Abstract
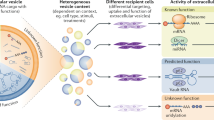
Exosomes are vesicles of endocytic origin released by many cells. These vesicles can mediate communication between cells, facilitating processes such as antigen presentation. Here, we show that exosomes from a mouse and a human mast cell line (MC/9 and HMC-1, respectively), as well as primary bone marrow-derived mouse mast cells, contain RNA. Microarray assessments revealed the presence of mRNA from approximately 1300 genes, many of which are not present in the cytoplasm of the donor cell. In vitro translation proved that the exosome mRNAs were functional. Quality control RNA analysis of total RNA derived from exosomes also revealed presence of small RNAs, including microRNAs. The RNA from mast cell exosomes is transferable to other mouse and human mast cells. After transfer of mouse exosomal RNA to human mast cells, new mouse proteins were found in the recipient cells, indicating that transferred exosomal mRNA can be translated after entering another cell. In summary, we show that exosomes contain both mRNA and microRNA, which can be delivered to another cell, and can be functional in this new location. We propose that this RNA is called “exosomal shuttle RNA” (esRNA).
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Van Niel, G., Porto-Carreiro, I., Simoes, S. & Raposo, G. Exosomes: a common pathway for a specialized function. J. Biochem. 140, 13–21 (2006).
Pan, B.-T. & Johnstone, R. M. Fate of the transferrin receptor during maturation of sheep reticulocytes in vitro: Selective externalization of the receptor. Cell 33, 967–978 (1983).
Thery, C. et al. Molecular characterization of dendritic cell-derived exosomes: selective accumulation of the heat shock protein hsc73. J. Cell Biol. 147, 599–610 (1999).
Raposo, G. et al. B lymphocytes secrete antigen-presenting vesicles. J. Exp. Med. 183, 1161–1172 (1996).
Blanchard, N. et al. TCR activation of human T cells induces the production of exosomes bearing the TCR/CD3/ζ complex. J. Immunol. 168, 3235–3241 (2002).
Raposo, G. et al. Accumulation of major histocompatibility complex class II molecules in mast cell secretory granules and their release upon degranulation. Mol. Biol. Cell. 8, 2631–2645 (1997).
Van Niel, G. et al. Intestinal epithelial cells secrete exosome-like vesicles. Gastroenterology 121, 337–349 (2001).
Mears, R. et al. Proteomic analysis of melanoma-derived exosomes by two-dimensional polyacrylamide gel electrophoresis and mass spectrometry. Proteomics 4, 4019–4031 (2004).
Karlsson, M. et al. “Tolerosomes” are produced by intestinal epithelial cells. Eur. J. Immunol. 31, 2892–2900 (2001).
Denzer, K. et al. Follicular dendritic cells carry MHC class II-expressing microvesicles at their surface. J. Immunol. 165, 1259–1265 (2000).
Clayton, A. et al. Adhesion and signaling by B cell-derived exosomes: the role of integrins. FASEB J. 18, 977–979 (2004).
Morelli, A. E. et al. Endocytosis, intracellular sorting and processing of exosomes by dendritic cells. Blood. 15, 3257–3266 (2004).
Caby, M.-P., Lankar, D., Vincendeau-Scherrer, C., Raposo, G. & Bonnerot, C. Exosomal-like vesicles are present in human blood plasma. Int. Immunol. 17, 879–87 (2005).
Segura, E. et al. ICAM-1 on exosomes from mature dendritic cells is critical for efficient naive T cell priming. Blood. 106, 216–23 (2005).
Skokos, D. et al. Mast cell-eependent B and T lymphocyte activation is mediated by the secretion of immunologically active exosomes. J Immunol. 166, 868–876 (2001).
Bess, J. W., Gorelick, R. J., Bosche, W. J., Henderson, L. E. & Arthur, L. O. Microvesicles are a source of contaminating cellular proteins found in purified HIV-1 preparations. Virology 230, 134–144 (1997).
Baj-Krzyworzeka, M. et al. Tumour-derived microvesicles carry several surface determinants and mRNA of tumour cells and transfer some of these determinants to monocytes. Cancer Immunol. Immunother. 55, 808–818 (2006).
Ratajczak, J. et al. Embryonic stem cell-derived microvesicles reprogram hematopoietic progenitors: evidence for horizontal transfer of mRNA and protein delivery. Leukemia. 20, 847–856 (2006).
Thomson, J. M. et al. Extensive post-transcriptional regulation of microRNAs and its implications for cancer. Genes Dev. 20, 2202–2207 (2006).
Wienholds, E. & Plasterk, R. H. MicroRNA function in animal development. FEBS Lett. 579, 5911–5922 (2005).
