Abstract
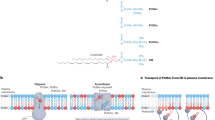
In all animal cells, phospholipids are asymmetrically distributed between the outer and inner leaflets of the plasma membrane1. This asymmetrical phospholipid distribution is disrupted in various biological systems. For example, when blood platelets are activated, they expose phosphatidylserine (PtdSer) to trigger the clotting system2,3. The PtdSer exposure is believed to be mediated by Ca2+-dependent phospholipid scramblases that transport phospholipids bidirectionally1,4, but its molecular mechanism is still unknown. Here we show that TMEM16F (transmembrane protein 16F) is an essential component for the Ca2+-dependent exposure of PtdSer on the cell surface. When a mouse B-cell line, Ba/F3, was treated with a Ca2+ ionophore under low-Ca2+ conditions, it reversibly exposed PtdSer. Using this property, we established a Ba/F3 subline that strongly exposed PtdSer by repetitive fluorescence-activated cell sorting. A complementary DNA library was constructed from the subline, and a cDNA that caused Ba/F3 to expose PtdSer spontaneously was identified by expression cloning. The cDNA encoded a constitutively active mutant of TMEM16F, a protein with eight transmembrane segments5. Wild-type TMEM16F was localized on the plasma membrane and conferred Ca2+-dependent scrambling of phospholipids. A patient with Scott syndrome6,7, which results from a defect in phospholipid scrambling activity8,9, was found to carry a mutation at a splice-acceptor site of the gene encoding TMEM16F, causing the premature termination of the protein.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Leventis, P. A. & Grinstein, S. The distribution and function of phosphatidylserine in cellular membranes. Annu. Rev. Biophys. 39, 407–427 (2010)
Zwaal, R. F., Comfurius, P. & Bevers, E. M. Lipid–protein interactions in blood coagulation. Biochim. Biophys. Acta 1376, 433–453 (1998)
Lentz, B. Exposure of platelet membrane phosphatidylserine regulates blood coagulation. Prog. Lipid Res. 42, 423–438 (2003)
Balasubramanian, K. & Schroit, A. Aminophospholipid asymmetry: a matter of life and death. Annu. Rev. Physiol. 65, 701–734 (2003)
Galietta, L. The TMEM16 protein family: a new class of chloride channels? Biophys. J. 97, 3047–3053 (2009)
Weiss, H. & Lages, B. Family studies in Scott syndrome. Blood 90, 475–476 (1997)
Zwaal, R., Comfurius, P. & Bevers, E. Scott syndrome, a bleeding disorder caused by defective scrambling of membrane phospholipids. Biochim. Biophys. Acta 1636, 119–128 (2004)
Williamson, P. et al. Phospholipid scramblase activation pathways in lymphocytes. Biochemistry 40, 8065–8072 (2001)
Wielders, S. J. et al. Absence of platelet-dependent fibrin formation in a patient with Scott syndrome. Thromb. Haemost. 102, 76–82 (2009)
Daleke, D. Phospholipid flippases. J. Biol. Chem. 282, 821–825 (2007)
Caputo, A. et al. TMEM16A, a membrane protein associated with calcium-dependent chloride channel activity. Science 322, 590–594 (2008)
Schroeder, B., Cheng, T., Jan, Y. & Jan, L. Expression cloning of TMEM16A as a calcium-activated chloride channel subunit. Cell 134, 1019–1029 (2008)
Yang, Y. et al. TMEM16A confers receptor-activated calcium-dependent chloride conductance. Nature 455, 1210–1215 (2008)
Schreiber, R. et al. Expression and function of epithelial anoctamins. J. Biol. Chem. 285, 7838–7845 (2010)
Shi, J. & Gilbert, G. Lactadherin inhibits enzyme complexes of blood coagulation by competing for phospholipid-binding sites. Blood 101, 2628–2636 (2003)
Hanayama, R., Tanaka, M., Miwa, K. & Nagata, S. Expression of developmental endothelial locus-1 in a subset of macrophages for engulfment of apoptotic cells. J. Immunol. 172, 3876–3882 (2004)
Emoto, K., Toyama-Sorimachi, N., Karasuyama, H., Inoue, K. & Umeda, M. Exposure of phosphatidylethanolamine on the surface of apoptotic cells. Exp. Cell Res. 232, 430–434 (1997)
Macia, E. et al. Dynasore, a cell-permeable inhibitor of dynamin. Dev. Cell 10, 839–850 (2006)
Toti, F., Satta, N., Fressinaud, E., Meyer, D. & Freyssinet, J. Scott syndrome, characterized by impaired transmembrane migration of procoagulant phosphatidylserine and hemorrhagic complications, is an inherited disorder. Blood 87, 1409–1415 (1996)
Kojima, H. et al. Production and characterization of transformed B-lymphocytes expressing the membrane defect of Scott syndrome. J. Clin. Invest. 94, 2237–2244 (1994)
