hypophysectomy is normalized by thyroid hormone. Gastroenterology 134, 1127–1136 (2008).
Lauritano, E. C. et al. Association between hypothyroidism and small intestinal bacterial overgrowth. J. Clin. Endocrinol. Metab. 92, 4180–4184 (2007).
Uzunlulu, M. et al. Prevalence of subclinical hypothyroidism in patients with metabolic syndrome. Endocr. J. 54, 71–76 (2007).
Targher, G. et al. Association between serum TSH, free T4 and serum liver enzyme activities in a large cohort of unselected outpatients. Clin. Endocrinol. (Oxf.) 68, 481–484 (2008).
Liangpunsakul, S. & Chalasani, N. Is hypothyroidism a risk factor for nonalcoholic steatohepatitis? J. Clin. Gastroenterol. 37, 340–343 (2003).
Reddy, A. et al. Hypothyroidism: a possible risk factor for liver cancer in patients with no known underlying cause of liver disease. Clin. Gastroenterol. Hepatol. 5, 118–123 (2007).
Bruck, R. et al. Induced hypothyroidism accelerates the regression of liver fibrosis in rats. J. Gastroenterol. Hepatol. 22, 2189–2194 (2007).
Chang, C. C. et al. Methimazole alleviates hepatic encephalopathy in bile-duct ligated cirrhotic rats. J. Chin. Med. Assoc. 69, 563–568 (2006).
Oren, R. et al. The effects of hypothyroidism on liver status of cirrhotic patients. J. Clin. Gastroenterol. 31, 162–163 (2000).
Ji, J. S. et al. Myxedema ascites: case report and literature review. J. Korean Med. Sci. 21, 761–764 (2006).
Duarte, D. R. et al. Myxedema ascites with elevated serum CA 125 concentration. Am. J. Med. Sci. 331, 103–104 (2006).
Kanehara, H. et al. Myxedema ascites with an extremely elevated CA125 level: a case report. Endocr. J. 54, 601–604 (2007).
Bittencourt, P. L. et al. Frequency of concurrent autoimmune disorders in patients with autoimmune hepatitis: effect of age, gender, and genetic background. J. Clin. Gastroenterol. 42, 300–305 (2008).
Sakauchi, F. et al. Comparison of the clinical features of Japanese patients with primary biliary cirrhosis in 1999 and 2004: utilization of clinical data when patients applied to receive public financial aid. J. Epidemiol. 17, 210–214 (2007).
Cindoruk, M. et al. The prevalence of autoimmune hepatitis in Hashimoto's thyroiditis in a Turkish population. Acta Gastroenterol. Belg. 65, 143–145 (2002).
Amenduni, T. et al. Unusual association of Hashimoto's thyroiditis with autoimmune hepatitis. Thyroid 17, 1307–1308 (2007).
Vogel, A. et al. Autoimmune regulator AIRE: evidence for genetic differences between autoimmune hepatitis and hepatitis as part of the autoimmune polyglandular syndrome type 1. Hepatology 33, 1047–1052 (2001).
Oki, K. et al. A case of polyglandular autoimmune syndrome type III complicated with autoimmune hepatitis. Endocr. J. 53, 705–709 (2006).
Antonelli, A. et al. Thyroid disorders in chronic hepatitis C virus infection. Thyroid 16, 563–572 (2006).
Fernandez-Soto, L. et al. Increased risk of autoimmune thyroid disease in hepatitis C vs hepatitis B before, during, and after discontinuing interferon therapy. Arch. Intern. Med. 158, 1445–1448 (1998).
Tomer, Y. et al. Interferon alpha treatment and thyroid dysfunction. Endocrinol. Metab. Clin. North Am. 36, 1051–1066 (2007).
Ghilardi, G. et al. Hypothyroid myopathy as a complication of interferon alpha therapy for chronic hepatitis C virus infection. Br. J. Rheumatol. 37, 1349–1351 (1998).
Deutsch, M. et al. Hashimoto encephalopathy with pegylated interferon alfa-2b and ribavirin. Ann. Pharmacother. 39, 1745–1748 (2005).
Berardi, S. et al. High incidence of allograft dysfunction in liver transplanted patients treated with pegylated-interferon alpha-2b and ribavirin for hepatitis C recurrence: possible de novo autoimmune hepatitis? Gut 56, 237–242 (2007).
Flohr, F. et al. Hypothyroidism in patients with hepatocellular carcinoma treated by transarterial chemoembolization. Hepatology 47, 2144 (2008).
Tamaskar, I. et al. Thyroid function test abnormalities in patients with metastatic renal cell carcinoma treated with sorafenib. Ann. Oncol. 19, 265–268 (2008).
