Abstract
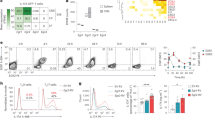
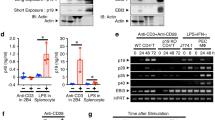
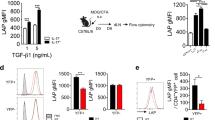
Studies have shown that transforming growth factor-β (TGF-β) and interleukin 6 (IL-6) are required for the lineage commitment of pathogenic IL-17-producing T helper cells (TH-17 cells). Unexpectedly, here we found that stimulation of myelin-reactive T cells with TGF-β plus IL-6 completely abrogated their pathogenic function despite upregulation of IL-17 production. Cells stimulated with TGF-β plus IL-6 were present in the spleen as well as the central nervous system, but they failed to upregulate the proinflammatory chemokines crucial for central nervous system inflammation. In addition, these cells produced IL-10, which has potent anti-inflammatory activities. In contrast, stimulation with IL-23 promoted expression of IL-17 and proinflammatory chemokines but not IL-10. Hence, TGF-β and IL-6 'drive' initial lineage commitment but also 'restrain' the pathogenic potential of TH-17 cells. Our findings suggest that full acquisition of pathogenic function by effector TH-17 cells is mediated by IL-23 rather than by TGF-β and IL-6.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Langrish, C.L. et al. IL-23 drives a pathogenic T cell population that induces autoimmune inflammation. J. Exp. Med. 201, 233–240 (2005).
Bettelli, E. et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 441, 235–238 (2006).
Murphy, C.A. et al. Divergent pro- and antiinflammatory roles for IL-23 and IL-12 in joint autoimmune inflammation. J. Exp. Med. 198, 1951–1957 (2003).
Ivanov, I.I. et al. The orphan nuclear receptor RORγt directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell 126, 1121–1133 (2006).
Liang, S.C. et al. Interleukin (IL)-22 and IL-17 are coexpressed by Th17 cells and cooperatively enhance expression of antimicrobial peptides. J. Exp. Med. 203, 2271–2279 (2006).
Aggarwal, S., Ghilardi, N., Xie, M.H., de Sauvage, F.J. & Gurney, A.L. Interleukin-23 promotes a distinct CD4 T cell activation state characterized by the production of interleukin-17. J. Biol. Chem. 278, 1910–1914 (2003).
Veldhoen, M., Hocking, R.J., Atkins, C.J., Locksley, R.M. & Stockinger, B. TGFβ in the context of an inflammatory cytokine milieu supports de novo differentiation of IL-17-producing T cells. Immunity 24, 179–189 (2006).
Mangan, P.R. et al. Transforming growth factor-β induces development of the TH17 lineage. Nature 441, 231–234 (2006).
Park, H. et al. A distinct lineage of CD4 T cells regulates tissue inflammation by producing interleukin 17. Nat. Immunol. 6, 1133–1141 (2005).
Batten, M. et al. Interleukin 27 limits autoimmune encephalomyelitis by suppressing the development of interleukin 17-producing T cells. Nat. Immunol. 7, 929–936 (2006).
Stumhofer, J.S. et al. Interleukin 27 negatively regulates the development of interleukin 17-producing T helper cells during chronic inflammation of the central nervous system. Nat. Immunol. 7, 937–945 (2006).
Kleinschek, M.A. et al. IL-25 regulates Th17 function in autoimmune inflammation. J. Exp. Med. 204, 161–170 (2007).
Laurence, A. et al. Interleukin-2 signaling via STAT5 constrains T helper 17 cell generation. Immunity 26, 371–381 (2007).
Mucida, D. et al. Reciprocal TH17 and regulatory T cell differentiation mediated by retinoic acid. Science 317, 256–260 (2007).
Veldhoen, M., Hocking, R.J., Flavell, R.A. & Stockinger, B. Signals mediated by transforming growth factor-β initiate autoimmune encephalomyelitis, but chronic inflammation is needed to sustain disease. Nat. Immunol. 7, 1151–1156 (2006).
Chen, Y., Hancock, W.W., Marks, R., Gonnella, P. & Weiner, H.L. Mechanisms of recovery from experimental autoimmune encephalomyelitis: T cell deletion and immune deviation in myelin basic protein T cell receptor transgenic mice. J. Neuroimmunol. 82, 149–159 (1998).
Kent, S.C. et al. Oral administration of myelin induces antigen-specific TGF-β1-secreting T cells in multiple sclerosis patients. Ann. NY Acad. Sci. 815, 412–422 (1997).
