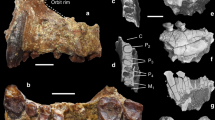
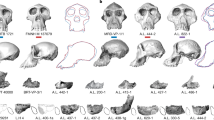
Abstract
Adapiform or ‘adapoid’ primates first appear in the fossil record in the earliest Eocene epoch (∼55 million years (Myr) ago), and were common components of Palaeogene primate communities in Europe, Asia and North America1. Adapiforms are commonly referred to as the ‘lemur-like’ primates of the Eocene epoch, and recent phylogenetic analyses have placed adapiforms as stem members of Strepsirrhini2,3,4, a primate suborder whose crown clade includes lemurs, lorises and galagos. An alternative view is that adapiforms are stem anthropoids5. This debate has recently been rekindled by the description of a largely complete skeleton of the adapiform Darwinius6, from the middle Eocene of Europe, which has been widely publicised as an important ‘link’ in the early evolution of Anthropoidea7. Here we describe the complete dentition and jaw of a large-bodied adapiform (Afradapis gen. nov.) from the earliest late Eocene of Egypt (∼37 Myr ago) that exhibits a striking series of derived dental and gnathic features that also occur in younger anthropoid primates—notably the earliest catarrhine ancestors of Old World monkeys and apes. Phylogenetic analysis of 360 morphological features scored across 117 living and extinct primates (including all candidate stem anthropoids) does not place adapiforms as haplorhines (that is, members of a Tarsius–Anthropoidea clade) or as stem anthropoids, but rather as sister taxa of crown Strepsirrhini; Afradapis and Darwinius are placed in a geographically widespread clade of caenopithecine adapiforms that left no known descendants. The specialized morphological features that these adapiforms share with anthropoids are therefore most parsimoniously interpreted as evolutionary convergences. As the largest non-anthropoid primate ever documented in Afro-Arabia, Afradapis nevertheless provides surprising new evidence for prosimian diversity in the Eocene of Africa, and raises the possibility that ecological competition between adapiforms and higher primates might have played an important role during the early evolution of stem and crown Anthropoidea in Afro-Arabia.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Godinot, M. A summary of adapiform systematics and phylogeny. Folia Primatol. (Basel) 69 (suppl. 1). 218–249 (1998)
Marivaux, L. et al. Anthropoid primates from the Oligocene of Pakistan (Bugti Hills): data on early anthropoid evolution and biogeography. Proc. Natl Acad. Sci. USA 102, 8436–8441 (2005)
Ross, C., Williams, B. & Kay, R. F. Phylogenetic analysis of anthropoid relationships. J. Hum. Evol. 35, 221–306 (1998)
Seiffert, E. R. et al. Basal anthropoids from Egypt and the antiquity of Africa’s higher primate radiation. Science 310, 300–304 (2005)
Rasmussen, D. T. in Anthropoid Origins (eds Fleagle, J. G. & Kay, R. F.) 335–360 (Plenum, 1994)
Franzen, J. L. et al. Complete primate skeleton from the middle Eocene of Messel in Germany: morphology and paleobiology. PLoS ONE 4, e5723 (2009)
Tudge, C. The Link: Uncovering Our Earliest Ancestor (Little, Brown & Company, 2009)
Seiffert, E. R., Simons, E. L. & Attia, Y. Fossil evidence for an ancient divergence of lorises and galagos. Nature 422, 421–424 (2003)
Seiffert, E. R. Revised age estimates for the later Paleogene mammal faunas of Egypt and Oman. Proc. Natl Acad. Sci. USA 103, 5000–5005 (2006)
Gingerich, P. D. Marine mammals (Cetacea and Sirenia) from the Eocene of Gebel Mokattam and Fayum, Egypt: stratigraphy, age, and paleoenvironments. Univ. Mich. Pap. Paleont. 30, 1–84 (1992)
Seiffert, E. R., Bown, T. M., Clyde, W. C. & Simons, E. L. in Elwyn L. Simons: A Search for Origins (eds Fleagle, J. G. & Gilbert, C. C.) 71–86 (Springer, 2008)
Conroy, G. C. Problems of body-weight estimation in fossil primates. Int. J. Primatol. 8, 115–137 (1987)
Kay, R. F. & Simons, E. L. The ecology of Oligocene African Anthropoidea. Int. J. Primatol. 1, 21–37 (1980)
Simons, E. L. Discovery of the oldest known anthropoidean skull from the Paleogene of Egypt. Science 247, 1567–1569 (1990)
