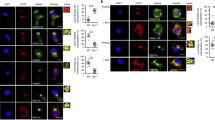
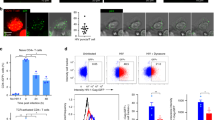
Abstract
Antiviral or antitumor immunity requires activation of cytotoxic CD8+ T cells by dendritic cells, which present viral or tumor antigens on major histocompatibility complex (MHC) class I molecules. The intracellular mechanisms facilitating MHC class I–restricted presentation of extracellular antigens ('cross-presentation') are unclear. Here we demonstrate that cross-presentation of soluble antigen occurred in an early endosomal compartment distinct from the endoplasmic reticulum where endogenous antigen is loaded onto MHC class I. Efficient cross-presentation required endotoxin-induced, Toll-like receptor 4– and signaling molecule MyD88–dependent relocation of the transporter associated with antigen processing, essential for loading of MHC class I, to early endosomes. Transport of cross-presented antigen from endosomes to the cell surface was inhibited by primaquine, which blocks endosomal trafficking. Thus, cross-presentation is spatially and mechanistically separated from endogenous MHC class I–restricted antigen presentation and is biased toward antigens containing microbial molecular patterns.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
den Haan, J.M., Lehar, S.M. & Bevan, M.J. CD8+ but not CD8− dendritic cells cross-prime cytotoxic T cells in vivo. J. Exp. Med. 192, 1685–1696 (2000).
Kurts, C., Cannarile, M., Klebba, I. & Brocker, T. Dendritic cells are sufficient to cross-present self-antigens to CD8 T cells in vivo. J. Immunol. 166, 1439–1442 (2001).
Bevan, M.J. Cross-priming. Nat. Immunol. 7, 363–365 (2006).
Villadangos, J.A., Heath, W.R. & Carbone, F.R. Outside looking in: the inner workings of the cross-presentation pathway within dendritic cells. Trends Immunol. 28, 45–47 (2007).
Kurts, C. et al. Constitutive class I-restricted exogenous presentation of self antigens in vivo. J. Exp. Med. 184, 923–930 (1996).
Kurts, C., Miller, J.F., Subramaniam, R.M., Carbone, F.R. & Heath, W.R. Major histocompatibility complex class I-restricted cross-presentation is biased towards high dose antigens and those released during cellular destruction. J. Exp. Med. 188, 409–414 (1998).
Li, M. et al. Cell-associated ovalbumin is cross-presented much more efficiently than soluble ovalbumin in vivo. J. Immunol. 166, 6099–6103 (2001).
Beutler, B., Hoebe, K., Du, X. & Ulevitch, R.J. How we detect microbes and respond to them: the Toll-like receptors and their transducers. J. Leukoc. Biol. 74, 479–485 (2003).
Kopp, E. & Medzhitov, R. Recognition of microbial infection by Toll-like receptors. Curr. Opin. Immunol. 15, 396–401 (2003).
Hemmi, H. & Akira, S. TLR signalling and the function of dendritic cells. Chem. Immunol. Allergy 86, 120–135 (2005).
Maurer, T. et al. CpG-DNA aided cross-presentation of soluble antigens by dendritic cells. Eur. J. Immunol. 32, 2356–2364 (2002).
Datta, S.K. et al. A subset of Toll-like receptor ligands induces cross-presentation by bone marrow-derived dendritic cells. J. Immunol. 170, 4102–4110 (2003).
Hoebe, K. et al. Upregulation of costimulatory molecules induced by lipopolysaccharide and double-stranded RNA occurs by Trif-dependent and Trif-independent pathways. Nat. Immunol. 4, 1223–1229 (2003).
Gil-Torregrosa, B.C. et al. Control of cross-presentation during dendritic cell maturation. Eur. J. Immunol. 34, 398–407 (2004).
Schulz, O. et al. Toll-like receptor 3 promotes cross-priming to virus-infected cells. Nature 433, 887–892 (2005).
Weck, M.M. et al. TLR ligands differentially affect uptake and presentation of cellular antigens. Blood 109, 3890–3894 (2007).
Abele, R. & Tampé, R. The ABCs of immunology: structure and function of TAP, the transporter associated with antigen processing. Physiology (Bethesda) 19, 216–224 (2004).
Ackerman, A.L. & Cresswell, P. Cellular mechanisms governing cross-presentation of exogenous antigens. Nat. Immunol. 5, 678–684 (2004).
