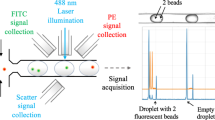
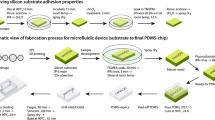
Abstract
Here we present a protocol for analyzing cell cultures using a photonic lab-on-a-chip (PhLoC). By using a broadband light source and a spectrometer, the spectrum of a given cell culture with an arbitrary population is acquired. The PhLoC can work in three different regimes: light scattering (using label-free cells), light scattering plus absorption (using stained cells) and, by subtraction of the two former regimes, absorption (without the scattering band). The acquisition time of the PhLoC is ∼30 ms. Hence, it can be used for rapid cell counting, dead/live ratio estimation or multiparametric measurements through the use of different dyes. The PhLoC, including microlenses, micromirrors and microfluidics, is simply fabricated in a single-mask process (by soft lithographic methods) using low-cost materials. Because of its low cost it can easily be implemented for point-of-care applications. From raw substrates to final results, this protocol can be completed in 29 h.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Yeo, L.Y., Chang, H.C., Chan, P.P.Y. & Friend, J.R. Microfluidic devices for bioapplications. Small 7, 12–48 (2011).
Dittrich, P.S. & Manz, A. Lab-on-a-chip: microfluidics in drug discovery. Nat. Rev. Drug. Discov. 5, 210–218 (2006).
Nagrath, S. et al. Isolation of rare circulating tumour cells in cancer patients by microchip technology. Nature 450, 1235–1239 (2007).
Gilleland, C.L., Rohde, C.B., Zeng, F. & Yanik, M.F. Microfluidic immobilization of physiologically active Caenorhabditis elegans. Nat. Protoc. 5, 1888–1902 (2010).
Yager, P. et al. Microfluidic diagnostic technologies for global public health. Nature 442, 412–418 (2006).
Martinez, A.W., Phillips, S.T. & Whitesides, G.M. Diagnostics for the developing world: microfluidic paper-based analytical devices. Anal. Chem. 82, 3–10 (2010).
Zhao, W. & van den Berg, A. Lab on paper. Lab. Chip 8, 1988–1991 (2008).
Centers for Disease Control and Prevention. Revised classification system for HIV infection and expanded surveillance case definition for AIDS among adolescents and Adults. Morbidity and Mortality Weekly Report 1–19 (18 December 1992).
Myers, F.B. & Lee, L.P. Innovations in optical microfluidic technologies for point-of-care diagnostics. Lab. Chip 8, 2015–2031 (2008).
Mayer, G. et al. Fluorescence-activated cell sorting for aptamer SELEX with cell mixtures. Nat. Protoc. 5, 1993–2004 (2010).
Llobera, A., Demming, S., Wilke, R. & Büttgenbach, S. Multiple internal reflection poly(dimethylsiloxane) systems for optical sensing. Lab. Chip 7, 1560–1566 (2007).
Godin, J. et al. Microfluidics and photonics for Bio-System-on-a-Chip: a review of advancements in technology towards a microfluidic flow cytometry chip. J. Biophoton. 1, 355–376 (2008).
Chung, T.D. & Kim, H.C. Recent advances in miniaturized microfluidic flow cytometry for clinical use. Electrophoresis 28, 4511–4520 (2007).
Qin, D., Xia, Y. & Whitesides, G.M. Soft lithography for micro- and nanoscale patterning. Nat. Protoc. 5, 491–502 (2010).
Llobera, A. et al. Monolithic PDMS passband filters for fluorescence detection. Lab. Chip 10, 1987–1992 (2010).
Llobera, A., Wilke, R. & Büttgenbach, S. Enhancement of the response of poly(dimethylsiloxane) hollow prisms through air mirrors for absorbance-based sensing. Talanta 75, 473–479 (2008).
Ibarlucea, B. et al. Cell screening using disposable photonic lab on a chip systems. Anal. Chem. 82, 4246–4251 (2010).
Merbs, S.L. & Nathans, J. Absorption-spectra of human cone pigments. Nature 356, 433–435 (1992).
Kajihara, D. et al. FRET analysis of protein conformational change through position-specific incorporation of fluorescent amino acids. Nat. Methods 3, 923–929 (2006).