Simons, E. L., Rasmussen, D. T. & Gingerich, P. D. New cercamoniine adapid from Fayum, Egypt. J. Hum. Evol. 29, 577–589 (1995)
Rasmussen, D. T. The phylogenetic position of Mahgarita stevensi: protoanthropoid or lemuroid? Int. J. Primatol. 11, 439–469 (1990)
Wilson, J. A. & Szalay, F. S. New adapid primate of European affinities from Texas. Folia Primatol. (Basel) 25, 294–312 (1976)
Stehlin, H. G. Die Säugetiere des schweizerischen Eocaens. Abh. Schweiz. Paläont. Ges. 41, 1299–1552 (1916)
Ross, C. F. in Anthropoid Origins (eds Fleagle, J. G. & Kay, R. F.) 469–548 (Plenum, 1994)
Simons, E. L. & Rasmussen, D. T. Skull of Catopithecus browni, an early Tertiary catarrhine. Am. J. Phys. Anthropol. 100, 261–292 (1996)
Miller, E. R. & Simons, E. L. Dentition of Proteopithecus sylviae, an archaic anthropoid from the Fayum, Egypt. Proc. Natl Acad. Sci. USA 94, 13760–13764 (1997)
Seiffert, E. R., Simons, E. L., Ryan, T. M. & Attia, Y. Additional remains of Wadilemur elegans, a primitive stem galagid from the late Eocene of Egypt. Proc. Natl Acad. Sci. USA 102, 11396–11401 (2005)
Swofford, D. L. PAUP* Phylogenetic Analysis Using Parsimony (*and Other Methods), Version 4 (Sinauer Associates, 1998)
Rose, K. D. et al. Early Eocene Primates from Gujarat, India. J. Hum. Evol. 56, 366–404 (2009)
Gheerbrant, E. & Rage, J. C. Paleobiogeography of Africa: How distinct from Gondwana and Laurasia? Palaeogeogr. Palaeoclimatol. Palaeoecol. 241, 224–246 (2006)
Tabuce, R. & Marivaux, L. Mammalian interchanges between Africa and Eurasia: an analysis of temporal constraints on plausible anthropoid dispersals during the Paleogene. Anthropol. Sci. 113, 27–32 (2005)
Chaimanee, Y., Suteethorn, V., Jaeger, J.-J. & Ducrocq, S. A new late Eocene anthropoid primate from Thailand. Nature 385, 429–431 (1997)
Jaeger, J.-J. et al. A new primate from the Middle Eocene of Myanmar and the Asian early origin of anthropoids. Science 286, 528–530 (1999)
Beard, K. C., Qi, T., Dawson, M. R., Wang, B. & Li, C. A diverse new primate fauna from middle Eocene fissure-fillings in southeastern China. Nature 368, 604–609 (1994)
Roos, C., Schmitz, J. & Zischler, H. Primate jumping genes elucidate strepsirrhine phylogeny. Proc. Natl Acad. Sci. USA 101, 10650–10654 (2004)
Dagosto, M., Gebo, D. L. & Beard, K. C. Revision of the Wind River faunas, early Eocene of central Wyoming. Part 14. Postcranium of Shoshonius cooperi (Mammalia, Primates). Ann. Carnegie Mus. 68, 175–211 (1999)
Gebo, D. L., Dagosto, M., Beard, K. C. & Qi, T. Middle Eocene primate tarsals from China: implications for haplorhine evolution. Am. J. Phys. Anthropol. 116, 83–107 (2001)
Kay, R. F., Williams, B. A., Ross, C. F., Takai, M. & Shigehara, N. in Anthropoid Origins: New Visions (eds Ross, C. F. & Kay, R. F.) 91–135 (Kluwer, 2004)
Masters, J. C. & Brothers, D. J. Lack of congruence between morphological and molecular data in reconstructing the phylogeny of the Galagonidae. Am. J. Phys. Anthropol. 117, 79–93 (2002)
Yoder, A. D. Relative position of the Cheirogaleidae in strepsirhine phylogeny: A comparison of morphological and molecular results and methods. Am. J. Phys. Anthropol. 94, 25–46 (1994)
Yoder, A. D., Irwin, J. A. & Payseur, B. A. Failure of the ILD to determine data combinability for slow loris phylogeny. Syst. Biol. 50, 408–424 (2001)
Franzen, J. L. Der sechste Messel-primate (Mammalia, Primates, Notharctidae, Cercamoniinae). Senckenbergiana Lethaea 80, 289–303 (2000)
Savage, R. J. G., Domning, D. P. & Thewissen, J. G. M. Fossil Sirenia of the west Atlantic and Caribbean region. V. The most primitive known sirenian, Prorastomus sirenoides Owen, 1855. J. Vert. Paleo. 14, 427–449 (1994)
Thewissen, J. G. M. & Williams, E. M. The early radiations of Cetacea (Mammalia): Evolutionary pattern and developmental correlations. Annu. Rev. Ecol. Syst. 33, 73–90 (2002)
Uhen, M. D. A new Xenophorus-like odontocete cetacean from the Oligocene of North Carolina and a discussion of the basal odontocete radiation. J. Syst. Palaeontology 6, 433–452 (2008)
Springer, M. S., Teeling, E. C., Madsen, O., Stanhope, M. J. & de Jong, W. W. Integrated fossil and molecular data reconstruct bat echolocation. Proc. Natl Acad. Sci. USA 98, 6241–6246 (2001)
