Abstract
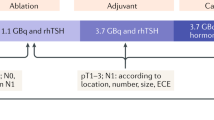
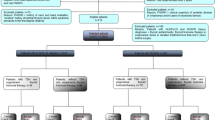
The postoperative administration of radioiodine can be avoided in low-risk patients with undetectable TSH-stimulated serum thyroglobulin and no lymph-node metastases detected at surgery. Sensitive methods for serum thyroglobulin determination can be used to avoid TSH stimulation 9–12 months after surgery in low-risk patients who have an undetectable serum thyroglobulin on levothyroxine treatment; the role of these sensitive assays in the period immediately after surgery needs to be established by further studies. Finally, a low activity of radioiodine (1.1 GBq) should be administered selectively in low-risk patients receiving levothyroxine treatment following injections of recombinant human TSH. These modifications of current protocols will improve the quality of life of patients, potentially decrease morbidity and considerably reduce the cost of treatment and follow-up.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Cooper, D. S. et al. Revised management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid 19, 1167–1214 (2009).
Pacini, F. et al. European consensus for the management of patients with differentiated thyroid cancer of the follicular epithelium. Eur. J. Endocrinol. 154, 787–803 (2006).
Smallridge, R. C. et al. Monitoring thyroglobulin in a sensitive immunoassay has comparable sensitivity to recombinant human TSH stimulated thyroglobulin in follow-up of thyroid cancer patients. J. Clin. Endocrinol. Metab. 92, 82–87 (2007).
Schlumberger, M. et al. Comparison of seven serum thyroglobulin assays in the follow-up of papillary and follicular thyroid cancer patients. J. Clin. Endocrinol. Metab. 92, 2487–2496 (2007).
Iervasi, A. et al. Clinical relevance of highly sensitive Tg assay in monitoring patients treated for differentiated thyroid cancer. Clin. Endocrinol. (Oxf.) 67, 434–441 (2007).
Rosario, P. W. & Purisch, S. Does a highly sensitive thyroglobulin (Tg) assay change the clinical management of low-risk patients with thyroid cancer with Tg on T4 < 1 ng/ml determined by traditional assays? Clin. Endocrinol. (Oxf.) 68, 338–342 (2008).
Spencer, C., Fatemi, S., Singer, P., Nicoloff, J. & Lopresti, J. Serum basal thyroglobulin measured by a second-generation assay correlates with the recombinant human thyrotropin-stimulated thyroglobulin response in patients treated for differentiated thyroid cancer. Thyroid 20, 587–595 (2010).
Rosario, P. W., Xavier, A. C. & Calsolari, M. R. Value of postoperative thyroglobulin and ultrasonography for the indication of ablation and 131I activity in patients with thyroid cancer and low risk of recurrence. Thyroid 21, 49–53 (2011).
Tala Jury, H. P. et al. Lack of association between urinary iodine excretion and successful thyroid ablation in thyroid cancer patients. J. Clin. Endocrinol. Metab. 95, 230–237 (2010).
Vaisman, A. et al. Application of post-surgical stimulated thyroglobulin for radioiodine remnant ablation selection in low-risk papillary thyroid carcinoma. Head Neck 32, 689–698 (2010).
Nascimento, C. L. et al. Persistent disease and recurrence in differentiated thyroid cancer patients with undetectable postoperative stimulated thyroglobulin level. Endocr. Relat. Cancer 18, 29–40 (2011).
Grunwald, F, Menzel, C, Fimmers, R., Zamora, P. O. & Biersack, H. J. Prognostic value of thyroglobulin after thyroidectomy before ablative radioiodine therapy in thyroid cancer. J. Nucl. Med. 37, 1962–1964 (1996).
Toubeau, M. et al. Predictive value for disease progression of serum thyroglobulin levels measured in the postoperative period and after 131I ablation therapy in patients with differentiated thyroid cancer. J. Nucl. Med. 45, 988–994 (2004).
