Abstract
In prostate cancer genomic rearrangements involving genes encoding ETS transcription factors are commonly present, with androgen-regulated transmembrane protease, serine 2 (TMPRSS2)-v-ets erythroblastosis virus E26 oncogen homologue (ERG) gene fusion occurring in 40–70%. Studies on the predictive value of ERG rearrangement as detected by in-situ hybridization or polymerase chain reaction have resulted in varying outcomes. The objective of this study was to correlate immunohistochemical ERG protein expression with clinico-pathological parameters at radical prostatectomy specimens, and to determine its predictive value for postoperative disease recurrence and progression in a prostate cancer screening cohort. Since androgen receptor is downregulated by ERG in cell lines, we also compared the expression of respective proteins. We selected 481 participants from the European Randomized Study of Screening for Prostate Cancer treated by radical prostatectomy for prostate adenocarcinoma. A tissue microarray was constructed containing representative cores of all prostate cancer specimens as well as 22 xenografts and seven cell lines. Immunohistochemical expression of ERG and androgen receptor was correlated with prostate-specific antigen (PSA), Gleason sum, pT-stage, surgical margins, biochemical recurrence, local recurrence, overall death and disease-specific death. ERG expression was detected in 284 patients (65%). Expression occurred significantly more frequent in patients with PSA ≤10 ng/ml (P=0.024). There was no significant association between ERG and Gleason sum, pT-stage or surgical margin status. PSA (P=0.011), Gleason sum (P=0.003), pT-stage (P=0.001) and surgical margin status (P<0.001) all had independent value for postoperative biochemical recurrence, while positive surgical margin (P=0.021) was the only independent predictor for local recurrence. ERG protein expression did not have prognostic value for the clinical end points in uni- and multivariate analyses. A positive correlation existed between ERG and androgen receptor expression in single tissue cores (P<0.001). In conclusion, immunohistochemical ERG expression has no predictive value for prostate cancer recurrence or progression after radical prostatectomy. Increasing ERG levels are associated with the upregulation of androgen receptor expression in clinical specimens.
Similar content being viewed by others
Main
Genomic rearrangement of androgen-regulated transmembrane protease, serine 2 (TMPRSS2) on 21q22.3 to v-ets erythroblastosis virus E26 oncogen homologue (ERG) on 21q22.2 is the most common genetic alteration in prostate cancer occurring in 40–70% of tumors.1 Less frequently, SLC45A3 and NDRG1 can serve as ERG fusion partners, whereas fusions of other ETS family members ETV1, ETV4 and ETV5 to over 10 different partner genes are found in approximately 10% of tumors.2, 3, 4
Various groups have investigated the prognostic relevance of TMPRSS2-ERG fusion in clinical prostate cancer with varying outcome. This discordance can be explained by diversity in patient cohorts, pathological specimens, clinical end points and ERG fusion detection. ERG rearrangement has so far been analyzed using either fluorescence in-situ hybridization (FISH) or quantitative polymerase chain reaction (QPCR). Advantageously to routine detection of ERG fusion, FISH can give qualitative information on the type of gene fusion, resulting either from translocation or deletion, which is reported to be of clinical relevance.5, 6, 7 Application of FISH, however, needs tissue-dependent optimization and can suffer from difficulty in interpretation, especially when only a few atypical glands are present. On the other hand, both quantitative and qualitative information on gene fusion can be obtained by QPCR. Hermans et al8 demonstrated that TMPRSS2 (exon 0)-ERG fusions were associated with less aggressive biological prostate cancer behavior. The disadvantage of QPCR for the detection of ERG rearrangement, however, is its requirement of frozen tissue containing a high percentage of prostate cancer cells.
Recently, Park et al9 described an ERG-specific antibody EPR3864, which reactivity showed excellent correlation with ERG rearrangement as determined by FISH in prostate cancer paraffin-embedded tissues. Likewise, van Leenders et al10 found a strong correlation between ERG protein expression and TMPRSS2-ERG expression using QPCR. Strong immunohistochemical staining was associated with high ERG transcript levels in radical prostatectomy specimens, indicating that semiquantitative ERG determination reflected molecular expression levels.10 Therefore, ERG immunohistochemistry is a simple methodology strongly indicative for the presence of ERG rearrangement.
