Abstract
Objective:
To establish the prevalence of neurodevelopmental disabilities among premature infants born in western Poland.
Study Design:
From 1999 to 2003, 640 extremely low birth weight (ELBW) and 777 very low birth weight (VLBW) infants were admitted to the tertiary care center in Poland. Discharged home were 302 and 671 of them, respectively. At 2 years corrected age, 165 ELBW and 285 VLBW children were diagnosed with neurosensory abnormality including cerebral palsy (CP) and evaluated with Bayley Scales of Infant Development II.
Result:
Mental developmental index (MDI) score <70 was recorded in 104 children (23.1%). There were 75 (16.7%) cases of CP, 8 (1.7%) deafness/hearing loss, 22 (4.8%) blindness and 14 (3.1%) of epilepsy. Patients with abnormal neurosensory status had significantly lower MDI compared with those with normal development (83.8±9 vs 54.7±10). Severe intraventricular haemorrhage, periventricular leucomalacia, ventricular dilatation and hydrocephalus were the most important risk factors for neurologic abnormality.
Conclusion:
Severe disability is more common among ELBW compared with VLBW infants and remains a major challenge for health-care providers.
Similar content being viewed by others
Introduction
As a result of improved quality of perinatal care, development and modernization of neonatal intensive care units, and implementation of the three level perinatal care, survival rates of premature infants in Poland improved significantly in recent years.1 Health-care providers were pushed to focus more on the growing population of children with history of severe prematurity and very low birth weight (VLBW). Number of recent studies regarding the improvement of perinatal and neonatal care indicated a decreased neonatal mortality but no actual decrease in frequency of cerebral palsy (CP) and other neurological disabilities.2, 3 CP is consistently the most common type of disability in children, with a prevalence of two cases per 1000 live births.3, 4
The goal of our study was to examine the neurosensory and cognitive status, to establish prevalence of neurological and developmental problems, and to assess the growth and neurodevelopmental outcome in the population of VLBW and extremely low birth weight (ELBW) infants born and cared for in the tertiary care center in western Poland at the age of corrected 2 years.
Methods
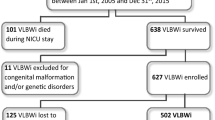
Between 1 January 1999 and 31 December 2003, 640 ELBW and 777 VLBW infants born at ⩽35 weeks of gestation were admitted to the neonatal intensive care unit of the tertiary care center of the University Hospital of the Medical University in Poznan, Poland. Of that group, 302 ELBW and 671 VLBW infants survived and were discharged home. Study group comprised 165 ELBW and 285 VLBW infants who reported to the follow-up clinic at 2 years corrected age (CA). Patients were stratified by birth weight (BW) and gestational age (GA). There were two BW strata: 500 to 999 g (n=165) and 1000 to 1500 g (n=285). All patients with BW >1500 g were excluded from the study. The GA of the study population ranged from 24 to 35 weeks of GA (24 to 26 weeks: n=105, 27 to 28 weeks: n=122 116, 29 to 30 weeks: n=116, 31 to 32 weeks: n=64 and 33 to 35 weeks: n=43). Number of known demographic, obstetric and neonatal risk factors for abnormal neurodevelopmental outcome was examined (Tables 1 and 2). Growth charts created on the basis of the anthropometric measurements of the newborns born in the Wielkopolska Region of Poland served as a reference for the assessment of the intrauterine growth.5 Patients with BW <10th percentile were considered small for GA (SGA).
The second edition of Bayley Scales of Infant Development was used as a tool to evaluate the development of the patients included in the study.6 Major neurologic abnormalities included CP (spastic diplegia, hemiplegia, tetraplegia), hypertonia and hypotonia, and mental developmental index (MDI) score <70. Shunt-dependent hydrocephalus without other neurologic abnormality was also considered impairment. Sensory abnormality included unilateral or bilateral blindness, deafness or hearing impairment.
