Abstract
Inflammatory bowel disease (IBD) is characterized by chronic inflammation of the digestive system and typically requires lifelong medical care. Recombinant human MFG-E8 (rhMFG-E8) is a 364-amino acid protein, which promotes apoptotic cell clearance and reduces inflammation. This study investigates the therapeutic effect of rhMFG-E8 on two well-established mouse models of IBD. Acute mucosal injury leading to colitis was caused by exposing C57BL/6 mice to 4% dextran sodium sulfate (DSS) in the drinking water over 7 days, and BALB/c mice to a single intrarectal dose of 2.75 mg of 2,4,6-trinitrobenzene sulfonic acid (TNBS). Upon clinical onset of colitis (day 2 in the DSS model and day 1 in the TNBS model), mice were treated with daily subcutaneous injections of rhMFG-E8 (60 or 120 μg/kg/day) or vehicle (saline) for 6 days. Treatment with rhMFG-E8 significantly attenuated colitis in both models in a dose-dependent way. Treatment of DSS-induced colitis with rhMFG-E8 (120 μg/kg/day) decreased weight loss by 59%, the colitis severity score by 71%, and colon shrinkage by 49% when compared with vehicle. Similarly, treatment of TNBS-induced colitis with rhMFG-E8 (120 μg/kg/day) decreased weight loss by 97%, the colitis severity score by 82%, and colon shrinkage by 62% when compared with vehicle. In both models, the colons of animals receiving rhMFG-E8 showed marked reduction in neutrophil infiltration, cytokine and chemokine expression, and apoptotic cell counts. In conclusion, rhMFG-E8 ameliorates DSS- and TNBS-induced colitis, suggesting that it has the potential to become a novel therapeutic agent for IBD.
Similar content being viewed by others
Main
Inflammatory bowel disease (IBD) refers to a group of chronic and recurring inflammatory conditions of the digestive system. The two major forms of IBD are ulcerative colitis (UC), characterized by inflammatory mucosal lesions in the distal colon, and Crohn’s disease (CD), characterized by transmural lesions that can develop anywhere in the gastrointestinal tract. IBD is relatively common, affecting 0.6–0.8% of the population in North America and Europe, and in the last 50 years the incidence rates for CD and UC have progressively increased worldwide.1 Chronic inflammation often leads to bleeding, obstruction, and fistula formation, and up to 75% of patients with CD and 25% of those with UC will require surgery.2 In the United States alone, IBD accounts annually for 700 000 physician visits, 100 000 hospitalizations, and 119 000 disability cases.3
IBD has no cure and generally require lifelong medical care. Traditional therapeutic approaches for IBD include aminosalicylates, antibiotics, glucocorticoids, and immunomodulators. Recently, FDA-approved biological agents targeting integrin subunit alpha-4 (natalizumab) and tumor necrosis factor alpha (TNF-α; infliximab, adalimumab, certolizumab pegol) have produced a leap in therapeutic efficacy.4 However, many patients are refractory to these biological treatments or develop intolerance over time, and one in five patients treated with biological agents will still require intestinal resection.5, 6 The failure of conventional and emerging therapeutic agents to induce lasting deep remission highlights a pressing need for better forms of treatment.
Milk fat globule-epidermal growth factor-factor VIII (MFG-E8) is a glycoprotein secreted by various cell types, including lamina propria macrophages in the jejuno-ileum and colon,7, 8 where it promotes epithelial homeostasis, enterocyte migration, and mucosal healing.8 MFG-E8 promotes efferocytosis (the phagocytic clearance of apoptotic cells) by bridging phosphatidylserine residues on the surface of apoptotic cells with αVβ3 and αVβ5 integrins on the surface of macrophages.7, 8 Apoptotic epithelial cells are increased in UC and CD,9, 10 leading to barrier loss and exposure to luminal contents.11 MFG-E8 also inhibits the release of pro-inflammatory cytokines by immune cells via attenuation of the NF-κB pathway12 and suppresses neutrophil migration into injured tissues.13 Moreover, there is an association between low levels of MFG-E8 and colitis. Colon samples from UC patients have decreased levels of MFG-E814 and mice deficient for MFG-E8 develop a more severe form of colitis,15 suggesting an intestinal protective role for MFG-E8.
The combination of MFG-E8’s intestinal protection and anti-inflammatory activities makes MFG-E8 a therapeutic candidate with the ideal profile to become an effective treatment for IBD. Therefore, we generated recombinant human MFG-E8 (rhMFG-E8)16 and used it to treat acute colitis induced by dextran sulfate sodium (DSS) and by 2,4,6-trinitrobenzene sulfonic acid (TNBS), two injury-repair models with features reminiscent of UC17 and CD,18, 19 respectively.
MATERIALS AND METHODS
Animals
Specific pathogen-free C57BL/6 and BALB/c mice were purchased from Taconic (Albany, NY, USA) and from Jackson Laboratory (Bar Harbor, ME, USA), respectively. Mice were maintained under 12-h light cycles, given free access to food and water, and allowed to acclimate for at least 1 week before any experimental manipulation. All experiments involving animals were reviewed and approved by the Feinstein Institute for Medical Research’s Institutional Animal Care and Use Committee.