Ciochon, R. L., Gingerich, P. D., Gunnell, G. F. & Simons, E. L. Primate postcrania from the late middle Eocene of Myanmar. Proc. Natl Acad. Sci. USA 98, 7672–7677 (2001)
Beard, K. C. et al. New sivaladapid primates from the Eocene Pondaung Formation of Myanmar and the anthropoid status of Amphipithecidae. Bull. Carnegie Mus. Nat. Hist. 39, 67–76 (2007)
Gunnell, G. F., Ciochon, R. L., Gingerich, P. D. & Holroyd, P. A. New assessment of Pondaungia and Amphipithecus (Primates) from the late middle Eocene of Myanmar, with a comment on “Amphipithecidae”. Contr. Mus. Paleont. Univ. Mich. 30, 337–372 (2002)
Beard, K. C. et al. Taxonomic status of purported primate frontal bones from the Eocene Pondaung Formation of Myanmar. J. Hum. Evol. 49, 468–481 (2005)
Ciochon, R. L. & Gunnell, G. F. Eocene primates from Myanmar: Historical perspectives on the origin of Anthropoidea. Evol. Anthropol. 11, 156–168 (2002)
MacPhee, R. D. E., Beard, K. C. & Qi, T. Significance of primate petrosal from middle Eocene fissure-fillings at Shanghuang, Jiangsu Province, People's Republic of China. J. Hum. Evol. 29, 501–514 (1995)
Ross, C. F. & Covert, H. H. The petrosal of Omomys carteri and the evolution of the primate basicranium. J. Hum. Evol. 39, 225–251 (2000)
Acknowledgements
We thank K. C. Beard, P. Chatrath, B. Engesser, M. Hellmund, J. Galkin, P. D. Gingerich, M. Godinot, G. Gunnell, E. Ladier, B. Marandat, L. Marivaux, K. D. Rose, I. Rutzky and P. Tassy for access to adapiform fossils and casts. F. Ankel-Simons and J. Fleagle provided comments on the manuscript. Collaborative palaeontological research in Egypt is made possible through cooperation with the Egyptian Mineral Resources Authority and the Egyptian Geological Museum. Fieldwork in Egypt is managed by P. Chatrath. This research has been funded by the Research Foundation of SUNY, and grants from the US National Science Foundation and The Leakey Foundation to E.R.S. and E.L.S. This is Duke Lemur Center publication 1155.
Author Contributions E.R.S. and E.L.S. recovered the fossils of Afradapis along with other members of their field crews in Egypt; E.R.S. assembled the character matrix and all figures, created Figs 2 and 3, ran the phylogenetic analyses, and compiled the Supplementary Information document; E.R.S. and J.M.G.P. analysed the fossils and wrote the manuscript; D.M.B. and J.M.G.P. CT-scanned fossils and provided three-dimensional reconstructions for Fig. 1. D.M.B. scored plesiadapiforms in the character matrix and helped to write the manuscript. E.L.S. helped to write the manuscript.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains dental measurements of Afradapis longicristatus gen. et sp. nov., full results of phylogenetic analyses, morphological character support for nodes within Strepsirrhini, the morphological character matrix, and the constraint tree used in the phylogenetic analysis. (PDF 1635 kb)
Rights and permissions
About this article
Cite this article
Seiffert, E., Perry, J., Simons, E. et al. Convergent evolution of anthropoid-like adaptations in Eocene adapiform primates. Nature 461, 1118–1121 (2009). https://doi.org/10.1038/nature08429
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature08429
This article is cited by
-
Mineralogy and geochemistry of elongated calcite-cemented concretions of the Upper Eocene Birket Qarun Formation (Fayum depression, Egypt): insights into concretions’ origin and paleoenvironmental implications
Euro-Mediterranean Journal for Environmental Integration (2024)
-
Modules and Mosaics in the Evolution of the Tetonius – Pseudotetonius Dentition
Journal of Mammalian Evolution (2020)
-
New Eocene primate from Myanmar shares dental characters with African Eocene crown anthropoids
Nature Communications (2019)
-
A new primate from the late Eocene of Vietnam illuminates unexpected strepsirrhine diversity and evolution in Southeast Asia
Scientific Reports (2019)
-
Comment to “Primates in the Eocene” by Gingerich (2012)
Palaeobiodiversity and Palaeoenvironments (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.