Abstract
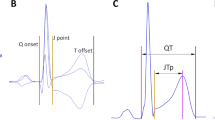
The duration of the QT interval as measured by 12-lead electrocardiography is a measure of myocardial repolarization and is widely used to describe cardiac abnormalities, to determine the presence of cardiac toxicity and to evaluate drug safety. In hypertension, the QT interval is a predictor of the risk of both coronary events and cardiovascular death, after adjusting for the effects of additional risk factors. The mechanism of QT interval prolongation is multifactorial and includes cardiomyocyte hypertrophy and increased left ventricular mass, with accompanying changes in left ventricular transmural dispersion of repolarization, as well as changes in the tone of the autonomic nervous system of some patients with hypertension and mechano-electrical feedback, although this mechanism is less likely. Antihypertensive drugs vary in their effect on QT interval duration. The mechanisms underlying their effect depend on changes in left ventricular mass and autonomic nervous system tone, as well as changes in the activity of cardiac ion channels. Although blood pressure reduction is the primary goal of antihypertensive drug therapy and although the choice of antihypertensive drug treatment regimens varies among different individuals, the data regarding the disparate effects of antihypertensive drugs on the duration of the QT interval warrant consideration when implementing long-term pharmacotherapy for hypertension.
Similar content being viewed by others
Introduction
Abnormalities of ventricular repolarization are an important component of both ECG diagnostics and medical decision-making. The duration of the QT interval as measured by 12-lead electrocardiography is a measure of repolarization and is widely used to describe cardiac abnormalities, to determine the presence of cardiac toxicity or to evaluate drug safety.1, 2, 3, 4, 5, 6 QT interval duration is a predictor of the occurrence of cardiovascular events,7, 8, 9, 10, 11 and increased QT duration is associated with the risk of sudden cardiac death in the hypertensive population, even among individuals without clinically recognized cardiac disease.12 The increased risk of cardiovascular events, including sudden cardiac death, is a well-described consequence of sustained arterial hypertension.13 Even small, non-specific abnormalities of repolarization may increase the risk of cardiovascular events in the hypertensive population.14
In subjects with uncomplicated hypertension, a prolonged QT interval (corrected for heart rate) carries a twofold increase in the risk of coronary events and cardiovascular death, after adjusting for the effects of age, sex, diabetes mellitus, serum cholesterol level, serum creatinine level, smoking, left ventricular hypertrophy (LVH) and 24-h systolic blood pressure (BP).15 In a hypertensive population with electrocardiographic LVH, an analysis of maximum QT intervals identified persons at an increased risk for cardiac mortality despite effective BP-lowering treatment.9 Prolonged QT intervals are a marker of cardiovascular morbidity and mortality in patients with resistant hypertension, even after taking into account traditional cardiovascular risk factors, including both hypertension and LVH.16 The question arises as to whether hypertension modulates the QT interval and therefore accounts for a mechanism by which hypertension increases the likelihood of the occurrence of fatal cardiac events despite the effects of antihypertensive agents on the QT interval.
The electrophysiological basis of the QT interval
The electrophysiological basis of the QT duration is the duration of the action potential of a single ventricular cardiomyocyte, as well as the presence of electrical heterogeneity in the form of either transmural or trans-septal dispersion of ventricular repolarization in an intact heart.17 An increase in duration of the QT interval may reflect an increase in the duration of the action potential in specific regions of the ventricle. Such increases reflect either an increase in inward current or a decrease in outward current.18 Most drugs that cause an increased QT interval duration prolong cardiomyocyte action potentials by blocking potassium channels.19 The potassium channels responsible for both the rapid (IKr) and the slow (IKs) activating components of the delayed rectifier potassium currents, in particular, modulate cardiac repolarization and have been implicated in both long QT syndrome and other forms of arrhythmia.20
In hypertension, the QT interval is viewed as a non-specific marker of cardiac pathology. The underlying cause of the prolongation of cardiac repolarization in hypertension remains controversial and reflects a large number of cardiac physiologic and pathophysiologic conditions. The dependence of the duration of the repolarization on the activity of potassium channels and potassium currents suggests that potassium currents should figure prominently in the relationship between the QT interval and hypertension. Interestingly, there is very little data regarding the relationship between QT and hypertension; however, the data suggest that a relationship between KATP channel polymorphisms and left ventricular size exists among hypertensive individuals.21
The mechanisms underlying the effect of hypertension on the QT interval
Left ventricular hypertrophy
Electrical remodeling in the setting of essential hypertension, as well as the consequent development of LVH, likely contributes to the pathophysiology of cardiac arrhythmias.22 From a clinical perspective, it may translate into a propensity for individuals to suffer sudden cardiac death.23, 24 Increased left ventricular mass (LVM) is associated with changes in cardiac repolarization.25, 26, 27 Several measures of cardiac repolarization, including QT duration, QT duration corrected for heart rate (QTc), QT variability and QT dispersion (the interlead differences in the duration of the QT interval) are associated with hypertensive heart disease, suggesting that increased LVM may modulate any QT parameter. Two decades ago, QT dispersion was enthusiastically studied as if it were an electrophysiological holy grail that explained the impacts of either diseases or drugs on the heart.28 Previous investigations describing QT abnormalities in the hypertensive population focused primarily on QT dispersion, whereas others focused on QT duration. As the two phenomena are linked by their relationship with both LVH and BP in the majority of studies,8, 25, 26, 27, 29, 30, 31, 32, 33, 34, 35, 36 they will be discussed together in this review.
