Abstract
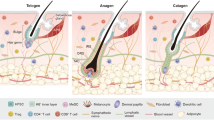
Although normally dormant, hair follicle stem cells (HFSCs) quickly become activated to divide during a new hair cycle. The quiescence of HFSCs is known to be regulated by a number of intrinsic and extrinsic mechanisms. Here we provide several lines of evidence to demonstrate that HFSCs utilize glycolytic metabolism and produce significantly more lactate than other cells in the epidermis. Furthermore, lactate generation appears to be critical for the activation of HFSCs as deletion of lactate dehydrogenase (Ldha) prevented their activation. Conversely, genetically promoting lactate production in HFSCs through mitochondrial pyruvate carrier 1 (Mpc1) deletion accelerated their activation and the hair cycle. Finally, we identify small molecules that increase lactate production by stimulating Myc levels or inhibiting Mpc1 carrier activity and can topically induce the hair cycle. These data suggest that HFSCs maintain a metabolic state that allows them to remain dormant and yet quickly respond to appropriate proliferative stimuli.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Hsu, Y. C., Pasolli, H. A. & Fuchs, E. Dynamics between stem cells, niche, and progeny in the hair follicle. Cell 144, 92–105 (2011).
Morris, R. J. & Potten, C. S. Highly persistent label-retaining cells in the hair follicles of mice and their fate following induction of anagen. J. Invest. Dermatol. 112, 470–475 (1999).
Fuchs, E. The tortoise and the hair: slow-cycling cells in the stem cell race. Cell 137, 811–819 (2009).
Fuchs, E., Merrill, B. J., Jamora, C. & DasGupta, R. At the roots of a never-ending cycle. Dev. Cell 1, 13–25 (2001).
Kloepper, J. E. et al. Mitochondrial function in murine skin epithelium is crucial for hair follicle morphogenesis and epithelial-mesenchymal interactions. J. Invest. Dermatol. 135, 679–689 (2015).
Hamanaka, R. B. & Chandel, N. S. Mitochondrial metabolism as a regulator of keratinocyte differentiation. Cell. Logist. 3, e25456 (2013).
Hamanaka, R. B. et al. Mitochondrial reactive oxygen species promote epidermal differentiation and hair follicle development. Sci. Signal. 6, ra8 (2013).
Baris, O. R. et al. The mitochondrial electron transport chain is dispensable for proliferation and differentiation of epidermal progenitor cells. Stem Cells 29, 1459–1468 (2011).
Blanpain, C., Lowry, W. E., Geoghegan, A., Polak, L. & Fuchs, E. Self-renewal, multipotency, and the existence of two cell populations within an epithelial stem cell niche. Cell 118, 635–648 (2004).
Tumbar, T. et al. Defining the epithelial stem cell niche in skin. Science 303, 359–363 (2004).
Morris, R. J. et al. Capturing and profiling adult hair follicle stem cells. Nat. Biotech. 22, 411–417 (2004).
Trempus, C. S. et al. Enrichment for living murine keratinocytes from the hair follicle bulge with the cell surface marker CD34. J. Invest. Dermatol. 120, 501–511 (2003).
Nguyen, H., Rendl, M. & Fuchs, E. Tcf3 governs stem cell features and represses cell fate determination in skin. Cell 127, 171–183 (2006).
Fromm, H. J. The nature of pyruvate involved in the enzymic formation of L-lactate in the rabbit-muscle lactate dehydrogenase reaction. Biochim. Biophys. Acta 99, 540–542 (1965).
Paus, R., Muller-Rover, S. & Botchkarev, V. A. Chronobiology of the hair follicle: hunting the ‘hair cycle clock’. J. Invest. Dermatol. Symp. Proc. 4, 338–345 (1999).
Chan, F. K., Moriwaki, K. & De Rosa, M. J. Detection of necrosis by release of lactate dehydrogenase activity. Methods Mol. Biol. 979, 65–70 (2013).
Xie, H. et al. Targeting lactate dehydrogenase-a inhibits tumorigenesis and tumor progression in mouse models of lung cancer and impacts tumor-initiating cells. Cell Metab. 19, 795–809 (2014).
White, A. C. et al. Defining the origins of Ras/p53-mediated squamous cell carcinoma. Proc. Natl Acad. Sci. USA 108, 7425–7430 (2011).