Shyu, A., Wilkinson, M. & van Hoof, A. Messenger RNA regulation: to translate or to degrade. EMBO J. 27, 471–481 (2008)
Kavathas, P. & Herzenberg, L. A. Amplification of a gene coding for human T-cell differentiation antigen. Nature 306, 385–387 (1983)
Suda, T., Takahashi, T., Golstein, P. & Nagata, S. Molecular cloning and expression of the Fas ligand: a novel member of the tumor necrosis factor family. Cell 75, 1169–1178 (1993)
Berridge, M., Bootman, M. & Roderick, H. Calcium signalling: dynamics, homeostasis and remodelling. Nature Rev. Mol. Cell. Biol. 4, 517–529 (2003)
Ferrera, L. et al. Regulation of TMEM16A chloride channel properties by alternative splicing. J. Biol. Chem. 284, 33360–33368 (2009)
Nagata, S., Hanayama, R. & Kawane, K. Autoimmunity and the clearance of dead cells. Cell 140, 619–630 (2010)
Huppertz, B., Bartz, C. & Kokozidou, M. Trophoblast fusion: fusogenic proteins, syncytins and ADAMs, and other prerequisites for syncytial fusion. Micron 37, 509–517 (2006)
Helming, L. & Gordon, S. Molecular mediators of macrophage fusion. Trends Cell Biol. 19, 514–522 (2009)
van den Eijnde, S. et al. Transient expression of phosphatidylserine at cell–cell contact areas is required for myotube formation. J. Cell Sci. 114, 3631–3642 (2001)
Williamson, P. et al. Transbilayer phospholipid movements in ABCA1-deficient cells. PLoS ONE 2, e729 (2007)
Morita, S., Kojima, T. & Kitamura, T. Plat-E: an efficient and stable system for transient packaging of retroviruses. Gene Ther. 7, 1063–1066 (2000)
Fukunaga, R., Ishizaka-Ikeda, E. & Nagata, S. Purification and characterization of the receptor for murine granulocyte colony-stimulating factor. J. Biol. Chem. 265, 14008–14015 (1990)
Aoki, Y., Uenaka, T., Aoki, J., Umeda, M. & Inoue, K. A novel peptide probe for studying the transbilayer movement of phosphatidylethanolamine. J. Biochem. 116, 291–297 (1994)
Hanayama, R. et al. Identification of a factor that links apoptotic cells to phagocytes. Nature 417, 182–187 (2002)
Kitamura, T. et al. Retrovirus-mediated gene transfer and expression cloning: powerful tools in functional genomics. Exp. Hematol. 31, 1007–1014 (2003)
Bernhagen, J. et al. MIF is a noncognate ligand of CXC chemokine receptors in inflammatory and atherogenic cell recruitment. Nature Med. 13, 587–596 (2007)
Acknowledgements
We thank T. Wiedmer for critical reading of our manuscript, and M. Fujii and M. Harayama for secretarial assistance. This work was supported in part by Grants-in-Aid for Specially Promoted Research (to S.N.) and for Young Scientists Start-up (to J.S.) from the Japan Society for the Promotion of Science. J.S. is supported by a research fellowship from the Japan Society for the Promotion of Science. P.J.S. was supported by grants from the National Institutes of Health, USA.
Author information
Authors and Affiliations
Contributions
J.S. designed and performed the experiments, and wrote the manuscript. M.U. provided biotin-labelled Ro09-0198 peptide. P.J.S. provided EBV-transformed cell lines from a patient and the patient’s parents, and commented on the manuscript. S.N. was responsible for the overall study design and for writing the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
This file contains Supplementary Figures 1-7 with legends. (PDF 1215 kb)
Rights and permissions
About this article
Cite this article
Suzuki, J., Umeda, M., Sims, P. et al. Calcium-dependent phospholipid scrambling by TMEM16F. Nature 468, 834–838 (2010). https://doi.org/10.1038/nature09583
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature09583
This article is cited by
-
Structural heterogeneity of the ion and lipid channel TMEM16F
Nature Communications (2024)
-
Plasma membrane damage limits replicative lifespan in yeast and induces premature senescence in human fibroblasts
Nature Aging (2024)
-
Context-specific regulation of extracellular vesicle biogenesis and cargo selection
Nature Reviews Molecular Cell Biology (2023)
-
Extracellular calcium functions as a molecular glue for transmembrane helices to activate the scramblase Xkr4
Nature Communications (2023)
-
The Effect of Extracellular Vesicles on Thrombosis
Journal of Cardiovascular Translational Research (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.