Abstract
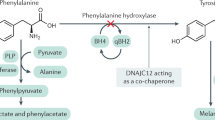
Phenylketonuria is the most prevalent inherited defect in amino acid metabolism. Owing to mutations in the gene encoding the enzyme phenylalanine hydroxylase, the essential amino acid phenylalanine cannot be hydroxylated to tyrosine and blood and tissue concentrations of phenylalanine increase. Untreated, phenylketonuria causes severe mental retardation, epilepsy and behavioral problems. The combined effect of neonatal screening and treatment has, however, meant that phenylketonuria is now a biochemical rather than a clinical diagnosis. Treatment consists of stringent dietary restriction of natural protein intake and supplementation of amino acids other than phenylalanine by a chemically manufactured protein substitute. Although clinical outcome on a phenylalanine-restricted diet is good, neuropsychological deficits are now known to exist in dietary-treated patients with phenylketonuria, and quality of life, nutritional condition and psychosocial outcome could probably also be improved. The need for new therapeutic approaches is being met by supplementation with tetrahydrobiopterin or large neutral amino acids, whilst development of the use of phenylalanine ammonia lyase, and, in the longer term, gene therapy and chaperone treatment holds promise. This Review provides an overview of the history of phenylketonuria, the challenges of treatment today and the treatment possibilities in the near future.
Key Points
-
Phenylketonuria was the first successfully treated inborn error of metabolism, and with treatment, prevention from mental retardation became possible
-
Essentially, phenylketonuria is now a biochemical rather than a clinical diagnosis, which is largely based on high blood phenylalanine concentrations or phenylalanine to tyrosine ratio found at neonatal screening
-
Conventional treatment for phenylketonuria consists of dietary restriction of phenylalanine, which necessitates a stringent restriction of natural protein with supplementation of all amino acids apart from phenylalanine
-
Problems in the outcome of phenylketonuria are almost completely restricted to the brain, which suggests that the blood–brain barrier is of major importance in the pathophysiology of phenylketonuria
-
Notwithstanding the enormous improvement of neurocognitive outcome on dietary treatment, intelligence quotient and neuropsychological tests show deficits that necessitate improvements in diagnosis and new treatment strategies
-
New therapeutic modalities, including large neutral amino acids and tetrahydrobiopterin, may in part replace dietary restriction, while phenylalanine ammonia lyase, chaperone treatment and gene therapy are in development
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Zschocke, J. Phenylketonuria mutations in Europe. Hum. Mutat. 21, 345–356 (2003).
Følling I. The discovery of phenylketonuria. Acta Paediatr. 83 (Suppl. 407), S4–S10 (1994).
Bickel, H., Gerrard, J. & Hickmans, E. M. Influence of phenylalanine intake on phenylketonuria. Lancet 265, 812–813 (1953).
Guthrie, R. & Susi, A. A simple phenylalanine method for detecting phenylketonuria in large populations of newborn infants. Pediatrics 32, 338–343 (1963).
Loeber, J. G. Neonatal screening in Europe; the situation in 2004. J. Inherit. Metab. Dis. 30, 430–438 (2007).
Zurflüh, M. R. et al. Screening for tetrahydrobiopterin deficiencies using dried blood spots on filter paper. Mol. Genet. Metab. 86 (Suppl. 1), S96–S103 (2005).
van Spronsen, F. J. & Enns, G. M. Future treatment strategies in phenylketonuria. Mol. Gen. Metab. 99 (Suppl. 1), S90–S95 (2010).
Smith, I., Beasley, M. G. & Ades, A. E. Intelligence and quality of dietary treatment in phenylketonuria. Arch. Dis. Child. 65, 472–478 (1990).
Verkerk, P. H., Vaandrager, G. J. & Sengers R. C. 15 years of national screening for phenylketonuria in The Netherlands; 4th Report of the National Commission for Management of Phenylketonuria [in Dutch]. Ned Tijdschr Geneeskd. 134, 2533–2536 (1990).
Gassió, R. et al. School performance in early and continuously treated phenylketonuria. Pediatr. Neurol. 33, 267–271 (2005).
Huijbregts, S. C. et al. Motor function under lower and higher controlled processing demands in early and continuously treated phenylketonuria. Neuropsychology 17, 369–379 (2003).
Christ, S. E, Huijbregts, S. C., de Sonneville, L. M. & White, D. A. Executive function in early-treated phenylketonuria: Profile and underlying mechanisms. Mol. Gen. Metab. 99 (Suppl. 1), S22–S32 (2010).