The androgen receptor pathway plays an important role in the development and maintenance of the normal prostate, as well as in prostate carcinogenesis.11, 12 Because many ETS fusion partners are androgen-regulated, ERG expression might be related to androgen receptor levels in prostate cancer.11, 12 On the other hand, it has been shown that ERG can downregulate the expression of androgen receptor and its target genes.13
In this study, we investigated whether ERG protein expression has predictive value for prostate cancer recurrence and progression after radical prostatectomy in a well-defined screening cohort. In addition, we analyzed the relation between androgen receptor and ERG expression in clinical specimens.
Materials and methods
Patient Information
We selected all men from the European Randomized Study of Screening for Prostate Cancer (ERSPC), Rotterdam section, who had undergone radical prostatectomy for prostate adenocarcinoma in Erasmus Medical Center between 1987 and 2010.14, 15 In this study, men aged between 55 and 74 years were invited for a screening visit every 4 years. Recruitment and randomization started in December 1993 and ended December 1999. Up until May 1997, patients with a serum prostate-specific antigen (PSA) level of ≥4.0 ng/ml, an abnormal digital rectal examination and/or abnormal transrectal ultrasound underwent lateralized sextant prostate needle-biopsies. As from May 1997, a biopsy was indicated by a PSA level of ≥3.0 ng/ml or abnormal digital rectal examination and/or transrectal ultrasound.
Directly after surgery, radical prostatectomy specimens were transported on ice to the pathology department. After fixation in neutral-buffered formaldehyde, the radical prostatectomy specimens were routinely cut in 4-mm transverse slices with additional perpendicular slicing of the apex and basis to allow optimal evaluation of surgical margins, and totally embedded in paraffin. Hematoxylin/eosin (HE) slides were microscopically evaluated by two board-certified pathologists with expertise in urological pathology (TvdK, GvL). At pathological examination, tumor areas were encircled at the glass slides, and Gleason sum, TNM stage and surgical margins were reported according to the international guidelines.
Clinical follow-up was recorded after each control at our outpatient clinic, and data were transmitted to the central study database. Postoperative biochemical recurrence was defined as an increase in serum PSA after two different measurements, at least 3 months apart. Suspicion on local recurrence was verified by a diagnostic needle-biopsy in each case. Death and death of disease was registered by the physician who last treated the patient. Use of samples for research purposes was approved by the Erasmus Medical Center Medical Ethics Committee according to the Medical Research Involving Human Subjects Act (MEC-2004-261).
Tissue Microarray Construction
Histological slides of all patients (n=509) were retrieved from the pathology archives together with corresponding paraffin blocks containing the largest tumor volume (with tumor of at least 0.5 cm in diameter per paraffin block). In 28 cases, the tumor diameter was less than 0.5 cm, which hampered sampling of three separate cores, or no paraffin tissue was available, resulting in 481 patients to be included. In addition, the following control specimens were selected: normal prostate tissues (n=10) from cystoprostatectomies, urothelial cell carcinomas (n=5), invasive ductal mammary adenocarcinomas (n=5), palliative transurethral resection of the prostate containing hormone-refractory prostate cancer (n=10), prostate cancer lymph node metastases (n=10) and placenta (n=1). In addition, we included seven relevant human prostate cancer cell lines and 22 human prostate cancer xenografts with known ERG genomic fusion status (Table 1).16, 17, 18 Cell lines were maintained in appropriate culture media. To facilitate inclusion of cell lines, confluent cell cultures were detached using EDTA, fixed in neutral-buffered formaldehyde, embedded in AGAR 2.5% (Sigma-Aldrich, St Louis, MO, USA), and subsequently in paraffin.
For tissue microarray (TMA) construction, three cylindrical cores (diameter 0.6 mm) were taken from representative areas in the paraffin block and transferred to recipient paraffin blocks (Beecher Microarrayer; Beecher Instruments, Sun Prairie, WI, USA). In total, nine TMA blocks were constructed each including 200 tissue and/or cell line cores.