Induction of Colitis Using DSS
Between days 0 and 7, male 8- to 12-week-old C57BL/6 mice received drinking water containing 4% DSS (MW 36 000–50 000; MP Biomedicals, Solon, OH, USA).20 A non-colitic control group received water without DSS. Five animals were studied in each group (non-colitic control, vehicle, and two doses of rhMFG-E8). Colitic and non-colitic mice were euthanized for tissue collection at day 8.
Induction of Colitis Using TNBS
Male 6- to 7-week-old BALB/c mice anesthetized with isoflurane were administered a single intrarectal dose of 2.75 mg of TNBS (Sigma-Aldrich Corp, St Louis, MO, USA) dissolved in 50% ethanol (day 0).21 Mice were held upside down for 5 min after administration of TNBS. A control group received 50% ethanol without TNBS. Ten animals were studied in each group (control, vehicle, and two doses of rhMFG-E8). Colitic and control mice were euthanized for tissue collection at day 7 post exposure to TNBS/ethanol.
Production of and Treatment with rhMFG-E8
Human Ex-M0438-B01 expression clone (GeneCopoeia Inc., Germantown, MD, USA) was expressed in E. coli, purified using metal affinity chromatography, and rendered endotoxin free by phase separation with Triton-X-114. The amino-acid composition of the purified protein was analyzed by LCMS/MS. Based on SDS-PAGE, rhMFG-E8 was over 99% pure and was identified as human MFG-E8 with more than 95% confidence.16 rhMFG-E8’s biological activity was confirmed using the pHrodo succinimidyl ester efferocytosis assay.16 Mice were randomly divided to receive a total of six daily subcutaneous injections of rhMFG-E8 (60 or 120 μg/kg/day), or vehicle (saline), and treatment started immediately after clinical onset of colitis (day 2 after the first administration of DSS; day 1 after administration of TNBS).
Colitis Severity Assessment and Tissue Collection
Mice were evaluated daily for changes in body weight, stool consistency, and the presence of blood in the stools (Table 1), which were combined to calculate an overall colitis clinical severity score, as previously described.15 After euthanasia, each colon was carefully removed and measured. The descending (in mice exposed to DSS and their non-colitic controls) or transverse (in mice exposed to TNBS and their non-colitic controls) colonic segments were promptly stored in 10% formalin and in −80 °C for further analysis.
Myeloperoxidase Activity Assay
Colon tissues were homogenized in KPO4 buffer containing 0.5% hexadecyltrimethylammonium bromide. Lysates were centrifuged and the supernatant was diluted in reaction solution. Hydrogen peroxide was added and the rate of optical density change over 2 min was measured spectrophotometrically at 460 nm.22
Quantitative Real-Time qPCR
Colonic total RNA was extracted with TRIzol (Life Technologies, Grand Island, NY, USA) and reverse-transcribed into cDNA using murine leukemia virus reverse transcriptase (Applied Biosystems, Foster, CA, USA). Quantitative real-time polymerase chain reactions (qPCRs) were carried out using SYBR Green PCR Master Mix and amplified in a 7300 thermocycler (Applied Biosystems). After normalization with mouse β-actin, expression was determined as fold change relative to the expression in non-colitic controls using the 2−ΔΔCt method.23 The primers used in this study are listed in Table 2.
Histological Studies
Colon tissues were fixed in 10% phosphate-buffered formalin, embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin (H&E). Histological examination was performed by an investigator blinded to the interventions. The crypt-epithelial injury score (range 0–16) was determined by multiplying the crypt and epithelium damage score (0=no damage, 1=basal third damaged, 2=basal two thirds damaged, 3=only surface epithelium intact, 4=entire crypt and epithelium lost) by a factor to reflect the percentage of tissue affected (1=0–25%, 2=26–50%, 3=51–75%, 4=76–100%).15
Evaluation of Colonic Cell Apoptosis by TUNEL Assay
Colon tissue sections were deparaffinized and digested with 20 μg/ml proteinase K for 20 min at room temperature. After washing, sections were stained with a green fluorescent-tagged terminal deoxynucleotide transferase dUTP nick-end labeling (TUNEL) kit (Roche Diagnostics, Indianapolis, IN, USA), counterstained with DAPI, and examined under a fluorescence microscope (Nikon Eclipse Ti-S, Melville, NY, USA). TUNEL-positive cells were counted in 10 fields ( × 200 magnification) per section, and groups were compared for the average number of cells per field.
Statistical Analysis
Data were compared using one-way ANOVA followed by the Student-Newman-Keuls method to detect pairwise differences, considering a significance threshold of P≤0.05. Unless otherwise specified, results are expressed as mean±s.e.m.
RESULTS
rhMFG-E8 Ameliorates DSS- and TNBS-Induced Colitis
We studied the effect of rhMFG-E8 on DSS- and TBNS-induced colitis, two injury-repair models of IBD. Mice were exposed either to DSS from day 0 to day 7, or to a single dose of TNBS on day 0, and disease severity was assessed using the colitis severity score—a composite index combining changes in body weight, stool consistency, and the presence of blood in the stools.15 In mice exposed to DSS and treated with vehicle, the colitis severity score increased markedly after day 4 and reached maximal severity at day 8. rhMFG-E8 decreased DSS colitis severity in a dose-dependent manner. In mice exposed to DSS and treated with rhMFG-E8, day 8 colitis severity scores were 54.5% (60 μg/kg/day) and 29.1% (120 μg/kg/day) of the scores of mice treated with vehicle (Figure 1a).