Statistical analysis
Analysis of variance, Mann–Whitney U-test, Kruskal–Wallis test and logistic regression with Bonferroni correction was used for analysis of the data. Differences on categorical data were analyzed using the χ2 test and Fischer's exact test. Attributions co-variations were calculated by the Pearson correlation coefficients and the Spearmen's rank correlation. Statistical analysis was carried out using STATISTICA (StatSoft, Tulsa, OK, USA, version 6.0). The P<0.05 was considered statistically significant.
Results
Maternal demographics and study population characteristics are presented in Table 1. The BW for the entire study population ranged from 560 to 1500 g. Mothers older than 35 years were more likely to deliver the newborn with BW 500 to 749 g (P=0.01). Selected neonatal morbidities in the study population are presented in Table 2. The length of hospital stay was (mean±s.d.) 67±27 days and ranged from 9 to 218 days. Sixty-nine of the studied infants had been discharged to community hospitals for further treatment and continued care before discharge home. Data on the neurosensory outcomes in the study population by BW strata at CA 2 years of age is presented in Table 3. Of all studied outcomes, only the incidence of CP was significantly associated with BW (P=0.02). Figure 1 illustrates the incidence of CP stratified by GA. The difference between the patients with gestational 24 to 26 weeks versus those born at 27 to 28 weeks of gestation and higher was statistically significant (P=0.002). Of the entire study group at 2 years CA, only 3 patients were not ambulatory, but 36 required support in the form of braces or walker. Selected factors and variables associated with increased risk of CP are presented in Table 4. In our study population being outborn, intraventricular haemorrhage (IVH) grade III/IV, periventricular leucomalacia (PVL), ventricular dilatation, shunt-dependent hydrocephalus, respiratory distress syndrome complicated by air leak, prolonged mechanical ventilation (>14 days), surgical patient ductus arteriousus (PDA) ligation, retinopathy of prematurity (ROP) stage III and bronchopulmonary dysplasia (BPD) increased the risk of developing CP. Newborns with BW <1000 g had significantly lower Apgar score at 1 min (P<0.05), and low Apgar score at 1 min was significantly associated with higher risk of CP.
At 24 months CA, the mean (±s.d.) MDI score of the whole study population was 78.0±14.9, and the mean (±s.d.) psychomotor developmental index (PDI) score was 78.2±15.1. Figure 2 illustrates the results of MDI and PDI scores for different BW strata. MDI and PDI stratified by GA is presented in Figures 3a and b, respectively. The difference between the GA groups was statistically significant for both MDI and PDI (P<0.05). Patients whose mothers received prenatal steroids had significantly higher MDI scores (P=0.03). Delivery via cesarean section, and delivery in the tertiary care were also associated with better MDI scores (P=0.01, and P=0.00, respectively). Lower MDI scores (<70) were associated with the postnatal steroid use (P=0.000). Maternal education, premature rupture of membranes, multiple pregnancy, parity and presentation did not have a significant effect on the MDI score.
The mean (±s.d.) MDI score in the group with normal neurosensory status was 83.8 (±9.0). The group with abnormal neurosensory status had mean (±s.d.) MDI of 54.7 (±10.1). In that group of patients, 80 (88.9%) children had MDI <70, and only 1 child in that group had MDI ⩾85. Similar results were observed for PDI. The mean (±s.d.) PDI score in the group with abnormal neurosensory status was 53.8±9.4. PDI <70 was recorded for 79 (87.8%) children, and only 1 child had PDI ⩾85.
Of the 450 children included in the study population 104 (23.1%) were SGA. At corrected 2 years of age, 51.6% of the children born appropriate for gestational age (AGA) and 64.2% of those born SGA had weight below the 10th percentile. Head circumference measured at the same visit was below the 10th percentile in 54.4% of children. In the group of children with normal development, 50.0% had body weight and 53.5% had head circumference below 10th percentile. Among children with CP, 72.0% had body weight below the 10th percentile and 68.0% had head circumference below 10th percentile. At 2 years CA, both head circumference and weight were statistically different between children with and without CP (P=0.004 and P=0.006, respectively). Children born SGA were not at increased risk for developing CP (P=0.76).
