Abstract
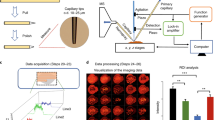
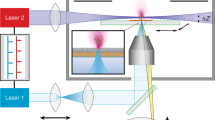
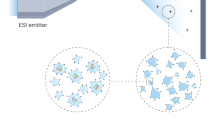
Desorption electrospray ionization (DESI) allows the direct analysis of ordinary objects or pre-processed samples under ambient conditions. Among other applications, DESI is used to identify and record spatial distributions of lipids and drug molecules in biological tissue sections. This technique does not require sample preparation other than production of microtome tissue slices and does not involve the use of ionization matrices. This greatly simplifies the procedure and prevents the redistribution of analytes during matrix deposition. Images are obtained by continuously moving the sample relative to the DESI sprayer and the inlet of the mass spectrometer. The timing of the protocol depends on the size of the surface to be analyzed and on the desired resolution. Analysis of organ tissue slices at 250 μm resolution typically takes between 30 min and 2 h.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Cooks, R.G., Ouyang, Z., Takats, Z. & Wiseman, J.M. Ambient mass spectrometry. Science 311, 1566–1570 (2006).
Takats, Z., Wiseman, J.M., Gologan, B. & Cooks, R.G. Mass spectrometry sampling under ambient conditions with desorption electrospray ionization. Science 306, 471–473 (2004).
Takats, Z., Gologan, B., Wiseman, J.M. & Cooks, R.G. Patent application number 20050230635. Purdue Research Foundation (West Lafayette, IN, USA, 2003).
Cody, R.B., Laramee, J.A. & Durst, H.D. Versatile new ion source for the analysis of materials in open air under ambient conditions. Anal. Chem. 77, 2297–2302 (2005).
Takats, Z., Cotte-Rodriguez, I., Talaty, N., Chen, H.W. & Cooks, R.G. Direct, trace level detection of explosives on ambient surfaces by desorption electrospray ionization mass spectrometry. Chem. Commun. 15, 1950–1952 (2005).
Shiea, J. et al. Electrospray-assisted laser desorption/ionization mass spectrometry for direct ambient analysis of solids. Rapid Commun. Mass Spectrom. 19, 3701–3704 (2005).
Nemes, P. & Vertes, A. Laser ablation electrospray ionization for atmospheric pressure, in vivo, and imaging mass spectrometry. Anal. Chem. 79, 8098–8106 (2007).
McEwen, C.N., McKay, R.G. & Larsen, B.S. Analysis of solids, liquids, and biological tissues using solids probe introduction at atmospheric pressure on commercial LC/MS instruments. Anal. Chem. 77, 7826–7831 (2005).
Laiko, V.V., Baldwin, M.A. & Burlingame, A.L. Atmospheric pressure matrix assisted laser desorption/ionization mass spectrometry. Anal. Chem. 72, 652–657 (2000).
Sampson, J.S., Hawkridge, A.M. & Muddiman, D.C. Generation and detection of multiply-charged peptides and proteins by matrix-assisted laser desorption electropsray ionization (MALDESI) Fourier transform ion cyclotron resonance mass spectrometry. J. Am. Soc. Mass Spectrom. 17, 1712–1716 (2006).
Cotte-Rodriguez, I., Takats, Z., Talaty, N., Chen, H.W. & Cooks, R.G. Desorption electrospray ionization of explosives on surfaces: sensitivity and selectivity enhancement by reactive desorption electrospray ionization. Anal. Chem. 77, 6755–6764 (2005).
Justes, D.R., Talaty, N., Cotte-Rodriguez, I. & Cooks, R.G. Detection of explosives on skin using ambient ionization mass spectrometry. Chem. Commun. 21, 2142–2144 (2007).
Wiseman, J.M., Ifa, D.R., Song, Q. & Cooks, R.G. Tissue imaging at atmospheric pressure using desorption electrospray ionization (DESI) mass spectrometry. Angew Chem. Int. Ed. Engl. 45, 7188–7192 (2006).