rhMFG-E8 ameliorates clinical signs of DSS- and TNBS-induced colitis. C57BL/6 mice were exposed to drinking water containing 4% DSS for 7 consecutive days, and received daily subcutaneous injections of rhMFG-E8 (60 or 120 μg/kg/day) or vehicle (saline) from day 2 to day 7 (n=5/group). BALB/c mice received a single intrarectal dose of TNBS at day 0, and received daily subcutaneous injections of rhMFG-E8 (60 or 120 μg/kg/day) or vehicle (saline) from day 1 to day 6 (n=10/group). (a) Mice exposed to DSS and treated with rhMFG-E8 experienced a dose-dependent reduction in the colitis clinical severity score, a composite measure of weight loss, stool consistency, and blood in the stools. (b) Mice exposed to TNBS and treated with rhMFG-E8 also had a dose-dependent reduction in the colitis clinical severity score, which peaked on day 1 and progressively decreased during the observation period. (c) At day 8, mice exposed to DSS and treated with vehicle had a significant reduction in body weight, while mice treated with rhMFG-E8 had a dose-dependent decrease in weight loss. (d) At day 7, mice exposed to TNBS and treated with vehicle also had a reduction in body weight, while mice treated with rhMFG-E8 also experienced a dose-dependent decrease in weight-loss. Ctrl=control; Veh=vehicle; 60=rhMFG-E8 60 μg/kg/day; 120=rhMFG-E8 120 μg/kg/day; *P<0.05 vs control; #P<0.05 vs vehicle.
In mice exposed to TNBS and treated with vehicle, the colitis severity score reached maximal severity at day 1 (Figure 1b). Control mice, receiving ethanol, developed a mild and transient form of colitis, an expected result of ethanol’s intrinsic irritant properties. Between days 1 and 7, colitis severity progressively decreased in all groups, but scores improved significantly faster in the groups receiving rhMFG-E8, in a dose-dependent manner. In mice exposed to TNBS and treated with rhMFG-E8, day 7 colitis severity scores were 81.0% (60 μg/kg/day) and 43.8% (120 μg/kg/day) of the scores of mice treated with vehicle (Figure 1b).
rhMFG-E8 also had a protective, dose-dependent effect on colitis induced weight loss. Compared with their starting weight, control non-colitic mice gained 3.4%, while mice exposed to DSS and treated with vehicle lost 25.0% of their body weight. Mice treated with rhMFG-E8 at 60 μg/kg/day lost 17.0% of their starting body weight, and mice treated with 120 μg/kg/day lost 10.2% (Figure 1c). Likewise, at day 7 TNBS control mice gained 3.2% while mice exposed to TNBS and treated with vehicle lost 19.3% of their starting body weight. Mice treated with rhMFG-E8 lost 9.2% (60 μg/kg/day) and 0.7% (120 μg/kg/day) of their starting body weight (Figure 1d). Therefore, treatment with rhMFG-E8 was associated with clinically significant amelioration of both the DSS and TNBS models of colitis.
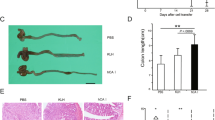
At the end of the observation period, all colons were collected and measured to determine whether rhMFG-E8 had an effect on the decrease in colon length and cecum size characteristically associated with colonic inflammation. Compared with the non-colitic control group (10.1±0.3 cm), colons of mice exposed to DSS and vehicle (6.3±0.2 cm) were shortened in length by 37.4%, while the colons of mice treated with rhMFG-E8 decreased by 28.7% in the group receiving 60 μg/kg/day (7.2±0.1 cm), and by 18.8% in the group receiving 120 μg/kg/day (8.2±0.2 cm) (Figure 2a). Similarly, colons of mice in the TNBS vehicle group (7.7±0.3 cm) were 27.8% shorter than those in the control group (10.7±0.3 cm), while the colons of mice treated with rhMFG-E8 were shortened by 19.3% in the group receiving 60 μg/kg/day (8.6±0.2 cm) and only 10.59% in the group receiving 120 μg/kg/day (9.5±0.2 cm) (Figure 2b). Prevention of colonic shortening suggests that treatment with rhMFG-E8 attenuated the submucosal edema and muscularis mucosa hypertrophy typical of the DSS and TNBS models of colitis.
rhMFG-E8 prevents colon length shortening in DSS- and TNBS-induced colitis of mice with DSS-induced colitis. (a) Colons of mice exposed to DSS treated with vehicle were significantly shortened, while those of mice treated with rhMFG-E8 exhibited a dose-dependent reduction in colon shortening (n=5/group). (b) While the colons of mice exposed to TNBS and treated with vehicle were significantly shortened, those of mice treated with rhMFG-E8 had a dose-dependent reduction in colonic shortening (n=10/group). Ctrl=control; Veh=vehicle; 60=rhMFG-E8 60 μg/kg/day; 120=rhMFG-E8 120 μg/kg/day; *P<0.05 vs control; #P<0.05 vs vehicle.
