Abstract
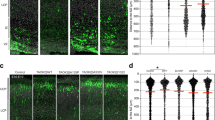
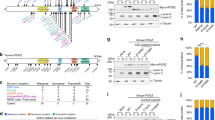
Over-inhibition is thought to be one of the underlying causes of the cognitive deficits in Ts65Dn mice, the most widely used model of Down syndrome. We found a direct link between gene triplication and defects in neuron production during embryonic development. These neurogenesis defects led to an imbalance between excitatory and inhibitory neurons and to increased inhibitory drive in the Ts65Dn forebrain. We discovered that Olig1 and Olig2, two genes that are triplicated in Down syndrome and in Ts65Dn mice, were overexpressed in the Ts65Dn forebrain. To test the hypothesis that Olig triplication causes the neurological phenotype, we used a genetic approach to normalize the dosage of these two genes and thereby rescued the inhibitory neuron phenotype in the Ts65Dn brain. These data identify seminal alterations during brain development and suggest a mechanistic relationship between triplicated genes and these brain abnormalities in the Ts65Dn mouse.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Epstein, C.J. Developmental genetics. Experientia 42, 1117–1128 (1986).
Korenberg, J.R. et al. Down syndrome: toward a molecular definition of the phenotype. Am. J. Med. Genet. Suppl. 7, 91–97 (1990).
Golden, J.A. & Hyman, B. Development of the superior temporal neocortex is anomalous in trisomy 21. J. Neuropathol. Exp. Neurol. 53, 513–520 (1994).
Schmidt-Sidor, B., Wisniewski, K.E., Shepard, T.H. & Sersen, E.A. Brain growth in Down syndrome subjects 15 to 22 weeks of gestational age and birth to 60 months. Clin. Neuropathol. 9, 181–190 (1990).
Weitzdoerfer, R., Dierssen, M., Fountoulakis, M. & Lubec, G. Fetal life in Down syndrome starts with normal neuronal density but impaired dendritic spines and synaptosomal structure. J. Neural Transm. Suppl. 61, 59–70 (2001).
Baxter, L.L., Moran, T.H., Richtsmeier, J.T., Troncoso, J. & Reeves, R.H. Discovery and genetic localization of Down syndrome cerebellar phenotypes using the Ts65Dn mouse. Hum. Mol. Genet. 9, 195–202 (2000).
Chakrabarti, L., Galdzicki, Z. & Haydar, T.F. Defects in embryonic neurogenesis and initial synapse formation in the forebrain of the Ts65Dn mouse model of Down syndrome. J. Neurosci. 27, 11483–11495 (2007).
Lorenzi, H.A. & Reeves, R.H. Hippocampal hypocellularity in the Ts65Dn mouse originates early in development. Brain Res. 1104, 153–159 (2006).
Belichenko, P.V. et al. Excitatory-inhibitory relationship in the fascia dentata in the Ts65Dn mouse model of Down syndrome. J. Comp. Neurol. 512, 453–466 (2009).
Belichenko, P.V. et al. Synaptic structural abnormalities in the Ts65Dn mouse model of Down syndrome. J. Comp. Neurol. 480, 281–298 (2004).
Kleschevnikov, A.M. et al. Hippocampal long-term potentiation suppressed by increased inhibition in the Ts65Dn mouse, a genetic model of Down syndrome. J. Neurosci. 24, 8153–8160 (2004).
Siarey, R.J., Stoll, J., Rapoport, S.I. & Galdzicki, Z. Altered long-term potentiation in the young and old Ts65Dn mouse, a model for Down syndrome. Neuropharmacology 36, 1549–1554 (1997).
Costa, A.C., Walsh, K. & Davisson, M.T. Motor dysfunction in a mouse model for Down syndrome. Physiol. Behav. 68, 211–220 (1999).
Hyde, L.A., Frisone, D.F. & Crnic, L.S. Ts65Dn mice, a model for Down syndrome, have deficits in context discrimination learning suggesting impaired hippocampal function. Behav. Brain Res. 118, 53–60 (2001).
Reeves, R.H. et al. A mouse model for Down syndrome exhibits learning and behavior deficits. Nat. Genet. 11, 177–184 (1995).
Kurt, M.A., Davies, D.C., Kidd, M., Dierssen, M. & Florez, J. Synaptic deficit in the temporal cortex of partial trisomy 16 (Ts65Dn) mice. Brain Res. 858, 191–197 (2000).
