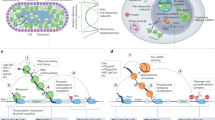
Key Points
-
Pausing is now recognized to be a pervasive feature of promoters in mammals and Drosophila melanogaster, and the evidence supporting this from genome-wide studies is presented.
-
The nomenclature of different promoter-associated RNA polymerase II (Pol II) species is explicitly defined in an effort to provide consistency in future literature.
-
The known mechanistic features of Pol II pausing and its release to productive elongation are described. Most genes are associated with factors that establish and release paused Pol II and therefore appear to progress through this step, although only a subset of genes appears to be directly regulated by pausing.
-
Multiple lines of evidence support the idea that Pol II and nucleosomes compete for promoter binding and suggest that a crucial role of paused Pol II involves maintenance of accessible promoter chromatin architecture.
-
Although pausing has been connected to extremely rapid and synchronous activation of genes, pausing is also highly associated with constitutively expressed genes that encode signalling and transcription factors. Pausing provides a mechanism to tune these key genes to cellular and external regulatory cues.
-
Pausing provides a point of regulation that is distinct from Pol II recruitment and initiation, and this may facilitate the integration of multiple cellular signals. Distinct signals that act through diverse targeted transcription factors can regulate different steps in the transcription pathway and provide a highly modulated transcriptional response at individual genes.
-
Pol II pausing and release occur at a point when 5′ end RNA processing and phosphorylation of the Pol II carboxy-terminal domain occurs. We speculate that by coupling RNA processing to the status and activity of Pol II itself, the cell ensures that nascent RNA is properly protected from degradation and efficiently matures into a functional mRNA.
Abstract
Recent years have witnessed a sea change in our understanding of transcription regulation: whereas traditional models focused solely on the events that brought RNA polymerase II (Pol II) to a gene promoter to initiate RNA synthesis, emerging evidence points to the pausing of Pol II during early elongation as a widespread regulatory mechanism in higher eukaryotes. Current data indicate that pausing is particularly enriched at genes in signal-responsive pathways. Here the evidence for pausing of Pol II from recent high-throughput studies will be discussed, as well as the potential interconnected functions of promoter-proximally paused Pol II.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Lis, J. Promoter-associated pausing in promoter architecture and postinitiation transcriptional regulation. Cold Spring Harb. Symp. Quant. Biol. 63, 347–356 (1998).
Core, L. J., Waterfall, J. J. & Lis, J. T. Nascent RNA sequencing reveals widespread pausing and divergent initiation at human promoters. Science 322, 1845–1848 (2008). GRO-seq maps the position, amount and orientation of transcriptionally engaged RNA polymerases genome-wide and shows peaks of promoter-proximal polymerase residing on ~30% of human genes.
Gilchrist, D. A. et al. Pausing of RNA polymerase II disrupts DNA-specified nucleosome organization to enable precise gene regulation. Cell 143, 540–551 (2010). Global analyses of Pol II pausing and nucleosome occupancy reveal that Pol II and nucleosomes compete for promoter occupancy to regulate gene expression coordinately.
Lee, C. et al. NELF and GAGA factor are linked to promoter-proximal pausing at many genes in Drosophila. Mol. Cell. Biol. 28, 3290–3300 (2008). Comprehensive analysis of promoter-associated Pol II in D. melanogaster using ChIP–chip and permanganate footprinting demonstrates that NELF-mediated pausing of Pol II is common in D. melanogaster.
Min, I. M. et al. Regulating RNA polymerase pausing and transcription elongation in embryonic stem cells. Genes Dev. 25, 742–754 (2011).
Muse, G. W. et al. RNA polymerase is poised for activation across the genome. Nature Genet. 39, 1507–1511 (2007). Genome-wide Pol II ChIP–chip assays, coupled with permanganate footprinting and genetic manipulation of NELF, indicated that Pol II pausing is widespread in D. melanogaster.
Nechaev, S. et al. Global analysis of short RNAs reveals widespread promoter-proximal stalling and arrest of Pol II in Drosophila. Science 327, 335–338 (2010).