Brossart, P. & Bevan, M.J. Presentation of exogenous protein antigens on major histocompatibility complex class I molecules by dendritic cells: pathway of presentation and regulation by cytokines. Blood 90, 1594–1599 (1997).
Norbury, C.C., Chambers, B.J., Prescott, A.R., Ljunggren, H.G. & Watts, C. Constitutive macropinocytosis allows TAP-dependent major histocompatibility complex class I presentation of exogenous soluble antigen by bone marrow-derived dendritic cells. Eur. J. Immunol. 27, 280–288 (1997).
Ackerman, A.L., Kyritsis, C., Tampé, R. & Cresswell, P. Early phagosomes in dendritic cells form a cellular compartment sufficient for cross presentation of exogenous antigens. Proc. Natl. Acad. Sci. USA 100, 12889–12894 (2003).
Norbury, C.C. et al. CD8+ T cell cross-priming via transfer of proteasome substrates. Science 304, 1318–1321 (2004).
Ackerman, A.L., Kyritsis, C., Tampé, R. & Cresswell, P. Access of soluble antigens to the endoplasmic reticulum can explain cross-presentation by dendritic cells. Nat. Immunol. 6, 107–113 (2005).
Imai, J., Hasegawa, H., Maruya, M., Koyasu, S. & Yahara, I. Exogenous antigens are processed through the endoplasmic reticulum-associated degradation (ERAD) in cross-presentation by dendritic cells. Int. Immunol. 17, 45–53 (2005).
Ackerman, A.L., Giodini, A. & Cresswell, P. A role for the endoplasmic reticulum protein retrotranslocation machinery during crosspresentation by dendritic cells. Immunity 25, 607–617 (2006).
Burgdorf, S., Kautz, A., Bohnert, V., Knolle, P.A. & Kurts, C. Distinct pathways of antigen uptake and intracellular routing in CD4 and CD8 T cell activation. Science 316, 612–616 (2007).
Song, R. & Harding, C.V. Roles of proteasomes, transporter for antigen presentation (TAP), and beta 2-microglobulin in the processing of bacterial or particulate antigens via an alternate class I MHC processing pathway. J. Immunol. 156, 4182–4190 (1996).
Houde, M. et al. Phagosomes are competent organelles for antigen cross-presentation. Nature 425, 402–406 (2003).
Guermonprez, P. et al. ER-phagosome fusion defines an MHC class I cross-presentation compartment in dendritic cells. Nature 425, 397–402 (2003).
Touret, N. et al. Quantitative and dynamic assessment of the contribution of the ER to phagosome formation. Cell 123, 157–170 (2005).
Burgdorf, S., Lukacs-Kornek, V. & Kurts, C. The mannose receptor mediates uptake of soluble but not of cell-associated antigen for cross-presentation. J. Immunol. 176, 6770–6776 (2006).
Riese, R.J. & Chapman, H.A. Cathepsins and compartmentalization in antigen presentation. Curr. Opin. Immunol. 12, 107–113 (2000).
Rodriguez, A., Regnault, A., Kleijmeer, M., Ricciardi-Castagnoli, P. & Amigorena, S. Selective transport of internalized antigens to the cytosol for MHC class I presentation in dendritic cells. Nat. Cell Biol. 1, 362–368 (1999).
Hotta, C., Fujimaki, H., Yoshinari, M., Nakazawa, M. & Minami, M. The delivery of an antigen from the endocytic compartment into the cytosol for cross-presentation is restricted to early immature dendritic cells. Immunology 117, 97–107 (2006).
Song, R., Porgador, A. & Harding, C.V. Peptide-receptive class I major histocompatibility complex molecules on TAP-deficient and wild-type cells and their roles in the processing of exogenous antigens. Immunology 97, 316–324 (1999).
Kyritsis, C. et al. Molecular mechanism and structural aspects of transporter associated with antigen processing inhibition by the cytomegalovirus protein US6. J. Biol. Chem. 276, 48031–48039 (2001).
Ciechanover, A., Schwartz, A.L. & Lodish, H.F. Sorting and recycling of cell surface receptors and endocytosed ligands: the asialoglycoprotein and transferrin receptors. J. Cell. Biochem. 23, 107–130 (1983).
Reid, P.A. & Watts, C. Cycling of cell-surface MHC glycoproteins through primaquine-sensitive intracellular compartments. Nature 346, 655–657 (1990).
van Weert, A.W., Geuze, H.J., Groothuis, B. & Stoorvogel, W. Primaquine interferes with membrane recycling from endosomes to the plasma membrane through a direct interaction with endosomes which does not involve neutralisation of endosomal pH nor osmotic swelling of endosomes. Eur. J. Cell Biol. 79, 394–399 (2000).
