Abstract
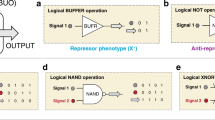
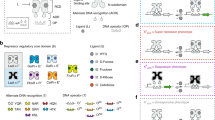
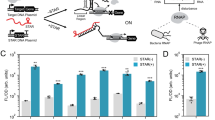
Synthetic genetic programs are built from circuits that integrate sensors and implement temporal control of gene expression1,2,3,4. Transcriptional circuits are layered by using promoters to carry the signal between circuits. In other words, the output promoter of one circuit serves as the input promoter to the next. Thus, connecting circuits requires physically connecting a promoter to the next circuit. We show that the sequence at the junction between the input promoter and circuit can affect the input-output response (transfer function) of the circuit5,6,7,8,9. A library of putative sequences that might reduce (or buffer) such context effects, which we refer to as 'insulator parts', is screened in Escherichia coli. We find that ribozymes that cleave the 5′ untranslated region (5′-UTR) of the mRNA are effective insulators. They generate quantitatively identical transfer functions, irrespective of the identity of the input promoter. When these insulators are used to join synthetic gene circuits, the behavior of layered circuits can be predicted using a mathematical model. The inclusion of insulators will be critical in reliably permuting circuits to build different programs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Kim, P.M. & Tidor, B. Limitations of quantitative gene regulation models: a case study. Genome Res. 13, 2391–2395 (2003).
Del Vecchio, D., Ninfa, A.J. & Sontag, E.D. Modular cell biology: retroactivity and insulation. Mol. Syst. Biol. 4, 161 (2008).
Grünberg, R. & Serrano, L. Strategies for protein synthetic biology. Nucleic Acids Res. 38, 2663–2675 (2010).
Tan, C., Marguet, P. & You, L. Emergent bistability by a growth-modulating positive feedback circuit. Nat. Chem. Biol. 5, 842–848 (2009).
Ellis, T., Wang, X. & Collins, J.J. Diversity-based, model-guided construction of synthetic gene networks with predicted functions. Nat. Biotechnol. 27, 465–471 (2009).
Anderson, J.C., Voigt, C.A. & Arkin, A.P. Environmental signal integration by a modular AND gate. Mol. Syst. Biol. 3, 133 (2007).
Wang, B., Kitney, R.I., Joly, N. & Buck, M. Engineering modular and orthogonal genetic logic gates for robust digital-like synthetic biology. Nat. Commun. 2, 508 (2011).
Hooshangi, S., Thiberge, S. & Weiss, R. Ultrasensitivity and noise propagation in a synthetic transcriptional cascade. Proc. Natl. Acad. Sci. USA 102, 3581–3586 (2005).
Pedraza, J.M. & van Oudenaarden, A. Noise propagation in gene networks. Science 307, 1965–1969 (2005).
Endy, D. Foundations for engineering biology. Nature 438, 449–453 (2005).
Voigt, C.A. Genetic parts to program bacteria. Curr. Opin. Biotechnol. 17, 548–557 (2006).
Bio FAB Group et al. Engineering life: building a fab for biology. Sci. Am. 294, 44–51 (2006).
Shetty, R.P., Endy, D. & Knight, T.F. Jr Engineering BioBrick vectors from BioBrick parts. J. Biol. Eng. 2, 5 (2008).
Anderson, J.C. et al. BglBricks: A flexible standard for biological part assembly. J. Biol. Eng. 4, 1 (2010).
Clancy, K. & Voigt, C.A. Programming cells: towards an automated 'Genetic Compiler'. Curr. Opin. Biotechnol. 21, 572–581 (2010).
Arkin, A. Setting the standard in synthetic biology. Nat. Biotechnol. 26, 771–774 (2008).
Rhodius, V.A., Mutalik, V.K. & Gross, C.A. Predicting the strength of UP-elements and full-length E. coli sE promoters. Nucleic Acids Res. 10.1093/nar/gkr1190 (2012).
Davis, J.H., Rubin, A.J. & Sauer, R.T. Design, construction and characterization of a set of insulated bacterial promoters. Nucleic Acids Res. 39, 1131–1141 (2011).
Carrier, T.A. & Keasling, J.D. Engineering mRNA stability in E. coli by the addition of synthetic hairpins using a 5′ cassette system. Biotechnol. Bioeng. 55, 577–580 (1997).
Qi, L., Haurwitz, R.E., Shao, W., Doudna, J.A. & Arkin, A.P. RNA processing enables predictable programming of gene expression. Nat. Biotechnol.advance online publication, 10.1038/nbt.2355 (16 September 2012).
Gardner, T.S., Cantor, C.R. & Collins, J.J. Construction of a genetic toggle switch in Escherichia coli. Nature 403, 339–342 (2000).
Elowitz, M.B. & Leibler, S. A synthetic oscillatory network of transcriptional regulators. Nature 403, 335–338 (2000).
Yokobayashi, Y., Weiss, R. & Arnold, F.H. Directed evolution of a genetic circuit. Proc. Natl. Acad. Sci. USA 99, 16587–16591 (2002).
Lou, C. et al. Synthesizing a novel genetic sequential logic circuit: a push-on push-off switch. Mol. Syst. Biol. 6, 350 (2010).
Lutz, R. & Bujard, H. Independent and tight regulation of transcriptional units in Escherichia coli via the LacR/O, the TetR/O and AraC/I1–I2 regulatory elements. Nucleic Acids Res. 25, 1203–1210 (1997).