Figure 4 illustrates the outcomes of all live—born infants, mortality and survival and follow-up at 2 years CA.
Discussion
This study showed that children in our study population have a high rate of neurosensory abnormalities. Somatic growth delay in early childhood was also a significant problem.
Data from different centers show that survival rate of ELBW and VLBW infants improved significantly with the introduction of the modern neonatal intensive, and is now approximately 80% of live births compared with only 20% reported 30 years ago.7, 8, 9, 10 Similar trend toward improved survival rate of those tiniest babies was also observed in Poland (survival of neonates with BW below 1000 g was 53.2% in 2007 compared with 43.3% in year 2000).1 Similarly, the results from our own unit show that the chances for survival in the group of tiniest neonates improved in the last two decades. In 1999, 18% of neonates born with BW 500 to 749 g and 52.4% of those with BW 750 to 999 g survived to discharge. In 2007, survival was 42.9 and 64.2% for the neonates born with BW 500 to 749 g and 750 to 999 g respectively (data not published). However, improved survival is often accompanied by increased incidence of abnormal neurological outcomes.2, 4, 7
The incidence of CP at 2 years CA in our study population was inversely proportional to GA and BW with the highest incidence in the group of the smallest patients with BW <1000 g. Our results are similar to those reported by Rutkowska et al.,11 who studied a cohort of patients born preterm in the province of Warsaw. Analysis of the data collected during EPIPAGE study also showed the results comparable to ours (at 5 years of age, 20 and 10% of children born at 24 to 27 weeks of gestation and 28 to 32 weeks of GA, respectively, had CP).12 However, in the study of Finnstrom et al.13 the overall incidence of CP for children with BW ⩽1000 g was only 7% compared with 22.4% in our unit. In the EPICure study, investigating the outcomes of children born in United Kingdom and Ireland at <26 weeks of gestation, 18% of the patients were diagnosed with CP at 30 months of age compared with 28.6% in studied group.14 Shorter experience in managing the tiniest neonates may be one the possible explanations for higher incidence of CP in our study population compared with other leading centers. Some investigators suggest a learning curve in the process of managing the very premature neonates, and longer experience translates into better outcomes.3, 4, 10
In our study population, quadriplegia was the most common form of CP followed by diplegia and hemiplegia. In contrast, Wood et al.14 reported that hemiplegia was the most prevalent form of CP in their population of patients born at <26 weeks of gestation. Their results are consistent with the trends observed in the last years. The percentage of patients with severe forms of CP decreased significantly, and some investigators believe that it is a reflection of both improved prenatal and neonatal care.3, 4, 10, 15 Higher than reported by other units, incidence of quadriplegia in spite of the comparable incidence of severe forms of intracranial pathologies (grade III and IV IVH, PVL) might be explained by the limitations of the imaging studies. Less severe forms of PVL cannot be easily visualized on head ultrasound. Since the conclusion of this study, now all neonates hospitalized in our unit with BW <1000 g have head magnetic resonance imaging performed before discharge home.
In keeping with the data published by other investigators, variables such as site of delivery (outside level III unit), low Apgar score at 1 min, BPD and severe cranial ultrasound abnormality (grade III/IV IVH, ventricular dilatation, shunt-dependent hydrocephalus and PVL) were associated with the increased the risk of CP in our study.16, 17, 18, 19, 20, 21 A significant number of the patients in our study population required surgical ligation of PDA. Constantinou et al.22 have previously suggested that surgical PDA ligation increases the risk of CP. We have found the same association in our study. Incidence of CP was higher in the group with surgical ligation compared with medical (Indomethacin) or no ligation.