Alvarez-Garcia, I. & Miska, E. A. MicroRNA functions in animal development and human disease. Development. 132, 4653–4662 (2005).
Krek, A. et al. Combinatorial microRNA target predictions. Nature Genet. 37, 495–500 (2005).
Nilsson, G. et al. Phenotypic characterization of the human mast-cell line HMC-1. Scand. J. Immunol. 39, 489–98 (1994).
Razin, E. et al. Interleukin 3: A differentiation and growth factor for the mouse mast cell that contains chondroitin sulfate E proteoglycan. J. Immunol. 132, 1479–1486 (1984).
Pisitkun, T., Shen, R.-F. & Knepper, M. A. Identification and proteomic profiling of exosomes in human urine. Proc. Natl Acad. Sci. USA 101, 13368–13373 (2004).
Thery, C. et al. Proteomic analysis of dendritic cell-derived exosomes: a secreted subcellular compartment distinct from apoptotic vesicles. J. Immunol. 166, 7309–7318 (2001).
Potolicchio, I. et al. Proteomic analysis of microglia-derived exosomes: metabolic role of the aminopeptidase CD13 in neuropeptide catabolism. J. Immunol. 175, 2237–2243 (2005).
Wubbolts, R. et al. Proteomic and biochemical analyses of human B cell-derived exosomes. J. Biol. Chem. 278, 10963–10972 (2003).
Mears, R. et al. Proteomic analysis of melanoma-derived exosomes by two-dimensional polyacrylamide gel electrophoresis and mass spectrometry. Proteomics. 4, 4019–4031 (2004).
Acknowledgements
We gratefully acknowledge Swegene Microarray Resource Centre at Lund University for help with the Affymetrix microarray processing and analysis and the Exiqon Company for analysing the microRNA. We thank core facility, Proteomics Resource Centre at Göteborg University (http://www.proteomics.cf.gu.se) for the help with LC-MS/MS and MALDI-TOF. The human mast cell line HMC-1 was kindly provided by G. Nilsson (Uppsala University) and J. Butterfield (Mayo Clinic). We have had exceedingly helpful discussions with E. Telemo and K. McNagny during the progress of the project. We thank B. R. Johansson, U. Nannmark and Y. Josefsson at The Electron Microscopy Unit, Inst Biomedicine, Göteborg University for the help with electron microscopy, and N. Almqvist for taking several of the electron microscopy photographs. We are also grateful to T. Nyström and A. Farwell for providing laboratory resources for some of the experiments at the Cell and Molecular Biology Laboratory at Göteborg University. The current experiments were funded by the Swedish Research Council (K2005-74X-13429-06A), the Swedish Heart and Lung foundation and the Swedish Asthma-Allergy Foundation. J.L. is funded by Herman Krefting's Foundation against Asthma/Allergy.
Author information
Authors and Affiliations
Contributions
H.V. and K.E., with contributions from A.B., performed all experiments. J.L. conceived the study and participated in the overall planning of the study, with support from all authors. All authors participated in the design of specific experiments and in writing the manuscript.
Corresponding author
Ethics declarations
Competing interests
J.L., H.V., K.E., M.S. and A.B. co-own the commercial development of a US patent application related to utilization of exosomes as a vector for therapy.
Supplementary information
Supplementary Information
Supplementary figures S1, S2, S3 and Supplementary methods (PDF 529 kb)
Supplementary Information
Supplementary Table S1 (PDF 92 kb)
Supplementary Information
Supplementary Table S2 (PDF 204 kb)
Supplementary Information
Supplementary Table S3 (PDF 82 kb)
Supplementary Information
Supplementary Table S4 (PDF 58 kb)
Supplementary Information
Supplementary Table S5 (PDF 82 kb)
Supplementary Information
Supplementary Table S6 (PDF 54 kb)
Rights and permissions
About this article
Cite this article
Valadi, H., Ekström, K., Bossios, A. et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol 9, 654–659 (2007). https://doi.org/10.1038/ncb1596
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1596
This article is cited by
-
Protein cargo in extracellular vesicles as the key mediator in the progression of cancer
Cell Communication and Signaling (2024)
-
Extracellular vesicles from UTX-knockout endothelial cells boost neural stem cell differentiation in spinal cord injury
Cell Communication and Signaling (2024)
-
Versatile extracellular vesicle-mediated information transfer: intercellular synchronization of differentiation and of cellular phenotypes, and future perspectives
Inflammation and Regeneration (2024)
-
LncRNA AL139294.1 can be transported by extracellular vesicles to promote the oncogenic behaviour of recipient cells through activation of the Wnt and NF-κB2 pathways in non-small-cell lung cancer
Journal of Experimental & Clinical Cancer Research (2024)
-
Non-bone-derived exosomes: a new perspective on regulators of bone homeostasis
Cell Communication and Signaling (2024)