Abstract
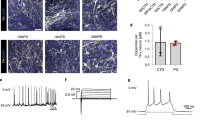
Familial dysautonomia (FD) is a debilitating disorder that affects derivatives of the neural crest (NC). For unknown reasons, people with FD show marked differences in disease severity despite carrying an identical, homozygous point mutation in IKBKAP, encoding IκB kinase complex–associated protein. Here we present disease-related phenotypes in human pluripotent stem cells (PSCs) that capture FD severity. Cells from individuals with severe but not mild disease show impaired specification of NC derivatives, including autonomic and sensory neurons. In contrast, cells from individuals with severe and mild FD show defects in peripheral neuron survival, indicating that neurodegeneration is the main culprit for cases of mild FD. Although genetic repair of the FD-associated mutation reversed early developmental NC defects, sensory neuron specification was not restored, indicating that other factors may contribute to disease severity. Whole-exome sequencing identified candidate modifier genes for individuals with severe FD. Our study demonstrates that PSC-based modeling is sensitive in recapitulating disease severity, which presents an important step toward personalized medicine.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Inoue, H., Nagata, N., Kurokawa, H. & Yamanaka, S. iPS cells: a game changer for future medicine. EMBO J. 33, 409–417 (2014).
Zeltner, N. & Studer, L. Pluripotent stem cell-based disease modeling: current hurdles and future promise. Curr. Opin. Cell Biol. 37, 102–110 (2015).
Bellin, M., Marchetto, M.C., Gage, F.H. & Mummery, C.L. Induced pluripotent stem cells: the new patient? Nat. Rev. Mol. Cell Biol. 13, 713–726 (2012).
Lee, G. et al. Modelling pathogenesis and treatment of familial dysautonomia using patient-specific iPSCs. Nature 461, 402–406 (2009).
Axelrod, F.B. Familial dysautonomia. Muscle Nerve 29, 352–363 (2004).
Slaugenhaupt, S.A. et al. Tissue-specific expression of a splicing mutation in the IKBKAP gene causes familial dysautonomia. Am. J. Hum. Genet. 68, 598–605 (2001).
Axelrod, F.B., Goldberg, J.D., Ye, X.Y. & Maayan, C. Survival in familial dysautonomia: Impact of early intervention. J. Pediatr. 141, 518–523 (2002).
Dong, J., Edelmann, L., Bajwa, A.M., Kornreich, R. & Desnick, R.J. Familial dysautonomia: detection of the IKBKAP IVS20+6T→C and R696P mutations and frequencies among Ashkenazi Jews. Am. J. Med. Genet. 110, 253–257 (2002).
Pearson, J., Pytel, B.A., Grover-Johnson, N., Axelrod, F. & Dancis, J. Quantitative studies of dorsal root ganglia and neuropathologic observations on spinal cords in familial dysautonomia. J. Neurol. Sci. 35, 77–92 (1978).
Pearson, J. & Pytel, B.A. Quantitative studies of sympathetic ganglia and spinal cord intermedio-lateral gray columns in familial dysautonomia. J. Neurol. Sci. 39, 47–59 (1978).
Axelrod, F.B. et al. Progressive sensory loss in familial dysautonomia. Pediatrics 67, 517–522 (1981).
Slaugenhaupt, S.A. et al. Rescue of a human mRNA splicing defect by the plant cytokinin kinetin. Hum. Mol. Genet. 13, 429–436 (2004).
Axelrod, F.B. et al. Kinetin improves IKBKAP mRNA splicing in patients with familial dysautonomia. Pediatr. Res. 70, 480–483 (2011).
Lee, G. et al. Large-scale screening using familial dysautonomia induced pluripotent stem cells identifies compounds that rescue IKBKAP expression. Nat. Biotechnol. 30, 1244–1248 (2012).
Miller, J.D. et al. Human iPSC-based modeling of late-onset disease via progerin-induced aging. Cell Stem Cell 13, 691–705 (2013).
