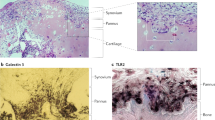
Abstract
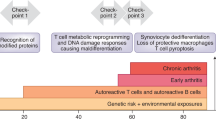
Rheumatoid arthritis is a complex systemic disease that ultimately leads to the progressive destruction of articular and periarticular structures. Novel data indicate that the innate immune system (through activation of Toll-like receptors) is involved in articular pathophysiology, including the recruitment of inflammatory cells, and that periarticular factors such as adipocytokines contribute to the perpetuation of joint inflammation. The deleterious process of joint destruction is mediated by intracellular signaling pathways involving transcription factors, such as nuclear factor κB, cytokines, chemokines, growth factors, cellular ligands, and adhesion molecules. Advances in molecular biology techniques have identified T-cell-independent and B-cell-independent pathways that operate at different stages of the disease. Cytokine-independent pathways appear to be responsible for maintaining basic disease activity that is not affected by currently available therapies. Using this knowledge in combination with gene-transfer and gene-silencing approaches, bench-to-bedside strategies will be developed, thus enabling the creation of novel treatments for rheumatoid arthritis.
Key Points
-
Molecular biology has unveiled numerous steps in the pathophysiology of rheumatoid arthritis, including pathways of cellular activation, inflammation, adhesion, and matrix degradation
-
The innate immune system is centrally involved in articular pathophysiology via Toll-like receptor signaling by macrophages, dendritic cells, and fibroblasts, which subsequently leads to the early recruitment of inflammatory cells
-
The deleterious process of joint destruction is mediated by activation of intracellular signaling pathways and leads to the production of cytokines, chemokines, growth factors, cellular ligands, and adhesion molecules
-
Periarticular factors, such as adipocytokines, can contribute to the perpetuation of joint inflammation and enhance the destructive potential of matrix-degrading synovial fibroblasts
-
Cytokine-independent pathways mediated by activated synovial fibroblasts appear to be responsible for maintaining persistent disease activity that is not affected by currently available therapies
-
T-cell-independent and B-cell-independent pathways that are operative at different stages of the disease can be targeted therapeutically by gene-transfer and gene-silencing approaches
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Neumann E et al. (2002) Identification of differentially expressed genes in rheumatoid arthritis by a combination of complementary DNA array and RNA arbitrarily primed-polymerase chain reaction. Arthritis Rheum 46: 52–63
Faour WH et al. (2005) Early growth response factor-1 mediates prostaglandin E2-dependent transcriptional suppression of cytokine-induced tumor necrosis factor-α gene expression in human macrophages and rheumatoid arthritis-affected synovial fibroblasts. J Biol Chem 280: 9536–9546
Garcia-Vicuna R et al. (2004) CC and CXC chemokine receptors mediate migration, proliferation, and matrix metalloproteinase production by fibroblast-like synoviocytes from rheumatoid arthritis patients. Arthritis Rheum 50: 3866–3877
Kasperkovitz PV et al. (2005) Fibroblast-like synoviocytes derived from patients with rheumatoid arthritis show the imprint of synovial tissue heterogeneity: evidence of a link between an increased myofibroblast-like phenotype and high-inflammation synovitis. Arthritis Rheum 52: 430–441
Firestein GS (2003) Evolving concepts of rheumatoid arthritis. Nature 423: 356–361
Ferrari-Lacraz S et al. (2004) Targeting IL-15 receptor-bearing cells with an antagonist mutant IL-15/Fc protein prevents disease development and progression in murine collagen-induced arthritis. J Immunol 173: 5818–5826
