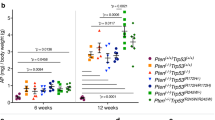
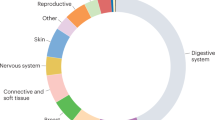
Abstract
Most cases of prostate cancer are now diagnosed as moderate-grade localized disease. These tumor specimens are important tools in the discovery and translation of prostate cancer research; however, unlike more advanced tumors, they are notoriously difficult to grow in the laboratory. We developed a system for efficiently xenografting localized human prostate cancer tissue, and we adapted this protocol to study the interactions between the specific subsets of epithelial and stromal cells. Fresh prostate tissues or isolated epithelial cells are recombined with mouse seminal vesicle mesenchyme (SVM) and grafted under the renal capsule of immunodeficient mice for optimum growth and survival. Alternatively, mouse mesenchyme can be replaced with human prostate fibroblasts in order to determine their contribution to tumor progression. Grafts can be grown for several months to determine the effectiveness of novel therapeutic compounds when administered to host mice, thereby paving the way for personalizing the treatment of individual prostate cancers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Toivanen, R., Taylor, R.A., Pook, D.W., Ellem, S.J. & Risbridger, G.P. Breaking through a roadblock in prostate cancer research: an update on human model systems. J. Steroid Biochem. Mol. Biol. 131, 122–131 (2012).
Maitland, N.J., Frame, F.M., Polson, E.S., Lewis, J.L. & Collins, A.T. Prostate cancer stem cells: do they have a basal or luminal phenotype? Horm. Cancer 2, 47–61 (2011).
Hayward, S.W., Rosen, M.A. & Cunha, G.R. Stromal-epithelial interactions in the normal and neoplastic prostate. Br. J. Urol. 79 (suppl. 2), 18–26 (1997).
Cunha, G.R. Epithelio-mesenchymal interactions in primordial gland structures which become responsive to androgenic stimulation. Anat. Rec. 172, 179–195 (1972).
Goldstein, A.S. et al. Purification and direct transformation of epithelial progenitor cells from primary human prostate. Nat. Protoc. 6, 656–667 (2011).
Lukacs, R.U., Goldstein, A.S., Lawson, D.A., Cheng, D. & Witte, O.N. Isolation, cultivation and characterization of adult murine prostate stem cells. Nat. Protoc. 5, 702–713 (2010).
Taylor, R.A. et al. Formation of human prostate tissue from embryonic stem cells. Nat. Methods 3, 179–181 (2006).
Taylor, R.A. et al. Lineage enforcement by inductive mesenchyme on adult epithelial stem cells across developmental germ layers. Stem Cells 27, 3032–3042 (2009).
Toivanen, R. et al. Brief report: a bioassay to identify primary human prostate cancer repopulating cells. Stem Cells 29, 1310–1314 (2011).
Collins, A.T., Habib, F.K., Maitland, N.J. & Neal, D.E. Identification and isolation of human prostate epithelial stem cells based on α(2)β(1)-integrin expression. J. Cell Sci. 114, 3865–3872 (2001).
Collins, A.T., Berry, P.A., Hyde, C., Stower, M.J. & Maitland, N.J. Prospective identification of tumorigenic prostate cancer stem cells. Cancer Res. 65, 10946–10951 (2005).
Richardson, G.D. et al. CD133, a novel marker for human prostatic epithelial stem cells. J. Cell Sci. 117, 3539–3545 (2004).
Hayashi, N., Cunha, G.R. & Wong, Y.C. Influence of male genital tract mesenchymes on differentiation of Dunning prostatic adenocarcinoma. Cancer Res. 50, 4747–4754 (1990).
Chung, L.W., Zhau, H.E. & Ro, J.Y. Morphologic and biochemical alterations in rat prostatic tumors induced by fetal urogenital sinus mesenchyme. Prostate 17, 165–174 (1990).
Tuxhorn, J.A. et al. Reactive stroma in human prostate cancer: induction of myofibroblast phenotype and extracellular matrix remodeling. Clin. Cancer Res. 8, 2912–2923 (2002).
Olumi, A.F. et al. Carcinoma-associated fibroblasts direct tumor progression of initiated human prostatic epithelium. Cancer Res. 59, 5002–5011 (1999).
Ayala, G. et al. Reactive stroma as a predictor of biochemical-free recurrence in prostate cancer. Clin. Cancer Res. 9, 4792–4801 (2003).
Jia, Z. et al. Diagnosis of prostate cancer using differentially expressed genes in stroma. Cancer Res. 71, 2476–2487 (2011).
Taylor, R.A. et al. Human epithelial basal cells are cells of origin of prostate cancer, independent of CD133 status. Stem Cells 30, 1087–1096 (2012).