Kim, T. Y. et al. Serum thyroglobulin levels at the time of 131I remnant ablation just after thyroidectomy are useful for early prediction of clinical recurrence in low-risk patients with differentiated thyroid carcinoma. J. Clin. Endocrinol. Metab. 90, 1440–1445 (2005).
Hay, I. D. et al. Papillary thyroid carcinoma managed at the Mayo Clinic during six decades (1940–1999): temporal trends in initial therapy and long-term outcome in 2444 consecutively treated patients. World J. Surg. 26, 879–885 (2002).
Jonklaas, J. et al. Radioiodine therapy in patients with stage I differentiated thyroid cancer. Thyroid 20, 1423–1424 (2010).
Durante, C. et al. Papillary Thyroid Cancer Study Group. Identification and optimal postsurgical follow-up of patients with very low-risk papillary thyroid microcarcinomas. J. Clin. Endocrinol. Metab. 95, 4882–4888 (2010).
Brassard, M. et al. Long-term follow-up of patients with papillary and follicular thyroid cancer: a prospective study on 715 patients. J. Clin. Endocrinol. Metab. 96, 1352–1359 (2011).
Malandrino, P. et al. Risk-adapted management of differentiated thyroid cancer assessed by a sensitive measurement of Basal serum thyroglobulin. J. Clin. Endocrinol. Metab. 96, 1703–1709 (2011).
Drummond, M. F., Sculpher, M., Torrance, G. W., O'Brien, B. J. & Stoddart, G. L. Methods for the Economic Evaluation of Health Care Programmes, 3rd edn (Oxford University Press, USA, 2005).
Bonnet, S. et al. Prophylactic lymph node dissection for papillary thyroid cancer less than 2 cm: implications for radioiodine treatment. J. Clin. Endocrinol. Metab. 94, 1162–1167 (2009).
Hwang, H. S. & Orloff, L. A. Efficacy of preoperative neck ultrasound in the detection of cervical lymph node metastasis from thyroid cancer. Laryngoscope 121, 487–491 (2011).
Leboulleux, S. et al. Ultrasound criteria of malignancy for cervical lymph nodes in patients followed up for differentiated thyroid cancer. J. Clin. Endocrinol. Metab. 92, 3590–3594 (2007).
Tuttle, R. M. & Leboeuf, R. Follow up approaches in thyroid cancer: a risk adapted paradigm. Endocrinol. Metab. Clin. North Am. 37, 419–435 (2008).
Baudin, E. et al. Positive predictive value of serum thyroglobulin levels, measured during the first year of follow-up following thyroid hormone withdrawal, in thyroid cancer patients. J. Clin. Endocrinol. Metab. 88, 1107–1111 (2003).
Chiovato, L. et al. Disappearance of humoral thyroid autoimmunity after complete removal of thyroid antigens. Ann. Intern. Med. 139, 346–351 (2003).
Catargi, B. et al. Comparison of four strategies of radioiodine ablation in patients with thyroid cancer with low-risk of recurrence: the randomized, prospective ESTIMABL study on 753 patients. Presented at the 35th International Thyroid Congress, Paris, September 2010.
Mallick, U. et al. HILO: multicentre randomised phase III clinical trial of high vs low dose radioiodine, with or without recombinant human thyroid stimulating hormone (rhTSH), for remnant ablation for differentiated thyroid cancer. Presented at the 35th International Thyroid Congress, Paris, September 2010.
Author information
Authors and Affiliations
Contributions
All authors researched the data for the article, provided a substantial contribution to discussions of the content, and reviewed and/or edited the manuscript before submission. M. Schlumberger wrote the article.
Corresponding author
Ethics declarations
Competing interests
M. Schlumberger declares an association with the following company: Genzyme (speakers bureau, grant/research support). The other authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Schlumberger, M., Borget, I., Nascimento, C. et al. Treatment and follow-up of low-risk patients with thyroid cancer. Nat Rev Endocrinol 7, 625–628 (2011). https://doi.org/10.1038/nrendo.2011.133
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2011.133
This article is cited by
-
Thyroid cancer radiotheragnostics: the case for activity adjusted 131I therapy
Clinical and Translational Imaging (2018)