Rahl, P. B. et al. c-Myc tegulates transcriptional pause release. Cell 141, 432–445 (2010). Investigation of transcription factor MYC reveals its important role in releasing paused Pol II through recruitment of P-TEFb.
Zeitlinger, J. et al. RNA polymerase stalling at developmental control genes in the Drosophila melanogaster embryo. Nature Genet. 39, 1512–1516 (2007). Genome-wide Pol II ChIP–chip assays indicated that Pol II pausing is widespread during early embryonic development in D. melanogaster.
Fraser, N. W., Sehgal, P. B. & Darnell, J. E. DRB-induced premature termination of late adenovirus transcription. Nature 272, 590–593 (1978).
Gariglio, P., Bellard, M. & Chambon, P. Clustering of RNA polymerase B molecules in the 5′ moiety of the adult beta-globin gene of hen erythrocytes. Nucleic Acids Res. 9, 2589–2598 (1981).
Gilmour, D. S. & Lis, J. T. RNA polymerase II interacts with the promoter region of the noninduced hsp70 gene in Drosophila melanogaster cells. Mol. Cell. Biol. 6, 3984–3989 (1986).
Rougvie, A. E. & Lis, J. T. The RNA polymerase II molecule at the 5′ end of the uninduced hsp70 gene of D. melanogaster is transcriptionally engaged. Cell 54, 795–804 (1988). Nuclear run-on assays demonstrate that the Pol II complex associated with the Hsp70 promoter is transcriptionally engaged but is unable to penetrate further into the gene without heat shock induction.
Giardina, C., Perez-Riba, M. & Lis, J. T. Promoter melting and TFIID complexes on Drosophila genes in vivo. Genes Dev. 6, 2190–2200 (1992).
Rasmussen, E. B. & Lis, J. T. In vivo transcriptional pausing and cap formation on three Drosophila heat shock genes. Proc. Natl Acad. Sci. USA 90, 7923–7927 (1993).
Grayhack, E. J., Yang, X. J., Lau, L. F. & Roberts, J. W. Phage lambda gene Q antiterminator recognizes RNA polymerase near the promoter and accelerates it through a pause site. Cell 42, 259–269 (1985).
Rougvie, A. E. & Lis, J. T. Postinitiation transcriptional control in Drosophila melanogaster. Mol. Cell. Biol. 10, 6041–6045 (1990).
Law, A., Hirayoshi, K., O'Brien, T. & Lis, J. T. Direct cloning of DNA that interacts in vivo with a specific protein: application to RNA polymerase II and sites of pausing in Drosophila. Nucleic Acids Res. 26, 919–924 (1998).
Strobl, L. J. & Eick, D. Hold back of RNA polymerase II at the transcription start site mediates down-regulation of c-Myc in vivo. EMBO J. 11, 3307–3314 (1992).
Krumm, A., Meulia, T., Brunvand, M. & Groudine, M. The block to transcriptional elongation within the human c-myc gene is determined in the promoter-proximal region. Genes Dev. 6, 2201–2213 (1992).
Plet, A., Eick, D. & Blanchard, J. M. Elongation and premature termination of transcripts initiated from c-Fos and c-Myc promoters show dissimilar patterns. Oncogene 10, 319–328 (1995).
Kao, S. Y., Calman, A. F., Luciw, P. A. & Peterlin, B. M. Anti-termination of transcription within the long terminal repeat of HIV-1 by Tat gene product. Nature 330, 489–493 (1987).
Stargell, L. A. & Struhl, K. Mechanisms of transcriptional activation in vivo: two steps forward. Trends Genet. 12, 311–315 (1996).
Ptashne, M. & Gann, A. Transcriptional activation by recruitment. Nature 386, 569–577 (1997).
Steinmetz, E. J. et al. Genome-wide distribution of yeast RNA polymerase II and its control by Sen1 helicase. Mol. Cell 24, 735–746 (2006).
Kim, T. H. et al. A high-resolution map of active promoters in the human genome. Nature 436, 876–880 (2005).