Immunohistochemistry
Tissue slides (5 μm) were mounted on aminoacetylsilane-coated glass slides (Starfrost, Berlin, Germany), deparaffinized in xylene and dehydrated in ethanol. Endogenous peroxidase activity was blocked by 1% hydrogen peroxide in methanol for 20 min. Microwave (700 W) pretreatment in tris(hydroxymethyl)aminomethane-EDTA (pH 9.0) was performed for 15 min. The slides were incubated with rabbit monoclonal ERG antibody (clone EPR3864; 1:100; Epitomics, Burlingame, CA, USA) and mouse monoclonal anti-androgen receptor (clone F39.4; 1:200; Avris Antibodies, Herford, Germany) overnight at 4°C, followed by chromogenic visualization using the EnVision DAKO kit (Dako, Glostrup, Denmark). After counterstaining with hematoxylin, slides were thoroughly washed, dehydrated, cleared in xylene and mounted in malinol (Chroma-Geselschaft, Körgen, Germany).
Immunohistochemical stainings for ERG and androgen receptor were visually examined as described previously.10 The intensity of ERG and androgen receptor expression was scored as negative (0; no staining), weak (1+; only visible at high magnification), moderate (2+; visible at low magnification) and strong (3+; striking at low magnification). Nuclear reactivity of the antibody in endothelial cells (ERG) and in stromal cells (androgen receptor) was used as internal control for the staining procedure.9 In case of staining heterogeneity, the highest level was used for statistical analysis. All cores were scored by two investigators (MH, GvL) in a blinded setting. In a combined session, consensus on expression value was reached in all cases.
Statistics
For statistical analyses, we defined a patient as ERG ‘positive’ if respective evaluable cores all demonstrated ERG protein expression. If all cores from a patient were negative, we labeled the case as ‘negative’. In case expression heterogeneity was present with both ERG-positive and -negative cancer cores taken from one radical prostatectomy, respective case was labeled ‘heterogeneous’. The heterogeneous cases were included in the group of ‘positive’ cases as well as studied as a separate group.
Statistical associations between expression of ERG (as categorical variable) and continuous clinico-pathological parameters (age and PSA at the time of diagnosis) were tested using Student’s t-test, and with categorical parameters (Gleason sum, pT-stage, surgical margins) using Pearson’s χ2 test. To determine whether ERG expression was predictive for biochemical recurrence, clinical recurrence, overall death or disease-specific death, we used uni- and multivariate Cox regression with stepwise backward entering of covariates. The proportionality assumption for ERG positive versus negative cases was visually assessed in Kaplan–Meier curves (not shown). To investigate the statistical association between ERG and androgen receptor expression in the same core, we used Pearson’s χ2 test. A P-value <0.05 was considered significant. All statistics were performed using SPSS 17 (SPSS, Chicago, IL, USA).
Results
Patient Characteristics
The mean age of the prostatectomy patients was 64.76 years (range 55.4–75.1 years). The Gleason sum was <7 in 265 (55%), 7 in 188 (39%) and >7 in 28 (6%) cases, respectively. In total 343 (71%) tumors were organ-confined (pT2) and 138 (29%) cases showed extra-prostatic expansion (pT3/4). Surgical margins were positive in 119 (25%) cases. The mean follow-up of our cohort was 107.3 months. The clinico-pathological characteristics are summarized in Table 2.
ERG Expression
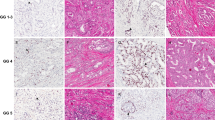
In 44 prostate cancer patients, no tumor was identified in any of the tissue cores. Immunohistochemical analysis of the remaining prostate cancer patients (n=437) demonstrated nuclear ERG expression in 239 cases (55%) and negative staining in 153 cases (35%). In 45 cases (10%), heterogeneous expression was observed, meaning that both areas with and without ERG expression were present within the same tumor. In all cases, endothelial cells were positive (1%, 2+; 99%, 3+), indicating that the immunohistochemical procedure was efficient and reliable. Different ERG and corresponding androgen receptor expression patterns are depicted in Figure 1.
Immunohistochemical expression of ERG (left column) and androgen receptor (AR) (right column) in corresponding prostate cancer specimens. (a) ERG intensity 3+; (b) AR intensity 2+; (c) ERG intensity 1+; (d) AR intensity 2+; (e) ERG negative; and (f) AR intensity 2+. In panels a, c and e, endothelial cells with intensity 3+ serve as internal controls. Original magnification × 400.
In 49 patients (10%), high-grade prostate intraepithelial neoplasia was present in the tissue cores, of which 18 cases (37%) demonstrated ERG expression, 29 (59%) were negative and two (4%) heterogeneous. Normal prostate epithelial glands were negative in all cases, except in one (0%, intensity 2+).