Chen, Y., Kuchroo, V.K., Inobe, J., Hafler, D.A. & Weiner, H.L. Regulatory T cell clones induced by oral tolerance: suppression of autoimmune encephalomyelitis. Science 265, 1237–1240 (1994).
Racke, M.K. et al. Prevention and treatment of chronic relapsing experimental allergic encephalomyelitis by transforming growth factor-β1. J. Immunol. 146, 3012–3017 (1991).
Kuruvilla, A.P. et al. Protective effect of transforming growth factor β1 on experimental autoimmune diseases in mice. Proc. Natl. Acad. Sci. USA 88, 2918–2921 (1991).
Zhou, L. et al. IL-6 programs TH-17 cell differentiation by promoting sequential engagement of the IL-21 and IL-23 pathways. Nat. Immunol. 8, 967–974 (2007).
Korn, T. et al. IL-21 initiates an alternative pathway to induce proinflammatory TH17 cells. Nature 448, 484–487 (2007).
Nurieva, R. et al. Essential autocrine regulation by IL-21 in the generation of inflammatory T cells. Nature 448, 480–483 (2007).
Zheng, Y. et al. Interleukin-22, a TH17 cytokine, mediates IL-23-induced dermal inflammation and acanthosis. Nature 445, 648–651 (2007).
Sutton, C., Brereton, C., Keogh, B., Mills, K.H. & Lavelle, E.C. A crucial role for interleukin (IL)-1 in the induction of IL-17-producing T cells that mediate autoimmune encephalomyelitis. J. Exp. Med. 203, 1685–1691 (2006).
Cua, D.J. et al. Interleukin-23 rather than interleukin-12 is the critical cytokine for autoimmune inflammation of the brain. Nature 421, 744–748 (2003).
Godiska, R., Chantry, D., Dietsch, G.N. & Gray, P.W. Chemokine expression in murine experimental allergic encephalomyelitis. J. Neuroimmunol. 58, 167–176 (1995).
Huang, D.R., Wang, J., Kivisakk, P., Rollins, B.J. & Ransohoff, R.M. Absence of monocyte chemoattractant protein 1 in mice leads to decreased local macrophage recruitment and antigen-specific T helper cell type 1 immune response in experimental autoimmune encephalomyelitis. J. Exp. Med. 193, 713–726 (2001).
Karpus, W.J. & Ransohoff, R.M. Chemokine regulation of experimental autoimmune encephalomyelitis: temporal and spatial expression patterns govern disease pathogenesis. J. Immunol. 161, 2667–2671 (1998).
Columba-Cabezas, S. et al. Induction of macrophage-derived chemokine/CCL22 expression in experimental autoimmune encephalomyelitis and cultured microglia: implications for disease regulation. J. Neuroimmunol. 130, 10–21 (2002).
Anderson, C.F., Oukka, M., Kuchroo, V.J. & Sacks, D. CD4+CD25−Foxp3− Th1 cells are the source of IL-10-mediated immune suppression in chronic cutaneous leishmaniasis. J. Exp. Med. 204, 285–297 (2007).
Jankovic, D. et al. Conventional T-bet+Foxp3− Th1 cells are the major source of host-protective regulatory IL-10 during intracellular protozoan infection. J. Exp. Med. 204, 273–283 (2007).
Bettelli, E. et al. IL-10 is critical in the regulation of autoimmune encephalomyelitis as demonstrated by studies of IL-10- and IL-4-deficient and transgenic mice. J. Immunol. 161, 3299–3306 (1998).
Cua, D.J., Groux, H., Hinton, D.R., Stohlman, S.A. & Coffman, R.L. Transgenic interleukin 10 prevents induction of experimental autoimmune encephalomyelitis. J. Exp. Med. 189, 1005–1010 (1999).
Cua, D.J. & Kastelein, R.A. TGF-β, a 'double agent' in the immune pathology war. Nat. Immunol. 7, 557–559 (2006).
Kullberg, M.C. et al. IL-23 plays a key role in Helicobacter hepaticus-induced T cell-dependent colitis. J. Exp. Med. 203, 2485–2494 (2006).
Hue, S. et al. Interleukin-23 drives innate and T cell-mediated intestinal inflammation. J. Exp. Med. 203, 2473–2483 (2006).
Uhlig, H.H. et al. Differential activity of IL-12 and IL-23 in mucosal and systemic innate immune pathology. Immunity 25, 309–318 (2006).