Fusaki, N., Ban, H., Nishiyama, A., Saeki, K. & Hasegawa, M. Efficient induction of transgene-free human pluripotent stem cells using a vector based on Sendai virus, an RNA virus that does not integrate into the host genome. Proc. Jpn. Acad., Ser. B, Phys. Biol. Sci. 85, 348–362 (2009).
Lee, G. et al. Isolation and directed differentiation of neural crest stem cells derived from human embryonic stem cells. Nat. Biotechnol. 25, 1468–1475 (2007).
Zeltner, N., Lafaille, F.G., Fattahi, F. & Studer, L. Feeder-free derivation of neural crest progenitor cells from human pluripotent stem cells. J. Vis. Exp. 87, e51609 (2014).
Chambers, S.M. et al. Combined small-molecule inhibition accelerates developmental timing and converts human pluripotent stem cells into nociceptors. Nat. Biotechnol. 30, 715–720 (2012).
Wainger, B.J. et al. Modeling pain in vitro using nociceptor neurons reprogrammed from fibroblasts. Nat. Neurosci. 18, 17–24 (2015).
Mica, Y., Lee, G., Chambers, S.M., Tomishima, M.J. & Studer, L. Modeling neural crest induction, melanocyte specification, and disease-related pigmentation defects in hESCs and patient-specific iPSCs. Cell Rep. 3, 1140–1152 (2013).
Fattahi, F. et al. Deriving human ENS lineages for cell therapy and drug discovery in Hirschsprung disease. Nature 531, 105–109 (2016).
Howard, M.J. Mechanisms and perspectives on differentiation of autonomic neurons. Dev. Biol. 277, 271–286 (2005).
Grundy, D. & Schemann, M. Enteric nervous system. Curr. Opin. Gastroenterol. 23, 121–126 (2007).
Schlosser, G. Induction and specification of cranial placodes. Dev. Biol. 294, 303–351 (2006).
Oh, Y. et al. Functional coupling with cardiac muscle promotes maturation of hPSC-derived sympathetic neurons. Cell Stem Cell 19, 95–106 (2016).
Dietrich, P., Alli, S., Shanmugasundaram, R. & Dragatsis, I. IKAP expression levels modulate disease severity in a mouse model of familial dysautonomia. Hum. Mol. Genet. 21, 5078–5090 (2012).
Morini, E. et al. Sensory and autonomic deficits in a new humanized mouse model of familial dysautonomia. Hum. Mol. Genet. 25, 1116–1128 (2016).
Ding, Q. et al. Enhanced efficiency of human pluripotent stem cell genome editing through replacing TALENs with CRISPRs. Cell Stem Cell 12, 393–394 (2013).
Mali, P. et al. RNA-guided human genome engineering via Cas9. Science 339, 823–826 (2013).
Adzhubei, I.A. et al. A method and server for predicting damaging missense mutations. Nat. Methods 7, 248–249 (2010).
Kumar, P., Henikoff, S. & Ng, P.C. Predicting the effects of coding non-synonymous variants on protein function using the SIFT algorithm. Nat. Protoc. 4, 1073–1081 (2009).
Itan, Y. et al. The human gene damage index as a gene-level approach to prioritizing exome variants. Proc. Natl. Acad. Sci. USA 112, 13615–13620 (2015).
Itan, Y. et al. The mutation significance cutoff: gene-level thresholds for variant predictions. Nat. Methods 13, 109–110 (2016).
Itan, Y. et al. HGCS: an online tool for prioritizing disease-causing gene variants by biological distance. BMC Genomics 15, 256 (2014).
Yang, L. et al. Human cardiovascular progenitor cells develop from a KDR+ embryonic-stem-cell-derived population. Nature 453, 524–528 (2008).
Dubois, N.C. et al. SIRPA is a specific cell-surface marker for isolating cardiomyocytes derived from human pluripotent stem cells. Nat. Biotechnol. 29, 1011–1018 (2011).