rhMFG-E8 Preserves Histological Integrity and Reduces Colonic Infiltration by Neutrophils in DSS- and TNBS-Induced Colitis
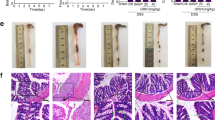
Inflammation of the mucosa and submucosa is one of the hallmarks of both UC and DSS-induced colitis. Thus, colon segments were examined histologically to determine the effects of rhMFG-E8 on histological integrity and the degree of inflammation. In the DSS model, sections from the descending colons of non-colitic controls had normal crypts and no inflammatory cells, while those of mice treated with vehicle featured numerous polymorphonuclear cells in the luminal space, mucosa, and submucosa, destruction of the normal crypt architecture, submucosal edema, and hyperplasia of the muscularis mucosa (Figure 3a). Treatment with rhMFG-E8 was associated with a clear dose-dependent reduction in the DSS-induced inflammatory infiltrate and tissue injury (Figure 3a). Compared with the vehicle group, the crypt-epithelial injury scores of mice treated with 60 μg/kg/day decreased by 20.9%, and those of mice treated with 120 μg/kg/day decreased by 73.3%, respectively (Figure 3a).
rhMFG-E8 prevents neutrophil infiltration and crypt damage in colons of mice with DSS- and TNBS-induced colitis. The descending colons of mice exposed to DSS (n=5/group) or transverse colons of mice exposed to TBNS (n=10/group) were collected at the end of the observation period for histological analysis. (a) The colons of control mice had normal architecture and cellular composition, while those of mice exposed to DSS and treated with vehicle had edema and infiltration by inflammatory cells in the mucosal and sub-mucosal layers, inflammatory exudate in the lumen, and destruction of the normal crypt structure. The colons of mice treated with rhMFG-E8 showed dose-dependent improvement toward normal colon histology and protective effect in the crypt-epithelial injury score. (b) Likewise, the colons of TNBS control mice also had normal architecture and cellular composition, while those of mice exposed to TNBS and treated with vehicle had edema and infiltration by inflammatory cells in the mucosal and sub-mucosal layers, inflammatory exudate in the lumen, and tissue damage. The colons of mice treated with rhMFG-E8 showed dose-dependent improvement toward normal colon histology rhMFG-E8 and a reduction in the crypt-epithelial injury score. H&E stain, 100 × ; Ctrl=control; Veh=vehicle; 60=rhMFG-E8 60 μg/kg/day; 120=rhMFG-E8 120 μg/kg/day; *P<0.05 vs control; #P<0.05 vs vehicle.
In the TNBS model, sections of the transverse colon segments from control mice had normal crypts and no inflammatory infiltrates, while those of mice treated with vehicle had transmural inflammation and neutrophil infiltration along with significant epithelial damage and marked disruption of the normal crypt architecture (Figure 3b). Tissues from mice treated with rhMFG-E8, on the other hand, had a dose-dependent reduction in inflammation and tissue injury (Figure 3b). Compared with mice treated with vehicle, the crypt-epithelial injury score decreased by 60.9 and 87.0% in mice treated with rhMFG-E8 at 60 and 120 μg/kg/day, respectively (Figure 3b).
Next, we measured myeloperoxidase (MPO) activity in the colons to quantify their infiltration by neutrophils. In the DSS model, the descending colon segments of vehicle-treated mice had a 260-fold increase in MPO activity compared with those of non-colitic controls (Figure 4a). Treatment with rhMFG-E8 was protective, and MPO activity in the colons of mice treated with 60 and 120 μg/kg/day was 24.7 and 49.8% lower, respectively, than that in the vehicle group (Figure 4a). Exposure to TNBS also resulted in marked neutrophil infiltration, and MPO activity in the transverse colons of mice treated with vehicle was 9.6-fold higher than that of controls (Figure 4b). As in the DSS model, rhMFG-E8 was associated with a significant, dose-dependent reduction in MPO activity. Compared with vehicle, treatment with 60 and 120 μg/kg/day decreased the MPO activity by 66.5 and 76.4%, respectively (Figure 4b). Therefore, rhMFG-E8 was associated with a significant, dose-dependent attenuation of histological damage to the colon and of its infiltration by neutrophils in both DSS- and TNBS-induced colitis.
rhMFG-E8 decreases the MPO activity in colons of mice with DSS- and TNBS-induced colitis. MPO activity was determined in the descending colons of mice exposed to DSS (n=5/group) or transverse colons of mice exposed to TBNS (n=10/group). rhMFG-E8 was associated with a dose-dependent reduction in the colon MPO activity in both (a) mice exposed to DSS (n=5/group) and in (b) mice exposed to TNBS (n=10/group) H&E stain, × 100; Ctrl=control; Veh=vehicle; 60=rhMFG-E8 60 μg/kg/day; 120=rhMFG-E8 120 μg/kg/day; *P<0.05 vs control; #P<0.05 vs vehicle.
rhMFG-E8 Decreases Expression of Pro-Inflammatory Cytokines and Chemokines in DSS- and TNBS-Induced Colitis
Histologically, treatment with rhMFG-E8 was associated with decreased signs of inflammation of the colon. Therefore, we measured the expression levels of TNF-α, interleukin (IL)-1β, and IL-6, three cytokines known to be elevated and have a crucial role driving up inflammation in both IBD and injury-repair models of colitis.24 As expected, expression levels of TNF-α, IL-1β, and IL-6 in the descending colons of mice exposed to DSS and treated with vehicle were increased by 18.6-, 29.4-, and 119.6-fold compared with those of non-colitic controls (Figure 5a). The descending colons from mice exposed to DSS and treated with rhMFG-E8 showed a significant dose-dependent reduction in the levels of these cytokines. In mice treated with 60 μg/kg/day, TNF-α, IL-1β, and IL-6 were 9.4-, 21.3-, and 85.4-fold; and in mice treated with 120 μg/kg/day, they were 5.2-, 5.7-, and 43.1-fold those of non-colitic controls, respectively (Figure 5a).