Buzayan, J.M., Gerlach, W.L. & Bruening, G. Satellite tobacco ringspot virus RNA: A subset of the RNA sequence is sufficient for autolytic processing. Proc. Natl. Acad. Sci. USA 83, 8859–8862 (1986).
Galdzicki, M., Rodriguez, C., Chandran, D., Sauro, H.M. & Gennari, J.H. Standard Biological Parts Knowledgebase. PLoS ONE 6, e17005 (2011).
Forster, A.C. & Symons, R.H. Self-cleavage of virusoid RNA is performed by the proposed 55-nucleotide active site. Cell 50, 9–16 (1987).
Di Serio, F., Daròs, J.A., Ragozzino, A. & Flores, R. A 451-nucleotide circular RNA from cherry with hammerhead ribozymes in its strands of both polarities. J. Virol. 71, 6603–6610 (1997).
Kaper, J.M., Tousignant, M.E. & Steger, G. Nucleotide sequence predicts circularity and self-cleavage of 300-ribonucleotide satellite of arabis mosaic virus. Biochem. Biophys. Res. Commun. 154, 318–325 (1988).
Hernández, C. & Flores, R. Plus and minus RNAs of peach latent mosaic viroid self-cleave in vitro via hammerhead structures. Proc. Natl. Acad. Sci. USA 89, 3711–3715 (1992).
Roossinck, M.J., Sleat, D. & Palukaitis, P. Satellite RNAs of plant viruses: structures and biological effects. Microbiol. Rev. 56, 265–279 (1992).
Beal, J., Lu, T. & Weiss, R. Automatic compilation from high-level biologically-oriented programming language to genetic regulatory networks. PLoS ONE 6, e22490 (2011).
Munsky, B., Neuert, G. & van Oudenaarden, A. Using gene expression noise to understand gene regulation. Science 336, 183–187 (2012).
Gillespie, D.T. Exact stochastic simulation of coupled chemical reactions. J. Phys. Chem. 81, 2340–2361 (1977).
Arkin, A., Ross, J. & McAdams, H.H. Stochastic kinetic analysis of developmental pathway bifurcation in phage l-infected Escherichia coli cells. Genetics 149, 1633–1648 (1998).
Munsky, B. & Khammash, M. The finite state projection algorithm for the solution of the chemical master equation. J. Chem. Phys. 124, 044104 (2006).
Munsky, B., Trinh, B. & Khammash, M. Listening to the noise: random fluctuations reveal gene network parameters. Mol. Syst. Biol. 5, 318 (2009).
Moon, T.S., Lou, C., Tamsir, A., Stanton B.C. & Voigt, C.A. Genetic programs constructed from layered logic gates in single cells. Nature advance online publication, 10.1038/nature11516 (7 October 2012).
Walczak, A.M., Mugler, A. & Wiggins, C.H. A stochastic spectral analysis of transcriptional regulatory cascades. Proc. Natl. Acad. Sci. USA 106, 6529–6534 (2009).
Ro, D.-K. et al. Production of the antimalarial drug precursor artemisinic acid in engineered yeast. Nature 440, 940–943 (2006).
Temme, K., Zhao, D. & Voigt, C.A. Refactoring the nitrogen fixation gene cluster from Klebsiella oxytoca. Proc. Natl. Acad. Sci. USA 109, 7085–7090 (2012).
Rhodius, V.A. & Mutalik, V.K. Predicting strength and function for promoters of the Escherichia coli alternative sigma factor, sigmaE. Proc. Natl. Acad. Sci. USA 107, 2854–2859 (2010).
Salis, H.M., Mirsky, E.A. & Voigt, C.A. Automated design of synthetic ribosome binding sites to control protein expression. Nat. Biotechnol. 27, 946–950 (2009).
Delcher, A.L., Harmon, D., Kasif, S., White, O. & Salzberg, S.L. Improved microbial gene identification with GLIMMER. Nucleic Acids Res. 27, 4636–4641 (1999).
Kelly, J.R. et al. Measuring the activity of BioBrick promoters using an in vivo reference standard. J. Biol. Eng. 3, 4 (2009).
Khvorova, A., Lescoute, A., Westhof, E. & Jayasena, S.D. Sequence elements outside the hammerhead ribozyme catalytic core enable intracellular activity. Nat. Struct. Biol. 10, 708–712 (2003).
Nelson, J.A., Shepotinovskaya, I. & Uhlenbeck, O.C. Hammerheads derived from sTRSV show enhanced cleavage and ligation rate constants. Biochemistry 44, 14577–14585 (2005).
Acknowledgements
C.A.V. is supported by Life Technologies, Defense Advanced Research Projects Agency Chronicle of Lineage Indicative of Origins (DARPA CLIO, N66001-12-C-4018), Office of Naval Research (N00014-10-1-0245), National Science Foundation (NSF) (CCF-0943385), National Institutes of Health (AI067699) and the NSF Synthetic Biology Engineering Research Center (SynBERC, SA5284-11210).
Author information
Authors and Affiliations
Contributions
C.A.V. conceived of and supervised the project. C.L. designed and performed the experiments. B.S. performed experiments with the McbR repressor. Y.-J.C. and C.L. performed the q-PCR experiments. B.M. and C.L. analyzed data. C.L., B.M., Y.-J.C. and C.A.V. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–21 and Supplementary Tables 1–3 (PDF 5261 kb)
Rights and permissions
About this article
Cite this article
Lou, C., Stanton, B., Chen, YJ. et al. Ribozyme-based insulator parts buffer synthetic circuits from genetic context. Nat Biotechnol 30, 1137–1142 (2012). https://doi.org/10.1038/nbt.2401
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.2401