Abstract
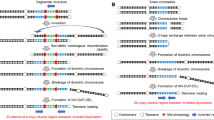
Chromosome instability is a hallmark of tumorigenesis. This study establishes that chromosome instability is also common during early human embryogenesis. A new array-based method allowed screening of genome-wide copy number and loss of heterozygosity in single cells. This revealed not only mosaicism for whole-chromosome aneuploidies and uniparental disomies in most cleavage-stage embryos but also frequent segmental deletions, duplications and amplifications that were reciprocal in sister blastomeres, implying the occurrence of breakage-fusion-bridge cycles. This explains the low human fecundity and identifies post-zygotic chromosome instability as a leading cause of constitutional chromosomal disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Geigl, J.B., Obenauf, A.C., Schwarzbraun, T. & Speicher, M.R. Defining 'chromosomal instability'. Trends Genet. 24, 64–69 (2008).
Lengauer, C., Kinzler, K.W. & Vogelstein, B. Genetic instability in colorectal cancers. Nature 386, 623–627 (1997).
Gao, C. et al. Chromosome instability, chromosome transcriptome, and clonal evolution of tumor cell populations. Proc. Natl. Acad. Sci. USA 104, 8995–9000 (2007).
Rajagopalan, H. & Lengauer, C. Aneuploidy and cancer. Nature 432, 338–341 (2004).
Delhanty, J.D. Mechanisms of aneuploidy induction in human oogenesis and early embryogenesis. Cytogenet. Genome Res. 111, 237–244 (2005).
Voullaire, L., Slater, H., Williamson, R. & Wilton, L. Chromosome analysis of blastomeres from human embryos by using comparative genomic hybridization. Hum. Genet. 106, 210–217 (2000).
Wells, D. & Delhanty, J.D. Comprehensive chromosomal analysis of human preimplantation embryos using whole genome amplification and single cell comparative genomic hybridization. Mol. Hum. Reprod. 6, 1055–1062 (2000).
Daphnis, D.D. et al. Analysis of the evolution of chromosome abnormalities in human embryos from Day 3 to 5 using CGH and FISH. Mol. Hum. Reprod. 14, 117–125 (2008).
Harper, J. et al. What next for preimplantation genetic screening? Hum. Reprod. 23, 478–480 (2008).
Le Caignec, C. et al. Single-cell chromosomal imbalances detection by array CGH. Nucleic Acids Res. 34, e68 (2006).
Fiegler, H. et al. High resolution array–CGH analysis of single cells. Nucleic Acids Res. 35, e15 (2007).
Iwamoto, K. et al. Detection of chromosomal structural alterations in single cells by SNP arrays: a systematic survey of amplification bias and optimized workflow. PLoS One 2, e1306 (2007).
Ballif, B.C., Yu, W., Shaw, C.A., Kashork, C.D. & Shaffer, L.G. Monosomy 1p36 breakpoint junctions suggest pre-meiotic breakage-fusion-bridge cycles are involved in generating terminal deletions. Hum. Mol. Genet. 12, 2153–2165 (2003).
Rossi, E. et al. Duplications in addition to terminal deletions are present in a proportion of ring chromosomes. Clues to the mechanisms of formation. J. Med. Genet. 45, 147–154 (2008).
Marshall, O.J., Chueh, A.C., Wong, L.H. & Choo, K.H. Neocentromeres: new insights into centromere structure, disease development, and karyotype evolution. Am. J. Hum. Genet. 82, 261–282 (2008).
Ballif, B.C. et al. Detecting sex chromosome anomalies and common triploidies in products of conception by array-based comparative genomic hybridization. Prenat. Diagn. 26, 333–339 (2006).
Kotzot, D. Abnormal phenotypes in uniparental disomy (UPD): fundamental aspects and a critical review with bibliography of UPD other than 15. Am. J. Med. Genet. 82, 265–274 (1999).
Kotzot, D. Complex and segmental uniparental disomy (UPD): review and lessons from rare chromosomal complements. J. Med. Genet. 38, 497–507 (2001).
Perry, J., Slater, H.R. & Choo, K.H. Centric fission—simple and complex mechanisms. Chromosome Res. 12, 627–640 (2004).
Pipiras, E., Coquelle, A., Bieth, A. & Debatisse, M. Interstitial deletions and intrachromosomal amplification initiated from a double-strand break targeted to a mammalian chromosome. EMBO J. 17, 325–333 (1998).
Artandi, S.E. et al. Telomere dysfunction promotes non-reciprocal translocations and epithelial cancers in mice. Nature 406, 641–645 (2000).
Bignell, G.R. et al. Architectures of somatic genomic rearrangement in human cancer amplicons at sequence-level resolution. Genome Res. 17, 1296–1303 (2007).
Munné, S. et al. Treatment-related chromosome abnormalities in human embryos. Hum. Reprod. 12, 780–784 (1997).
Weghofer, A. et al. The impact of LH-containing gonadotropins on diploidy rates in preimplantation embryos: long protocol stimulation. Hum. Reprod. 23, 499–503 (2008).