rhMFG-E8 decreases the expression of pro-inflammatory cytokines in mice with DSS- and TNBS-induced colitis. The descending colons of mice exposed to DSS (n=5/group) or transverse colons of mice exposed to TBNS (n=10/group) were collected at the end of the observation period, submitted to RNA extraction, cDNA synthesis, and qPCR amplification. (a) The expression of TNF-α, IL-1β, and IL-6 was significantly elevated in mice exposed to DSS and treated with vehicle, compared with control mice, and treatment with rhMFG-E8 was associated with a dose-dependent reduction in the expression level of these pro-inflammatory cytokines. (b) The expression of TNF-α, IL-1β, and IL-6 was also elevated in mice exposed to TNBS and treated with vehicle, compared with control mice, and treatment with rhMFG-E8 was also associated with a dose-dependent decrease in the expression level of these pro-inflammatory cytokines. Ctrl=control; Veh=vehicle; 60=rhMFG-E8 60 μg/kg/day; 120=rhMFG-E8 120 μg/kg/day; *P<0.05 vs control; #P<0.05 vs vehicle.
Likewise, mRNA expression levels of TNF-α, IL-1β, and IL-6 were 3.5-, 3.0-, and 29.9-fold higher in the transverse colons of mice exposed to TNBS and treated with vehicle compared with those of mice not exposed to TNBS (Figure 5b). Treatment with rhMFG-E8 was protective and associated with a dose-dependent reduction in the levels of these cytokines. In mice exposed to TNBS and treated with rhMFG-E8, TNF-α, IL-1β, and IL-6 mRNA levels were expressed at 1.9-, 1.4-, and 9.1-fold (60 μg/kg/day), and 1.2-, 1.0-, and 4.3-fold (120 μg/kg/day) the levels of non-colitic controls, respectively (Figure 5b).
Treatment with rhMFG-E8 was also associated with a marked reduction in neutrophil infiltration. Thus, we next determined the expression levels of keratinocyte chemoattractant (KC) (CXCL1) and macrophage inflammatory protein-2 (MIP-2) (CXCL2), two chemokines important for neutrophil infiltration and known to be elevated in IBD and injury-repair models of colitis.24 Expression levels of KC and MIP-2 in mice exposed to DSS and treated with vehicle increased by 56.7- and 196.9-fold, respectively, compared with non-colitic controls (Figure 6a). In mice exposed to DSS and treated with rhMFG-E8, however, levels of KC and MIP-2 were 23.5- and 100.6-fold (60 μg/kg/day), and 13.6- and 74.5-fold (120 μg/kg/day) the levels of non-colitic controls, respectively (Figure 6a).
rhMFG-E8 decreases the expression of neutrophil chemoattractants in mice with DSS- and TNBS-induced colitis. cDNA from the descending colons of mice exposed to DSS (n=5/group) or transverse colons of mice exposed to TBNS (n=10/group) was used for qPCR amplification. (a) Expression of the neutrophil chemoattractants KC and MIP-2 was increased in mice exposed to DSS and treated with vehicle, and decreased dose-dependently in mice treated with rhMFG-8. (b) KC and MIP-2 were also increased in the colons of mice exposed to TNBS and treated with vehicle, and decreased dose-dependently in mice treated with rhMFG-8. Ctrl=control; Veh=vehicle; 60=rhMFG-E8 60 μg/kg/day; 120=rhMFG-E8 120 μg/kg/day; *P<0.05 vs control; #P<0.05 vs vehicle.
Similarly to what was observed in the DSS model, the expression levels of KC and MIP-2 in mice exposed to TNBS and treated with vehicle increased by 6.8- and 4.1-fold, respectively, compared with mice exposed to TNBS and treated with vehicle (Figure 6b). In mice exposed to TNBS and treated with rhMFG-E8, however, levels of KC and MIP-2 were 4.1- and 1.9-fold (60 μg/kg/day), and 2.1- and 1.4-fold (120 μg/kg/day) the levels of controls, respectively (Figure 6b).
Therefore, treatment with rhMFG-E8 significantly reduced the expression of neutrophil chemoattractants and pro-inflammatory cytokines that initiate and perpetuate inflammation in IBD.
rhMFG-E8 Decreases Apoptotic Cell Counts in DSS- and TNBS-Induced Colitis
Increased apoptotic events are commonly observed in IBD and injury-repair models of colitis9, 10, 25, 26 and thought to contribute to the pathogenesis of colonic inflammation.11 MFG-E8 is known to promote the clearance of apoptotic cells. Hence, we quantified apoptotic events in the colon using TUNEL. The descending colons of non-colitic controls had 2.5±0.9 TUNEL-positive apoptotic cells per field, while those of mice receiving DSS and treated with vehicle had 69.9±12.5 apoptotic cells per field—a 28-fold increase (Figure 7a). Apoptotic cells were mainly detected in the mucosal layer, along with the destruction of the normal architecture of the colonic epithelium. The colons of mice exposed to DSS and treated with rhMFG-E8 had an average of 24.5±3.4 (60 μg/kg/day) and 9.7±1.7 (120 μg/kg/day) apoptotic cells/field, reflecting 65.0 and 86.2% less apoptotic cells than in the vehicle group, respectively (Figure 7a).