Miranda-Carus ME et al. (2004) IL-15 and the initiation of cell contact-dependent synovial fibroblast-T lymphocyte cross-talk in rheumatoid arthritis: effect of methotrexate. J Immunol 173: 1463–1476
Neumann E et al. (2005) The RANK/RANKL/OPG system in rheumatoid arthritis: new insights from animal models. Arthritis Rheum 52: 2960–2967
Schmutz C et al. (2005) Chemokine receptors in the rheumatoid synovium: upregulation of CXCR5. Arthritis Res Ther 7: R217–R229
Takemura S et al. (2001) Lymphoid neogenesis in rheumatoid synovitis. J Immunol 167: 1072–1080
Zheng B et al. (2005) CXCL13 neutralization reduces the severity of collagen-induced arthritis. Arthritis Rheum 52: 620–626
Van der Voort R et al. (2005) Elevated CXCL16 expression by synovial macrophages recruits memory T cells into rheumatoid joints. Arthritis Rheum 52: 1381–1391
Franz JK et al. (1998) Interleukin-16 produced by synovial fibroblasts mediates chemoattraction to CD4+ T-cells in rheumatoid arthritis. Eur J Immunol 28: 2661–2671
Sawai H et al. (2005) T cell costimulation by fractalkine-expressing synoviocytes in rheumatoid arthritis. Arthritis Rheum 52: 1392–1401
Ohata J et al. (2005) Fibroblast-like synoviocytes of mesenchymal origin express functional B cell-activating factor of the TNF family in response to proinflammatory cytokines. J Immunol 174: 864–870
Tarner IH et al. (2005) The different stages of synovitis: acute vs chronic, early vs late and non-erosive vs erosive. Best Pract Res Clin Rheumatol 19: 19–35
Bijlsma JW et al. (2005) Clinical aspects of immune neuroendocrine mechanism in rheumatic diseases. Rheum Dis Clin North Am 31: xiii–xvi
Zeisel MB et al. (2005) MMP-3 expression and release by rheumatoid arthritis fibroblast-like synoviocytes induced with a bacterial ligand of integrin α5β1. Arthritis Res Ther 7: R118–R126
Seibl R et al. (2004) Expression and regulation of Toll-like receptor 2 in rheumatoid arthritis synovium. Am J Pathol 162: 1221–1227
Pierer M et al. (2004) Chemokine secretion of rheumatoid arthritis synovial fibroblasts stimulated by Toll-like receptor 2 ligands. J Immunol 172: 1256–1265
Roelofs MF et al. (2005) The expression of toll-like receptors 3 and 7 in rheumatoid arthritis synovium is increased and costimulation of toll-like receptors 3, 4, and 7/8 results in synergistic cytokine production by dendritic cells. Arthritis Rheum 52: 2313–2322
Neidhart M et al. (2000) Retrotransposable L1 elements expressed in rheumatoid arthritis synovial tissue: association with genomic DNA hypomethylation and influence on gene expression. Arthritis Rheum 43: 2634–2447
Ospelt C et al. (2004) Toll-like receptors in rheumatoid arthritis joint destruction mediated by two distinct pathways. Ann Rheum Dis 63 (Suppl II): ii90–ii91
Kurowska M et al. (2002) Fibroblast-like synoviocytes from rheumatoid arthritis patients express functional IL-15 receptor complex: endogenous IL-15 in autocrine fashion enhances cell proliferation and expression of Bcl-x(L) and Bcl-2. J Immunol 169: 1760–1767
Schedel J et al. (2002) FLICE-inhibitory protein expression in synovial fibroblasts and at sites of cartilage and bone erosion in rheumatoid arthritis. Arthritis Rheum 46: 1512–1518
Amano T et al. (2003) Synoviolin/Hrd1, an ubiquitin ligase, as a novel pathogenic factor for arthropathy. Genes Dev 17: 2436–2449
Franz JK et al. (2000) Expressions of sentrin, a novel anti-apoptopic molecule at sites of synovial invasion in rheumatoid arthritis. Arthritis Rheum 43: 544–607
Miyashita T et al. (2004) Osteoprotegerin (OPG) acts as an endogenous decoy receptor in tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)-mediated apoptosis of fibroblast-like synovial cells. Clin Exp Immunol 137: 430–436
Schedel J et al. (2004) Discrepancy between mRNA and protein expression of tumour suppressor maspin in synovial tissue may contribute to synovial hyperplasia in rheumatoid arthritis. Ann Rheum Dis 63: 1205–1211