Jaks, V. et al. Lgr5 marks cycling, yet long-lived, hair follicle stem cells. Nat. Genet. 40, 1291–1299 (2008).
Kellenberger, A. J. & Tauchi, M. Mammalian target of rapamycin complex 1 (mTORC1) may modulate the timing of anagen entry in mouse hair follicles. Exp. Dermatol. 22, 77–80 (2013).
Bricker, D. K. et al. A mitochondrial pyruvate carrier required for pyruvate uptake in yeast, Drosophila, and humans. Science 337, 96–100 (2012).
Schell, J. C. et al. A role for the mitochondrial pyruvate carrier as a repressor of the Warburg effect and colon cancer cell growth. Mol. Cell 56, 400–413 (2014).
Snippert, H. J. et al. Lgr6 marks stem cells in the hair follicle that generate all cell lineages of the skin. Science 327, 1385–1389 (2010).
Patterson, J. N. et al. Mitochondrial metabolism of pyruvate is essential for regulating glucose-stimulated insulin secretion. J. Biol. Chem. 289, 13335–13346 (2014).
Wang, N. et al. The expression and role of c-Myc in mouse hair follicle morphogenesis and cycling. Acta Histochem. 114, 199–206 (2012).
Bull, J. J. et al. Ectopic expression of c-Myc in the skin affects the hair growth cycle and causes an enlargement of the sebaceous gland. Br. J. Dermatol. 152, 1125–1133 (2005).
Zanet, J. et al. Endogenous Myc controls mammalian epidermal cell size, hyperproliferation, endoreplication and stem cell amplification. J. Cell Sci. 118, 1693–1704 (2005).
Schell, J. C. et al. Control of intestinal stem cell function and proliferation by mitochondrial pyruvate metabolism. Nat. Cell Biol. http://dx.doi.org/10.1038/ncb3593 (2017).
Hsu, P. & Qu, C. K. Metabolic plasticity and hematopoietic stem cell biology. Curr. Opin. Hematol. 20, 289–294 (2013).
Harris, J. M. et al. Glucose metabolism impacts the spatiotemporal onset and magnitude of HSC induction in vivo. Blood 121, 2483–2493 (2013).
Takubo, K. et al. Regulation of glycolysis by Pdk functions as a metabolic checkpoint for cell cycle quiescence in hematopoietic stem cells. Cell Stem Cell 12, 49–61 (2013).
Simsek, T. et al. The distinct metabolic profile of hematopoietic stem cells reflects their location in a hypoxic niche. Cell Stem Cell 7, 380–390 (2010).
Shin, J. et al. Single-cell RNA-Seq with waterfall reveals molecular cascades underlying adult neurogenesis. Cell Stem Cell 17, 360–372 (2015).
Ito, M. et al. Stem cells in the hair follicle bulge contribute to wound repair but not to homeostasis of the epidermis. Nat. Med. 11, 1351–1354 (2005).
Wang, L., Siegenthaler, J. A., Dowell, R. D. & Yi, R. Foxc1 reinforces quiescence in self-renewing hair follicle stem cells. Science 351, 613–617 (2016).
White, A. C. et al. Stem cell quiescence acts as a tumour suppressor in squamous tumours. Nat. Cell Biol. 16, 99–107 (2014).
Acknowledgements
We would like to acknowledge the significant technical support of M. Neebe, J. Cinkornpumin and A. Liu on this project. We are also particularly grateful to members of the Banerjee laboratory for guidance and development of the Ldh activity assay. A.F. and A.C.W. were supported by a fellowship from the Eli and Edythe Broad Center for Regenerative Medicine at UCLA. A.C.W. and M.G. were supported by a fellowship from the Tumor Cell Biology programme at UCLA (NIH). A.C.W. was also supported by a training grant from CIRM. D.J. was supported by awards from a New Idea Award from the Leukemia Lymphoma Society, the Jonsson Comprehensive Cancer Center, the UCLA Clinical Translational Science Institute UL1TR000124, the Prostate Cancer SPORE at UCLA P50 CA092131, and the Eli & Edythe Broad Center for Regenerative Medicine & Stem Cell Research. N.A.G. is a postdoctoral trainee supported by the UCLA Scholars in Oncologic Molecular Imagining program (NCI/NIH grant R25T CA098010). A.S.K. was supported by a UCLA Dissertation Year Fellowship. H.A.C. was supported by National Institute of General Medical Sciences R01-GM081686 and R01-GM0866465. J.R. was supported by NIH (RO1GM094232). H.R.C. was supported by a Research Scholar Grant, RSG-16-111-01-MPC, from the American Cancer Society and the Eli & Edythe Broad Center of Regenerative Medicine and Stem Cell Research at UCLA and Rose Hills Foundation Research Award. W.E.L. was supported by NIH-NIAMS (5R01AR57409), an Impact award from CTSI and the Jonsson Comprehensive Cancer Foundation, and The Gaba Fund through the Eli & Edythe Broad Center of Regenerative Medicine at UCLA.
