Abstract
Microglial cells have important roles in maintaining brain homeostasis, and they are implicated in multiple brain diseases. There is currently interest in investigating microglial migration that results in cell accumulation at focal sites of injury. Here we describe a protocol for rapidly triggering and monitoring microglial migration by using a micropipette assay. This protocol is an adaptation of the axon turning assay using microglial cells. Chemoattractants released from the micropipette tip produce a chemotactic gradient that induces robust microglial migration. In combination with microscopic imaging, this assay allows simultaneous recording of cell movement and subcellular compartment trafficking, along with quantitative analysis. The actual handling time for the assay takes ∼2–3 h in total. The protocol is simple, inexpensive and convenient to set up, and it can be adopted to examine cell migration in multiple cell types, including cancer cells with a wide range of chemical signals.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Nimmerjahn, A., Kirchhoff, F. & Helmchen, F. Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science 308, 1314–1318 (2005).
Davalos, D. et al. ATP mediates rapid microglial response to local brain injury in vivo. Nat. Neurosci. 8, 752–758 (2005).
Ifuku, M. et al. Bradykinin-induced microglial migration mediated by B1-bradykinin receptors depends on Ca2+ influx via reverse-mode activity of the Na+/Ca2+ exchanger. J. Neurosci. 27, 13065–13073 (2007).
Honda, S. et al. Extracellular ATP or ADP induce chemotaxis of cultured microglia through Gi/o-coupled P2Y receptors. J. Neurosci. 21, 1975–1982 (2001).
Rappert, A. et al. Secondary lymphoid tissue chemokine (CCL21) activates CXCR3 to trigger a Cl– current and chemotaxis in murine microglia. J. Immunol. 168, 3221–3226 (2002).
Walter, L. et al. Nonpsychotropic cannabinoid receptors regulate microglial cell migration. J. Neurosci. 23, 1398–1405 (2003).
Zhang, Z.J. et al. Regulated ATP release from astrocytes through lysosome exocytosis. Nat. Cell Biol. 9, 945–953 (2007).
Junger, W.G. Immune cell regulation by autocrine purinergic signalling. Nat. Rev. Immunol. 11, 201–212 (2011).
Elliott, M.R. et al. Nucleotides released by apoptotic cells act as a find-me signal to promote phagocytic clearance. Nature 461, 282–286 (2009).
Koizumi, S., Ohsawa, K., Inoue, K. & Kohsaka, S. Purinergic receptors in microglia: functional modal shifts of microglia mediated by P2 and P1 receptors. Glia 61, 47–54 (2013).
Wu, L.J., Vadakkan, K.I. & Zhuo, M. ATP-induced chemotaxis of microglial processes requires P2Y receptor-activated initiation of outward potassium currents. Glia 55, 810–821 (2007).
Nakajima, K. et al. Identification of elastase as a secretory protease from cultured rat microglia. J. Neurochem. 58, 1401–1408 (1992).
Siddiqui, T.A., Lively, S., Vincent, C. & Schlichter, L.C. Regulation of podosome formation, microglial migration and invasion by Ca2+-signaling molecules expressed in podosomes. J. Neuroinflamm. 9, 250 (2012).
Lively, S. & Schlichter, L.C. The microglial activation state regulates migration and roles of matrix-dissolving enzymes for invasion. J. Neuroinflamm. 10, 75 (2013).
Dou, Y. et al. Microglial migration mediated by ATP-induced ATP release from lysosomes. Cell Res. 22, 1022–1033 (2012).
Zigmond, S.H. Ability of polymorphonuclear leukocytes to orient in gradients of chemotactic factors. J. Cell Biol. 75, 606–616 (1977).
Boyden, S. The chemotactic effect of mixtures of antibody and antigen on polymorphonuclear leucocytes. J. Exp. Med. 115, 453–466 (1962).
Zicha, D., Dunn, G.A. & Brown, A.F. A new direct-viewing chemotaxis chamber. J. Cell Sci. 99, 769–775 (1991).
Goncharova, E.A., Goncharov, D.A. & Krymskaya, V.P. Assays for in vitro monitoring of human airway smooth muscle (ASM) and human pulmonary arterial vascular smooth muscle (VSM) cell migration. Nat. Protoc. 1, 2933–2939 (2006).
Liang, C.C., Park, A.Y. & Guan, J.L. In vitro scratch assay: a convenient and inexpensive method for analysis of cell migration in vitro. Nat. Protoc. 2, 329–333 (2007).
Chaubey, S., Ridley, A.J. & Wells, C.M. Using the Dunn chemotaxis chamber to analyze primary cell migration in real time. Methods Mol. Biol. 769, 41–51 (2011).
Nakajima, K., Tsuzaki, N., Shimojo, M., Hamanoue, M. & Kohsaka, S. Microglia isolated from rat brain secrete a urokinase-type plasminogen activator. Brain Res. 577, 285–292 (1992).
Lohof, A.M., Quillan, M., Dan, Y. & Poo, M.M. Asymmetric modulation of cytosolic camp activity induces growth cone turning. J. Neurosci. 12, 1253–1261 (1992).
Hosmane, S. et al. Toll/interleukin-1 receptor domain-containing adapter inducing interferon-β mediates microglial phagocytosis of degenerating axons. J. Neurosci. 32, 7745–7757 (2012).