Khovidhunkit, W. et al. Development of overt autoimmune hyperthyroidism in a patient therapeutically immunosuppressed after liver transplantation. Thyroid 10, 829–832 (2000).
Kryczka, W. et al. Thyroid gland dysfunctions during antiviral therapy of chronic hepatitis C. Med. Sci. Monit. 7 (Suppl. 1), 221–225 (2001).
Indolfi, G. et al. Thyroid function and antithyroid autoantibodies in untreated children with vertically acquired chronic hepatitis C virus infection. Clin. Endocrinol. (Oxf.) 68, 117–121 (2008).
Koh, L. K. et al. Interferon-alpha induced thyroid dysfunction: three clinical presentations and a review of the literature. Thyroid 7, 891–896 (1997).
Rodríguez-Torres, M. et al. Thyroid dysfunction (TD) among chronic hepatitis C patients with mild and severe hepatic fibrosis. Ann. Hepatol. 7, 72–77 (2008).
El-Serag, H. B. et al. Diagnosis and treatment of hepatocellular carcinoma. Gastroenterology 134, 1752–1763 (2008).
Brobeck, J. R. Mechanism of the development of obesity in animals with hypothalamic lesions. Physiol. Rev. 26, 541–559 (1946).
Johannsson, G. & Bengtsson, B. A. Growth hormone and the metabolic syndrome. J. Endocrinol. Invest. 22, 41 (1999).
Maison, P. et al. Evidence for distinct effects of GH and IGF-I in the metabolic syndrome. Diabet. Med. 24, 1012–1018 (2007).
Svensson, J. et al. Malignant disease and cardiovascular morbidity in hypopituitary adults with or without growth hormone replacement therapy. J. Clin. Endocrinol. Metab. 89, 3306–3312 (2004).
Sigurjónsdóttir, H. A. et al. GH effect on enzyme activity of 11β HSD in abdominal obesity is dependent on treatment duration. Eur. J. Endocrinol. 154, 69–74 (2006).
Clodfelter, K. H. et al. Role of STAT5a in regulation of sex-specific gene expression in female but not male mouse liver revealed by microarray analysis. Physiol. Genomics 31, 63–74 (2007).
Takano, S. et al. Effect of growth hormone on fatty liver in panhypopituitarism. Arch. Dis. Child. 76, 537–538 (1997).
Tai, T. S. et al. Metabolic effects of growth hormone therapy in an Alström syndrome patient. Horm. Res. 60, 297–301 (2003).
Takahashi, Y. et al. Growth hormone reverses nonalcoholic steatohepatitis in a patient with adult growth hormone deficiency. Gastroenterology 132, 938–943 (2007).
Nyunt, A. et al. Adult cirrhosis due to untreated congenital hypoituitarism. J. R. Soc. Med. 98, 316–317 (2005).
Lonardo, A. et al. Growth hormone plasma levels in nonalcoholic fatty liver disease. Am. J. Gastroenterol. 97, 1071–1072 (2002).
Ichikawa, T. et al. Non-alcoholic steatohepatitis and hepatic steatosis in patients with adult onset growth hormone deficiency Gut. 52, 914 (2003).
Adams, L. A. et al. Nonalcoholic fatty liver disease among patients with hypothalamic and pituitary dysfunction. Hepatology 39, 909–914 (2004).
Wang, H. T. et al. Expression of growth hormone receptor and its mRNA in hepatic cirrhosis. World J. Gastroenterol. 9, 765–770 (2003).
Chen, S. et al. Protective effects of recombinant human growth hormone on cirrhotic rats. World J. Gastroenterol. 10, 2894–2897 (2004).
Theiss, A. L. et al. Growth hormone reduces the severity of fibrosis associated with chronic intestinal inflammation. Gastroenterology 129, 204–219 (2005).
Castiella, A. et al. Hypertransaminasaemia and Addison's disease. Eur. J. Gastroenterol. Hepatol. 10, 891–892 (1998).
Olsson, R. G. et al. Liver involvement in Addison's disease. Am. J. Gastroenterol. 85, 435–438 (1990).
Boulton, R. et al. Subclinical Addison's disease: a cause of persistent abnormalities in transaminase values. Gastroenterology 109, 1321–1327 (1995).
Rizvi, A. A. & Kerrick, J. G. Liver involvement and abnormal iron variables in undiagnosed Addison's disease. Endocr. Pract. 7, 181–188 (2001).
Milionis, H. J. et al. Unexplained hypertransaminasemia: a clue to diagnosis of Addison's disease. Eur. J. Gastroenterol. Hepatol. 14, 1285–1286 (2002).