Retinopathy of prematurity is one of the main causes of blindness in children. In 1999, half of the cases of blindness in children in Poland were secondary to ROP.23 Our results compare favorably with other units in Poland, but are worse than reported by the centers from other European countries and the United States. Incidence of blindness is in the middle of spectrum most commonly quoted in the literature.10, 14, 24, 25 One of the possible explanations for slightly worse ROP results in our unit in comparison with other leading centers is the fact that in the initial period of our study there were limited number of ophthalmologists experienced in recognizing and treating ROP. With longer experience in managing extremely immature neonates and better availability of laser therapy, our own results improved significantly in subsequent years.23 Mental and motor development in our study population was assessed using Bayley Scales of Infant Development II. As presented by Vohr et al.26 in their analysis, the outcomes may vary significantly even between units with seemingly comparable level of care. Our results are similar to what Hack et al.27 observed in their cohort of children born at <28 weeks of gestation, but worse than the patients who participated in the EPICure study.14 Prenatal steroids and delivery via cesarean section was associated with higher MDI. Neonates born via cesarean section had better 1′ Apgar score compared with those born vaginally. It is possible that mode of delivery is at least partially a reflection of the level of care. Often patients who delivered vaginally had no or limited prenatal care, while those born via cesarean section were closely supervised (data not published). Contrary to prenatal steroids, postnatal steroid use was associated with lower MDI scores in the group of patients with BW <1000 g and GA <27 weeks. Variables, such as, maternal education, vaginal bleeding, premature rupture of membranes, multiple pregnancy, parity and fetal presentation had no statistically significant effect on MDI score. Similar to the reports of other investigators, there was a significant difference between MDI and PDI scores of children with and without neurosensory deficits in our study.15, 27 Effective early after discharge intervention might be one of the possible explanations why our results in the group of children with neurosensory deficits are worse than reported by Hack et al.27 In Poland, high costs of transportation and living far away from the center of the early intervention program is in some cases a limiting factor in an access to the early therapy.
One of the important problems and challenges in the management of the extremely premature patients is poor weight gain and inadequate growth including head circumference persisting long past beyond neonatal period. Children born SGA were significantly more likely to have growth failure compared with those born AGA. Only few SGA patients had weight 50th percentile at 2-year of follow-up, and catch-up growth was practically absent in that group. Hack et al.28 also concluded that in some cases catch-up growth is very delayed (school age or late teenage years) or may not occur at all. Compared with studies of Peterson et al.29 who also studied a cohort of patients with BW <1500 g, the percentage of patients with abnormally low head circumference in our study population was much higher. Nutrition that is one of the known factors affecting the growth and head circumference, and could potentially explain the observed difference, was not studied in our group. Follow-up rate for the entire group was 48.1% (54.3% ELBW and 42.3% VLBW).
One of the important limitations of our study is the significant number of patients lost to follow-up. We can only speculate, with the support of some data from the literature that good outcomes deterred parents from overcoming the barriers for follow-up that they felt not necessary.30
Conclusion
Our results show that improved survival of neonates with ELBW and VLBW is accompanied by high rates of abnormal neurological outcomes. Inborn patients in our study population had significantly lower incidence of CP. Every effort should be made to assure a delivery of all VLBW and ELBW neonates in the specialized level III perinatal centers. Surgical PDA ligation was associated with the higher risk of CP in our study population. The protocol of early prophylactic administration of indomethacin, which was introduced in our unit since the conclusion of the study might decrease the incidence of PDA requiring surgical ligation and incidence of CP. Prolonged mechanical ventilation increases the risk of CP. We speculate that much wider use of gentle ventilation in our unit might result in improved neurodevelopmental outcome.
References
Yearbook of Poland. (http://www.stat.gov.pl) 2008; 330–333.
