Abstract
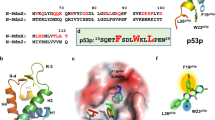
In tumors that retain wild-type p53, its tumor-suppressor function is often impaired as a result of the deregulation of HDM-2, which binds to p53 and targets it for proteasomal degradation. We have screened a chemical library and identified a small molecule named RITA (reactivation of p53 and induction of tumor cell apoptosis), which bound to p53 and induced its accumulation in tumor cells. RITA prevented p53–HDM-2 interaction in vitro and in vivo and affected p53 interaction with several negative regulators. RITA induced expression of p53 target genes and massive apoptosis in various tumor cells lines expressing wild-type p53. RITA suppressed the growth of human fibroblasts and lymphoblasts only upon oncogene expression and showed substantial p53-dependent antitumor effect in vivo. RITA may serve as a lead compound for the development of an anticancer drug that targets tumors with wild-type p53.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Vogelstein, B., Lane, D. & Levine, A.J. Surfing the p53 network. Nature 408, 307–310 (2000).
Olivier, M. et al. The IARC TP53 database: new online mutation analysis and recommendations to users. Hum. Mutat. 19, 607–614 (2002).
Chene, P. Inhibiting the p53–MDM2 interaction: an important target for cancer therapy. Nat. Rev. Cancer 3, 102–109 (2003).
Prives, C. Signaling to p53: breaking the MDM2-p53 circuit. Cell 95, 5–8 (1998).
Montes de Oca Luna, R., Wagner, D.S. & Lozano, G. Rescue of early embryonic lethality in mdm2-deficient mice by deletion of p53. Nature 378, 203–206 (1995).
Vousden, K.H. Activation of the p53 tumor suppressor protein. Biochim. Biophys. Acta 1602, 47–59.(2002).
Evan, G. & Littlewood, T. A matter of life and cell death. Science 281, 1317–1322 (1998).
Bottger, A. et al. Design of a synthetic Mdm2-binding mini protein that activates the p53 response in vivo. Curr. Biol. 7, 860–869 (1997).
Chene, P. et al. A small synthetic peptide, which inhibits the p53-hdm2 interaction, stimulates the p53 pathway in tumour cell lines. J. Mol. Biol. 299, 245–253 (2000).
Midgley, C.A. et al. An N-terminal p14ARF peptide blocks Mdm2-dependent ubiquitination in vitro and can activate p53 in vivo. Oncogene 19, 2312–2323 (2000).
Lai, Z. et al. Differentiation of Hdm2-mediated p53 ubiquitination and Hdm2 autoubiquitination activity by small molecular weight inhibitors. Proc. Natl. Acad. Sci. USA 99, 14734–14739 (2002).
Zhao, J. et al. The initial evaluation of non-peptidic small-molecule HDM2 inhibitors based on p53–HDM2 complex structure. Cancer Lett. 183, 69–77 (2002).
Vassilev, L.T. et al. In vivo activation of the p53 pathway by small-molecule antagonists of MDM2. Science 303, 844–848 (2004).
Bunz, F. et al. Requirement for p53 and p21 to sustain G2 arrest after DNA damage. Science 282, 1497–1501 (1998).
Bunz, F. et al. Disruption of p53 in human cancer cells alters the responses to therapeutic agents. J. Clin. Invest. 104, 263–269 (1999).
Rigler, R. et al. Specific binding of proinsulin C-peptide to human cell membranes. Proc. Natl. Acad. Sci. USA 96, 13318–13323 (1999).
Yakovleva, T. et al. p53 Latency. C-terminal domain prevents binding of p53 core to target but not to nonspecific DNA sequences. J. Biol. Chem. 276, 15650–15658 (2001).
Wang, L., Grossman, S.R. & Kieff, E. Epstein-Barr virus nuclear protein 2 interacts with p300, CBP, and PCAF histone acetyltransferases in activation of the LMP1 promoter. Proc. Natl. Acad. Sci. USA 97, 430–435 (2000).