Piccirillo and colleagues37 described their findings of QT abnormalities in hypertensive subjects with LVH and speculated that greater ventricular mass results in decreased repolarization synchronization in myocardial cells. This may explain not only the QT interval prolongation observed in previous studies but also the increased QT dispersion noted in said studies;8, 25, 26, 27, 32, 38, 39, 40 however, clear evidence confirming this interesting hypothesis is lacking. Another explanation for the relationship between QT changes and LVH is that cardiomyocyte hypertrophy is often associated with changes in either the expression or the activity of ion channels (primarily potassium and calcium channels). These changes may affect the action potential durations41 and include specific alternations such as decreased potassium currents, dysregulation of intracellular calcium and gap junction dysfunction.42, 43, 44 Other factors may contribute to QT prolongation in the setting of LVH, including fibrosis, myocardial ischemia and diabetes mellitus.45, 46
Although LVH is an important factor in numerous cardiac diseases, several studies have noted the presence of QT prolongation in hypertensive patients, both with LVH and without LVH,37, 39 although this issue remains controversial.40 Uncomplicated hypertensive patients may have increased cardiac mass;47 therefore, the link between LVM and the QT interval may exist prior to the development of LVH. Experimental models have confirmed the presence of QT prolongation in the setting of hypertension, particularly in spontaneously hypertensive rats and models of both essential hypertension and LVH.3, 48, 49 It remains unclear when hypertension-induced increases in LVM and QT prolongation begin to manifest, and the extent to which other factors such as cardiac fibrosis, diabetes mellitus or ischemic heart disease accelerate the changes in the QT interval in the setting of LVH.
Changes in the duration of the QT interval secondary to alterations of the autonomic nervous system in the setting of hypertension
Prolongation of the QT interval has been linked to both alterations in sympathetic drive50 and imbalances of the autonomic nervous system.33, 51 Systemic catecholamines may alter ventricular repolarization52 and prolong the duration of the QT interval, and also increase QT dispersion, even among healthy subjects.52, 53 The regulation of the QT interval by the autonomic nervous system has been demonstrated by several studies. Despite their comparable BPs, hypertensive patients with anxiety exhibit significantly longer QT intervals and broader QT dispersion than their counterparts without anxiety.33 This finding is supported by evidence that the magnitude of beat-to-beat QT variability is related to cardiac sympathetic activation in hypertensive patients.47
Marfella and colleagues54 determined that both the prolongation of cardiac repolarization and morning sympathetic overactivity coexist in hypertensive patients at the time of each patient’s maximum morning BP. Interestingly, subjects with essential hypertension and increased LVM exhibited increased QT durations primarily during the awake period, including the time period during which morning catecholamine levels and BP were each elevated, but not later in the day or at night. The reason for these findings is complex because not all individuals with hypertension exhibit increased catecholamine synthesis and release, nor are they as sensitive to the effects of catecholamines on the heart.55, 56
Changes in QT interval duration secondary to acute alterations in BP
In experimental hypertension, elevated BP correlates with QT prolongation independent of associated LVM.3 This finding suggests that ventricular loading may affect ventricular repolarization. Indeed, decades ago, a mechano-electrical feedback mechanism57, 58 was implicated in action potential duration disturbances.59 The experimental data were confusing, however. Although, there appears to be agreement that mechano-electrical feedback modulates action potential duration in muscle preparations, this phenomenon may not apply to the entirety of the heart.58 Moreover, Dean and Lab57 demonstrated that BP reduction with nitroprusside is associated with increased action potential durations, a finding that opposes the contention that BP elevation prolongs the durations of action potentials. However, it has been suggested that the dynamicity of the duration of the QT interval is influenced by circadian BP patterns.60 It is widely known that circadian rhythms are involved in BP regulation.61 Moreover, Marfella and colleagues54 demonstrated that QT duration increases in conjunction with morning BP peaks in hypertensive individuals. Following chronic antihypertensive drug therapy, subsequent decreases in BP may be more strongly associated with reductions in the QT interval, as well as small decreases in LV mass.62 Given the controversy surrounding the experimental data,58 the mechanism underlying changes in the duration of the QT interval secondary to alterations in BP remain to be elucidated.