Perez-Cremades, D. et al. Alteration of inhibitory circuits in the somatosensory cortex of Ts65Dn mice, a model for Down's syndrome. J. Neural Transm. 117, 445–455 (2010).
Costa, A.C. & Grybko, M.J. Deficits in hippocampal CA1 LTP induced by TBS, but not HFS, in the Ts65Dn mouse: a model of Down syndrome. Neurosci. Lett. 382, 317–322 (2005).
Fernandez, F. et al. Pharmacotherapy for cognitive impairment in a mouse model of Down syndrome. Nat. Neurosci. 10, 411–413 (2007).
Haydar, T.F. Advanced microscopic imaging methods to investigate cortical development and the etiology of mental retardation. Ment. Retard. Dev. Disabil. Res. Rev. 11, 303–316 (2005).
Miller, M.W. Cogeneration of retrogradely labeled corticocortical projection and GABA-immunoreactive local circuit neurons in cerebral cortex. Brain Res. 355, 187–192 (1985).
Valcanis, H. & Tan, S.S. Layer specification of transplanted interneurons in developing mouse neocortex. J. Neurosci. 23, 5113–5122 (2003).
DeFelipe, J. Neocortical neuronal diversity: chemical heterogeneity revealed by colocalization studies of classic neurotransmitters, neuropeptides, calcium-binding proteins and cell surface molecules. Cereb. Cortex 3, 273–289 (1993).
Kawaguchi, Y. & Kubota, Y. GABAergic cell subtypes and their synaptic connections in rat frontal cortex. Cereb. Cortex 7, 476–486 (1997).
Markram, H. et al. Interneurons of the neocortical inhibitory system. Nat. Rev. Neurosci. 5, 793–807 (2004).
Wonders, C.P. & Anderson, S.A. The origin and specification of cortical interneurons. Nat. Rev. Neurosci. 7, 687–696 (2006).
Hobert, O. & Westphal, H. Functions of LIM-homeobox genes. Trends Genet. 16, 75–83 (2000).
Liodis, P. et al. Lhx6 activity is required for the normal migration and specification of cortical interneuron subtypes. J. Neurosci. 27, 3078–3089 (2007).
Shirasaki, R. & Pfaff, S.L. Transcriptional codes and the control of neuronal identity. Annu. Rev. Neurosci. 25, 251–281 (2002).
Eisenstat, D.D. et al. DLX-1, DLX-2, and DLX-5 expression define distinct stages of basal forebrain differentiation. J. Comp. Neurol. 414, 217–237 (1999).
Grigoriou, M., Tucker, A.S., Sharpe, P.T. & Pachnis, V. Expression and regulation of Lhx6 and Lhx7, a novel subfamily of LIM homeodomain encoding genes, suggests a role in mammalian head development. Development 125, 2063–2074 (1998).
Porteus, M.H., Bulfone, A., Ciaranello, R.D. & Rubenstein, J.L. Isolation and characterization of a novel cDNA clone encoding a homeodomain that is developmentally regulated in the ventral forebrain. Neuron 7, 221–229 (1991).
Sussel, L., Marin, O., Kimura, S. & Rubenstein, J.L. Loss of Nkx2.1 homeobox gene function results in a ventral to dorsal molecular respecification within the basal telencephalon: evidence for a transformation of the pallidum into the striatum. Development 126, 3359–3370 (1999).
Alifragis, P., Liapi, A. & Parnavelas, J.G. Lhx6 regulates the migration of cortical interneurons from the ventral telencephalon, but does not specify their GABA phenotype. J. Neurosci. 24, 5643–5648 (2004).
Lu, Q.R. et al. Common developmental requirement for Olig function indicates a motor neuron/oligodendrocyte connection. Cell 109, 75–86 (2002).
Takebayashi, H. et al. Dynamic expression of basic helix-loop-helix Olig family members: implication of Olig2 in neuron and oligodendrocyte differentiation and identification of a new member, Olig3. Mech. Dev. 99, 143–148 (2000).
Zhou, Q. & Anderson, D.J. The bHLH transcription factors OLIG2 and OLIG1 couple neuronal and glial subtype specification. Cell 109, 61–73 (2002).
Bertrand, N., Castro, D.S. & Guillemot, F. Proneural genes and the specification of neural cell types. Nat. Rev. Neurosci. 3, 517–530 (2002).
Ma, Q. Transcriptional regulation of neuronal phenotype in mammals. J. Physiol. (Lond.) 575, 379–387 (2006).