Porgador, A., Yewdell, J.W., Deng, Y., Bennink, J.R. & Germain, R.N. Localization, quantitation, and in situ detection of specific peptide-MHC class I complexes using a monoclonal antibody. Immunity 6, 715–726 (1997).
Bancherau, J. & Steinman, R.M. Dendritic cells and the control of immunity. Nature 392, 245–252 (1998).
Cella, M., Engering, A., Pinet, V., Pieters, J. & Lanzavecchia, A. Inflammatory stimuli induce accumulation of MHC class II complexes on dendritic cells. Nature 388, 782–787 (1997).
Turley, S.J. et al. Transport of peptide-MHC class II complexes in developing dendritic cells. Science 288, 522–527 (2000).
Trombetta, E.S., Ebersold, M., Garrett, W., Pypaert, M. & Mellman, I. Activation of lysosomal function during dendritic cell maturation. Science 299, 1400–1403 (2003).
Blander, J.M. & Medzhitov, R. Toll-dependent selection of microbial antigens for presentation by dendritic cells. Nature 440, 808–812 (2006).
Watanabe, J., Miyazaki, Y., Zimmerman, G.A., Albertine, K.H. & McIntyre, T.M. Endotoxin contamination of ovalbumin suppresses murine immunologic responses and development of airway hyper-reactivity. J. Biol. Chem. 278, 42361–42368 (2003).
Gagnon, E. et al. Endoplasmic reticulum-mediated phagocytosis is a mechanism of entry into macrophages. Cell 110, 119–131 (2002).
Blander, J.M. & Medzhitov, R. Regulation of phagosome maturation by signals from toll-like receptors. Science 304, 1014–1018 (2004).
Van Meirvenne, S. et al. Efficient genetic modification of murine dendritic cells by electroporation with mRNA. Cancer Gene Ther. 9, 787–797 (2002).
Burgdorf, S., Leister, P. & Scheidtmann, K.H. TSG101 interacts with apoptosis-antagonizing transcription factor and enhances androgen receptor-mediated transcription by promoting its monoubiquitination. J. Biol. Chem. 279, 17524–17534 (2004).
Acknowledgements
We thank M.C. Nussenzweig (Rockefeller University) for mannose receptor-deficient mice; W.R. Heath (The Walter and Eliza Hall Institute of Medical Research) for OT-I and OT-II mice; R. Germain (National Institutes of Health) for 25-D1.16; A. Haas for the transferrin receptor antibody; and P. Knolle for critically reading the manuscript. We acknowledge the technical support of the flow cytometry core facility of the Institute of Molecular Medicine and Experimental Immunology and the House for Experimental Therapy of the Medical Faculty of Bonn University. Supported by the BONFOR program of Friedrich-Wilhelms University (O-173.0009 to S.B.) and by the Deutsche Forschungsgemeinschaft (SFB704 to C.K. and Ta157/7-1 to R.T.).
Author information
Authors and Affiliations
Contributions
S.B. designed experiments, did research, analyzed and interpreted data, and wrote the paper; C.S. and A.K. did experiments; R.T. contributed critical analytical tools and analyzed and interpreted data; C.K. designed experiments, analyzed and interpreted data, and wrote the paper.
Corresponding authors
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–12 (PDF 2528 kb)
Rights and permissions
About this article
Cite this article
Burgdorf, S., Schölz, C., Kautz, A. et al. Spatial and mechanistic separation of cross-presentation and endogenous antigen presentation. Nat Immunol 9, 558–566 (2008). https://doi.org/10.1038/ni.1601
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1601
This article is cited by
-
Lymph node-targeted neoantigen nanovaccines potentiate anti-tumor immune responses of post-surgical melanoma
Journal of Nanobiotechnology (2022)
-
Lipid A analog CRX-527 conjugated to synthetic peptides enhances vaccination efficacy and tumor control
npj Vaccines (2022)
-
Single-molecule imaging of glycan–lectin interactions on cells with Glyco-PAINT
Nature Chemical Biology (2021)
-
Über Salz und Immunität – Eine Geschichte von Mr. Hyde
Der Pathologe (2019)
-
A novel probe to assess cytosolic entry of exogenous proteins
Nature Communications (2018)