The effect of antihypertensive agents in the modulation of the duration of the QT interval
The ability of certain pharmacologic agents to alter the QT interval is well accepted.6, 19, 63, 64 Specific agents, primarily antiarrhythmic drugs, are able to reduce the duration of the QT interval via different pharmacological mechanisms.65, 66 The potential for antihypertensive drugs to alter the QT interval must be considered within the context of the effect of BP reduction on LV mass, as well as the effects of specific drugs on the autonomic nervous system, which in turn affects the duration of the QT interval and exerts additional effects on the heart. As suggested above, if LVH produces changes in the duration of the QT interval, one may expect that a reduction in LVM that coincides with a sustained BP decrease will be important in decreasing the duration of the QT interval. Indeed, the LIFE study demonstrated that antihypertensive therapy reduced the QT interval and suggested that this occurred exclusively in patients who experienced a concomitant reduction in their echocardiographic LVH.32 LVH regression secondary to antihypertensive drug therapy has also been associated with a reduced QTc dispersion and QTc variability, which some investigators have linked to the development of cardiac arrhythmias.67 Antihypertensive drugs are not equivalent in their modulation of the QT interval despite similar antihypertensive (that is, BP reducing) and antihypertrophic (that is, cardiac mass reducing) medications.
These discordances between drugs and their ability to reduce BP and affect changes in the QTc interval have been reported clinically in hypertensive populations,31, 67, 68, 69, 70 and have been documented in animal models of hypertension.3 Another consideration is that the time course for changes in the QT interval is different than those regarding changes either in LV mass or in BP. Chronic treatment with the angiotensin-converting enzyme inhibitor (ACEi) enalapril elicited a significant reduction in BP following 8 weeks of treatment; a reduction in LVM was evident following 1 year of treatment, and a reduction in the duration of the QT interval was evident following 3 years of treatment.30 These findings suggest that hypertrophy may prolong the duration of an action potential; however, its regression may not immediately result in AP normalization.71
In a comparative study involving amlodipine and irbesartan, both drugs decreased BP, but only irbesartan decreased the duration of the QT interval.31 Interestingly, a positive correlation between decreased BP and decreased QT duration was noted in this study. Similar results were described by Fogari et al.68 in hypertensive patients with type 2 diabetes mellitus, without LVH on echocardiography. Despite similar BP-lowering effects, the direct renin inhibitor aliskiren, but not the calcium channel blocker (CCB) amlodipine, decreased the duration of the QT interval in these patients. Therefore, pharmacotherapies with similar effects on hypertension will not exert identical effects on cardiac repolarization. When considering the effects of antihypertensive treatment, we must consider not only their effects on BP and LVH but also their effects on the rest of the body. Beta-blockers (BB), ACEi and angiotensin receptor blockers (ARB) may affect the sympathetic system, and may therefore potentiate the positive effects of BP control and LVM reduction. Diuretics may affect electrolyte balance,72 which impacts the QT interval. Dihydropyridine CCB may potentially activate the sympathetic nervous system;73 therefore, the positive effects exerted by this medication on BP may be masked by its additional pharmacological effects.
ACE inhibitors and ARBs
The majority of studies have provided evidence that drugs inhibiting the renin-angiotensin-aldosterone system, particularly ACEis and ARBs, exert beneficial effects on the duration of the QT interval (Table 1). These drugs reduce the QT interval.30, 31, 32, 69, 70 Experimental studies have also demonstrated that ACEis reduce the QT interval.1, 3, 48, 74 The effects of either an ACEi or an ARB on the QT interval are exerted via mechanisms that regulate also the QT interval. The effects of ACEis and ARBs on cardiac hypertrophy and cardiac fibrosis may have proven to be important effects.75, 76, 77, 78 ACEis and ARBs may affect the electrophysiological mechanisms altered by angiotensin.79 The inhibitory effect exerted by angiotensin II on the rapidly activating components of delayed rectifier potassium current (IKr) has been described previously and may result in prolonged action potential duration.80
One of the principal mechanisms that contributes to cardiac electrical disturbances is the dysregulation of intracellular calcium cycling.81 The intracellular calcium within the sarcoplasmic reticulum is released via ryanodine receptors (RyR2) during systole and is restored during diastole via the sarco/endoplasmic reticulum Ca2+ ATPase (SERCA2a) pump.82 In heart failure, that is, at a high angiotensin II state, both SERCA2a and RyR2 are suppressed.83, 84 The decrease in SERCA2a activity, which is triggered by angiotensin II, may lead to increased diastolic calcium and arrhythmogenesis. Indeed, this effect has been described previously in the setting of various pathological conditions, including hypertensive cardiomyopathy, in which the administration of either ACEIs or ARBs normalizes the abnormal intracellular calcium handling and increases SERCA2a expression.85, 86 Whether these ACEis or ARBs antagonize the effect of angiotensin on SERCA2a remains unclear.