Gentile J. K., Ten Hoedt, A. E. & Bosch, A. M. Psychosocial aspects of PKU: hidden disabilities. Mol. Gen. Metab. 99 (Suppl. 1), S64–S67 (2010).
van Spronsen, F. J. & Burgard, P. The truth of treating patients with phenylketonuria after childhood: the need for a new guideline. J. Inherit. Metab. Dis. 31, 673–679 (2008).
Albrecht, J., Garbade, S. F. & Burgard, P. Neuropsychological speed tests and blood phenylalanine levels in patients with phenylketonuria: a meta-analysis. Neurosci. Biobehav. Rev. 33, 414–421 (2009).
Koch, R. et al. Phenylketonuria in adulthood: A collaborative study. J. Inherit. Metab. Dis. 25, 333–346 (2002).
Pietz, J. et al. Psychiatric disorders in adult patients with early-treated phenylketonuria. Pediatrics 99, 345–350 (1997).
Lee, P. J., McKitterick, K., Channon, S. & Leach, A. Improvements in neuropsychometric outcome when re-introducing diet in adulthood in phenylketonuria (PKU). J. Inherit. Metab. Dis. 30 (Suppl. 1), 14 (2007).
Koch, R., Trefz, F. & Waisbren, S. Psychosocial issues and outcomes in maternal PKU. Mol. Gen. Metab. 99 (Suppl. 1), S68–S74 (2010).
Laclair, C. E., Ney, D. M., MacLeod, E. L. & Etzel, M. R. Purification and use of glycomacropeptide for nutritional management of phenylketonuria. J. Food Sci. 74, E199–E206 (2009).
Ney, D. M. et al. Nutritional management of PKU with glycomacropeptide from cheese whey. J. Inherit. Metab. Dis. 32, 32–39 (2009).
Lim, K., van Calcar, S. C., Nelson, K. L., Gleason, S. T. & Ney, D. M. Acceptable low-phenylalanine foods and beverages can be made with glycomacropeptide from cheese whey for individuals with PKU. Mol. Genet. Metab. 92, 176–178 (2007).
van Calcar, S. C. et al. Improved nutritional management of phenylketonuria by using a diet containing glycomacropeptide compared with amino acids. Am. J. Clin. Nutr. 89, 1068–1077 (2009).
Andersen, A. E. & Avins, L. Lowering brain phenylalanine levels by giving other large neutral amino acids. A new experimental therapeutic approach to phenylketonuria. Arch. Neurol. 33, 684–686 (1976).
Pietz, J. et al. Large neutral amino acids block phenylalanine transport into brain tissue in patients with phenylketonuria. J. Clin. Invest. 103, 1169–1178 (1999).
Moats, R. A., Moseley, K. D., Koch, R. & Nelson, M. Jr. Brain phenylalanine concentrations in phenylketonuria: research and treatment of adults. Pediatrics 112, 1575–1579 (2003).
Schindeler, S. et al. The effects of large neutral amino acid supplements in PKU: an MRS and neuropsychological study. Mol. Genet. Metab. 91, 48–54 (2007).
Lou, H. Large doses of tryptophan and tyrosine as potential therapeutic alternative to dietary phenylalanine restriction in phenylketonuria. Lancet 2, 150–151 (1985).
Matalon, R. et al. Double blind placebo control trial of large neutral amino acids in treatment of PKU: effect on blood phenylalanine. J. Inherit. Metab. Dis. 30, 153–158 (2007).
Kaufman, S. & Milstien, S. Phenylketonuria and its variants. Ann. Clin. Lab. Sci. 7, 178–185 (1977).
Kure, S. et al. Tetrahydrobiopterin-responsive phenylalanine hydroxylase deficiency. J. Pediatr. 135, 375–378 (1999).
Burton, B. K. et al. The response of patients with phenylketonuria and elevated serum phenylalanine to treatment with oral sapropterin dihydrochloride (6R-tetrahydrobiopterin): a phase II, multicentre, open-label, screening study. J. Inherit. Metab. Dis. 30, 700–707 (2007).
Muntau, A. C. et al. Tetrahydrobiopterin as an alternative treatment for mild phenylketonuria. N. Engl. J. Med. 347, 2122–2132 (2002).
Levy, H., Burton, B., Cederbaum, S. & Scriver, C. Recommendations for evaluation of responsiveness to tetrahydrobiopterin (BH(4)) in phenylketonuria and its use in treatment. Mol. Genet. Metab. 92, 287–291 (2007).
Bernegger, C. & Blau, N. High frequency of tetrahydrobiopterin-responsiveness among hyperphenylalaninemias: a study of 1,919 patients observed from 1988 to 2002. Mol. Genet. Metab. 77, 304–313 (2002).