Weston, D.J., Bateman, R., Wilson, I.D., Wood, T.R. & Creaser, C.S. Direct analysis of pharmaceutical drug formulations using ion mobility spectrometry/quadrupole-time-of-flight mass spectrometry combined with desorption electrospray ionization. Anal. Chem. 77, 7572–7580 (2005).
Rodriguez-Cruz, S.E. Rapid analysis of controlled substances using desorption electrospray ionization mass spectrometry. Rapid Commun. Mass Spectrom. 20, 53–60 (2006).
Kauppila, T.J., Talaty, N., Kuuranne, T., Kostiainen, R. & Cooks, R.G. Rapid analysis of metabolites and drugs of abuse from urine samples by desorption electrospray ionization-mass spectrometry. Analyst 132, 868–875 (2007).
Morlock, G. & Ueda, Y. New coupling of planar chromatography with direct analysis in real time mass spectrometry. J. Chromatogr. A 1143, 273–251 (2007).
Van Berkel, G.J., Ford, M.J. & Deibel, M.A. Thin-layer chromatography and mass spectrometry coupled using desorption electrospray ionization. Anal. Chem. 77, 1207–1215 (2005).
Mulligan, C.C., MacMillan, DK., Noll, R.J. & Cooks, R.G. Fast analysis of high energy compounds and agricultural chemicals in water with desorption electrospray ionization mass spectrometry. Rapid Commun. Mass Spectrom. 21, 3729–3736 (2007).
Myung, S. et al. Coupling desorption electrospray ionization with ion mobility/mass spectrometry for analysis of protein structure: Evidence for desorption of folded and denatured states. J. Phys. Chem. B 110, 5045–5051 (2006).
Hu, Q.Z., Talaty, N., Noll, R.J. & Cooks, R.G. Desorption electrospray ionization using an Orbitrap mass spectrometer: exact mass measurements on drugs and peptides. Rapid Commun. Mass Spectrom. 20, 3403–3408 (2006).
Takats, Z., Wiseman, J.M. & Cooks, R.G. Ambient mass spectrometry using desorption electrospray ionization (DESI): instrumentation, mechanisms and applications in forensics, chemistry, and biology. J. Mass Spectrom. 40, 1261–1275 (2005).
Ifa, D.R., Manicke, N.E., Rusine, A.L. & Cooks, R.G. Quantitative analysis of small molecules by desorption electrospray ionization (DESI) mass spectrometry from PTFE surfaces. Rapid Commun. Mass Spectrom. 22, 503–510 (2008).
Ifa, D.R., Gumaelius, L.M., Eberlin, L.S., Manicke, N.E. & Cooks, R.G. Forensic analysis of inks by imaging desorption electrospray ionization (DESI) mass spectrometry. Analyst 132, 461–467 (2007).
Van Berkel, G.J. & Kertesz, V. Automated sampling and imaging of analytes separated on thin-layer chromatography plates using desorption electrospray ionization mass spectrometry. Anal. Chem. 78, 4938–4944 (2006).
Wiseman, J.M., Puolitaival, S.M., Takats, Z., Cooks, R.G. & Caprioli, R.M. Mass spectrometric profiling of intact biological tissue by using desorption electrospray ionization. Angew Chem. Int. Ed. Engl. 44, 7094–7097 (2005).
McDonnell, L.A. & Heeren, R.M.A. Imaging mass spectrometry. Mass Spectrom. Rev. 26, 606–643 (2007).
Touboul, D., Kollmer, F., Niehuis, E., Brunelle, A. & Laprevote, O. Improvement of biological time-of-flight-secondary ion mass spectrometry imaging with a bismuth cluster ion source. J. Am. Soc. Mass Spectrom. 16, 1608–1618 (2005).
Touboul, D. et al. Tissue molecular ion imaging by gold cluster ion bombardment. Anal. Chem. 76, 1550–1559 (2004).
Stoeckli, M., Chaurand, P., Hallahan, D.E. & Caprioli, R.M. Imaging mass spectrometry: a new technology for the analysis of protein expression in mammalian tissues. Nat. Med. 7, 493–496 (2001).