Author information
Authors and Affiliations
Contributions
A.F., J.S., A.S.K., D.J., M.M., M.G. and D.B. performed experiments. A.F., A.S.K., J.L.Z., N.A.G. performed analysis and compiled data. P.S., D.E. and J.R. provided key reagents essential to the work. T.G. and H.A.C. provided important insight and advice. H.R.C. and W.E.L. provided oversight and were financially responsible for the work. A.F., H.R.C. and W.E.L. were responsible for assembling the figures and writing the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The use of RCGD423 to promote hair growth is covered by a provisional patent application filed by UC Regents and this technology has been licensed by Carthronix LLC. W.E.L. is a member of the board of advisers and a shareholder of Carthronix LLC. None of the work in this study was supported by Carthronix. The use of UK-5099 to promote hair growth is covered by a separate provisional patent filed by UC Regents with W.E.L. and H.R.C. as inventors.
Integrated supplementary information
Supplementary Figure 1 Validation of key reagents and assays.
a, top, IHC with antibody recognizing specifically Ldha (same as used in Fig. 1a). bottom, IHC with antibody recognizing multiple isoforms of Ldh protein. Scale bars indicate 20 micrometers. b, the sorting strategy employed to isolate two populations of cells from the bulge. This particular sort was used to isolate the protein samples shown by western blot in Fig. 1b. c, Validation of colorimetric Ldh enzyme activity assay. The highest Ldh enzyme activity was observed in HFSC bulge and in the muscle. Activity indicated by purple stain; pink color is nuclear fast red counterstain. In absence of substrate lactate there was no detectable activity (purple stain). Right, Additional validation of colorimetric Ldh enzyme activity assay. Enzyme activity inhibited by treating skin with HCl before addition of staining solution with substrate lactate. No Ldh activity (purple stain) detected. Skin in which enzyme activity is not inhibited by Hydrochloric Acid (HCl) shows highest Ldh enzyme activity in HFSC bulge and in the muscle. Scale bars indicate 50 micrometers.
Supplementary Figure 2 Validation of hair cycle stage.
a, Analysis of RNA-seq data to validate that HFSCs in telogen-anagen transition were in fact in such a transition. The telogen-anagen transition is known to be driven by Shh (Gli factors are targets) and Wnt (Lef1, Axin, Ccnd1 are targets) signaling, and correlate with increased proliferation (Ki-67 and Pcna). In addition, Sox4 was previously identified as a regulator of the telogen-anagen transition. n = 3 mice per timepoint. Shown as mean ± s.e.m. Paired t-test was performed, P < 0.05. b, staining for Ki-67 marks dividing cells during various stages of the hair cycle. Brackets indicate the HFSC niche. Scale bars indicate 100 micrometers.
Supplementary Figure 3 Long term deletion of Ldha in HFSCs.
