Abstract
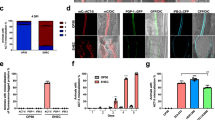
Enteropathogenic Escherichia coli (EPEC) is a bacterial pathogen that causes infantile diarrhea worldwide1. EPEC injects a bacterial protein, translocated intimin receptor (Tir), into the host-cell plasma membrane where it acts as a receptor for the bacterial outer membrane protein, intimin2. The interaction of Tir and intimin triggers a marked rearrangement of the host actin cytoskeleton into pedestals beneath adherent bacteria. On delivery into host cells, EPEC Tir is phosphorylated on tyrosine 474 of the intracellular carboxy-terminal domain, an event that is required for pedestal formation3. Despite its essential role, the function of Tir tyrosine phosphorylation has not yet been elucidated. Here we show that tyrosine 474 of Tir directly binds the host-cell adaptor protein Nck, and that Nck is required for the recruitment of both neural Wiskott–Aldrich-syndrome protein (N-WASP) and the actin-related protein (Arp)2/3 complex to the EPEC pedestal, directly linking Tir to the cytoskeleton. Cells with null alleles of both mammalian Nck genes are resistant to the effects of EPEC on the actin cytoskeleton. These results implicate Nck adaptors as host-cell determinants of EPEC virulence.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Todd, E. C. World Health Stat. Q. 50, 30–50 (1997).
Kenny, B. et al. Cell 91, 511–520 (1997).
Kenny, B. Mol. Microbiol. 31, 1229–1241 (1999).
Garrity, P. A. et al. Cell 85, 639–650 (1996).
Rockow, S., Tang, J., Xiong, W. & Li, W. Oncogene 12, 2351–2359 (1996).
Chen, M., She, H., Kim, A., Woodley, D. T. & Li, W. Mol. Cell. Biol. 20, 7867–7880 (2000).
Frischknecht, F. et al. Nature 401, 926–929 (1999).
Songyang, Z. et al. Cell 72, 767–778 (1993).
Goosney, D. et al. Curr. Biol. 10, 735–738 (2000).
Braverman, L. E. & Quilliam, L. A. J. Biol. Chem. 274, 5542–5549 (1999).
Coutinho, S. et al. Blood 96, 618–624 (2000).
DeVinney, R. et al. Infect. Immun. 67, 2389–2398 (1999).
Kalman, D. et al. Nature Cell Biol. 1, 389–391 (1999).
Rohatgi, R., Nollau, P., Ho, H. Y., Kirschner, M. W. & Mayer, B. J. J. Biol. Chem. 276, 26448–26452 (2001).
Anton, I. M., Lu, W., Mayer, B. J., Ramesh, N. & Geha, R. S. J. Biol. Chem. 273, 20992–20995 (1998).
Wunderlich, L., Goher, A., Farago, A., Downward, J. & Buday, L. Cell. Signal. 11, 253–262 (1999).
Rao, Y. & Zipursky, S. L. Proc. Natl Acad. Sci. USA 95, 2077–2082 (1998).
Freeman, N. L. et al. Cell. Motil. Cytoskel. 47, 307–318 (2000).
Critchley, D. R. Curr. Opin. Cell Biol. 12, 133–139 (2000).
Marches, O. et al. Infect. Immun. 68, 2171–2182 (2000).
Moreau, V. et al. Nature Cell Biol. 2, 441–448 (2000).
Frischknecht, F. & Way, M. Trends Cell Biol. 11, 30–38 (2001).
Griffin, P. M. et al. Ann. Int. Med. 109, 705–712 (1988).
Levine, M. M. et al. Lancet 1, 1119–1122 (1978).
Fortineau, N. et al. in 98th Annual General Meeting of the American Society for Microbiology (Am. Soc. Microbiol., Washington DC, 1998).
Henkemeyer, M. et al. Oncogene 9, 1001–1014 (1994).
Kameshita, I. & Fujisawa, H. Anal. Biochem. 183, 139–143 (1989).
Lin, D., Gish, G. D., Songyang, Z. & Pawson, T. J. Biol. Chem. 274, 3726–3733 (1999).
Mizushima, S. & Nagata, S. Nucleic Acids Res. 18, 5322 (1990).
Acknowledgements
We thank P. Nash for assistance with phosphopeptide analysis; the Kirschner lab for the N-WASP antibody; E. Gouin for the Arp3 antibody; M. Gold (UBC) for helpful discussions; and members of the Finlay laboratory for comments on the manuscript. This work was supported by grants from the Canadian Institutes of Health Research (CIHR) and Howard Hughes International Research Scholar Awards to B.B.F. and T.P. S.G is supported by a postdoctoral fellowship from the MRC/CIHR, F.B. is supported by a postdoctoral fellowship from the Deutsche Forschungsgemeinschaft (DFG). B.B.F and T.P are Distinguished Scientists of the CIHR.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary figure
Figure S1 A dominant negative Nck 1 protein blocks EPEC pedestal formation (PDF 76 kb)
Rights and permissions
About this article
Cite this article
Gruenheid, S., DeVinney, R., Bladt, F. et al. Enteropathogenic E. coli Tir binds Nck to initiate actin pedestal formation in host cells. Nat Cell Biol 3, 856–859 (2001). https://doi.org/10.1038/ncb0901-856
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb0901-856
This article is cited by
-
Identification and characterization of a large family of superbinding bacterial SH2 domains
Nature Communications (2018)
-
GWAS analysis of QTL for enteric septicemia of catfish and their involved genes suggest evolutionary conservation of a molecular mechanism of disease resistance
Molecular Genetics and Genomics (2017)
-
Plasticity of the brush border — the yin and yang of intestinal homeostasis
Nature Reviews Gastroenterology & Hepatology (2016)