Cho, H. et al. Microfluidic chemotaxis platform for differentiating the roles of soluble and bound amyloid-β on microglial accumulation. Sci. Rep. 3, 1823 (2013).
Beaudoin, G.M.J. et al. Culturing pyramidal neurons from the early postnatal mouse hippocampus and cortex. Nat. Protoc. 7, 1741–1754 (2012).
Chen, Y. et al. Isolation and culture of rat and mouse oligodendrocyte precursor cells. Nat. Protoc. 2, 1044–1051 (2007).
Acknowledgements
This work was supported by grants from the Major State Basic Research Program of China (no. 2011CB504400), the National Natural Science Foundation of China (nos. 31190060, 81221003, 30730037, 30870834, 91232000), the Program for Changjiang Scholars and Innovative Research Teams in University (PCSIRT) and the Fundamental Research Funds for the Central Universities.
Author information
Authors and Affiliations
Contributions
H.W., Y.L., H. Li, C.C., Y.D., H. Lou, Z.G. and S.D. contributed to protocol development. H.W. performed all of the experiments and data analysis. H.W., M.S.H., Z.G., X.L. and S.D. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
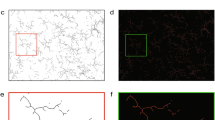
Supplementary Figure 1 Diffusion profile of Sulforhodamine 101 (SR101) when ejected from the tip of a micropipette.
(A) Schematic of fluorescence detection position. Circles in different colors denote different distances from the tip of the micropipette. Scale bar, 50 μm. (B) Quantitative analysis of SR 101 fluorescence intensities. Fluorescence intensities at three different points on each circle were collected and used for data analysis. Data were normalized to the average fluorescence intensity measured at the position 15 μm from the source after SR101 ejection for 10 min. The arrow marks the time point when the pulsatile application of SR101 was started. Error bars indicate s.e.m. (C) The diffusion profile of SR101 around ∼300 μm from the micropipette tip at different time points is illustrated in a three dimensional surface plot, which was generated using ImageJ with the Interactive 3D Surface Plot plugin. X axis and Y axis indicate distance from the tip of micropipette. Note that the diffusion profile of SR 101 is relatively stable 5 min after its application up to more than 30 min.
Supplementary Figure 2 Double immunofluorescence staining for CD11b (microglial marker) and GFAP (astrocytic marker) in purified primary microglia obtained in Steps 11–13.
Note that most of the cells were CD11b positive and GFAP negative. Scale bar, 20 μm.
Supplementary information
Supplementary Figure 1
Diffusion profile of Sulforhodamine 101 (SR101) when ejected from the tip of a micropipette. (PDF 411 kb)
Supplementary Figure 2
Double immunofluorescence staining for CD11b (microglial marker) and GFAP (astrocytic marker) in purified primary microglia obtained in Steps 11–13. (PDF 158 kb)
Microglial migration induced by application of 1 mM ATPγS.
Time-lapse images were acquired by a confocal microscope (Olympus FV1000, Japan) with a 20x air objective (UPLSAPO 20×, 0.75 NA). 1 mM ATPγS was ejected from the micropipette after ten frames (300 s) and frames were acquired every 30 seconds. Scale bar, 50 μm. The video speed is six frames per second. (MOV 1537 kb)
Microglial migration induced by application of 10 mM ATPγS.
Time-lapse images were acquired by a confocal microscope (Olympus FV1000, Japan) with a 20x air objective (UPLSAPO 20×, 0.75 NA). Image frames were acquired every 30 seconds and 10 mM ATPγS was ejected from the micropipette after ten frames (300 s). Note that cells in juxtaposition to the tip do not migrate well. Arrows indicate cells with no obvious directed migration (indicated by the arrowheads) or migrate towards the opposite direction (indicated by the arrow). Scale bars, 50 μm. The video speed is six frames per second. (MOV 1410 kb)
Lysosomal trafficking during microglial migration induced by 1 mM ATPγS.
Microglia lysosomes were labeled with 500 nM LysoTracker Red for 30 min before imaging. The LysoTracker Red is presented as red color overlaid on the bright field images. Time-lapse images were acquired by a confocal microscope (Olympus FV1000, Japan) with a 60×water immersion objective (UPLSAPO 60×W, 1.2 NA). 1 mM ATPγS was ejected from the micropipette after the first frame (20 s) and frames were acquired every 20 seconds. The micropipette is positioned at the lower left corner as indicated by the orange arrow. Scale bar, 10 μm. The video speed is two frames per second. (AVI 2171 kb)
Lysosomal tracks of individual microglia during migration induced by 1 mM ATPγS.
An individual microglia with a magnified field from Supplementary Video 3 (as indicated in Fig. 5A). The trajectories of lysosome were reconstructed using Imaris software (Bitplane). The time course of trajectories are presented as pseudocolor indicated at the lower right corner. Scale bar, 5 μm. The video speed is two frames per second. (AVI 2369 kb)
Rights and permissions
About this article
Cite this article
Wu, Hj., Liu, Yj., Li, Hq. et al. Analysis of microglial migration by a micropipette assay. Nat Protoc 9, 491–500 (2014). https://doi.org/10.1038/nprot.2014.015
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2014.015
This article is cited by
-
Systemic pro-inflammatory response facilitates the development of cerebral edema during short hypoxia
Journal of Neuroinflammation (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.