Zhao, H., Nolley, R., Chen, Z. & Peehl, D.M. Tissue slice grafts: an in vivo model of human prostate androgen signaling. Am. J. Pathol. 177, 229–239 (2010).
van Weerden, W.M. & Romijn, J.C. Use of nude mouse xenograft models in prostate cancer research. Prostate 43, 263–271 (2000).
Wang, Y. et al. Development and characterization of efficient xenograft models for benign and malignant human prostate tissue. Prostate 64, 149–159 (2005).
Priolo, C. et al. Establishment and genomic characterization of mouse xenografts of human primary prostate tumors. Am. J. Pathol. 176, 1901–1913 (2010).
Wang, Y. et al. An orthotopic metastatic prostate cancer model in SCID mice via grafting of a transplantable human prostate tumor line. Lab. Invest. 85, 1392–1404 (2005).
Lin, D. et al. ASAP1, a gene at 8q24, is associated with prostate cancer metastasis. Cancer Res. 68, 4352–4359 (2008).
Goldstein, A.S., Huang, J., Guo, C., Garraway, I.P. & Witte, O.N. Identification of a cell of origin for human prostate cancer. Science 329, 568–571 (2010).
Ricke, W.A. et al. Prostatic hormonal carcinogenesis is mediated by in situ estrogen production and estrogen receptor alpha signaling. FASEB J. 22, 1512–1520 (2008).
Wang, Y. et al. A human prostatic epithelial model of hormonal carcinogenesis. Cancer Res. 61, 6064–6072 (2001).
Hu, W.Y. et al. Estrogen-initiated transformation of prostate epithelium derived from normal human prostate stem-progenitor cells. Endocrinology 152, 2150–2163 (2011).
Lin, D. et al. Development of metastatic and non-metastatic tumor lines from a patient′s prostate cancer specimen-identification of a small subpopulation with metastatic potential in the primary tumor. Prostate 70, 1636–1644 (2010).
Logothetis, C.J., Navone, N.M. & Lin, S.H. Understanding the biology of bone metastases: key to the effective treatment of prostate cancer. Clin. Cancer Res. 14, 1599–1602 (2008).
Raheem, O. et al. A novel patient-derived intrafemoral xenograft model of bone metastatic prostate cancer that recapitulates mixed osteolytic and osteoblastic lesions. J. Transl. Med. 9, 185 (2011).
Kerger, M. et al. Microscopic assessment of fresh prostate tumour specimens yields significantly increased rates of correctly annotated samples for downstream analysis. Pathology 44, 204–208 (2012).
Szot, G.L., Koudria, P. & Bluestone, J.A. Transplantation of pancreatic islets into the kidney capsule of diabetic mice. J. Vis. Exp. 9, e404 (2007).
Ritner, C. & Bernstein, H.S. Fate mapping of human embryonic stem cells by teratoma formation. J. Vis. Exp. 42, e2036 (2010).
Joesting, M.S. et al. Identification of SFRP1 as a candidate mediator of stromal-to-epithelial signaling in prostate cancer. Cancer Res. 65, 10423–10430 (2005).
Acknowledgements
We would like to thank L. Harewood and M. Simms for providing specimens, A. Meeker and D. Berman for invaluable advice in developing the protocol, S. Hayward for the generous gift of BPH-1 cells and J. Barlow for help in drafting the manuscript. Funding was obtained from the Australian National Health and Medical Research Council (Fellowship to M.G.L.: 1035721, Project Grant: 606492, Enabling Grant: 614296), the Prostate Cancer Foundation of Australia (November Young Investigator Grants to M.G.L. and R.A.T.), the Victorian Cancer Agency (CAPTIV), Yorkshire Cancer Research Programme Support (N.J.M. and A.T.C.) and the Peter and Lyndy White Foundation.
Author information
Authors and Affiliations
Consortia
Contributions
All the authors contributed to protocol development; J.P., S.N., M.M.P., D.W.P. and M.F. acquired, processed or analyzed the surgical specimens; R.T., B.N., M.G.R. and H.W. performed and analyzed experiments; M.G.L., R.A.T., R.T., A.T.C., N.J.M. and G.P.R. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Lawrence, M., Taylor, R., Toivanen, R. et al. A preclinical xenograft model of prostate cancer using human tumors. Nat Protoc 8, 836–848 (2013). https://doi.org/10.1038/nprot.2013.043
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2013.043
This article is cited by
-
The future of patient-derived xenografts in prostate cancer research
Nature Reviews Urology (2023)
-
Using the Microwell-mesh to culture microtissues in vitro and as a carrier to implant microtissues in vivo into mice
Scientific Reports (2021)
-
Oxytocin receptor antagonists as a novel pharmacological agent for reducing smooth muscle tone in the human prostate
Scientific Reports (2021)
-
A humanized orthotopic tumor microenvironment alters the bone metastatic tropism of prostate cancer cells
Communications Biology (2021)
-
Alterations in the methylome of the stromal tumour microenvironment signal the presence and severity of prostate cancer
Clinical Epigenetics (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.