Six of nine (67%) evaluable prostate cancer lymph node metastases showed ERG expression, while three were negative (33%); for one patient, no tumor was present in the three cores. In castration-resistant prostate cancer treated by palliative transurethral resection, ERG was uniformly expressed in four patients (40%), with heterogeneous expression in three (30%) and negative staining in three patients (30%).
To validate ERG immunohistochemistry, we also included a large series of human prostate cancer cell lines and xenografts with known ERG fusion status in the TMA. Nuclear ERG protein expression was found in VCaP, which has a known TMPRSS2-ERG fusion,16 while other cell lines without ERG fusion were negative for ERG staining (Table 1). From the 22 xenografts analyzed, VCaP, DuCaP, PC82, PC295 and PC310 showed uniform ERG expression, while the other 17 were negative. As shown in Table 1, ERG immunohistochemistry was concordant with TMPRSS2-ERG fusion status. The control tissues derived from urothelial cell carcinoma, breast adenocarcinoma and placenta were all negative for ERG.
Clinico-Pathological Correlations
The relation of ERG immunohistochemical expression and clinico-pathological parameters is depicted in Table 3. Expression of ERG occurred significantly more frequent in patients with PSA ≤10 ng/ml (P=0.024). There was no statistically significant relation between ERG immunohistochemistry and Gleason sum, pT-stage or surgical margin status. PSA at the time of diagnosis (P=0.011), Gleason sum (P=0.003), pT-stage (P=0.001) and surgical margin status (P<0.001) all had independent predictive value for postoperative biochemical recurrence in multivariate analysis (Table 4), while positive surgical margin (P=0.021) was the only independent predictor for local recurrence (Table 5). ERG protein expression did neither have a prognostic value for biochemical nor local recurrence after radical prostatectomy in uni- and multivariate analysis. While age (P<0.001) and PSA (P=0.001) both were independently predictive for overall death, pathological parameters and ERG immunohistochemistry were not (Table 6). Serum PSA (P=0.022) was the only independent prognostic factor for disease-specific death, with Gleason sum (P=0.052) and surgical margin status (P=0.070) approaching significance (data not shown). The low number of events (n=9), however, limits the power of this analysis. Since multiple end points were tested, P-values should be interpreted cautiously. If patients with heterogeneous expression (n=45) were analyzed as a separate group, we neither were able to find a significant correlation between ERG and follow-up parameters (data not shown).
Androgen Receptor Expression
To compare androgen receptor and ERG expression, we investigated both proteins in the same tissue cores. Of the 969 evaluable cores, androgen receptor expression was weak (1+) in 96 (10%), moderate (2+) in 861 (89%) and strong (3+) in 78 (8%) cases. As depicted in Figure 2, a positive correlation existed between the level of nuclear androgen receptor and ERG, with increased androgen receptor expression at higher ERG levels (P<0.001). The androgen receptor expression level of the ERG-negative cases was not statistically different from cases with moderate (2+) ERG expression (P=0.32).
Discussion
Since its discovery in 2005, several groups have investigated the clinical significance of TMPRSS2-ERG fusion for prostate cancer behavior with variable outcome.8, 19, 20, 21, 22, 23 This variability might be due to differences in study cohorts, clinical end points and methodologies for detecting gene fusion. For example, RT-PCR-detected TMPRSS2-ERG fusion was found in more than 70% of patients surgically treated by radical prostatectomy for prostate cancer.24 In other studies, TMPRSS2-ERG fusion detected by FISH was present in only 15–30% of prostate cancer diagnosed at transurethral resection for benign prostate hyperplasia.6, 25 While prostate cancer at diagnostic prostate needle-biopsies showed TMPRSS2-ERG fusion in 46% by FISH,26 our group recently found nuclear ERG overexpression in 61% using immunohistochemistry.10
The description of a novel ERG antibody in 2010 highly facilitates detection of ERG genomic fusions in clinical specimens.9 Nuclear staining by the EPR3864 antibody showed excellent concordance with the presence of TMPRSS2-ERG fusion detected by FISH or QPCR.9, 10 Although crossreactivity of the EPR3864 antibody with ETS family member FLI-1 has been reported by the manufacturer, we demonstrated lack of FLI-1 expression in prostate cancer, indicating that the antibody actually detects ERG protein in this disease.10 In this study, we found concordance of nuclear ERG expression and TMPRSS2-ERG fusion in seven human prostate cancer cell lines and 22 xenografts, with exception of xenografts PC133, PC324 and PC339, which have breakpoint in intron 1 of TMPRSS2.16, 17, 18