Aldosterone has been linked to cardiac hypertrophy with prolongation of the QT interval, as well as electrical instability. This has been explained by the downregulation of the potassium channels responsible for the repolarization currents (Ito, IK1, Ikur), as well as the downregulation of L-type Ca2+ channels, which suggest that aldosterone exerts direct proarrhythmic effects.87, 88 Aldosterone antagonists, eplerenone in animals87 and spironolactone in humans,89 have been found to modulate the duration of the QT interval. Another contributing and potentially crucial mechanism of reducing the QT interval following ACEi and ARB therapy is the modulation of autonomic nervous system function by decreasing sympathetic drive. Angiotensin II modulates the autonomic nervous system via various mechanisms, including the release of catecholamines, the stimulation of both the systemic and the peripheral sympathetic nervous system, and the modulation of centrally regulated vagal tone.31, 90, 91 Consequently, drugs reducing the influence of either angiotensin or aldosterone may exert significant benefits by reducing sympathetic drive and will likely also exert beneficial effects regarding QT interval regulation.
Beta-blockers
Both sympathetic drive and catecholamines alter the electrical activity of the ventricles via the modulation of repolarization,53, 92 and play a crucial role in the development of hypertension in specific patients with hypertension. Beta-adrenergic stimulation prolongs the QT interval in some cases as a result of the induction of myocardial ischemia.5 Beta-blockade may also decrease the duration of the QT interval and normalize ventricular repolarization in a dose-dependent manner, without having any effect on either LVM or BP.29, 93, 94
Although the effects of beta-blockade on the QT interval in the setting of long QT syndrome appears to be minimal,95 previous studies have suggested that the potential of this drug class to reduce the QT interval may also benefit hypertensive patients (Table 2). Particularly, clinical studies involving carvedilol,96 atenolol69 and nadolol,97 as well as bisoprolol,70 demonstrated effects in patients with prolonged QT intervals. Carvedilol and its analogues, in particular, may have additional antiarrhythmic properties.98, 99 Its modulation of cardiac electrogenesis appears to be complex, and may be related to a combination of beta-blocking effects and the modulation of a variety of ion channels (including the direct inhibition of ryanodine receptors) and currents. Additionally, this hypothesis appears to be supported by its antioxidant, antiischemic and antihypertrophic effects.100 Controversially, some experimental studies in the past demonstrated QT prolongation following the administrations of BBs.101, 102 The ability of BBs to shorten the duration of the QT interval, which has made possible their use in the treatment of long QT syndrome,103 strengthens the case that BBs reduce the duration of the QT interval. The QT-modulating actions of BB may be an additional benefit that complements their antihypertensive effects.