Guenther, M. G., Levine, S. S., Boyer, L. A., Jaenisch, R. & Young, R. A. A chromatin landmark and transcription initiation at most promoters in human cells. Cell 130, 77–88 (2007).
Larschan, E. et al. X chromosome dosage compensation via enhanced transcriptional elongation in Drosophila. Nature 471, 115–118 (2011).
Kephart, D. D., Marshall, N. F. & Price, D. H. Stability of Drosophila RNA polymerase II elongation complexes in vitro. Mol. Cell. Biol. 12, 2067–2077 (1992).
Nechaev, S. & Adelman, K. Promoter-proximal Pol II: when stalling speeds things up. Cell Cycle 7, 1539–1544 (2008).
Hargreaves, D. C., Horng, T. & Medzhitov, R. Control of inducible gene expression by signal-dependent transcriptional elongation. Cell 138, 129–145 (2009).
Schones, D. E. et al. Dynamic regulation of nucleosome positioning in the human genome. Cell 132, 887–898 (2008).
Peterlin, B. M. & Price, D. H. Controlling the elongation phase of transcription with P-TEFb. Mol. Cell 23, 297–305 (2006).
Gromak, N., West, S. & Proudfoot, N. J. Pause sites promote transcriptional termination of mammalian RNA polymerase II. Mol. Cell. Biol. 26, 3986–3996 (2006).
Proudfoot, N. J. Ending the message: poly(A) signals then and now. Genes Dev. 25, 1770–1782 (2011).
Carrillo Oesterreich, F., Preibisch, S. & Neugebauer, K. M. Global analysis of nascent RNA reveals transcriptional pausing in terminal exons. Mol. Cell 40, 571–581 (2010).
de la Mata, M. et al. A slow RNA polymerase II affects alternative splicing in vivo. Mol. Cell 12, 525–532 (2003).
Juven-Gershon, T. & Kadonaga, J. T. Regulation of gene expression via the core promoter and the basal transcriptional machinery. Dev. Biol. 339, 225–229 (2010).
Roeder, R. G. Transcriptional regulation and the role of diverse coactivators in animal cells. FEBS Lett. 579, 909–915 (2005).
Marshall, N. F. & Price, D. H. Control of formation of two distinct classes of RNA polymerase II elongation complexes. Mol. Cell. Biol. 12, 2078–2090 (1992).
Wada, T. et al. DSIF, a novel transcription elongation factor that regulates RNA polymerase II processivity, is composed of human Spt4 and Spt5 homologs. Genes Dev. 12, 343–356 (1998).
Yamaguchi, Y. et al. NELF, a multisubunit complex containing RD, cooperates with DSIF to repress RNA polymerase II elongation. Cell 97, 41–51 (1999). Biochemical assays reveal the presence and identity of the NELF complex and elucidate its role in inhibiting early transcription elongation.
Narita, T. et al. Human transcription elongation factor NELF: identification of novel subunits and reconstitution of the functionally active complex. Mol. Cell. Biol. 23, 1863–1873 (2003).
Cheng, B. et al. Functional association of Gdown1 with RNA polymerase II poised on human genes. Mol. Cell 45, 38–50 (2012).
Brannan, K. et al. mRNA decapping factors and the exonuclease Xrn2 function in widespread premature termination of RNA polymerase II transcription. Mol. Cell 46, 311–324 (2012).
Marshall, N. F. & Price, D. H. Purification of P-TEFb, a transcription factor required for the transition into productive elongation. J. Biol. Chem. 270, 12335–12338 (1995). This pioneering paper describes the purification and characterization of the kinase P-TEFb.
Wada, T., Takagi, T., Yamaguchi, Y., Watanabe, D. & Handa, H. Evidence that P-TEFb alleviates the negative effect of DSIF on RNA polymerase II-dependent transcription in vitro. EMBO J. 17, 7395–7403 (1998).
Czudnochowski, N., Bosken, C. A. & Geyer, M. Serine-7 but not serine-5 phosphorylation primes RNA polymerase II CTD for P-TEFb recognition. Nature Commun. 3, 842 (2012).