Pap T et al. (2000) Activation of synovial fibroblasts in rheumatoid arthritis: lack of expression of the tumour suppressor PTEN at sites of invasive growth and destruction. Arthritis Res 2: 59–64
Distler JH et al. (2005) The induction of matrix metalloproteinase and cytokine expression in synovial fibroblasts stimulated with immune cells microparticles. Proc Natl Acad Sci USA 102: 2892–2897
Smolen JS et al. (2003) Therapeutic strategies for rheumatoid arthritis. Nat Rev Drug Discov 2: 473–488
Müller-Ladner U et al. (1997) Expression of alternatively spliced CS-1 fibronectin isoform and its counter-receptor VLA-4 in rheumatoid arthritis synovium. J Rheumatol 24: 1873–1880
Johnston A et al. (2005) The anti-inflammatory action of methotrexate is not mediated by lymphocyte apoptosis, but by the suppression of activation and adhesion molecules. Clin Immunol 114: 154–163
Valencia X et al. (2004) Cadherin-11 provides specific cellular adhesion between fibroblast-like synoviocytes. J Exp Med 200: 1673–1679
Neidhart M et al. (1999) Synovial fluid CD146 (MUC18) a marker for synovial membrane angiogenesis in rheumatoid arthritis. Arthritis Rheum 42: 622–630
Giatromanolaki A et al. (2001) The angiogenic pathway 'vascular endothelial growth factor/flk-1(KDR)-receptor' in rheumatoid arthritis and osteoarthritis. J Pathol 194: 101–108
Afuwape AO et al. (2003) Adenoviral delivery of soluble VEGF receptor 1 (sFlt-1) abrogates disease activity in murine collagen-induced arthritis. Gene Ther 10: 1950–1960
Nakahara H et al. (2003) Anti-interleukin-6 receptor antibody therapy reduces vascular endothelial growth factor production in rheumatoid arthritis. Arthritis Rheum 48: 1521–1529
Park YW et al. (2004) Thrombospondin 2 functions as an endogenous regulator of angiogenesis and inflammation in rheumatoid arthritis. Am J Pathol 165: 2087–2098
Takahara K et al. (2004) Autocrine/paracrine role of the angiopoietin-1 and -2 /Tie2 system in cell proliferation and chemotaxis of cultured fibroblastic synoviocytes in rheumatoid arthritis. Hum Pathol 35: 150–158
Pap T et al. (2000) Differential expression pattern of membrane-type matrix metalloproteinases in rheumatoid arthritis. Arthritis Rheum 43: 1226–1232
Judex MO and Mueller BM (2005) Plasminogen activation/plasmin in rheumatoid arthritis: matrix degradation and more. Am J Pathol 166: 645–647
Yang YH et al. (2005) Reduction of arthritis severity in protease-activated receptor-deficient mice. Arthritis Rheum 52: 1325–1332
Schäffler A et al. (2003) Adipocytokines in synovial fluid. JAMA 290: 1709–1710
Bokarewa M et al. (2005) Resistin, an adipokine with potent proinflammatory properties. J Immunol 174: 5789–5795
Van Lent P et al. (2005) Expression of the novel metalloproteinase inhibitor RECK (reversion inducing cysteine-rich protein with Kazal motifs) in inflamed membranes of rheumatoid arthritis patients. Ann Rheum Dis 64: 368–374
Fassbender HG et al. (1980) Transformation of synovial cells in rheumatoid arthritis. Verh Dtsch Ges Pathol 64: 193–212
Gay S et al. (1993) Molecular and cellular mechanisms of joint destruction in rheumatoid arthritis: two cellular mechanisms explain joint destruction? Ann Rheum Dis 52 (Suppl 1): S39–S47
Neidhart M et al. (2005). Galectin-3 is induced in rheumatoid arthritis synovial fibroblasts after adhesion to cartilage oligomeric matrix protein. Ann Rheum Dis 64: 419–424
Acknowledgements
The work is funded, in part, by grants from the German Research Society (DFG) and the Swiss National Fund.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Müller-Ladner, U., Pap, T., Gay, R. et al. Mechanisms of Disease: the molecular and cellular basis of joint destruction in rheumatoid arthritis. Nat Rev Rheumatol 1, 102–110 (2005). https://doi.org/10.1038/ncprheum0047
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncprheum0047