Calcium channel blockers
Dihydropyridine CCBs do not appear to exert a beneficial effect on QT interval duration (Table 3). Historically, the CCB bepridil is known to carry risks of both QT prolongation104 and the potentially fatal arrhythmia torsade de pointes.105 This may be the result of its concomitant actions on potassium channels.36, 105 There is some clinical evidence that dihydropyridine CCBs may prolong the QT interval in subjects with normal sinus node function;106 experimental data support this finding.3 When comparing various antihypertensive treatments in a hypertensive population, CCBs often fail to reduce cardiac repolarization abnormalities despite their ability to reduce both BP and LVH31, 68, 70, 107 and may actually trigger QT prolongation and subsequent arrhythmias in hypertensive subjects.108
CCBs mediate their antihypertensive effects via vasodilatation by blocking the L-type calcium channels found in smooth muscle cells. However, their overall beneficial efficacy may be significantly decreased by counter-regulatory cardiac and renal activation because of baroreflex mechanisms.109 Therapy with dihydropyridine CCBs increases norepinephrine plasma concentrations, although differences in intensity have been described when comparing particular agents and formulations.73, 109, 110, 111, 112 De Champlain et al.110 observed a transient increase in norepinephrine levels with a slow-release nifedipine preparation, and a sustained increase in norepinephrine levels following chronic treatment with amlodipine, suggesting the existence of differences in sympathetic activation between the two preparations. Similarly, Leenen et al.112 noted that extended release felodipine significantly increased supine and standing plasma noradrenaline, which was not observed with either sustained release nifedipine or the ACEi enalapril. The interaction between CCBs and the sympathetic and parasympathetic nervous systems is also important. Karas et al.113 reported increased sympathetic activity during the day and decreased parasympathetic activity during the night following therapy with amlodipine; an effect that correlated with increased plasma norepinephrine levels. In contrast, the ARB telmisartan increased parasympathetic activity without affecting the norepinephrine levels during both the night and the day, whereas ramipril increased parasympathetic activity only during the day.114 Only CCBs that elicit a concomitant blockade of the N-type calcium channels located at sympathetic nerve endings, which enables them to directly inhibit sympathetic neurotransmitter release and suppress sympathetic activity, may avoid reflex activation.114, 115 Interestingly, only these CCBs may successfully decrease cardiac repolarization.116
An alternative explanation for unfavorable effects of CCBs on repolarization is their possible effects on potassium channels, as dihydropyridines not only suppress Ca2+ entry but also exert inhibitory effects on potassium channels.117, 118 By contrast, the nondihydropyridine CCB verapamil shortened the duration of the QT interval, whereas diltiazem did not change the duration of the QT interval in the setting of angina pectoris,104 and appeared to prolong the interval in a small sample of hypertensive patients119 (Table 3). Verapamil significantly shortened the QT interval at low heart rates in patients with structural normal hearts within 2 months after oral verapamil was prescribed for paroxysmal atrioventricular nodal re-entrant tachycardia.120 Verapamil reduces both the action potential duration and the transmural dispersion of repolarization via direct action on the heart,121, 122 as well as via central nervous system action, which mediates decreases in BP.123 There is no evidence regarding the pro-arrhythmogenic potential of dihydropyridine CCBs. Any pro-arrhythmogenic risk is likely to be marginal, as the blockade of L-type calcium current (ICa, L) may subsequently reduce intracellular calcium release and early after-depolarization, and suppress ventricular arrhythmogenesis.124, 125 The antiarrhythmic effects of both verapamil and diltiazem are well known.
Diuretics
The effect of diuretics on the QT interval is complex. Some types of diuretics exert direct effects on the heart, as demonstrated by their actions in isolated cardiomyocytes,126 and these effects may be observed in the setting of LVH.127 Diuretics, particularly loop diuretics and thiazide diuretics, produce electrolytic disturbances such as hypokalemia and hypomagnesemia.72 These electrolyte disturbances may result in QT interval prolongation. The potassium-sparing diuretic amiloride reduced both the duration of the QTc and dispersion, and increased serum potassium concentrations in patients with NYHA II-III.128 Similarly, when added to an ACEi or an ARB, both spironolactone and amiloride shortened the QTc interval concomitantly to reduction of cardiac fibrosis and increased serum potassium levels in stroke survivors.129 When comparing diuretics with other antihypertensive agents, they either exert no effect70 or occasionally prolong repolarization130, 131 (Table 4). Indapamide131, 132, 133 and furosemide130 have each been associated with both QT prolongation and the subsequent development of torsade de pointes or Brugada syndrome or syncope. The net effect of diuretics on the duration of the QT interval is the net effect of their favorable actions on BP32 and their effects on electrolyte homeostasis.
Centrally acting antihypertensive agents
Consistent with the evidence that sympathetic activity plays an important role in the regulation of the duration of the QT interval, central alpha-2 agonists would be expected to reduce the QT interval. However, only limited evidence supporting this exists in the literature. Clonidine attenuates QT prolongation in some experimental models of rat134 and rabbit,135 and its effects on QT variability have been suggested in humans.136 The relationship between these effects and BP remains questionable. Even less information is available regarding QT modulation by imidazoline receptor agonists (moxonidine, rilmenidine).
Direct vasodilators
Direct vasodilators may also have an influence on QT interval measurements,34 but a link to their BP-lowering effects is missing. Nicorandil, in particular, shortens ventricular repolarization if prolonged experimentally137 and modulates QT interval changes in humans.35, 138 However, nicorandil acts directly on ATP-sensitive K+ channels; this action is predictable and is likely not associated with BP changes.
