Abstract
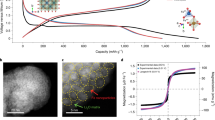
In lithium-ion batteries, the critical need for high-energy-density, low-cost storage for applications ranging from wearable computing to megawatt-scale stationary storage has created an unmet need for facile methods to produce high-density, low-tortuosity, kinetically accessible storage electrodes. Here we show that magnetic control of sacrificial features enables the creation of directional pore arrays in lithium-ion electrodes. The directional pores result in faster charge transport kinetics and enable electrodes with more than threefold higher area capacity (for example, >12 mAh cm−2 versus <4 mAh cm−2 in conventional electrodes) at practical charge–discharge rates. We demonstrate these capabilities in laboratory cells under various test conditions, including an electric vehicle model drive cycle.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Gaudillere, C., Garcia-Fayos, J. & Serra, J. M. Enhancing oxygen permeation through hierarchically-structured perovskite membranes elaborated by freeze-casting. J. Mater. Chem. A 2, 3828–3833 (2014).
Deville, S., Saiz, E. & Tomsia, A. P. Freeze casting of hydroxyapatite scaffolds for bone tissue engineering. Biomaterials 27, 5480–5489 (2006).
Wicklein, B. et al. Thermally insulating and fire-retardant lightweight anisotropic foams based on nanocellulose and graphene oxide. Nature Nanotech. 10, 277–283 (2015).
Dong, X., Jin, W., Xu, N. & Li, K. Dense ceramic catalytic membranes and membrane reactors for energy and environmental applications. Chem. Commun. 47, 10886–10902 (2011).
Lai, W. et al. Ultrahigh-energy-density microbatteries enabled by new electrode architecture and micropackaging design. Adv. Mater. 22, E139–E144 (2010).
Nemani, V. P., Harris, S. J. & Smith, K. C. Design of bi-tortuous, anisotropic graphite anodes for fast ion-transport in Li-ion batteries. J. Electrochem. Soc. 162, A1415–A1423 (2015).
Gallagher, K. G. & Nelson, P. A. in Lithium-Ion Batteries (ed. Pistoia, G. ) 97–126 (Elsevier, 2014).
Patry, G., Romagny, A., Martinet, S. & Froelich, D. Cost modeling of lithium-ion battery cells for automotive applications. Energy Sci. Eng. 3, 71–82 (2015).
Doyle, M. & Newman, J. Analysis of capacity-rate data for lithium batteries using simplified models of the discharge process. J. Appl. Electrochem. 27, 846–856 (1997).
Johns, P. A., Roberts, M. R., Wakizaka, Y., Sanders, J. H. & Owen, J. R. How the electrolyte limits fast discharge in nanostructured batteries and supercapacitors. Electrochem. Commun. 11, 2089–2092 (2009).
Ebner, M., Chung, D.-W., Garcia, R. E. & Wood, V. Tortuosity anisotropy in lithium-ion battery electrodes. Adv. Energy Mater. 4, 1301278 (2014).
Kehrwald, D., Shearing, P. R., Brandon, N. P., Sinha, P. K. & Harris, S. J. Local tortuosity inhomogeneities in a lithium battery composite electrode. J. Electrochem. Soc. 158, A1393–A1399 (2011).
Chung, D.-W., Ebner, M., Ely, D. R., Wood, V. & Garcia, R. E. Validity of the Bruggeman relation for porous electrodes. Modelling Simul. Mater. Sci. Eng. 21, 074009 (2013).
Ebner, M. & Wood, V. Tool for tortuosity estimation in lithium ion battery porous electrodes. J. Electrochem. Soc. 162, A3064–A3070 (2015).
Thorat, I. V. et al. Quantifying tortuosity in porous Li-ion battery materials. J. Power Sources 188, 592–600 (2009).
Zacharias, N. A. et al. Direct measurements of effective ionic transport in porous Li-ion electrodes. J. Electrochem. Soc. 160, A306–A311 (2013).
Bae, C.-J., Erdonmez, C. K., Halloran, J. W. & Chiang, Y.-M. Design of battery electrodes with dual-scale porosity to minimize tortuosity and maximize performance. Adv. Mater. 25, 1254–1258 (2012).
Behr, S., Amin, R., Chiang, Y. M. & Tomsia, A. P. Highly structured, additive free lithium-ion cathodes by freeze-casting technology. Ceram. Forum Int. 92, E39–E43 (2015).
Erb, R. M., Libanori, R., Rothfuchs, N. & Studart, A. R. Composites reinforced in three dimensions by using low magnetic fields. Science 335, 199–204 (2012).
Dreyfus, R., Lacoste, D., Bibette, J. & Baudry, J. Measuring colloidal forces with the magnetic chaining technique. Eur. Phys. J. E 28, 113–123 (2009).
Sander, J. S., Erb, R. M., Denier, C. & Studart, A. R. Magnetic transport, mixing and release of cargo with tailored nanoliter droplets. Adv. Mater. 24, 2582–2587 (2012).
Sommer, M. R., Erb, R. M. & Studart, A. R. Injectable materials with magnetically controlled anisotropic porosity. ACS Appl. Mater. Interfaces 4, 5086–5091 (2012).
Erb, R. M., Segmehl, J., Schaffner, M. & Studart, A. R. Temporal response of magnetically labeled platelets under dynamic magnetic fields. Soft Matter 9, 498–505 (2013).
Chiang, Y. M. & Hellweg, B. Reticulated and controlled porosity battery structures. US Patent 7,781,098 (2010) and 8,277,975 B2 (2012).
Acknowledgements
This work was supported by the Assistant Secretary for Energy Efficiency and Renewable Energy, Office of Vehicle Technologies of the US Department of Energy under Contract no. DE-AC02-05CH11231, Subcontract no. 7056592 under the Batteries for Advanced Transportation Technologies (BATT) Program. J.S.S. thanks the Swiss National Science Foundation for financial Support (Grant Numbers P300P2_154584 and P2EZP2_148768).
Author information
Authors and Affiliations
Contributions
J.S.S., R.M.E. and Y.-M.C. conceived the research, designed the experiments, and wrote the manuscript. J.S.S., L.L. and A.G. carried out the experiments and analysed the data.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1–5 (PDF 1003 kb)
Supplementary Video 1
Out-of-plane alignment of magnetic emulsion droplets in water. The movie shows chaining and alignment of magnetic emulsion droplets in a dc magnetic field. (MP4 2905 kb)
Supplementary Video 2
Out-of-plane alignment of magnetic emulsion droplets in water. The movie shows chaining and alignment of magnetic emulsion droplets in a dc magnetic field. (MP4 2520 kb)
Rights and permissions
About this article
Cite this article
Sander, J., Erb, R., Li, L. et al. High-performance battery electrodes via magnetic templating. Nat Energy 1, 16099 (2016). https://doi.org/10.1038/nenergy.2016.99
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nenergy.2016.99
This article is cited by
-
Regulating electrochemical performances of lithium battery by external physical field
Rare Metals (2024)
-
Iron oxide/CNT-based artificial nacre for electromagnetic interference shielding
Nano Research (2024)
-
Redox-homogeneous, gel electrolyte-embedded high-mass-loading cathodes for high-energy lithium metal batteries
Nature Communications (2022)
-
Ultra-high-energy lithium-ion batteries enabled by aligned structured thick electrode design
Rare Metals (2022)
-
Novel 3D grid porous Li4Ti5O12 thick electrodes fabricated by 3D printing for high performance lithium-ion batteries
Journal of Advanced Ceramics (2022)