Harper, J.C. et al. ESHRE PGD consortium data collection VII: cycles from January to December 2004 with pregnancy follow-up to October 2005. Hum. Reprod. 23, 741–755 (2008).
Rubio, C. et al. Chromosomal abnormalities and embryo development in recurrent miscarriage couples. Hum. Reprod. 18, 182–188 (2003).
Munné, S. et al. Increased rate of aneuploid embryos in young women with previous aneuploid conceptions. Prenat. Diagn. 24, 638–643 (2004).
Baart, E.B. et al. Preimplantation genetic screening reveals a high incidence of aneuploidy and mosaicism in embryos from young women undergoing IVF. Hum. Reprod. 21, 223–233 (2006).
Braude, P., Bolton, V. & Moore, S. Human gene expression first occurs between the four- and eight-cell stages of preimplantation development. Nature 332, 459–461 (1988).
Macklon, N.S., Geraedts, J.P. & Fauser, B.C. Conception to ongoing pregnancy: the 'black box' of early pregnancy loss. Hum. Reprod. Update 8, 333–343 (2002).
Pflueger, S. Cytogenetics of spontaneous abortion. in The Principles of Clinical Cytogenetics (eds. Gersen, S. & Keagle, M.) 317–343 (Humana Press, New Jersey, 1999).
Fritz, B. et al. Cytogenetic analyses of culture failures by comparative genomic hybridisation (CGH)—re-evaluation of chromosome aberration rates in early spontaneous abortions. Eur. J. Hum. Genet. 9, 539–547 (2001).
Benkhalifa, M. et al. Array comparative genomic hybridization profiling of first-trimester spontaneous abortions that fail to grow in vitro. Prenat. Diagn. 25, 894–900 (2005).
Schinzel, A. Catalogue of Unbalanced Chromosome Aberrations in Man (Walter de Gruyter, New York, 2001).
Staessen, C. et al. Comparison of blastocyst transfer with or without preimplantation genetic diagnosis for aneuploidy screening in couples with advanced maternal age: a prospective randomized controlled trial. Hum. Reprod. 19, 2849–2858 (2004).
Li, M. et al. Fluorescence in situ hybridization reanalysis of day-6 human blastocysts diagnosed with aneuploidy on day 3. Fertil. Steril. 84, 1395–1400 (2005).
Munné, S. et al. Self-correction of chromosomally abnormal embryos in culture and implications for stem cell production. Fertil. Steril. 84, 1328–1334 (2005).
Yurov, Y.B. et al. Aneuploidy and confined chromosomal mosaicism in the developing human brain. PLoS One 2, e558 (2007).
Acknowledgements
We thank all families who participated in the study, the Leuven University Fertility Center for technical assistance and S. Jackmaert for her help with the SNP arrays. We are also grateful to the Mapping Core and Map finishing groups of the Wellcome Trust Sanger Institute for initial BAC clone supply and verification and to the microarray facility of the Flanders Interuniversity Institute for Biotechnology for their help in spotting the arrays. We would like to thank C. Spiessens, E. Legius, T. de Ravel de l'Argentière, H. van Esch and K. Devriendt for the critical reading of the manuscript. This work was made possible by grants from the Institute for the Promotion of Innovation through Science and Technology (IWT-Flanders) (SBO-60848) and GOA/2006/12 and Center of Excellence SymBioSys (Research Council K.U.Leuven EF/05/007) to J.R.V. and Fonds de la Recherche Scientifique to M.V. E.V. was supported by the Institute for the Promotion of Innovation through Science and Technology in Flanders.
Author information
Authors and Affiliations
Contributions
E.V., T.V. and J.R.V. designed the experiments and wrote the manuscript. E.V., T.V., C.L.C. and C.M. conducted the experiments. M. Ampe, G.V., P.K. and Y.M. developed the statistical algorithms and software for data analysis. M. Amyere, M.V. and F.S. provided expertise on SNP typing. J.-P.F. performed the genetic counseling of the couples opting for PGD. T.D. and S.D. performed IVF and provided the embryos. J.R.V. supervised.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1–3, Supplementary Tables 1 and 2 and Supplementary Methods (PDF 12875 kb)
Rights and permissions
About this article
Cite this article
Vanneste, E., Voet, T., Le Caignec, C. et al. Chromosome instability is common in human cleavage-stage embryos. Nat Med 15, 577–583 (2009). https://doi.org/10.1038/nm.1924
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.1924
This article is cited by
-
Experience of copy number variation sequencing applied in spontaneous abortion
BMC Medical Genomics (2024)
-
Association of early cleavage, morula compaction and blastocysts ploidy of IVF embryos cultured in a time-lapse system and biopsied for genetic test for aneuploidy
Scientific Reports (2024)
-
Genome-wide equine preimplantation genetic testing enabled by simultaneous haplotyping and copy number detection
Scientific Reports (2024)
-
Single-cell lineage tracing with endogenous markers
Biophysical Reviews (2024)
-
Nanopore sequencing for detecting reciprocal translocation carrier status in preimplantation genetic testing
BMC Genomics (2023)