Conclusion
The observation that a prolonged QT interval (corrected for heart rate) increases the risk of both coronary events and cardiovascular death in hypertensive patients, even after adjusting for the effects of other cardiovascular risk factors,15 should serve as an incentive for clinicians to focus their attention on the QT interval in the assessment of hypertension. The mechanism underlying QT interval prolongation is multifactorial and includes cardiomyocyte hypertrophy, increased LVM, with consequent changes in left ventricular transmural dispersion of the repolarization, as well as changes in autonomic nervous system tone in patients with hypertension, and less likely a mechano-electrical feedback. Although BP reduction is the primary goal of antihypertensive drug therapy, and the type of antihypertensive drug treatment varies in different individuals, the data regarding the disparate effects of antihypertensive drugs warrant consideration when planning long-term pharmacotherapy for hypertension.
References
Cernecka H, Ochodnicka-Mackovicova K, Kucerova D, Kmecova J, Nemcekova V, Doka G, Kyselovic J, Krenek P, Ochodnicky P, Klimas J . Enalaprilat increases PPARbeta/delta expression, without influence on PPARalpha and PPARgamma, and modulate cardiac function in sub-acute model of daunorubicin-induced cardiomyopathy. Eur J Pharmacol 2013; 714: 472–477.
Jankyova S, Kmecova J, Cernecka H, Mesarosova L, Musil P, Brnoliakova Z, Kyselovic J, Babal P, Klimas J . Glucose and blood pressure lowering effects of Pycnogenol(R) are inefficient to prevent prolongation of QT interval in experimental diabetic cardiomyopathy. Pathol Res Pract 2012; 208: 452–457.
Klimas J, Stankovicova T, Kyselovic J, Bacharova L . Prolonged QT interval is associated with blood pressure rather than left ventricular mass in spontaneously hypertensive rats. Clin Exp Hypertens 2008; 30: 475–485.
Klimas J, Vaja V, Vercinska M, Kyselovic J, Krenek P . Discrepant regulation of QT (QTc) interval duration by calcium channel blockade and angiotensin converting enzyme inhibition in experimental hypertension. Basic Clin Pharmacol Toxicol 2012; 111: 279–288.
Kmecova J, Klimas J . Heart rate correction of the QT duration in rats. Eur J Pharmacol 2010; 641: 187–192.
Rabkin SW . Aging effects on QT interval: Implications for cardiac safety of antipsychotic drugs. J Geriatr Cardiol 2014; 11: 20–25.
Day CP, McComb JM, Campbell RW . QT dispersion: an indication of arrhythmia risk in patients with long QT intervals. Br Heart J 1990; 63: 342–344.
Dimopoulos S, Nicosia F, Turini D, Zulli R . Prognostic evaluation of QT-dispersion in elderly hypertensive and normotensive patients. Pacing Clin Electrophysiol 2009; 32: 1381–1387.
Oikarinen L, Nieminen MS, Viitasalo M, Toivonen L, Jern S, Dahlof B, Devereux RB, Okin PM . LIFE Study Investigators. QRS duration and QT interval predict mortality in hypertensive patients with left ventricular hypertrophy: the Losartan Intervention for Endpoint Reduction in Hypertension Study. Hypertension 2004; 43: 1029–1034.
Panikkath R, Reinier K, Uy-Evanado A, Teodorescu C, Gunson K, Jui J, Chugh SS . Electrocardiographic predictors of sudden cardiac death in patients with left ventricular hypertrophy. Ann Noninvasive Electrocardiol 2013; 18: 225–229.
Schouten EG, Dekker JM, Meppelink P, Kok FJ, Vandenbroucke JP, Pool J . QT interval prolongation predicts cardiovascular mortality in an apparently healthy population. Circulation 1991; 84: 1516–1523.
Siscovick DS, Raghunathan TE, Rautaharju P, Psaty BM, Cobb LA, Wagner EH . Clinically silent electrocardiographic abnormalities and risk of primary cardiac arrest among hypertensive patients. Circulation 1996; 94: 1329–1333.
Rabkin SW, Mathewson FA, Tate RB . Long term changes in blood pressure and risk of cerebrovascular disease. Stroke 1978; 9: 319–327.
Schillaci G, Pirro M, Pasqualini L, Vaudo G, Ronti T, Gemelli F, Marchesi S, Reboldi G, Porcellati C, Mannarino E . Prognostic significance of isolated, non-specific left ventricular repolarization abnormalities in hypertension. J Hypertens 2004; 22: 407–414.
Schillaci G, Pirro M, Ronti T, Gemelli F, Pucci G, Innocente S, Porcellati C, Mannarino E . Prognostic impact of prolonged ventricular repolarization in hypertension. Arch Intern Med 2006; 166: 909–913.
Salles GF, Cardoso CR, Muxfeldt ES . Prognostic value of ventricular repolarization prolongation in resistant hypertension: a prospective cohort study. J Hypertens 2009; 27: 1094–1101.