rhMFG-E8 is associated with a reduction in the number of apoptotic cells in mice with DSS- and TNBD-induced colitis. The descending colons of mice exposed to DSS (n=5/group) or transverse colons of mice exposed to TBNS (n=10/group) were collected at the end of the observation period and submitted to the TUNEL assay for detection of apoptotic cells. While few apoptotic cells were detected in the descending colons of controls, mice exposed to DSS (a) or TBNS (b) and treated with vehicle had numerous apoptotic cells, mainly in the mucosal and sub-mucosal layers. Compared with the vehicle group, mice treated with rhMFG-E8 had a significant dose-dependent reduction in the number of apoptotic cells. Images: TUNEL-positive apoptotic cells are stained green and nuclei are counterstained blue with DAPI, × 100. Lower panel: number of apoptotic cells per × 200 magnification field. Ctrl=control; Veh=vehicle; 60=rhMFG-E8 60 μg/kg/day; 120=rhMFG-E8 120 μg/kg/day; *P<0.05 vs control; #P<0.05 vs vehicle.
Similarly, there were 112.7-fold more TUNEL-positive apoptotic cells in the transverse colons of mice exposed to TNBS and treated with vehicle had than in those of controls (Figure 7b), and treatment with rhMFG-E8 was associated with a near-complete normalization in the amount of apoptotic cells. The colons of mice exposed to TNBS and treated with rhMFG-E8 92.5% (60 μg/kg/day) and 97.5% (120 μg/kg/day) fewer apoptotic events than those of mice treated with vehicle, respectively (Figure 7b). These results indicate that treatment with rhMFG-E8 was associated with a marked decrease in the number of apoptotic events in the colon.
DISCUSSION
Our data demonstrate that treatment with rhMFG-E8 has dose-dependent beneficial effects on relevant clinical, immunological, and histological parameters in both DSS- and TNBS-induced colitis, two well-established experimental models widely used to screen therapeutic compounds for the treatment of colitis.17 This is the first time the human form of MFG-E8 is shown to ameliorate IBD, and the first time MFG-E8 was used after induction of colitis and during its most severe phase.
Recombinant murine (rm) MFG-E8 can prevent colitis when given before disease induction7, 27 and can attenuate damage when given during the recovery phase of the disease,15 suggesting a potential for the use of MFG-E8 as a therapeutic agent for IBD. However, rodent proteins are not suitable for clinical use. Therefore, we generated recombinant human (rh) MFG-E8 and confirmed its ability to promote the phagocytic clearance of apoptotic cells.16 We then induced colitis and tested rhMFG-E8 in a more clinically relevant setting, starting treatment after induction of colitis and at time points when clinical signs of colitis, such as loose stools, bleeding, and weight loss, were already starting to be evident. We selected the subcutaneous route for rhMFG-E8 administration because it is a frequently used route for protein and peptide medications (such as insulin).
The mechanism through which MFG-E8 operates to ameliorate colitis is likely to comprise a combination of MFG-E8’s known effects on the clearance of apoptotic cells,7, 8 attenuation of the NF-κB pathway,12 suppression of neutrophil migration to sites of inflammation,13 and promotion of intestinal mucosal homeostasis and healing.8 MFG-E8 promotes the phagocytic clearance of apoptotic cells by bridging phosphatidylserine residues on the surface of apoptotic cells with αVβ3 and αVβ5 integrins on the surface of macrophages.28 An increased number of apoptotic crypt epithelial cells is present in UC,9 CD,10 and animal models of IBD.25, 26 In IBD, intestinal epithelial cell apoptosis disrupts the tight junction barrier, exposing the immune system to luminal contents and leading to mucosal inflammation.11 In our study, the colons of mice treated with rhMFG-E8 had significantly less apoptotic cells, suggesting that increased clearance of apoptotic cells may have contributed to amelioration of colitis by rhMFG-E8.
Inflammation has a key role in IBD’s perpetuation, severity, and complications, and infiltration with inflammatory cells is a characteristic finding of IBD. Accordingly, we observed a large number of neutrophils and increased expression levels of pro-inflammatory cytokines and chemokines in the intestines of animals with colitis. It is fair to hypothesize that the decreased levels of the neutrophil-attracting chemokines KC (CXCL1) and MIP-2 (CXCL2) in the colons of mice treated with rhMFG-E8 may have contributed to the reduced neutrophil infiltration in these mice. How exactly rhMFG-E8 suppressed the expression of pro-inflammatory cytokines and chemokines in the colon is unclear, but MFG-E8 binding to αvβ3/αvβ5 integrin receptors has already been shown to attenuate inflammation via a number of mechanisms, including reduction of CXCR2 expression in neutrophils,13 promotion of anti-inflammatory macrophage reprogramming,29 and suppression of the MAPK, p38, STAT-3, and NF-κB pathways.12, 30
In addition to its effects on apoptotic cell clearance and inflammation, MFG-E8 is also important for the maintenance of intestinal epithelial homeostasis, basal-apical enterocyte migration along the colonic crypts, and mucosal healing.8 The structure of the mucosa and crypts was significantly preserved in mice treated with rhMFG-E8, suggesting that effects on intestinal homeostasis and healing may also have contributed to part of its protective effect on colitis.
rhMFG-E8 was given daily until tissue collection, and our histological, gene expression, and TUNEL findings are fully compatible with MFG-E8’s known effects on cells and tissues. Although there is a possibility that our findings may not have been a direct result from MFG-E8 activity, it is clear that they did result from treatment of colitic mice with rhMFG-E8.
