Abstract
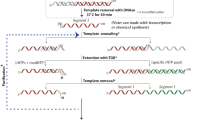
Site-specific incorporation of labeled nucleotides is an extremely useful synthetic tool for many structural studies (e.g., NMR, electron paramagnetic resonance (EPR), fluorescence resonance energy transfer (FRET), and X-ray crystallography) of RNA. However, specific-position-labeled RNAs >60 nt are not commercially available on a milligram scale. Position-selective labeling of RNA (PLOR) has been applied to prepare large RNAs labeled at desired positions, and all the required reagents are commercially available. Here, we present a step-by-step protocol for the solid–liquid hybrid phase method PLOR to synthesize 71-nt RNA samples with three different modification applications, containing (i) a 13C15N-labeled segment; (ii) discrete residues modified with Cy3, Cy5, or biotin; or (iii) two iodo-U residues. The flexible procedure enables a wide range of downstream biophysical analyses using precisely localized functionalized nucleotides. All three RNAs were obtained in <2 d, excluding time for preparing reagents and optimizing experimental conditions. With optimization, the protocol can be applied to other RNAs with various labeling schemes, such as ligation of segmentally labeled fragments.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Lu, K., Miyazaki, Y. & Summers, M.F. Isotope labeling strategies for NMR studies of RNA. J. Biomol. NMR 46, 113–125 (2010).
Duss, O., Lukavsky, P.J. & Allain, F.H. Isotope labeling and segmental labeling of larger RNAs for NMR structural studies. Adv. Exp. Med. Biol. 992, 121–144 (2012).
Alvarado, L.J. et al. Chemo-enzymatic synthesis of selectively (1)(3)C/(1)(5)N-labeled RNA for NMR structural and dynamics studies. Methods Enzymol. 549, 133–162 (2014).
Nelissen, F.H. et al. Multiple segmental and selective isotope labeling of large RNA for NMR structural studies. Nucleic Acids Res. 36, e89 (2008).
Lin, L., Sheng, J. & Huang, Z. Nucleic acid X-ray crystallography via direct selenium derivatization. Chem. Soc. Rev. 40, 4591–4602 (2011).
Cate, J.H. & Doudna, J.A. Solving large RNA structures by X-ray crystallography. Methods Enzymol. 317, 169–180 (2000).
Moroder, H., Kreutz, C., Lang, K., Serganov, A. & Micura, R. Synthesis, oxidation behavior, crystallization and structure of 2′-methylseleno guanosine containing RNAs. J. Am. Chem. Soc. 128, 9909–9918 (2006).
Gasnier, M., Dennis, C., Vaurs-Barriere, C. & Chazaud, C. Fluorescent mRNA labeling through cytoplasmic FISH. Nat. Protoc. 8, 2538–2547 (2013).
Hikida, Y., Kimoto, M., Yokoyama, S. & Hirao, I. Site-specific fluorescent probing of RNA molecules by unnatural base-pair transcription for local structural conformation analysis. Nat. Protoc. 5, 1312–1323 (2010).
Pitchiaya, S., Heinicke, L.A., Custer, T.C. & Walter, N.G. Single molecule fluorescence approaches shed light on intracellular RNAs. Chem. Rev. 114, 3224–3265 (2014).
Mairal, T. et al. Aptamers: molecular tools for analytical applications. Anal. Bioanal. Chem. 390, 989–1007 (2008).
Soontornworajit, B. & Wang, Y. Nucleic acid aptamers for clinical diagnosis: cell detection and molecular imaging. Anal. Bioanal. Chem. 399, 1591–1599 (2011).
Paredes, E., Evans, M. & Das, S.R. RNA labeling, conjugation and ligation. Methods 54, 251–259 (2011).
Caruthers, M.H. The chemical synthesis of DNA/RNA: our gift to science. J. Biol. Chem. 288, 1420–1427 (2013).
Caruthers, M.H. A brief review of DNA and RNA chemical synthesis. Biochem. Soc. Trans. 39, 575–580 (2011).
Lang, K. & Micura, R. The preparation of site-specifically modified riboswitch domains as an example for enzymatic ligation of chemically synthesized RNA fragments. Nat. Protoc. 3, 1457–1466 (2008).
Moore, M.J. & Query, C.C. Joining of RNAs by splinted ligation. Methods Enzymol. 317, 109–123 (2000).
Kurschat, W.C., Muller, J., Wombacher, R. & Helm, M. Optimizing splinted ligation of highly structured small RNAs. RNA 11, 1909–1914 (2005).
Liu, Y. et al. Synthesis and applications of RNAs with position-selective labelling and mosaic composition. Nature 522, 368–372 (2015).
Liu, Y., Sousa, R. & Wang, Y.X. Specific labeling: an effective tool to explore the RNA world. Bioessays 38, 192–200 (2016).
Zuo, X. et al. Solution structure of the cap-independent translational enhancer and ribosome-binding element in the 3′ UTR of turnip crinkle virus. Proc. Natl. Acad. Sci. USA 107, 1385–1390 (2010).