Nerup, J. Addison's disease: a review of some clinical, pathological and immunological features. Dan. Med. Bull. 21, 201–217 (1974).
Thompson, C. B. Apoptosis in the pathogenesis and treatment of disease. Science 267, 1456–1462 (1995).
Charlton, M. et al. Low circulating levels of dehydroepiandrosterone in histologically advanced nonalcoholic fatty liver disease. Hepatology 47, 484–492 (2008).
Harry, R. et al. The clinical importance of adrenal insufficiency in acute hepatic dysfunction. Hepatology 36, 395–402 (2002).
Tsai, M. H. et al. Adrenal insufficiency in patients with cirrhosis, severe sepsis and septic shock. Hepatology 43, 673–681 (2006).
Fernandez, J. et al. Adrenal insufficiency in patients with cirrhosis and septic shock: effect of treatment with hydrocortisone on survival. Hepatology 44, 1288–1295 (2006).
Marik, P. E. et al. The hepatoadrenal syndrome: a common yet unrecognized clinical condition. Crit. Care Med. 33, 1254–1259 (2005).
O'Beirne, J. et al. Adrenal insufficiency in liver disease. What is the evidence? J. Hepatol. 47, 418–423 (2007).
Marik, P. E. Adrenal insufficiency: the link between low apolipoprotein A-1 levels and poor outcome in the critically ill? Crit. Care Med. 32, 1977–1978 (2004).
Yaguchi, H. et al. Involvement of high density lipoprotein as substrate cholesterol for steroidogenesis by bovine adrenal fasciculo-reticularis cells. Life Sci. 62, 1387–1395 (1988).
Sckiyama, K. D. et al. Circulating proinflammatory cytokines (IL-1 β, TNF-α, and IL-6) and IL-1 receptor antagonist (IL-1Ra) in fulminant hepatic failure and acute hepatitis. Clin. Exp. Immunol. 98, 71–77 (1994).
Stahn, C. et al. Molecular mechanisms of glucocorticoid action and selective glucocorticoid receptor agonists. Mol. Cell. Endocrinol. 275, 71–78 (2007).
Schäche, H. et al. Selective glucocorticoid receptor agonists (SEGRAs): novel ligands with an improved therapeutic index. Mol. Cell. Endocrinol. 275, 109–117 (2007).
Jacobson, P. B. et al. Hepatic glucocorticoid receptor antagonism is sufficient to reduce elevated hepatic glucose output and improve glucose control in animal models of type 2 diabetes. J. Pharm. Exp. Ther. 314, 191–200 (2005).
Von Geldern, T. W. et al. Liver-selective glucocorticoid antagonists: a novel treatment for type 2 diabetes. J. Med. Chem. 47, 4213–4230 (2004).
Rautiainen, H. et al. Budesonide combined with UDCA to improve liver histology in primary biliary cirrhosis: a three-year randomized trial. Hepatology 41, 747–752 (2005).
Arenas, F. et al. Combination of ursodeoxycholic acid and glucocorticoids upregulates the AE2 alternate promoter in human liver cells. J. Clin. Invest. 118, 695–709 (2008).
Fassnacht, M. et al. Beyond adrenal and ovarian androgen generation: increased peripheral 5 alpha-reductase activity in women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 88, 2760–2766 (2003).
Setji, T. L. & Brown, A. J. Polycystic ovary syndrome: diagnosis and treatment. Am. J. Med. 120, 128–132 (2007).
The Rotterdam ESHRE/ASRM-Sponsored PCOS consensus workshop group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum. Reprod. 19, 41–47 (2004).
Glueck, C. J. et al. Incidence and treatment of metabolic syndrome in newly referred women with confirmed polycystic ovarian syndrome. Metabolism 52, 908–915 (2003).
Apridonidze, T. et al. Prevalence and characteristics of the metabolic syndrome in women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 90, 1929–1935 (2005).
Carmina, E. Need for liver evaluation in polycystic ovary syndrome. J. Hepatol. 47, 313–315 (2007).
Ducluzeau, P. H. et al. Glucose-to-insulin ratio rather than sex hormone-binding globulin and adiponectin levels is the best predictor of insulin resistance in nonobese women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 88, 3626–3631 (2003).
Lewy, V. D. et al. Early metabolic abnormalities in adolescent girls with polycystic ovarian syndrome. J. Pediatr. 138, 38–44 (2001).
Pirwany, I. R. et al. Lipids and lipoprotein subfractions in women with PCOS: relationship to metabolic and endocrine parameters. Clin. Endocrinol. (Oxf.) 54, 447–453 (2001).