Chaurand, P., Schwartz, S.A., Reyzer, M.L. & Caprioli, R.M. Imaging mass spectrometry: principles and potentials. Toxicol. Pathol. 33, 92–101 (2005).
Khatib-Shahidi, S., Andersson, M., Herman, J.L., Gillespie, T.A. & Caprioli, R.M. Direct molecular analysis of whole-body animal tissue sections by imaging MALDI mass spectrometry. Anal. Chem. 78, 6448–6456 (2006).
Hsieh, Y., Chen, J. & Korfmacher, W.A. Mapping pharmaceuticals in tissues using MALDI imaging mass spectrometry. J. Pharmacol. Toxicol. Methods 55, 193–200 (2007).
Luxembourg, S.L., Mize, T.H., McDonnell, L.A. & Heeren, R.M.A. High-spatial resolution mass spectrometric imaging of peptide and protein distributions on a surface. Anal. Chem. 76, 5339–5344 (2004).
Ifa, D.R., Wiseman, J.M., Song, Q. & Cooks, R.G. Development of capabilities for imaging mass spectrometry under ambient conditions with desorption electrospray ionization (DESI). Int. J. Mass Spectrom. 259, 8–15 (2007).
Ostrowski, S.G., Van Bell, C.T., Winograd, N. & Ewing, A.G. Mass spectrometric imaging of highly curved membranes during Tetrahymena mating. Science 305, 71–73 (2004).
McDonnell, L.A. et al. Subcellular imaging mass spectrometry of brain tissue. J. Mass Spectrom. 40, 160–168 (2005).
Todd, P.J., Schaaff, T.G., Chaurand, P. & Caprioli, R.M. Organic ion imaging of biological tissue with secondary ion mass spectrometry and matrix-assisted laser desorption/ionization. J. Mass Spectrom. 36, 355–369 (2001).
Schueler, B., Sander, P. & Reed, D.A. A Time-of-flight secondary ion-microscope. Vacuum 41, 1661–1664 (1990).
Northen, T.R. et al. Clathrate nanostructures for mass spectrometry. Nature 449, 1033–1037 (2007).
Venter, A. & Cooks, R.G. Desorption electrospray ionization in a small tight pressure enclosure. Anal. Chem. 79, 6398–6403 (2007).
Acknowledgements
The authors acknowledge funding from the Office of Naval Research (Research Tools program, grant N000140510454) from which this work was supported. The authors gratefully acknowledge the assistance of Candice Kissinger and Simon Katner of Bioanalytical Systems Inc. (West Lafayette, IN) for graciously providing animal tissues and performing the surgical and dosing procedures in accordance with the local animal care and use guidelines.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Dr Justin Wiseman, formerly of Purdue University, is now employed by PROSOLIA Inc., Indianapolis, a company which sells DESI ion sources. None of the other authors have competing financial interests.
Rights and permissions
About this article
Cite this article
Wiseman, J., Ifa, D., Venter, A. et al. Ambient molecular imaging by desorption electrospray ionization mass spectrometry. Nat Protoc 3, 517–524 (2008). https://doi.org/10.1038/nprot.2008.11
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2008.11
This article is cited by
-
Desorption electrospray ionization and matrix-assisted laser desorption/ionization as imaging approaches for biological samples analysis
Analytical and Bioanalytical Chemistry (2023)
-
Mammalian ovarian lipid distributions by desorption electrospray ionization–mass spectrometry (DESI-MS) imaging
Analytical and Bioanalytical Chemistry (2020)
-
Monitoring of changes in lipid profiles during PLK1 knockdown in cancer cells using DESI MS
Analytical and Bioanalytical Chemistry (2016)
-
A Comparison of DESI-MS and LC-MS for the Lipidomic Profiling of Human Cancer Tissue
Journal of the American Society for Mass Spectrometry (2016)
-
Direct Electrospray Ionization Mass Spectrometric Profiling of Real-World Samples via a Solid Sampling Probe
Journal of the American Society for Mass Spectrometry (2013)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.