Lin, J., Chen, J., Elenbaas, B. & Levine, A.J. Several hydrophobic amino acids in the p53 amino-terminal domain are required for transcriptional activation, binding to mdm-2 and the adenovirus 5 E1B 55-kDa protein. Genes Dev. 8, 1235–1246 (1994).
Kussie, P.H. et al. Structure of the MDM2 oncoprotein bound to the p53 tumor suppressor transactivation domain. Science 274, 948–953. (1996).
Burch, L.R., Midgley, C.A., Currie, R.A., Lane, D.P. & Hupp, T.R. Mdm2 binding to a conformationally sensitive domain on p53 can be modulated by RNA. FEBS Lett. 472, 93–98 (2000).
Kanovsky, M. et al. Peptides from the amino terminal mdm-2-binding domain of p53, designed from conformational analysis, are selectively cytotoxic to transformed cells. Proc. Natl. Acad. Sci. USA 98, 12438–12443 (2001).
Gu, W. & Roeder, R.G. Activation of p53 sequence-specific DNA binding by acetylation of the p53 C-terminal domain. Cell 90, 595–606 (1997).
Grossman, S.R. et al. Polyubiquitination of p53 by a ubiquitin ligase activity of p300. Science 300, 342–344 (2003).
Bergamaschi, D. et al. iASPP oncoprotein is a key inhibitor of p53 conserved from worm to human. Nat. Genet. 33, 162–167 (2003).
Mihara, M. et al. p53 has a direct apoptogenic role at the mitochondria. Mol. Cell 11, 577–590 (2003).
Nikolaev, A.Y., Li, M., Puskas, N., Qin, J. & Gu, W. Parc: a cytoplasmic anchor for p53. Cell 112, 29–40 (2003).
Pandhare, J., Dash, C., Rao, M. & Deshpande, V. Slow tight binding inhibition of proteinase K by a proteinaceous inhibitor: conformational alterations responsible for conferring irreversibility to the enzyme-inhibitor complex. J. Biol. Chem. 278, 48735–48744 (2003).
Jabbur, J.R. et al. Mdm-2 binding and TAF(II)31 recruitment is regulated by hydrogen bond disruption between the p53 residues Thr18 and Asp21. Oncogene 21, 7100–7113 (2002).
Iyer, N.G. et al. p300 regulates p53-dependent apoptosis after DNA damage in colorectal cancer cells by modulation of PUMA/p21 levels. Proc. Natl. Acad. Sci. USA 101, 7386–7391 (2004).
Grossman, S.R. et al. p300/MDM2 complexes participate in MDM2-mediated p53 degradation. Mol. Cell 2, 405–415 (1998).
Liu, W.L., Midgley, C., Stephen, C., Saville, M. & Lane, D.P. Biological significance of a small highly conserved region in the N terminus of the p53 tumour suppressor protein. J. Mol. Biol. 313, 711–731 (2001).
Friedlander, P., Legros, Y., Soussi, T. & Prives, C. Regulation of mutant p53 temperature-sensitive DNA binding. J. Biol. Chem. 271, 25468–25478 (1996).
Hansen, S., Hupp, T.R. & Lane, D.P. Allosteric regulation of the thermostability and DNA binding activity of human p53 by specific interacting proteins. CRC Cell Transformation Group. J. Biol. Chem. 271, 3917–3924 (1996).
Cho, Y., Gorina, S., Jeffrey, P.D. & Pavletich, N.P. Crystal structure of a p53 tumor suppressor-DNA complex: understanding tumorigenic mutations. Science 265, 346–355 (1994).
Jeffrey, P.D., Gorina, S. & Pavletich, N.P. Crystal structure of the tetramerization domain of the p53 tumor suppressor at 1.7 angstroms. Science 267, 1498–14502 (1995).
Lee, W. et al. Solution structure of the tetrameric minimum transforming domain of p53. Nat. Struct. Biol. 1, 877–890 (1994).