Antzelevitch C . Ionic, molecular, and cellular bases of QT-interval prolongation and torsade de pointes. Europace 2007; 9 (Suppl 4): iv4–15.
Roden DM . Cellular basis of drug-induced torsades de pointes. Br J Pharmacol 2008; 154: 1502–1507.
Roden DM . Drug-induced prolongation of the QT interval. N Engl J Med 2004; 350: 1013–1022.
Guo J, Wang T, Yang T, Xu J, Li W, Fridman MD, Fisher JT, Zhang S . Interaction between the cardiac rapidly (IKr) and slowly (IKs) activating delayed rectifier potassium channels revealed by low K+-induced hERG endocytic degradation. J Biol Chem 2011; 286: 34664–34674.
Reyes S, Terzic A, Mahoney DW, Redfield MM, Rodeheffer RJ, Olson TM . K(ATP) channel polymorphism is associated with left ventricular size in hypertensive individuals: a large-scale community-based study. Hum Genet 2008; 123: 665–667.
Armoundas AA, Wu R, Juang G, Marban E, Tomaselli GF . Electrical and structural remodeling of the failing ventricle. Pharmacol Ther 2001; 92: 213–230.
Messerli FH . Hypertension and sudden cardiac death. Am J Hypertens 1999; 12: 181S–188S.
Straus SM, Kors JA, De Bruin ML, van der Hooft CS, Hofman A, Heeringa J, Deckers JW, Kingma JH, Sturkenboom MC, Stricker BH, Witteman JC . Prolonged QTc interval and risk of sudden cardiac death in a population of older adults. J Am Coll Cardiol 2006; 47: 362–367.
Chapman N, Mayet J, Ozkor M, Lampe FC, Thom SA, Poulter NR . QT intervals and QT dispersion as measures of left ventricular hypertrophy in an unselected hypertensive population. Am J Hypertens 2001; 14: 455–462.
Mayet J, Shahi M, McGrath K, Poulter NR, Sever PS, Foale RA, Thom SA . Left ventricular hypertrophy and QT dispersion in hypertension. Hypertension 1996; 28: 791–796.
Zoghi M, Gurgun C, Yavuzgil O, Akilli A, Turkoglu C, Kultursay H, Akin M . QT dispersion in patients with different etiologies of left ventricular hypertrophy: the significance of QT dispersion in endurance athletes. Int J Cardiol 2002; 84: 153–159.
Celik T, Iyisoy A, Kursaklioglu H, Turhan H . QT dispersion: electrophysiological holy grail or the greatest fallacy in the surface ECG? Int J Cardiol 2007; 117: 404–405.
Galetta F, Franzoni F, Magagna A, Femia FR, Pentimone F, Santoro G, Carpi A . Effect of nebivolol on QT dispersion in hypertensive patients with left ventricular hypertrophy. Biomed Pharmacother 2005; 59: 15–19.
Gonzalez-Juanatey JR, Garcia-Acuna JM, Pose A, Varela A, Calvo C, Cabezas-Cerrato J, de la Peña MG . Reduction of QT and QTc dispersion during long-term treatment of systemic hypertension with enalapril. Am J Cardiol 1998; 81: 170–174.
Lim PO, Nys M, Naas AA, Struthers AD, Osbakken M, MacDonald TM . Irbesartan reduces QT dispersion in hypertensive individuals. Hypertension 1999; 33: 713–718.
Oikarinen L, Nieminen MS, Toivonen L, Viitasalo M, Wachtell K, Papademetriou V, Jern S, Dahlöf B, Devereux RB, Okin PM, LIFE Study Investigators. Relation of QT interval and QT dispersion to regression of echocardiographic and electrocardiographic left ventricular hypertrophy in hypertensive patients: the Losartan Intervention For Endpoint Reduction (LIFE) study. Am Heart J 2003; 145: 919–925.
Piccirillo G, Viola E, Nocco M, Santagada E, Durante M, Bucca C, Marigliano V . Autonomic modulation and QT interval dispersion in hypertensive subjects with anxiety. Hypertension 1999; 34: 242–246.
Suzuki M, Nishizaki M, Arita M, Ashikaga T, Yamawake N, Kakuta T, Numano F, Hiraoka M . Increased QT dispersion in patients with vasospastic angina. Circulation 1998; 98: 435–440.
Ueda H, Hayashi T, Tsumura K, Yoshimaru K, Nakayama Y, Yoshikawa J . Intravenous nicorandil can reduce QT dispersion and prevent bradyarrhythmia during percutaneous transluminal coronary angioplasty of the right coronary artery. J Cardiovasc Pharmacol Ther 2004; 9: 179–184.
