Abstract
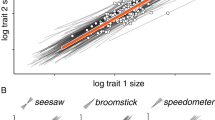
As perceived by Darwin, evolutionary adaptation by the processes of mutation and selection is difficult to understand for complex features that are the product of numerous traits acting in concert, for example the eye or the apparatus of flight. Typically, mutations simultaneously affect multiple phenotypic characters. This phenomenon is known as pleiotropy. The impact of pleiotropy on evolution has for decades been the subject of formal analysis1,2,3,4,5,6. Some authors have suggested that pleiotropy can impede evolutionary progress (a so-called ‘cost of complexity’5). The plausibility of various phenomena attributed to pleiotropy depends on how many traits are affected by each mutation and on our understanding of the correlation between the number of traits affected by each gene substitution and the size of mutational effects on individual traits. Here we show, by studying pleiotropy in mice with the use of quantitative trait loci (QTLs) affecting skeletal characters, that most QTLs affect a relatively small subset of traits and that a substitution at a QTL has an effect on each trait that increases with the total number of traits affected. This suggests that evolution of higher organisms does not suffer a ‘cost of complexity’ because most mutations affect few traits and the size of the effects does not decrease with pleiotropy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Fisher, R. A. The Genetical Theory of Natural Selection (Clarendon, Oxford, 1930)
Rechenberg, I. Evolutionsstrategie: Optimierung technischer Systeme nach Prinzipien der biologischen Evolution (Fromman-Holzboog, Stuttgart, 1973)
Turelli, M. Effects of pleiotropy on predictions concerning mutation-selection balance for polygenic traits. Genetics 111, 165–195 (1985)
Wagner, G. P. The influence of variation and developmental constraints on the rate of multivariate phenotypic evolution. J. Evol. Biol. 1, 45–66 (1988)
Orr, H. A. Adaptation and the cost of complexity. Evolution Int. J. Org. Evolution 54, 13–20 (2000)
Wingreen, N. S., Miller, J. & Cox, E. C. Scaling of mutational effects in models of pleiotropy. Genetics 164, 1221–1228 (2003)
Chai, C. K. Analysis of quantitative inheritance of body size in mice II: gene action and segregation. Genetics 41, 165–178 (1956)
Eppig, J. T., Bult, C. J., Kadin, J. A., Richardson, J. E. & Blake, J. A. and the members of the Mouse Genome Database Group. The mouse genome data base (MGD): from genes to mice – a community resource for mouse biology. Nucleic Acids Res. 33, D471–D475 (2005)
Kenney-Hunt, J. P. et al. Pleiotropic patterns of quantitative trait loci for seventy murine skeletal traits. Genetics (in the press)
Knott, S. A. & Haley, C. S. Multitrait least squares for quantitative trait loci detection. Genetics 156, 899–911 (2000)
Xu, S. Theoretical basis of the Beavis effect. Genetics 165, 2259–2268 (2003)
Wagner, G. P. Multivariate mutation-selection balance with constrained pleiotropic effects. Genetics 122, 223–234 (1989)
Waxman, D. & Peck, J. R. Pleiotropy and preservation of perfection. Science 279, 1210–1213 (1998)
Waddington, C. H. The Strategy of Genes (Macmillan, New York, 1957)
Hermisson, J. & Wagner, G. P. The population genetic theory of hidden variation and genetic robustness. Genetics 168, 2271–2284 (2004)
Wagner, G. P. & Altenberg, L. Complex adaptation and the evolution of evolvability. Evolution Int. J. Org. Evolution 50, 967–976 (1996)
Hansen, T. F. Is modularity necessary for evolvability? Remarks on the relationship between pleiotropy and evolvability. Biosystems 69, 83–94 (2003)
Wagner, G. P., Pavlicev, M. & Cheverud, J. M. The road to modularity. Nature Rev. Genet. 8, 921–931 (2007)
Welch, J. J. & Waxman, D. Modularity and the cost of complexity. Evolution Int. J. Org. Evolution 57, 1723–1734 (2003)
Martin, G. & Lenormand, T. A general multivariate extension of Fisher’s geometrical model and the distribution of mutation fitness effects across species. Evolution Int. J. Org. Evolution 60, 893–907 (2006)
Cheverud, J. M. et al. Quantitative trait loci for murine growth. Genetics 142, 1305–1319 (1996)
Vaughn, T. T. et al. Mapping quantitative trait loci for murine growth: a closer look at genetic architecture. Genet. Res. 74, 313–322 (1999)
Cheverud, J. M. et al. Genetic architecture of adiposity in the cross of LG/J and SM/J inbred mice. Mamm. Genome 12, 3–12 (2001)
Haley, C. S. & Knott, S. A. A simple regression method for mapping quantitative trait loci in line crosses using flanking markers. Heredity 69, 315–324 (1992)
Cheverud, J. M. A simple correction for multiple comparisons in interval mapping genome scans. Heredity 87, 52–58 (2001)
Ehrich, T. H. et al. Pleiotropic effects on mandibular morphology I: developmental morphological integration and differential dominance. J. Exp. Zool. B 296, 58–78 (2003)
Acknowledgements
We thank A. Pyle, A. Kondrashov and B. Walsh for suggestions that have improved this manuscript. We thank the members of the Wagner and Cheverud laboratories for critical discussion. J.P. and D.W. thank members of the evolution group at Sussex. J.M.C. is funded by the National Institutes of Health and the National Science Foundation (NSF), G.P.W. acknowledges funding from the NSF, the Humboldt Foundation and the John Templeton Foundation, M.P. is funded by the Austrian Science Foundation (FWF) Fellowship, and the work of D.W. was supported by the Leverhulme Trust.
Author Contributions G.P.W. conceived this study, participated in the statistical analysis and wrote the manuscript. J.P.K.-H. collected the morphological data and performed the QTL analysis. M.P. did the statistical analyses. J.M.C. was responsible for generating the mouse populations and the genotype data used in the original mapping and advised on the pleiotropic scaling analysis. J.P. and D.W. performed a theoretical analysis of the scaling of trait effects with pleiotropy. All authors participated in the preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary Information
The file contains Supplementary Figures S1-S4 with Legends, Supplementary Table T1 and Supplementary Notes with additional references. (PDF 356 kb)
Rights and permissions
About this article
Cite this article
Wagner, G., Kenney-Hunt, J., Pavlicev, M. et al. Pleiotropic scaling of gene effects and the ‘cost of complexity’. Nature 452, 470–472 (2008). https://doi.org/10.1038/nature06756
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature06756
This article is cited by
-
Genomewide architecture of adaptation in experimentally evolved Drosophila characterized by widespread pleiotropy
Journal of Genetics (2024)
-
Natural variation in Drosophila shows weak pleiotropic effects
Genome Biology (2022)
-
Pleiotropy data resource as a primer for investigating co-morbidities/multi-morbidities and their role in disease
Mammalian Genome (2022)
-
Solving the grand challenge of phenotypic integration: allometry across scales
Genetica (2022)
-
Favorable pleiotropic loci for fiber yield and quality in upland cotton (Gossypium hirsutum)
Scientific Reports (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.