We identified ERG expression in 65% of prostate cancer patients treated by radical prostatectomy. The prevalence of ERG overexpression is well in line with our earlier study on ERG immunohistochemistry in 61% of prostate cancer diagnosed on needle-biopsies and earlier studies on TMPRSS2-ERG fusion in prostate cancer.10 One-tenth of prostate cancer revealed heterogeneity of ERG expression between different tissue cores taken from the same tumor, which corresponds with other studies on genomic fusion heterogeneity in prostate cancer.10, 27
Correlation of ERG immunohistochemistry with clinico-pathological parameters in this population of 481 prostate cancer patients revealed that expression of ERG occurred significantly more frequent in patients with PSA ≤10 ng/ml (P=0.024). There was no statistically significant relation between ERG immunohistochemistry and age, Gleason sum, pT-stage or surgical margin status. While an association of ERG fusion with low Gleason sum was identified in other prostate cancer cohorts,28, 29 others did not.30 Using logistic regression, we did not find a statistically significant predictive value of immunohistochemical ERG expression on radical prostatectomy with subsequent biochemical or clinical recurrence, overall death or disease-specific death. Although predictive value of TMPRSS2-ERG fusion for biochemical recurrence and death has been reported by some studies,6, 7, 31, 32 ERG fusion with deletion and duplication of the 5′ end (Edel and 2+Edel) has specifically been associated with worse outcome.6, 7, 31, 32 Obviously, immunohistochemistry is not able to identify the genetic aberration leading to ERG overexpression. It remains to be elucidated whether TMPRSS2-ERG fusion by deletion or duplication is causally related to worse outcome, or merely reflect general genetic imbalance.
As transcription of the ERG fusion partner TMPRSS2 is regulated by androgen receptor, it is hypothesized that androgen receptor and ERG signaling are functionally related. Such a mechanism was proposed because androgen receptor signaling induced TOP2B-mediated double-strands DNA breaks with de novo TMPRSS2-ERG fusion in LAPC4 and LNCaP cells.33 On the other hand, ERG inhibits androgen receptor signaling by reducing receptor expression and activity, together with the induction of repressive epigenetic programs.13 In this study, we investigated the relationship between the ERG and androgen receptor pathways as reflected by the respective protein expression in clinical specimens. Our results showed that increased androgen receptor expression is significantly correlated with higher ERG levels in single cores. Moreover, androgen receptor expression levels in ERG-negative cases were statistically similar to those in cases with moderate ERG expression. To avoid scoring artifacts, we used endothelial and stromal cells as internal controls for ERG and androgen receptor, respectively, in each case. These findings suggest that there is no repressive effect of ERG signaling on androgen receptor expression in clinical specimens. The fact that androgen receptor expression in ERG-negative cores was similar to its expression in moderate ERG-positive cases furthermore reflects a more complex relation of both important pathways.
This study including 481 patients is one of the largest study cohorts on ERG prognostic value until now and included the well-defined European Randomized Study of Screening for Prostate Cancer population. This screening study has shown that PSA-based screening reduced the rate of death from prostate cancer.15 Part of the screening protocol is pathological revision of all radical prostatectomy specimens by a pathologist with expertise in urological pathology. In addition, the protocol ensures uniformity in tumor detection, and monitoring of relevant clinical, pathological and follow-up parameters.
While ERG immunohistochemistry is easy to perform and interpret, it does not reveal qualitative information on the ERG genomic fusion. It cannot distinguish whether fusion to TMPRSS2 or another fusion partner causes ERG overexpression.9, 34, 35 Likewise, with immunohistochemistry no distinction can be made between genomic deletion, translocation or duplication leading to ERG overexpression. While immunohistochemical analysis was performed by visual semiquantitative scoring by two independent researchers and not by automated imaging, we have previously shown that semiquantitative scoring correlated with quantitative ERG mRNA levels.9
Conclusions
Immunohistochemical ERG expression in a well-defined screening cohort of 481 prostate cancer patients revealed no statistical relationship between ERG expression and tumor recurrence or death. Therefore, we conclude that immunohistochemical ERG expression does not have a role in the prognostic stratification of prostate cancer patients after radical prostatectomy.