Jang, M. et al. The bromodomain protein Brd4 is a positive regulatory component of P-TEFb and stimulates RNA polymerase II-dependent transcription. Mol. Cell 19, 523–534 (2005).
Yang, Z. et al. Recruitment of P-TEFb for stimulation of transcriptional elongation by the bromodomain protein Brd4. Mol. Cell 19, 535–545 (2005).
Barboric, M., Nissen, R. M., Kanazawa, S., Jabrane-Ferrat, N. & Peterlin, B. M. NF-κB binds P-TEFb to stimulate transcriptional elongation by RNA polymerase II. Mol. Cell 8, 327–337 (2001).
Eberhardy, S. & Farnham, P. Myc recruits P-TEFb to mediate the final step in the transcriptional activation of the cad promoter. J. Biol. Chem. 277, 40156–40162 (2002).
Takahashi, H. et al. Human mediator subunit MED26 functions as a docking site for transcription elongation factors. Cell 146, 92–104 (2011).
Mueller, D. et al. A role for the MLL fusion partner ENL in transcriptional elongation and chromatin modification. Blood 110, 4445–4454 (2007).
Lin, C. et al. AFF4, a component of the ELL/P-TEFb elongation complex and a shared subunit of MLL chimeras, can link transcription elongation to leukemia. Mol. Cell 37, 429–437 (2010).
Sobhian, B. et al. HIV-1 Tat assembles a multifunctional transcription elongation complex and stably associates with the 7SK snRNP. Mol. Cell 38, 439–451 (2010).
He, N. et al. HIV-1 Tat and host AFF4 recruit two transcription elongation factors into a bifunctional complex for coordinated activation of HIV-1 transcription. Mol. Cell 38, 428–438 (2010).
Ni, Z. et al. P-TEFb is critical for the maturation of RNA polymerase II into productive elongation in vivo. Mol. Cell. Biol. 28, 1161–1170 (2008).
Workman, J. L. Nucleosome displacement in transcription. Genes Dev. 20, 2009–2017 (2006).
Boeger, H., Griesenbeck, J., Strattan, J. S. & Kornberg, R. D. Nucleosomes unfold completely at a transcriptionally active promoter. Mol. Cell 11, 1587–1598 (2003).
Reinke, H. & Horz, W. Histones are first hyperacetylated and then lose contact with the activated PHO5 promoter. Mol. Cell 11, 1599–1607 (2003).
Wu, C. The 5′ ends of Drosophila heat shock genes in chromatin are hypersensitive to DNase I. Nature 286, 854–860 (1980).
Costlow, N. & Lis, J. T. High-resolution mapping of DNase I-hypersensitive sites of Drosophila heat shock genes in Drosophila melanogaster and Saccharomyces cerevisiae. Mol. Cell. Biol. 4, 1853–1863 (1984).
Shopland, L. S., Hirayoshi, K., Fernandes, M. & Lis, J. T. HSF access to heat shock elements in vivo depends critically on promoter architecture defined by GAGA factor, TFIID, and RNA polymerase II binding sites. Genes Dev. 9, 2756–2769 (1995).
Gilchrist, D. A. et al. NELF-mediated stalling of Pol II can enhance gene expression by blocking promoter-proximal nucleosome assembly. Genes Dev. 22, 1921–1933 (2008).
Leibovitch, B. A. et al. GAGA factor and the TFIID complex collaborate in generating an open chromatin structure at the Drosophila melanogaster hsp26 promoter. Mol. Cell. Biol. 22, 6148–6157 (2002).
Lee, H., Kraus, K. W., Wolfner, M. F. & Lis, J. T. DNA sequence requirements for generating paused polymerase at the start of hsp70. Genes Dev. 6, 284–295 (1992).
Iyer, V. & Struhl, K. Poly(dA:dT), a ubiquitous promoter element that stimulates transcription via its intrinsic DNA structure. EMBO J. 14, 2570–2579 (1995).