Doses of rmMFG-E8 used in proof-of-concept studies were lower than the doses of rhMFG-E8 used in this study, probably because a lower dose is required to prevent7, 27 or treat disease in the recovery phase15 as opposed to the early clinical onset. It is also possible that the human form of MFG-E8 may be less potent in the mouse, as the homology between human and mouse MGF-E8 is only 56%. Nevertheless, rhMFG-E8 is clearly active in rodents, as we have demonstrated previously31, 32, 33, 34, 35 and in the present study. Therefore, rhMFG-E8 is expected to have comparable beneficial effects in humans with IBD.
The in vivo use of rhMFG-E8 appears to be safe, and it has been used for ischemia and reperfusion, acute irradiation syndrome, and sepsis with no measurable changes in hepatic and renal function tests.31, 32, 33, 34, 35 We have injected mice with up to 2560 μg/kg rhMFG-E8 intravenously, and observed no alteration in liver transaminases (AST and ALT) 24 h later (unpublished observations). More comprehensive studies, however, will need to be conducted to exclude any toxicity.
Over the years, more than 50 mouse models of IBD have been generated in an attempt to better recapitulate human disease. Some chronic models of IBD (such as IL-10 knockout mice and the T-cell transfer model) have immunologic, clinical, and histological features more representative of human UC and CD,36 and additional studies investigating rhMFG-E8’s efficacy in such models are therefore warranted. Ultimately, independent of its exact mechanism of action, our results support rhMFG-E8’s potential to be developed as a novel therapeutic agent for UC and CD.
References
Molodecky NA, Soon IS, Rabi DM et al. Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology 2012;142:46–54.
Sonnenberg A . Disability from inflammatory bowel disease among employees in West Germany. Gut 1989;30:367–370.
CDC. Inflammatory Bowel Disease (IBD) Atlanta: Centers for Disease Control and Prevention; 2012 (updated 15 July 2011; cited 23 October 2013). Available from http://www.cdc.gov/ibd/.
Rutgeerts P, Vermeire S, Van Assche G . Biological therapies for inflammatory bowel diseases. Gastroenterology 2009;136:1182–1197.
Lichtenstein GR . Comprehensive review: antitumor necrosis factor agents in inflammatory bowel disease and factors implicated in treatment response. Therap Adv Gastroenterol 2013;6:269–293.
Peyrin-Biroulet L, Lemann M . Review article: remission rates achievable by current therapies for inflammatory bowel disease. Aliment Pharmacol Ther 2011;33:870–879.
Aziz MM, Ishihara S, Mishima Y et al. MFG-E8 attenuates intestinal inflammation in murine experimental colitis by modulating osteopontin-dependent alphavbeta3 integrin signaling. J Immunol 2009;182:7222–7232.
Bu HF, Zuo XL, Wang X et al. Milk fat globule-EGF factor 8/lactadherin plays a crucial role in maintenance and repair of murine intestinal epithelium. J Clin Invest 2007;117:3673–3683.
Hagiwara C, Tanaka M, Kudo H . Increase in colorectal epithelial apoptotic cells in patients with ulcerative colitis ultimately requiring surgery. J Gastroenterol Hepatol 2002;17:758–764.
Di Sabatino A, Ciccocioppo R, Luinetti O et al. Increased enterocyte apoptosis in inflamed areas of Crohn's disease. Dis Colon Rectum 2003;46:1498–1507.
Su L, Nalle SC, Shen L et al. TNFR2 activates MLCK-dependent tight junction dysregulation to cause apoptosis-mediated barrier loss and experimental colitis. Gastroenterology 2013;145:407–415.
Aziz M, Jacob A, Matsuda A et al. Pre-treatment of recombinant mouse MFG-E8 downregulates LPS-induced TNF-alpha production in macrophages via STAT3-mediated SOCS3 activation. PLoS ONE 2011;6:e27685.
Aziz M, Matsuda A, Yang WL et al. Milk fat globule-epidermal growth factor-factor 8 attenuates neutrophil infiltration in acute lung injury via modulation of CXCR2. J Immunol 2012;189:393–402.
Zhao QJ, Yu YB, Zuo XL et al. Milk fat globule-epidermal growth factor 8 is decreased in intestinal epithelium of ulcerative colitis patients and thereby causes increased apoptosis and impaired wound healing. Mol Med 2012;18:497–506.
Chogle A, Bu HF, Wang X et al. Milk fat globule-EGF factor 8 is a critical protein for healing of dextran sodium sulfate-induced acute colitis in mice. Mol Med 2011;17:502–507.
Qiang X, Li J, Wu R et al. Expression and characterization of recombinant human milk fat globule-EGF factor VIII. Int J Mol Med 2011;28:1071–1076.
Wirtz S, Neufert C, Weigmann B et al. Chemically induced mouse models of intestinal inflammation. Nat Protoc 2007;2:541–546.
Scheiffele F, Fuss IJ . Induction of TNBS colitis in mice. Curr Protoc Immunol 2002;Chapter 15:Unit 15.9.
Alex P, Zachos NC, Nguyen T et al. Distinct cytokine patterns identified from multiplex profiles of murine DSS and TNBS-induced colitis. Inflamm Bowel Dis 2009;15:341–352.