Miyoshi, G., Butt, S.J., Takebayashi, H. & Fishell, G. Physiologically distinct temporal cohorts of cortical interneurons arise from telencephalic Olig2-expressing precursors. J. Neurosci. 27, 7786–7798 (2007).
Kahlem, P. et al. Transcript level alterations reflect gene dosage effects across multiple tissues in a mouse model of Down syndrome. Genome Res. 14, 1258–1267 (2004).
Contestabile, A. et al. Cell cycle alteration and decreased cell proliferation in the hippocampal dentate gyrus and in the neocortical germinal matrix of fetuses with down syndrome and in Ts65Dn mice. Hippocampus 17, 665–678 (2007).
Segal, D.J. & McCoy, E.E. Studies on Down's syndrome in tissue culture. I. Growth rates and protein contents of fibroblast cultures. J. Cell. Physiol. 83, 85–90 (1974).
Clark, S., Schwalbe, J., Stasko, M.R., Yarowsky, P.J. & Costa, A.C. Fluoxetine rescues deficient neurogenesis in hippocampus of the Ts65Dn mouse model for Down syndrome. Exp. Neurol. 200, 256–261 (2006).
Freund, T.F. & Buzsaki, G. Interneurons of the hippocampus. Hippocampus 6, 347–470 (1996).
Dierssen, M. et al. Alterations of neocortical pyramidal cell phenotype in the Ts65Dn mouse model of Down syndrome: effects of environmental enrichment. Cereb. Cortex 13, 758–764 (2003).
Gibson, J.R., Bartley, A.F., Hays, S.A. & Huber, K.M. Imbalance of neocortical excitation and inhibition and altered UP states reflect network hyperexcitability in the mouse model of fragile X syndrome. J. Neurophysiol. 100, 2615–2626 (2008).
Polleux, F. & Lauder, J.M. Toward a developmental neurobiology of autism. Ment. Retard. Dev. Disabil. Res. Rev. 10, 303–317 (2004).
Cortez, M.A. et al. Infantile spasms and Down syndrome: a new animal model. Pediatr. Res. 65, 499–503 (2009).
Harashima, C. et al. Abnormal expression of the G protein–activated inwardly rectifying potassium channel 2 (GIRK2) in hippocampus, frontal cortex and substantia nigra of Ts65Dn mouse: a model of Down syndrome. J. Comp. Neurol. 494, 815–833 (2006).
Acknowledgements
We wish to thank V. Gallo, J. Corbin, and members of Corbin and Haydar laboratories for discussions and critical reading of the manuscript. This work was supported by a Dana Foundation Neuro-Immuno Imaging grant (T.F.H.), RO1 HD05780 (T.F.H. and Z.G.), the National Down Syndrome Society (L.C.), the Jerome Lejeune Foundation (Z.G.), Uniformed Services University of the Health Sciences (Z.G.) and a gift from Robin and Rob Wilder. Imaging was supported by the Intellectual and Developmental Disabilities Research Center (P30 HD40677).
Author information
Authors and Affiliations
Contributions
L.C. performed the experiments, analyzed the data, wrote the manuscript and generated the figures. T.K.B., N.P.C. (both contributed equally), J.T.R.I. and Z.G. performed the electrophysiology experiments, analyzed data and revealed the physiological phenotype. R.S.E.C. generated the RNA probes. T.F.H. generated the hypothesis, designed experiments, analyzed data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 and Supplementary Tables 1–5 (PDF 6756 kb)
Rights and permissions
About this article
Cite this article
Chakrabarti, L., Best, T., Cramer, N. et al. Olig1 and Olig2 triplication causes developmental brain defects in Down syndrome. Nat Neurosci 13, 927–934 (2010). https://doi.org/10.1038/nn.2600
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2600
This article is cited by
-
Understanding the genetic mechanisms and cognitive impairments in Down syndrome: towards a holistic approach
Journal of Neurology (2024)
-
Atypical electrophysiological and behavioral responses to diazepam in a leading mouse model of Down syndrome
Scientific Reports (2021)
-
Spatiotemporal development of spinal neuronal and glial populations in the Ts65Dn mouse model of Down syndrome
Journal of Neurodevelopmental Disorders (2019)
-
Unbalanced dendritic inhibition of CA1 neurons drives spatial-memory deficits in the Ts2Cje Down syndrome model
Nature Communications (2019)
-
Prenatal treatment with EGCG enriched green tea extract rescues GAD67 related developmental and cognitive defects in Down syndrome mouse models
Scientific Reports (2019)