Wang, Y.X., Zuo, X., Wang, J., Yu, P. & Butcher, S.E. Rapid global structure determination of large RNA and RNA complexes using NMR and small-angle X-ray scattering. Methods 52, 180–191 (2010).
Stupina, V.A. et al. The 3′ proximal translational enhancer of Turnip crinkle virus binds to 60S ribosomal subunits. RNA 14, 2379–2393 (2008).
Reining, A. et al. Three-state mechanism couples ligand and temperature sensing in riboswitches. Nature 499, 355–359 (2013).
Serganov, A. et al. Structural basis for discriminative regulation of gene expression by adenine- and guanine-sensing mRNAs. Chem. Biol. 11, 1729–1741 (2004).
Leipply, D. & Draper, D.E. Effects of Mg2+ on the free energy landscape for folding a purine riboswitch RNA. Biochemistry 50, 2790–2799 (2011).
Milligan, J.F. & Uhlenbeck, O.C. Synthesis of small RNAs using T7 RNA-polymerase. Methods Enzymol. 180, 51–62 (1989).
Pokrovskaya, I.D. & Gurevich, V.V. In vitro transcription: preparative RNA yields in analytical scale reactions. Anal. Biochem. 220, 420–423 (1994).
Sano, T., Vajda, S. & Cantor, C.R. Genetic engineering of streptavidin, a versatile affinity tag. J. Chromatogr. B Biomed. Sci. Appl. 715, 85–91 (1998).
Kao, C., Zheng, M. & Rudisser, S. A simple and efficient method to reduce nontemplated nucleotide addition at the 3 terminus of RNAs transcribed by T7 RNA polymerase. RNA 5, 1268–1272 (1999).
Guo, Q., Nayak, D., Brieba, L.G. & Sousa, R. Major conformational changes during T7RNAP transcription initiation coincide with, and are required for, promoter release. J. Mol. Biol. 353, 256–270 (2005).
Tang, G.Q., Roy, R., Bandwar, R.P., Ha, T. & Patel, S.S. Real-time observation of the transition from transcription initiation to elongation of the RNA polymerase. Proc. Natl. Acad. Sci. USA 106, 22175–22180 (2009).
Huang, J., Brieba, L.G. & Sousa, R. Misincorporation by wild-type and mutant T7 RNA polymerases: identification of interactions that reduce misincorporation rates by stabilizing the catalytically incompetent open conformation. Biochemistry 39, 11571–11580 (2000).
He, B. et al. Rapid mutagenesis and purification of phage RNA polymerases. Protein Expr. Purif. 9, 142–151 (1997).
Pluthero, F.G. Rapid purification of high-activity Taq DNA polymerase. Nucleic Acids Res. 21, 4850–4851 (1993).
Otwinowski, Z. & Minor, W. Processing of diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997).
Zhang, J. & Ferr-D'Amaré, A.R. Dramatic improvement of crystals of large RNAs by cation replacement and dehydration. Structure 22, 1363–1371 (2014).
Sheldrick, G.M. Experimental phasing with SHELXC/D/E: combining chain tracing with density modification. Acta Crystallogr. D Biol. Crystallogr. D66, 479–485 (2010).
Korbie, D.J. & Mattick, J.S. Touchdown PCR for increased specificity and sensitivity in PCR amplification. Nat. Protoc. 3, 1452–1456 (2008).
Acknowledgements
This work was supported by the Intramural Research Programs of the National Cancer Institute (Y.-X.W.). E.H. and D.J.N gratefully acknowledge funding for this work by the National Science Foundation (CHE 1266416 and PHY 1125844) and the National Institute for Standards and Technology. We thank S. Frey from Abteilung Zelluläre Logistik, Max-Planck-Institut für Biophysikalische Chemie, Göttingen, Germany, for providing us with the plasmid for His-tagged Taq DNA polymerase and the preparation protocol, and D. Draper from Johns Hopkins University for providing us with the plasmid for His-tagged T7 RNA polymerase.
Author information
Authors and Affiliations
Contributions
Y.L. performed RNA synthesis, NMR experiments, and X-ray crystallography experiments, and wrote the manuscript; E.H. and D.N. designed and performed smFRET experiments; P.Y. performed enzyme preparation; R.S. provided critical advice on PLOR; J.R.S., K.T. and X.Z. performed X-ray crystallography experiments and data analysis; and Y.-X.W. designed PLOR. All authors revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Liu, Y., Holmstrom, E., Yu, P. et al. Incorporation of isotopic, fluorescent, and heavy-atom-modified nucleotides into RNAs by position-selective labeling of RNA. Nat Protoc 13, 987–1005 (2018). https://doi.org/10.1038/nprot.2018.002
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2018.002
This article is cited by
-
Expedient production of site specifically nucleobase-labelled or hypermodified RNA with engineered thermophilic DNA polymerases
Nature Communications (2024)
-
Observation of structural switch in nascent SAM-VI riboswitch during transcription at single-nucleotide and single-molecule resolution
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.