Baker, S.J., Markowitz, S., Fearon, E.R., Willson, J.K. & Vogelstein, B. Suppression of human colorectal carcinoma cell growth by wild-type p53. Science 249, 912–915 (1990).
D'Orazi, G. et al. Exogenous wt-p53 protein is active in transformed cells but not in their non-transformed counterparts: implications for cancer gene therapy without tumor targeting. J. Gene Med. 2, 11–21 (2000).
Nieves-Neira, W. et al. DNA protein cross-links produced by NSC 652287, a novel thiophene derivative active against human renal cancer cells. Mol. Pharmacol. 56, 478–484 (1999).
Rivera, M.I. et al. Selective toxicity of the tricyclic thiophene NSC 652287 in renal carcinoma cell lines: differential accumulation and metabolism. Biochem. Pharmacol. 57, 1283–1295 (1999).
Selivanova, G. et al. The single-stranded DNA end binding site of p53 coincides with the C- terminal regulatory region. Nucleic Acids Res. 24, 3560–3567 (1996).
Schuhmacher, M. et al. Control of cell growth by c-Myc in the absence of cell division. Curr. Biol. 9, 1255–1258 (1999).
Felsher, D.W. & Bishop, J.M. Transient excess of MYC activity can elicit genomic instability and tumorigenesis. Proc. Natl. Acad. Sci. USA 96, 3940–3944 (1999).
Vigo, E. et al. CDC25A phosphatase is a target of E2F and is required for efficient E2F-induced S phase. Mol. Cell. Biol. 19, 6379–6395 (1999).
Hengstermann, A., Linares, L.K., Ciechanover, A., Whitaker, N.J. & Scheffner, M. Complete switch from Mdm2 to human papillomavirus E6-mediated degradation of p53 in cervical cancer cells. Proc. Natl. Acad. Sci. USA 98, 1218–1223 (2001).
Acknowledgements
We thank the Drug Synthesis & Chemistry Branch, Developmental Therapeutics Program, Division of Cancer Treatment and Diagnosis, National Cancer Institute, for the library of low molecular weight compounds. We are indebted to all our colleagues for sharing cell lines and reagents with us. This work was supported by project grants from the Swedish Cancer Society (Cancerfonden), Swedish Research Council, Graduate Research School for Genomics and Bioinformatics (Stockholm), Lars Hiertas Minne and Robert Lundbergs Foundations and the Cancer Society of Stockholm. P.B. is a recipient of a fellowship from Wenner-Gren Foundation, Stockholm.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
RITA did not induce p53 and did not suppress the growth of mouse cell lines in a p53-dependent manner. (PDF 297 kb)
Supplementary Fig. 2
Studies of RITA interaction with proteins using fluorescence correlation spectroscopy. (PDF 960 kb)
Supplementary Fig. 3
RITA did not inhibit the binding of p53 to the monoclonal antibody DO1 and Bcl-xL. (PDF 435 kb)
Supplementary Table 1
Growth suppression induced by RITA in different cell lines (PDF 83 kb)
Rights and permissions
About this article
Cite this article
Issaeva, N., Bozko, P., Enge, M. et al. Small molecule RITA binds to p53, blocks p53–HDM-2 interaction and activates p53 function in tumors. Nat Med 10, 1321–1328 (2004). https://doi.org/10.1038/nm1146
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1146
This article is cited by
-
Drugging p53 in cancer: one protein, many targets
Nature Reviews Drug Discovery (2023)
-
Cellular senescence in glioma
Journal of Neuro-Oncology (2023)
-
Transition of amyloid/mutant p53 from tumor suppressor to an oncogene and therapeutic approaches to ameliorate metastasis and cancer stemness
Cancer Cell International (2022)
-
Should mutant TP53 be targeted for cancer therapy?
Cell Death & Differentiation (2022)
-
A role for bioinorganic chemistry in the reactivation of mutant p53 in cancer
JBIC Journal of Biological Inorganic Chemistry (2022)