Kaplan, N. et al. The DNA-encoded nucleosome organization of a eukaryotic genome. Nature 458, 362–366 (2009).
Jones, P. A. Functions of DNA methylation: islands, start sites, gene bodies and beyond. Nature Rev. Genet. 13, 484–492 (2012).
Tillo, D. et al. High nucleosome occupancy is encoded at human regulatory sequences. PLoS ONE 5, e9129 (2010).
Valouev, A. et al. Determinants of nucleosome organization in primary human cells. Nature 474, 516–520 (2011).
Ramirez-Carrozzi, V. R. et al. A unifying model for the selective regulation of inducible transcription by CpG islands and nucleosome remodeling. Cell 138, 114–128 (2009).
Gilchrist, D. A. & Adelman, K. Coupling polymerase pausing and chromatin landscapes for precise regulation of transcription. Biochim. Biophys. Acta 1819, 700–706 (2012).
Bernstein, B. E. et al. A bivalent chromatin structure marks key developmental genes in embryonic stem cells. Cell 125, 315–326 (2006).
Mikkelsen, T. S. et al. Genome-wide maps of chromatin state in pluripotent and lineage-committed cells. Nature 448, 553–560 (2007).
Chopra, V. S. et al. The polycomb group mutant esc leads to augmented levels of paused Pol II in the Drosophila embryo. Mol. Cell 42, 837–844 (2011).
Weake, V. & Workman, J. Inducible gene expression: diverse regulatory mechanisms. Nature Rev. Genet. 11, 426–437 (2010).
Bryant, G. O. & Ptashne, M. Independent recruitment in vivo by Gal4 of two complexes required for transcription. Mol. Cell 11, 1301–1309 (2003).
Yudkovsky, N., Ranish, J. A. & Hahn, S. A transcription reinitiation intermediate that is stabilized by activator. Nature 408, 225–229 (2000).
Bai, L., Charvin, G., Siggia, E. D. & Cross, F. R. Nucleosome-depleted regions in cell-cycle-regulated promoters ensure reliable gene expression in every cell cycle. Dev. Cell 18, 544–555 (2010).
Hendrix, D. A., Hong, J. W., Zeitlinger, J., Rokhsar, D. S. & Levine, M. S. Promoter elements associated with RNA Pol II stalling in the Drosophila embryo. Proc. Natl Acad. Sci. USA 105, 7762–7767 (2008).
Boettiger, A. N. & Levine, M. Synchronous and stochastic patterns of gene activation in the Drosophila embryo. Science 325, 471–473 (2009).
Aida, M. et al. Transcriptional pausing caused by NELF plays a dual role in regulating immediate-early expression of the junB gene. Mol. Cell. Biol. 26, 6094–6104 (2006).
Adelman, K. et al. Immediate mediators of the inflammatory response are poised for gene activation through RNA polymerase II stalling. Proc. Natl Acad. Sci. USA 106, 18207–18212 (2009).
Nissen, R. M. & Yamamoto, K. R. The glucocorticoid receptor inhibits NFκB by interfering with serine-2 phosphorylation of the RNA polymerase II carboxy-terminal domain. Genes Dev. 14, 2314–2329 (2000).
Kininis, M. et al. Genomic analyses of transcription factor binding, histone acetylation, and gene expression reveal mechanistically distinct classes of estrogen-regulated promoters. Mol. Cell. Biol. 27, 5090–5104 (2007).
Lin, C. et al. Dynamic transcriptional events in embryonic stem cells mediated by the super elongation complex (SEC). Genes Dev. 25, 1486–1498 (2011).
Gilchrist, D. A. et al. Regulating the regulators: the pervasive effects of Pol II pausing on stimulus-responsive gene networks. Genes Dev. 26, 933–944 (2012).
Blau, J. et al. Three functional classes of transcriptional activation domain. Mol. Cell. Biol. 16, 2044–2055 (1996).
Egloff, S., Dienstbier, M. & Murphy, S. Updating the RNA polymerase CTD code: adding gene-specific layers. Trends Genet. 28, 333–341 (2012).