Coburn LA, Gong X, Singh K et al. L-arginine supplementation improves responses to injury and inflammation in dextran sulfate sodium colitis. PLoS ONE 2012;7:e33546.
Lee T, Lee E, Irwin R et al. beta-Arrestin-1 deficiency protects mice from experimental colitis. Am J Pathol 2013;182:1114–1123.
Day YJ, Marshall MA, Huang L et al. Protection from ischemic liver injury by activation of A2A adenosine receptors during reperfusion: inhibition of chemokine induction. Am J Physiol Gastrointest Liver Physiol 2004;286:G285–G293.
Livak KJ, Schmittgen TD . Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001;25:402–408.
Yan Y, Kolachala V, Dalmasso G et al. Temporal and spatial analysis of clinical and molecular parameters in dextran sodium sulfate induced colitis. PLoS ONE 2009;4:e6073.
Dirisina R, Katzman RB, Goretsky T et al. p53 and PUMA independently regulate apoptosis of intestinal epithelial cells in patients and mice with colitis. Gastroenterology 2011;141:1036–1045.
Araki Y, Bamba T, Mukaisho K et al. Dextran sulfate sodium administered orally is depolymerized in the stomach and induces cell cycle arrest plus apoptosis in the colon in early mouse colitis. Oncol Rep 2012;28:1597–1605.
Otani A, Ishihara S, Aziz MM et al. Intrarectal administration of milk fat globule epidermal growth factor-8 protein ameliorates murine experimental colitis. Int J Mol Med 2012;29:349–356.
Hanayama R, Tanaka M, Miyasaka K et al. Autoimmune disease and impaired uptake of apoptotic cells in MFG-E8-deficient mice. Science 2004;304:1147–1150.
Brissette MJ, Lepage S, Lamonde AS et al. MFG-E8 released by apoptotic endothelial cells triggers anti-inflammatory macrophage reprogramming. PLoS ONE 2012;7:e36368.
Miksa M, Amin D, Wu R et al. Maturation-induced down-regulation of MFG-E8 impairs apoptotic cell clearance and enhances endotoxin response. Int J Mol Med 2008;22:743–748.
Shah KG, Wu R, Jacob A et al. Recombinant human milk fat globule-EGF factor 8 produces dose-dependent benefits in sepsis. Intens Care Med 2012;38:128–136.
Cheyuo C, Jacob A, Wu R et al. Recombinant human MFG-E8 attenuates cerebral ischemic injury: its role in anti-inflammation and anti-apoptosis. Neuropharmacology 2012;62:890–900.
Ajakaiye MA, Jacob A, Wu R et al. Recombinant human MFG-E8 attenuates intestinal injury and mortality in severe whole body irradiation in rats. PLoS ONE 2012;7:e46540.
Matsuda A, Jacob A, Wu R et al. Milk fat globule—EGF factor VIII ameliorates liver injury after hepatic ischemia-reperfusion. J Surg Res 2013;180:e37–e46.
Zhang F, Shah KG, Qi L et al. Milk fat globule epidermal growth factor-factor 8 mitigates inflammation and tissue injury after hemorrhagic shock in experimental animals. J Trauma Acute Care Surg 2012;72:861–869.
Valatas V, Vakas M, Kolios G . The value of experimental models of colitis in predicting efficacy of biological therapies for inflammatory bowel diseases. Am J Physiol Gastrointest Liver Physiol 2013;305:G763–G785.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
One of the authors (PW) is an inventor of the pending PCT applications #WO/2006/122327: ‘Milk fat globule epidermal growth factor-factor VIII and sepsis’, #WO/2009/064448: ‘Prevention and treatment of inflammation and organ injury after ischemia/reperfusion using MFG-E8’, and WO/2012/149254: ‘MFG-E8 and uses thereof’. These patent applications cover the fundamental concept of using MFG-E8 for the treatment of inflammatory diseases. TheraSource LLC holds the exclusive option to license the technology from the Feinstein Institute for Medical Research. PW is a co-founder of TheraSource LLC.
Additional information
Mice with chemically induced colitis reminiscent of inflammatory bowel disease (IBD) that are subsequently treated with recombinant human MFG-E8 show decreased colitis severity, accompanied by decreases in numbers of neutrophils, the amounts of proinflammatory cytokines and chemokines, and the numbers of apoptotic cells in the animals' colons. MFG-E8 therefore has potential as a new drug for IBD.
Rights and permissions
About this article
Cite this article
Zhang, Y., Brenner, M., Yang, WL. et al. Recombinant human MFG-E8 ameliorates colon damage in DSS- and TNBS-induced colitis in mice. Lab Invest 95, 480–490 (2015). https://doi.org/10.1038/labinvest.2015.32
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/labinvest.2015.32
This article is cited by
-
Proteome-wide Mendelian randomization highlights AIF1 and HLA-DQA2 as targets for primary sclerosing cholangitis
Hepatology International (2024)
-
SLC26A3 (DRA) prevents TNF-alpha-induced barrier dysfunction and dextran sulfate sodium-induced acute colitis
Laboratory Investigation (2018)
-
RETRACTED ARTICLE: Adiponectin administration alleviates DSS-induced colonic inflammation in Caco-2 cells and mice
Inflammation Research (2018)
-
Role of milk fat globule-epidermal growth factor 8 in osteoimmunology
BoneKEy Reports (2016)
-
Phagocytosis of apoptotic cells in homeostasis
Nature Immunology (2015)