References
Tomlins SA, Rhodes DR, Perner S, et al. Recurrent fusion of TMPRSS2 and ETS transcription factor genes in prostate cancer. Science 2005;310:644–648.
Helgeson BE, Tomlins SA, Shah N, et al. Characterization of TMPRSS2:ETV5 and SLC45A3:ETV5 gene fusions in prostate cancer. Cancer Res 2008;68:73–80.
Tomlins SA, Bjartell A, Chinnaiyan AM, et al. ETS gene fusions in prostate cancer: from discovery to daily clinical practice. Eur Urol 2009;56:275–286.
Tomlins SA, Laxman B, Varambally S, et al. Role of the TMPRSS2-ERG gene fusion in prostate cancer. Neoplasia 2008;10:177–188.
Albadine R, Latour M, Toubaji A, et al. TMPRSS2-ERG gene fusion status in minute (minimal) prostatic adenocarcinoma. Mod Pathol 2009;22:1415–1422.
Attard G, Clark J, Ambroisine L, et al. Duplication of the fusion of TMPRSS2 to ERG sequences identifies fatal human prostate cancer. Oncogene 2008;27:253–263.
Mehra R, Tomlins SA, Yu J, et al. Characterization of TMPRSS2-ETS gene aberrations in androgen-independent metastatic prostate cancer. Cancer Res 2008;68:3584–3590.
Hermans KG, Boormans JL, Gasi D, et al. Overexpression of prostate-specific TMPRSS2(exon 0)-ERG fusion transcripts corresponds with favorable prognosis of prostate cancer. Clin Cancer Res 2009;15:6398–6403.
Park K, Tomlins SA, Mudaliar KM, et al. Antibody-based detection of ERG rearrangement-positive prostate cancer. Neoplasia 2010;12:590–598.
van Leenders GJLH, Boormans JL, Vissers CJ, et al. Antibody EPR3864 is specific for ERG genomic fusions in prostate cancer: implications for pathology practice. Mod Pathol 2011;24:1128–1138.
Balk SP, Knudsen KE . AR, the cell cycle, and prostate cancer. Nucl Recept Signal 2008;6:e001.
Richter E, Srivastava S, Dobi A . Androgen receptor and prostate cancer. Prostate Cancer Prostatic Dis 2007;10:114–118.
Yu J, Mani RS, Cao Q, et al. An integrated network of androgen receptor, polycomb, and TMPRSS2-ERG gene fusions in prostate cancer progression. Cancer Cell 2010;17:443–454.
Roobol MJ, Kirkels WJ, Schroder FH . Features and preliminary results of the Dutch centre of the ERSPC (Rotterdam, the Netherlands). BJU Int 2003;92 (Suppl 2):48–54.
Schroder FH, Hugosson J, Roobol M, et al. Screening and prostate-cancer mortality in a randomized European study. N Engl J Med 2009;360:1320–1328.
Hermans KG, van Marion R, van Dekken H, et al. TMPRSS2:ERG fusion by translocation or interstitial deletion is highly relevant in androgen-dependent prostate cancer, but is bypassed in late-stage androgen receptor-negative prostate cancer. Cancer Res 2006;66:10658–10663.
Mertz KD, Setlur SR, Dhanasekaran SM, et al. Molecular characterization of TMPRSS2-ERG gene fusion in the NCI-H660 prostate cancer cell line: a new perspective for an old model. Neoplasia 2007;9:200–206.
Saramaki OR, Harjula AE, Martikainen PM, et al. TMPRSS2:ERG fusion identifies a subgroup of prostate cancers with a favorable prognosis. Clin Cancer Res 2008;14:3395–3400.
Boormans JL, Hermans KG, Made AC, et al. Expression of the androgen-regulated fusion gene TMPRSS2-ERG does not predict response to endocrine treatment in hormone-naive, node-positive prostate cancer. Eur Urol 2010;57:830–835.
Perner S, Mosquera JM, Demichelis F, et al. TMPRSS2-ERG fusion prostate cancer: an early molecular event associated with invasion. Am J Surg Pathol 2007;31:882–888.
Nam RK, Sugar L, Yang W, et al. Expression of the TMPRSS2:ERG fusion gene predicts cancer recurrence after surgery for localised prostate cancer. Br J Cancer 2007;97:1690–1695.