a, K15-CrePR;Ldhafl/fl animals treated with Mifepristone during telogen (day 50) were allowed to develop for 6 months. None of the K15-CrePR;Ldhafl/fl mice showed complete hair regrowth, compared to control animals that all grew their hair coats back completely. Images are representative of at least 12 animals per genotype. b, Histological examination of the long term K15-CrePR;Ldhafl/fl mice showed that Ldha-null HFSCs remained in telogen while WT HFSCs went through anagen and then returned to telogen. This is apparent from thick sections (50 micron, right) that show an increased number of club hairs in the WT relative to Ldha-null follicles. Scale bars indicate 100 micrometers (left), and 20 micrometers (middle and right). c, IHC for HFSC marker Sox9 showed that deletion of Ldha from HFSCs does not affect their presence in the bulge even after 6 months. In addition, IHC and Ldh activity assay demonstrate that the deletion of Ldha was sustained. Because of the mosaicism of the deletion, in some portions of K15-CrePR;Ldhafl/fl skin Ldha was not deleted. Shown on the bottom row is tissue from hair bearing skin in the K15-CrePR;Ldhafl/fl mice where Ldha was still expressed, showing that new hair growth in K15-CrePR;Ldhafl/fl mice was due to lack of deletion of Ldha caused by the mosaic approach used to mediate Cre recombination. Scale bars indicate 20 micrometers. d, To determine how various signaling pathways previously linked to the hair cycle are affected by loss of Ldha in HFSCs, we performed IHC for markers that indicate activity of these pathways in telogen and telogen-anagen transition. Note that pStat5 appears to be suppressed in normal telogen-anagen transition, and this does not seem to occur in Ldha-null HFSCs. pStat1 and pStat3 did not seem to be affected by loss of Ldha. Expression of Gli3, a target of Shh signaling, is typically induced in an activated hair germ derived from HFSCs, but Ldha-null HFSCs do not make an active hair germ. Activation of the Wnt pathway is indicated by nuclear localization of β-catenin, and very little nuclear β-catenin was detected in Ldha-null HFSCs. Scale bars indicate 6 micrometers.
Supplementary Figure 4 Long term deletion of Mpc1 in HFSCs.
a, Six months after initiation of deletion of Mpc1 in HFSCs (K15-CrePR;Mpc1fl/fl), mice lacking Mpc1 show no deleterious effects as measured by the hair cycle (left), pathology (middle, H and E), or staining for HFSCs (right, Sox9). Scale bars indicate 100 micrometers in middle panel, and 50 micrometers in right panel. Images are representative of at least 12 animals per genotype. b, To demonstrate that the deletion of Mpc1 promotes proliferation specifically in HFSCs, we used K15-CrePR;Ldhafl/fl mice bearing a lox-stop-lox-Tomato allele to look at K15 + HFSCs and proliferation with and without Mpc1 deletion (left). In addition, we took advantage of the ires-GFP within the Lgr5-CreER allele to stain for Ki-67 and GFP and look for co-localization with and without Mpc1 deletion (right). White brackets denote bulge area. Scale bars represent 20 micrometers. c, Deletion of Mpc1 in mice bearing the Lgr6-CreER allele shows no premature induction of the hair cycle. d, Ldh activity assay on sorted HFSCs from either control or Lgr6-CreER mediated Mpc1 deletion mice showed increased activity in cells lacking Mpc1. n = 6 mice per genotype pooled from 2 independent experiments. Shown as mean ± s.e.m. Paired t-test was performed, P < 0.05.
Supplementary Figure 5 Stimulation of Jak-Stat signaling and the hair cycle.
RCGD423 was applied topically to shaved mice at day 50. 48 hours after treatment, the skin was harvested and prepared for IHC. IHC with the indicated antibodies demonstrates relative activity of Stat signaling in vehicle versus RCGD423 treated skin. Scale bars indicate 20 micrometers.
Supplementary information
Supplementary Information
Supplementary Information (PDF 6518 kb)
Supplementary Information
Life Sciences Reporting Summary
Supplementary Table 1
Supplementary Information
Rights and permissions
About this article
Cite this article
Flores, A., Schell, J., Krall, A. et al. Lactate dehydrogenase activity drives hair follicle stem cell activation. Nat Cell Biol 19, 1017–1026 (2017). https://doi.org/10.1038/ncb3575
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb3575
This article is cited by
-
Autophagy induces hair follicle stem cell activation and hair follicle regeneration by regulating glycolysis
Cell & Bioscience (2024)
-
Deciphering the molecular mechanisms of stem cell dynamics in hair follicle regeneration
Experimental & Molecular Medicine (2024)
-
Local and systemic mechanisms that control the hair follicle stem cell niche
Nature Reviews Molecular Cell Biology (2024)
-
Traditional Chinese Medicine Shi-Bi-Man regulates lactic acid metabolism and drives hair follicle stem cell activation to promote hair regeneration
Chinese Medicine (2023)
-
Lactate-dependent transcriptional regulation controls mammalian eye morphogenesis
Nature Communications (2023)