Wilson-Costello D, Friedman H, Minich N, Fanaroff AA, Hack M . Improved survival rates with increased neurodevelopmental disability for extremely low birth weight infants in the 1990s. Pediatrics 2005; 115: 997–1003.
Platt MJ, Cans C, Johnson A, Surman G, Topp M, Torrioli MG et al. Trends in cerebral palsy among infants of very low birthweight (<1500 g) or born prematurely (<32 weeks) in 16 European centres: a database study. Lancet 2007; 369: 43–50.
Robertson CM, Watt MJ, Yasui Y . Changes in the prevalence of cerebral palsy for children born very prematurely within a population-based program over 30 years. JAMA 2007; 297: 2733–2740.
Gadzinowski J, Kaliszewska-Drozdowska MD, Kosinska M, Mazela J, Stoinska B . Birth weight and gestational age of newborns from Wielkopolski and Lubuski regions. Ginekol Pol 2003; 74: 186–192.
Bayley N . Manual for the Bayley Scales of Infant Development. 2nd edn. Psychological Corporation: San Antonio, TX, 1993.
Fanaroff AA, Hack M, Walsh MC . The NICHD neonatal research network: changes in practice and outcomes during the first 15 years. Semin Perinatol 2003; 27: 281–287.
Horbar JD, Badger GJ, Carpenter JH, Fanaroff AA, Kilpatrick S, LaCorte M et al. Trends in mortality and morbidity for very low birth weight infants, 1991–1999. Pediatrics 2002; 110: 143–151.
Larroque B, Breart G, Kaminski M, Dehan M, Andre M, Burguet A et al. Survival of very preterm infants: EPIPAGE, a population based cohort study. Arch Dis Child Fetal Neonatal Ed 2004; 89: F139–F144.
Wilson-Costello D, Friedman H, Minich N, Siner B, Taylor G, Schluchter M et al. Improved neurodevelopmental outcomes for extremely low birth weight infants in 2000–2002. Pediatrics 2007; 119: 37–45.
Rutkowska M, Helwich E, Szamotulska K, Rudzinska-Chazan M, Kulakowska Z, Jeziorek A et al. A two-year evaluation of the development of preterm babies born in the region of Warsaw: a prospective cohort study prematurities. Med Wieku Rozwoj 2005; 9: 249–269.
Foix-L’Helias L, Marret S, Ancel PY, Marchand L, Arnaud C, Fresson J et al. Impact of the use of antenatal corticosteroids on mortality, cerebral lesions and 5-year neurodevelopmental outcomes of very preterm infants: the EPIPAGE cohort study. BJOG 2008; 115: 275–282.
Finnstrom O, Otterblad Olausson P, Sedin G, Serenius F, Svenningsen N, Thiringer K et al. Neurosensory outcome and growth at three years in extremely low birthweight infants: follow-up results from the Swedish national prospective study. Acta Paediatr 1998; 87: 1055–1060.
Wood NS, Marlow N, Costeloe K, Gibson AT, Wilkinson AR . Neurologic and developmental disability after extremely preterm birth. EPICure Study Group. N Engl J Med 2000; 343: 378–384.
Vohr BR, Wright LL, Poole WK, McDonald SA . Neurodevelopmental outcomes of extremely low birth weight infants <32 weeks’ gestation between 1993 and 1998. Pediatrics 2005; 116: 635–643.
Grether JK, Nelson KB, Emery 3rd ES, Cummins SK . Prenatal and perinatal factors and cerebral palsy in very low birth weight infants. J Pediatr 1996; 128: 407–414.
Saigal S, Rosenbaum P, Hattersley B, Milner R . Decreased disability rate among 3-year-old survivors weighing 501 to 1000 grams at birth and born to residents of a geographically defined region from 1981 to 1984 compared with 1977 to 1980. J Pediatr 1989; 114: 839–846.
Skidmore MD, Rivers A, Hack M . Increased risk of cerebral palsy among very low-birthweight infants with chronic lung disease. Dev Med Child Neurol 1990; 32: 325–332.