Nam RK, Sugar L, Wang Z, et al. Expression of TMPRSS2:ERG gene fusion in prostate cancer cells is an important prognostic factor for cancer progression. Cancer Biol Ther 2007;6:40–45.
Morris DS, Tomlins SA, Montie JE, et al. The discovery and application of gene fusions in prostate cancer. BJU Int 2008;102:276–282.
Clark J, Merson S, Jhavar S, et al. Diversity of TMPRSS2-ERG fusion transcripts in the human prostate. Oncogene 2007;26:2667–2673.
Demichelis F, Fall K, Perner S, et al. TMPRSS2:ERG gene fusion associated with lethal prostate cancer in a watchful waiting cohort. Oncogene 2007;26:4596–4599.
Mosquera JM, Mehra R, Regan MM, et al. Prevalence of TMPRSS2-ERG fusion prostate cancer among men undergoing prostate biopsy in the United States. Clin Cancer Res 2009;15:4706–4711.
Zhang S, Pavlovitz B, Tull J, et al. Detection of TMPRSS2 gene deletions and translocations in carcinoma, intraepithelial neoplasia, and normal epithelium of the prostate by direct fluorescence in situ hybridization. Diagn Mol Pathol 2010;19:151–156.
Darnel AD, Lafargue CJ, Vollmer RT, et al. TMPRSS2-ERG fusion is frequently observed in Gleason pattern 3 prostate cancer in a Canadian cohort. Cancer Biol Ther 2009;8:125–130.
Fine SW, Gopalan A, Leversha MA, et al. TMPRSS2-ERG gene fusion is associated with low Gleason scores and not with high-grade morphological features. Mod Pathol 2010;23:1325–1333.
Rajput AB, Miller MA, De Luca A, et al. Frequency of the TMPRSS2:ERG gene fusion is increased in moderate to poorly differentiated prostate cancers. J Clin Pathol 2007;60:1238–1243.
Perner S, Demichelis F, Beroukhim R, et al. TMPRSS2:ERG fusion-associated deletions provide insight into the heterogeneity of prostate cancer. Cancer Res 2006;66:8337–8341.
Yoshimoto M, Joshua AM, Cunha IW, et al. Absence of TMPRSS2:ERG fusions and PTEN losses in prostate cancer is associated with a favorable outcome. Mod Pathol 2008;21:1451–1460.
Haffner MC, Aryee MJ, Toubaji A, et al. Androgen-induced TOP2B-mediated double-strand breaks and prostate cancer gene rearrangements. Nat Genet 2010;42:668–675.
Esgueva R, Demichelis F, Rubin MA . TMPRSS2-ERG gene fusions are infrequent in prostatic ductal adenocarcinomas. Mod Pathol 2009;22:1398–1399; author reply 9–40.
Tomlins SA, Laxman B, Dhanasekaran SM, et al. Distinct classes of chromosomal rearrangements create oncogenic ETS gene fusions in prostate cancer. Nature 2007;448:595–599.
Acknowledgements
This research was sponsored by CTMM, project PCMM, project number 03O-203.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Hoogland, A., Jenster, G., van Weerden, W. et al. ERG immunohistochemistry is not predictive for PSA recurrence, local recurrence or overall survival after radical prostatectomy for prostate cancer. Mod Pathol 25, 471–479 (2012). https://doi.org/10.1038/modpathol.2011.176
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.2011.176
Keywords
This article is cited by
-
Combined loss of TFF3 and PTEN is associated with lethal outcome and overall survival in men with prostate cancer
Journal of Cancer Research and Clinical Oncology (2019)
-
C-MYC, HIF-1α, ERG, TKT, and GSTP1: an Axis in Prostate Cancer?
Pathology & Oncology Research (2019)
-
Dysregulation and prognostic potential of 5-methylcytosine (5mC), 5-hydroxymethylcytosine (5hmC), 5-formylcytosine (5fC), and 5-carboxylcytosine (5caC) levels in prostate cancer
Clinical Epigenetics (2018)
-
Nuclear receptor ERRα and transcription factor ERG form a reciprocal loop in the regulation of TMPRSS2:ERG fusion gene in prostate cancer
Oncogene (2018)
-
Ethnicity and ERG frequency in prostate cancer
Nature Reviews Urology (2018)