Willenborg, D.O., Fordham, S., Bernard, C.C., Cowden, W.B. & Ramshaw, I.A. IFN-γ plays a critical down-regulatory role in the induction and effector phase of myelin oligodendrocyte glycoprotein-induced autoimmune encephalomyelitis. J. Immunol. 157, 3223–3227 (1996).
Bailey, S.L., Schreiner, B., McMahon, E.J. & Miller, S.D. CNS myeloid DCs presenting endogenous myelin peptides 'preferentially' polarize CD4+ TH-17 cells in relapsing EAE. Nat. Immunol. 8, 172–180 (2007).
Vanderlugt, C.J. & Miller, S.D. Epitope spreading. Curr. Opin. Immunol. 8, 831–836 (1996).
Leibundgut-Landmann, S. et al. Syk- and CARD9-dependent coupling of innate immunity to the induction of T helper cells that produce interleukin 17. Nat Immunol (2007).
Marie, J.C., Letterio, J.J., Gavin, M. & Rudensky, A.Y. TGF-β1 maintains suppressor function and Foxp3 expression in CD4+CD25+ regulatory T cells. J. Exp. Med. 201, 1061–1067 (2005).
Fantini, M.C. et al. Cutting edge: TGF-β induces a regulatory phenotype in CD4+CD25– T cells through Foxp3 induction and down-regulation of Smad7. J. Immunol. 172, 5149–5153 (2004).
Chen, W. et al. Conversion of peripheral CD4+CD25− naive T cells to CD4+CD25+ regulatory T cells by TGF-β induction of transcription factor Foxp3. J. Exp. Med. 198, 1875–1886 (2003).
Li, M.O., Wan, Y.Y. & Flavell, R.A. T cell-produced transforming growth factor-β1 controls T cell tolerance and regulates Th1- and Th17-cell differentiation. Immunity 26, 579–591 (2007).
McGeachy, M.J., Stephens, L.A. & Anderton, S.M. Natural recovery and protection from autoimmune encephalomyelitis: contribution of CD4+CD25+ regulatory cells within the central nervous system. J. Immunol. 175, 3025–3032 (2005).
Baecher-Allan, C. & Hafler, D.A. Suppressor T cells in human diseases. J. Exp. Med. 200, 273–276 (2004).
Viglietta, V., Baecher-Allan, C., Weiner, H.L. & Hafler, D.A. Loss of functional suppression by CD4+CD25+ regulatory T cells in patients with multiple sclerosis. J. Exp. Med. 199, 971–979 (2004).
Kukreja, A. et al. Multiple immuno-regulatory defects in type-1 diabetes. J. Clin. Invest. 109, 131–140 (2002).
Sakaguchi, S. Naturally arising CD4+ regulatory t cells for immunologic self-tolerance and negative control of immune responses. Annu. Rev. Immunol. 22, 531–562 (2004).
Acknowledgements
We thank B. Desai and S. Jungers for assistance with flow cytometry; J. Mattson for real-time PCR data; B. Joyce-Shaikh for technical assistance; and R. Kastelein for comments.
Author information
Authors and Affiliations
Contributions
M.J.M. designed and did all experiments and prepared the manuscript; K.S.B.-J. assisted with in vitro experiments; Y.C. and C.M.T. assisted with in vivo experiments; W.B. and T.M. did quantitative PCR analysis; and D.J.C. did cerebroventricular injections, supervised the studies and preparation of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors are employed by or were previously employed by Schering-Plough Biopharma.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 (PDF 412 kb)
Rights and permissions
About this article
Cite this article
McGeachy, M., Bak-Jensen, K., Chen, Y. et al. TGF-β and IL-6 drive the production of IL-17 and IL-10 by T cells and restrain TH-17 cell–mediated pathology. Nat Immunol 8, 1390–1397 (2007). https://doi.org/10.1038/ni1539
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1539
This article is cited by
-
Neuromorphic electro-stimulation based on atomically thin semiconductor for damage-free inflammation inhibition
Nature Communications (2024)
-
Pertussis toxin-induced inhibition of Wnt/β-catenin signaling in dendritic cells promotes an autoimmune response in experimental autoimmune uveitis
Journal of Neuroinflammation (2023)
-
Cerebral microvascular endothelial cell-derived extracellular vesicles regulate blood − brain barrier function
Fluids and Barriers of the CNS (2023)
-
Increased synovial immunohistochemistry reactivity of TGF-β1 in erosive peripheral psoriatic arthritis
BMC Musculoskeletal Disorders (2023)
-
Central role for neurally dysregulated IL-17A in dynamic networks of systemic and local inflammation in combat casualties
Scientific Reports (2023)