Berthold, M. et al. KNIME: The Konstanz Information Miner. in Data Analysis, Machine Learning and Applications (eds. Preisach, C., Burkhardt, H., Schmidt-Thieme, L. & Decker, R.) 319–326 (Springer Berlin Heidelberg, 2008).
Ooi, Y.H., Oh, D.J. & Rhee, D.J. Analysis of α2-adrenergic receptors and effect of brimonidine on matrix metalloproteinases and their inhibitors in human ciliary body. Invest. Ophthalmol. Vis. Sci. 50, 4237–4243 (2009).
Trapnell, C., Pachter, L. & Salzberg, S.L. TopHat: discovering splice junctions with RNA-Seq. Bioinformatics 25, 1105–1111 (2009).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Engström, P.G. et al. Systematic evaluation of spliced alignment programs for RNA-seq data. Nat. Methods 10, 1185–1191 (2013).
Acknowledgements
We thank J.-F. Brunet (Ecole Normale Supérieure) for providing us with the PHOX2A antibody, S. Irion and M. Tomishima for critical review of the manuscript, A. Hudon for general help, H. Ralph (Weill Cornell Cell Screening Core) for image quantification and the Bioinformatics, Integrated Genomics Operations (funded by the NCI Cancer Center Support Grant (CCSG, P30 CA08748), Cycle for Survival and the Marie-Josée and Henry R. Kravis Center for Molecular Oncology) and Flow Cytometry Core Facilities at Sloan Kettering Institute. This work was supported by the Swiss National Science Foundation (N.Z.); the US National Institutes of Health (P30CA08748), New York State Stem Cell Science (NYSTEM) (C026446 and C026447) and the Tri-institutional Stem Cell Initiative (Starr Foundation) (L.S.); and a Robertson Investigator Award from New York Stem Cell Foundation, Maryland Stem Cell Research Funding (MSCRF/TEDCO) and the Adrienne Helis Malvin Medical Research Foundation (G.L.).
Author information
Authors and Affiliations
Contributions
N.Z.: design and conception of the study, writing of the manuscript, cell maintenance, reprogramming, GO term analysis, directed differentiation and survival assays, protocol optimization, gene targeting of PSCs and cellular and molecular assays. F.F.: AN differentiation protocol establishment and execution and survival assays in ANs. J.T.: RNA-sequencing data analysis. N.C.D.: design and execution of cardiac mesoderm differentiations. B.Z.: data quantification of scratch assay. N.S. and M.A.S.: western blotting. G.L.: reprogramming of S3 PSCs and mentoring. J.-L.C., L. Shang and F.L.: advice and analysis of whole-exome sequencing. L. Studer: design and conception of the study, data interpretation, writing of the manuscript and mentoring.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures and Tables
Supplementary Figures 1–17 and Supplementary Tables 1 and 3 (PDF 18148 kb)
Supplementary Table 2
List of candidate gene analysis from whole-exome sequencing (XLSX 191 kb)
Rights and permissions
About this article
Cite this article
Zeltner, N., Fattahi, F., Dubois, N. et al. Capturing the biology of disease severity in a PSC-based model of familial dysautonomia. Nat Med 22, 1421–1427 (2016). https://doi.org/10.1038/nm.4220
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.4220
This article is cited by
-
Norepinephrine transporter defects lead to sympathetic hyperactivity in Familial Dysautonomia models
Nature Communications (2022)
-
Towards bridging the translational gap by improved modeling of human nociception in health and disease
Pflügers Archiv - European Journal of Physiology (2022)
-
Human pluripotent stem cell-derived functional sympathetic neurons express ACE2 and RAAS components: a framework for studying the effect of COVID-19 on sympathetic responsiveness
Clinical Autonomic Research (2022)
-
Therapeutic manipulation of IKBKAP mis-splicing with a small molecule to cure familial dysautonomia
Nature Communications (2021)
-
The sympathetic nervous system in development and disease
Nature Reviews Neuroscience (2021)