Gao, X.H., Cui, Y.Y., Levenson, R.M., Chung, L.W.K. & Nie, S.M. In vivo cancer targeting and imaging with semiconductor quantum dots. Nat. Biotechnol. 22, 969–976 (2004).
Llobera, A., Wilke, R. & Büttgenbach, S. Poly(dimethylsiloxane) hollow Abbe prism with microlenses for detection based on absorption and refractive index shift. Lab. Chip 4, 24–27 (2004).
Mayers, B.T., Vezenov, D.V., Vullev, V.I. & Whitesides, G.M. Arrays and cascades of fluorescent liquid-liquid waveguides: broadband light sources for spectroscopy in microchannels. Anal. Chem. 77, 1310–1316 (2005).
Llobera, A., Wilke, R. & Büttgenbach, S. Optimization of poly(dimethylsiloxane) hollow prisms for optical sensing. Lab. Chip 5, 506–511 (2005).
Bhattacharya, S., Datta, A., Berg, J.M. & Gangopadhyay, S. Studies on surface wettability of poly(dimethyl)siloxane (PDMS) and glass under oxygen-plasma treatment and correlation with bond strength. J. Microelectromech. Syst. 14, 590–597 (2005).
Blanchard, D., Ligrani, P., Gale, B. & Harvey, I. Micro-structure mechanical failure characterization using rotating Couette flow in a small gap. J. Micromech. Microeng. 15, 792–801 (2005).
Martin, C. et al. Stress and aging minimization in photoplastic AFM probes. Microel. Eng. 86, 1226–1229 (2009).
Feng, R. & Farris, R.J. Influence of processing conditions on the thermal and mechanical properties of SU-8 negative photoresist coatings. J. Micromech. Microeng. 13, 80–88 (2003).
Cheng, L.L., Luk, Y.Y., Murphy, C.J., Israel, B.A. & Abbott, N.L. Compatibility of lyotropic liquid crystals with viruses and mammalian cells that support the replication of viruses. Biomaterials 26, 7173–7182 (2005).
Llobera, A. Device for particle analysis and counting. WIPO patent application no. PCT/ES2011/070011 (11 January 2010).
Demming, S. et al. Poly(dimethylsiloxane) photonic microbioreactors based on segmented waveguides for local absorbance measurement. Electrophoresis 32, 431–439 (2011).
Long, G.L. & Wineforder, J.D. Limit of detection: a closer look at the IUPAC definition. Anal. Chem. 55, A712–A724 (1983).
Cui, X. et al. Lensless high-resolution on-chip optofluidic microscopes for Caenorhabditis elegans and cell imaging. PNAS 105, 10670–10675 (2008).
Acknowledgements
The research leading to these results has received funding from the European Research Council under the European Community's Seventh Framework Programme (FP7/2007-2013)/ERC grant agreement no. 209243 and also from the IST Programme (P. CEZANNE, IST-2-IP-031867). We thank the research group FOR 856, mikroPART 'Mikrosysteme für partikuläre Life-Science-Produkte', for support of this work. Discussions with A. Voigt and G. Gruetzner of Microresist (Germany) regarding the fabrication steps are highly appreciated.
AUTHOR CONTRIBUTIONS
J.V.-P., E.F.-R. and B.I. conducted the experiments. E.F.-R. and C.N. prepared the cell culture. S.D., S.B. and A.L. designed and fabricated the PhLoC. J.A.P., C.D., S.B. and A.L. defined the broadband screening protocol. A.L. supervised the project at all stages.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vila-Planas, J., Fernández-Rosas, E., Ibarlucea, B. et al. Cell analysis using a multiple internal reflection photonic lab-on-a-chip. Nat Protoc 6, 1642–1655 (2011). https://doi.org/10.1038/nprot.2011.383
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2011.383
This article is cited by
-
Biofunctionalized all-polymer photonic lab on a chip with integrated solid-state light emitter
Light: Science & Applications (2015)
-
A guiding light: spectroscopy on digital microfluidic devices using in-plane optical fibre waveguides
Analytical and Bioanalytical Chemistry (2015)
-
Photonic lab-on-chip (PhLOC) for enzyme-catalyzed reactions in continuous flow
Microfluidics and Nanofluidics (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.