Chen, M. J. et al. Low sex hormone-binding globulin is associated with low high-density lipoprotein cholesterol and metabolic syndrome in women with PCOS. Hum. Reprod. 21, 2266–2271 (2006).
Schwimmer, J. B. et al. Abnormal aminotransferase activity in women with polycystic ovary syndrome. Fertil. Steril. 83, 494–497 (2005).
Setji, T. L. et al. Nonalcoholic steatohepatitis and nonalcoholic fatty liver disease in young women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 91, 1741–1747 (2006).
Cerda, C. et al. Nonalcoholic fatty liver disease in women with polycystic ovary syndrome. J. Hepatol. 47, 412–417 (2007).
Gambarin-Gelwan, M. et al. Prevalence of nonalcoholic fatty liver disease in women with polycystic ovary syndrome. Clin. Gastroenterol. Hepatol. 5, 496–501 (2007).
Toso, C. et al. Hepatocellular adenoma and polycystic ovary syndrome. Liver Int. 23, 35–37 (2003).
Brown, A. J. et al. Polycystic ovary syndrome and severe nonalcoholic steatohepatitis: beneficial effect of modest weight loss and exercise on liver biopsy findings. Endocr. Pract. 11, 319–324 (2005).
Rautio, K. et al. Effects of metformin and ethinyl estradiol–cyproterone acetate on lipid levels in obese and nonobese women with polycystic ovary syndrome. Eur. J. Endocrinol. 152, 269–275 (2005).
Luque-Ramírez, M. et al. Comparison of ethinyl-estradiol plus cyproterone acetate versus metformin effects on classic metabolic cardiovascular risk factors in women with the polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 92, 2453–2461 (2007).
Lemay, A. et al. Rosiglitazone and ethinyl estradiol/cyproterone acetate as single and combined treatment of overweight women with polycystic ovary syndrome and insulin resistance. Hum. Reprod. 21, 121–128 (2006).
Morin-Papunen, L. et al. Metformin reduces serum C-reactive protein levels in women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 88, 4649–4654 (2003).
Palomba, S. et al. Clomiphene citrate, metformin or both as first-step approach in treating anovulatory infertility in patients with polycystic ovary syndrome (PCOS): a systematic review of head-to-head randomized controlled studies and meta-analysis. Clin. Endocrinol. (Oxf.) [doi:10.1111/j.1365–22652008.03369.x] (2008).
Dunaif, A. Drug insight: insulin-sensitizing drugs in the treatment of polycystic ovary syndrome—a reappraisal. Nat. Clin. Pract. Endocrinol. Metab. 4, 272–283 (2008).
Goldenberg, N. & Glueck, C. Medical therapy in women with polycystic ovarian syndrome before and during pregnancy and lactation. Minerva Ginecol. 60, 63–75 (2008).
Nawaz, F. H. et al. Does continuous use of metformin throughout pregnancy improve pregnancy outcomes in women with polycystic ovarian syndrome? J. Obstet. Gynaecol. Res. 34, 832–837 (2008).
Brettenthaler, N. et al. Effect of the insulin sensitizer pioglitazone on insulin resistance, hyperandrogenism, and ovulatory dysfunction in women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 89, 3835–3840 (2004).
Dahabreh, I. J. & Economopoulos, K. Meta-analysis of rare events: an update and sensitivity analysis of cardiovascular events in randomized trials of rosiglitazone. Clin. Trials 5, 116–120 (2008).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Loria, P., Carulli, L., Bertolotti, M. et al. Endocrine and liver interaction: the role of endocrine pathways in NASH. Nat Rev Gastroenterol Hepatol 6, 236–247 (2009). https://doi.org/10.1038/nrgastro.2009.33
Issue Date:
DOI: https://doi.org/10.1038/nrgastro.2009.33
This article is cited by
-
Perspectives on Precision Medicine Approaches to NAFLD Diagnosis and Management
Advances in Therapy (2021)
-
High regenerative capacity of the liver and irreversible injury of male reproductive system in carbon tetrachloride-induced liver fibrosis rat model
EPMA Journal (2018)
-
NAFLD as a Sexual Dimorphic Disease: Role of Gender and Reproductive Status in the Development and Progression of Nonalcoholic Fatty Liver Disease and Inherent Cardiovascular Risk
Advances in Therapy (2017)
-
Digestive system in psoriasis: an update
Archives of Dermatological Research (2017)
-
Thyroid hormone suppresses expression of stathmin and associated tumor growth in hepatocellular carcinoma
Scientific Reports (2016)