Wilson-Costello D, Borawski E, Friedman H, Redline R, Fanaroff AA, Hack M . Perinatal correlates of cerebral palsy and other neurologic impairment among very low birth weight children. Pediatrics 1998; 102: 315–322.
Furman L, Hack M, Watts C, Borawski-Clark E, Baley J, Amini S et al. Twenty-month outcome in ventilator-dependent, very low birth weight infants born during the early years of dexamethasone therapy. J Pediatr 1995; 126: 434–440.
Vohr BR, Wright LL, Dusick AM, Mele L, Verter J, Steichen JJ et al. Neurodevelopmental and functional outcomes of extremely low birth weight infants in the National Institute of Child Health and Human Development Neonatal Research Network, 1993–1994. Pediatrics 2000; 105: 1216–1226.
Constantinou JC, Adamson-Macedo EN, Mirmiran M, Ariagno RL, Fleisher BE . Neurobehavioral assessment predicts differential outcome between VLBW and ELBW preterm infants. J Perinatol 2005; 25: 788–793.
Gotz-Wieckowska ASA, Gadzinowski J . Wystêpowanie, przebieg i wyniki leczenia retinopatii wczesniaków w Katedrze i Klinice Neonatologii w Poznaniu. Postepy w neonatologii 2006; 1: 109–111.
Gilbert C, Fielder A, Gordillo L, Quinn G, Semiglia R, Visintin P et al. Characteristics of infants with severe retinopathy of prematurity in countries with low, moderate, and high levels of development: implications for screening programs. Pediatrics 2005; 115: e518–e525.
Costeloe K, Hennessy E, Gibson AT, Marlow N, Wilkinson AR . The EPICure study: outcomes to discharge from hospital for infants born at the threshold of viability. Pediatrics 2000; 106: 659–671.
Vohr BR, Wright LL, Dusick AM, Perritt R, Poole WK, Tyson JE et al. Center differences and outcomes of extremely low birth weight infants. Pediatrics 2004; 113: 781–789.
Hack M, Wilson-Costello D, Friedman H, Taylor GH, Schluchter M, Fanaroff AA . Neurodevelopment and predictors of outcomes of children with birth weights of less than 1000 g: 1992–1995. Arch Pediatr Adolesc Med 2000; 154: 725–731.
Hack M, Schluchter M, Cartar L, Rahman M, Cuttler L, Borawski E . Growth of very low birth weight infants to age 20 years. Pediatrics 2003; 112: e30–e38.
Peterson J, Taylor HG, Minich N, Klein N, Hack M . Subnormal head circumference in very low birth weight children: neonatal correlates and school-age consequences. Early Hum Dev 2006; 82: 325–334.
Castro L, Yolton K, Haberman B, Roberto N, Hansen NI, Ambalavanan N et al. Bias in reported neurodevelopmental outcomes among extremely low birth weight survivors. Pediatrics 2004; 114 (2): 404–410.
Acknowledgements
We thank Agnieszka Montgomery our psychologist for Bayley developmental panel.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
This work is licensed under the Creative Commons Attribution-NonCommercial-No Derivative Works 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/3.0/
About this article
Cite this article
Stoinska, B., Gadzinowski, J. Neurological and developmental disabilities in ELBW and VLBW: follow-up at 2 years of age. J Perinatol 31, 137–142 (2011). https://doi.org/10.1038/jp.2010.75
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jp.2010.75
Keywords
This article is cited by
-
Neurologic Complications of Poverty: the Associations Between Poverty as a Social Determinant of Health and Adverse Neurologic Outcomes
Current Neurology and Neuroscience Reports (2021)
-
The Impact of Psoriasis on Pregnancy Outcomes
Journal of Investigative Dermatology (2012)
-
Does a Medical Home Influence the Effect of Low Birthweight on Health Outcomes?
Maternal and Child Health Journal (2012)