Hennermann, J. B., Bührer, C., Blau, N., Vetter, B. & Mönch, E. Long-term treatment with tetrahydrobiopterin increases phenylalanine tolerance in children with severe phenotype of phenylketonuria. Mol. Genet. Metab. 86 (Suppl. 1), S86–S90 (2005).
Dobrowolski, S. F. et al. A limited spectrum of phenylalanine hydroxylase mutations is observed in phenylketonuria patients in western Poland and implications for treatment with 6R tetrahydrobiopterin. J. Hum. Genet. 54, 335–339 (2009).
Pérez, B. et al. Kinetic and stability analysis of PKU mutations identified in BH4-responsive patients. Mol. Genet. Metab. 86 (Suppl. 1), S11–S16 (2005).
Gersting, S. W. et al. Loss of function in phenylketonuria is caused by impaired molecular motions and conformational instability. Am. J. Hum. Genet. 83, 5–17 (2008).
Blau, N. et al. Optimizing the use of sapropterin (BH4) in the management of phenylketonuria. Mol. Genet. Metab. 96, 158–163 (2009).
Sarkissian, C. N. et al. A different approach to treatment of phenylketonuria: phenylalanine degradation with recombinant phenylalanine ammonia lyase. Proc. Natl Acad. Sci. USA 96, 2339–2344 (1999).
Sarkissian, C. N. et al. Preclinical evaluation of multiple species of PEGylated recombinant phenylalanine ammonia lyase for the treatment of phenylketonuria. Proc. Natl Acad. Sci. USA 105, 20894–20899 (2008).
Kang, T. S. et al. Converting an injectable protein therapeutic into an oral form: phenylalanine ammonia lyase for phenylketonuria. Mol. Genet. Metab. 99, 4–9 (2010).
Harding, C. O. et al. Complete correction of hyperphenylalaninemia following liver-directed, recombinant AAV2/8 vector-mediated gene therapy in murine phenylketonuria. Gene Ther. 13, 457–462 (2006).
Ding, Z., Georgiev, P. & Thöny, B. Administration-route and gender-independent long-term therapeutic correction of phenylketonuria (PKU) in a mouse model by recombinant adeno-associated virus 8 pseudotyped vector-mediated gene transfer. Gene Ther. 13, 587–593 (2006).
Ding, Z. et al. Correction of murine PKU following AAV-mediated intramuscular expression of a complete phenylalanine hydroxylating system. Mol. Ther. 16, 673–681 (2008).
Rebuffat, A., Harding, C. O., Ding, Z. & Thöny, B. Comparison of adeno-associated virus pseudotype 1, 2, and 8 vectors administered by intramuscular injection in the treatment of murine phenylketonuria. Hum. Gene Ther. 21, 463–477 (2010).
Thöny, B. Long-term correction of murine phenylketonuria by viral gene transfer: liver versus muscle. J. Inherit. Metab. Dis. doi:10.1007/s10545-010-9044-3.
Gregersen, N., Bross, P., Vang, S. & Christensen, J. H. Protein misfolding and human disease. Annu. Rev. Genomics Hum. Genet. 7, 103–124 (2006).
Cohen, F. E. & Kelly, J. W. Therapeutic approaches to protein-misfolding diseases. Nature 426, 905–909 (2003).
Pey, A. L. et al. Identification of pharmacological chaperones as potential therapeutic agents to treat phenylketonuria. J. Clin. Invest. 118, 2858–2867 (2008).
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author declares associations with the following companies: Merck Serono (Consultant, Speakers Bureau, Grant/research support), Nutricia (Consultant, Speakers Bureau, Grant/research support).
Rights and permissions
About this article
Cite this article
van Spronsen, F. Phenylketonuria: a 21st century perspective. Nat Rev Endocrinol 6, 509–514 (2010). https://doi.org/10.1038/nrendo.2010.125
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2010.125
This article is cited by
-
Expression of phenylalanine ammonia lyase as an intracellularly free and extracellularly cell surface-immobilized enzyme on a gut microbe as a live biotherapeutic for phenylketonuria
Science China Life Sciences (2023)
-
Dietary and metabolic effects on the oral status of patients with phenylketonuria: a nation-based cross-sectional study
Clinical Oral Investigations (2023)
-
Genetische Diagnostik der Epilepsien: Empfehlung der Kommission Epilepsie und Genetik der Deutschen Gesellschaft für Epileptologie (DGfE)
Clinical Epileptology (2023)
-
Metabolic control during the neonatal period in phenylketonuria: associations with childhood IQ
Pediatric Research (2022)
-
Lost in print: difficulty in reading online information pertaining to phenylketonuria
Journal of Community Genetics (2022)