Ghosh, A., Shuman, S. & Lima, C. D. Structural insights to how mammalian capping enzyme reads the CTD code. Mol. Cell 43, 299–310 (2011).
Mandal, S. S. et al. Functional interactions of RNA-capping enzyme with factors that positively and negatively regulate promoter escape by RNA polymerase II. Proc. Natl Acad. Sci. USA 101, 7572–7577 (2004).
Moore, M. J. & Proudfoot, N. J. Pre-mRNA processing reaches back to transcription and ahead to translation. Cell 136, 688–700 (2009).
Buratowski, S. Progression through the RNA polymerase II CTD cycle. Mol. Cell 36, 541–546 (2009).
Levine, M. Paused, R. N. A. Polymerase II as a developmental checkpoint. Cell 145, 502–511 (2011).
Gould, T. J., Verkhusha, V. V. & Hess, S. T. Imaging biological structures with fluorescence photoactivation localization microscopy. Nature Protoc. 4, 291–308 (2009).
Rasmussen, E. B. & Lis, J. T. Short transcripts of the ternary complex provide insight into RNA polymerase II elongational pausing. J. Mol. Biol. 252, 522–535 (1995).
Acknowledgements
We thank the members of the Lis and Adelman laboratories for their helpful discussions on this Review. Funding for this work was provided by US National Institutes of Health (NIH) grant GM25232 to J.L. and the Intramural Research Program of the NIH National Institute of Environmental Health Sciences (Z01 ES101987) to K.A.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Related links
FURTHER INFORMATION
Nature Reviews Genetics Series on Modes of transcriptional regulation
Glossary
- Pre-initiation complex
-
(PIC). An entry form of Pol II in a complex with general transcription factors in which the polymerase is bound to the promoter DNA but has not yet initiated RNA synthesis.
- Heat shock genes
-
(Hsp genes). These genes are a set of highly conserved genes that encode molecular chaperones. These genes are rapidly induced in cells or organisms in response to various cellular stresses, including a several-degree increase in temperature.
- Long terminal repeat
-
(LTR). In HIV, this promoter resides in a region of LTRs. Transcription from this promoter produces both viral proteins and new RNA genomes.
- Ligation-meditated PCR
-
(LM-PCR). A technique that can be used to map the ends of DNA fragments precisely from a specific region of the genome. Small DNA linkers are added to ends of DNA samples and then primers that are complementary to this linker are combined with a sequence-specific primer to amplify the DNA of interest by PCR.
- CpG islands
-
Regions of higher-than-normal CpG sequence content that are on average 1,000 base pairs in length. Such regions contain ~70% of all mammalian promoters, including both genes that are highly regulated and broadly expressed.
- Polycomb
-
Regulate chromatin structure to contribute to epigenetic inheritance of a repressed state. They form several complexes, which are broadly defined as Polycomb repressive complexes 1 and 2 (PRC1 and PRC2), and these are thought to compact chromatin structure.
- Bivalent genes
-
Exhibit histone modifications that are characteristic of both gene repression and activation. These genes display low levels of Pol II occupancy and activity and are hypothesized to be poised for activation during development.
Rights and permissions
About this article
Cite this article
Adelman, K., Lis, J. Promoter-proximal pausing of RNA polymerase II: emerging roles in metazoans. Nat Rev Genet 13, 720–731 (2012). https://doi.org/10.1038/nrg3293
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrg3293
This article is cited by
-
Tissue-specific RNA Polymerase II promoter-proximal pause release and burst kinetics in a Drosophila embryonic patterning network
Genome Biology (2024)
-
NELF and PAF1C complexes are core transcriptional machineries controlling colon cancer stemness
Oncogene (2024)
-
Transcription directionality is licensed by Integrator at active human promoters
Nature Structural & Molecular Biology (2024)
-
RNA polymerase II pausing is essential during spermatogenesis for appropriate gene expression and completion of meiosis
Nature Communications (2024)
-
ERK2-topoisomerase II regulatory axis is important for gene activation in immediate early genes
Nature Communications (2023)