Yoshiga Y, Shimizu A, Yamagata T, Hayano T, Ueyama T, Ohmura M, Itagaki K, Kimura M, Matsuzaki M . Beta-blocker decreases the increase in QT dispersion and transmural dispersion of repolarization induced by bepridil. Circ J 2002; 66: 1024–1028.
Piccirillo G, Germano G, Quaglione R, Nocco M, Lintas F, Lionetti M, Moisè A, Ragazzo M, Marigliano V, Cacciafesta M . QT-interval variability and autonomic control in hypertensive subjects with left ventricular hypertrophy. Clin Sci (Lond) 2002; 102: 363–371.
Malik M, Batchvarov VN . Measurement, interpretation and clinical potential of QT dispersion. J Am Coll Cardiol 2000; 36: 1749–1766.
Perkiomaki JS, Ikaheimo MJ, Pikkujamsa SM, Rantala A, Lilja M, Kesaniemi YA, Huikuri HV . Dispersion of the QT interval and autonomic modulation of heart rate in hypertensive men with and without left ventricular hypertrophy. Hypertension 1996; 28: 16–21.
Tomiyama H, Doba N, Fu Y, Kushiro T, Hisaki R, Shinozaki Y, Kanmatsuse K, Kajiwara N, Yoshida H, Hinohara S . Left ventricular geometric patterns and QT dispersion in borderline and mild hypertension: their evolution and regression. Am J Hypertens 1998; 11: 286–292.
Hill JA . Electrical remodeling in cardiac hypertrophy. Trends Cardiovasc Med 2003; 13: 316–322.
Cerbai E, Barbieri M, Li Q, Mugelli A . Ionic basis of action potential prolongation of hypertrophied cardiac myocytes isolated from hypertensive rats of different ages. Cardiovasc Res 1994; 28: 1180–1187.
Shorofsky SR, Aggarwal R, Corretti M, Baffa JM, Strum JM, Al-Seikhan BA, Kobayashi YM, Jones LR, Wier WG, Balke CW . Cellular mechanisms of altered contractility in the hypertrophied heart: big hearts, big sparks. Circ Res 1999; 84: 424–434.
Tomaselli GF, Marban E . Electrophysiological remodeling in hypertrophy and heart failure. Cardiovasc Res 1999; 42: 270–283.
Frohlich ED . Fibrosis and ischemia: the real risks in hypertensive heart disease. Am J Hypertens 2001; 14: 194S–199S.
Marfella R, Nappo F, De Angelis L, Siniscalchi M, Rossi F, Giugliano D . The effect of acute hyperglycaemia on QTc duration in healthy man. Diabetologia 2000; 43: 571–575.
Baumert M, Schlaich MP, Nalivaiko E, Lambert E, Sari CI, Kaye DM, Elser MD, Sanders P, Lambert G . Relation between QT interval variability and cardiac sympathetic activity in hypertension. Am J Physiol Heart Circ Physiol 2011; 300: H1412–H1417.
Baillard C, Mansier P, Ennezat PV, Mangin L, Medigue C, Swynghedauw B, Chevalier B . Converting enzyme inhibition normalizes QT interval in spontaneously hypertensive rats. Hypertension 2000; 36: 350–354.
Lu HR, Yu F, Dai DZ, Remeysen P, De Clerck F . Reduction in QT dispersion and ventricular arrhythmias by ischaemic preconditioning in anaesthetized, normotensive and spontaneously hypertensive rats. Fundam Clin Pharmacol 1999; 13: 445–454.
Zipes DP . The long QT interval syndrome. A Rosetta stone for sympathetic related ventricular tachyarrhythmias. Circulation 1991; 84: 1414–1419.
Kaftan AH, Kaftan O . QT intervals and heart rate variability in hypertensive patients. Jpn Heart J 2000; 41: 173–182.
Yee KM, Lim PO, Ogston SA, Struthers AD . Effect of phenylephrine with and without atropine on QT dispersion in healthy normotensive men. Am J Cardiol 2000; 85: 69–74.
Hansen O, Johansson BW, Gullberg B . Metabolic, hemodynamic, and electrocardiographic responses to increased circulating adrenaline: effects of pretreatment with class 1 antiarrhythmics. Angiology 1991; 42: 990–1001.
Marfella R, Gualdiero P, Siniscalchi M, Carusone C, Verza M, Marzano S, Esposito K, Giugliano D . Morning blood pressure peak, QT intervals, and sympathetic activity in hypertensive patients. Hypertension 2003; 41: 237–243.
Podrid PJ, Fuchs T, Candinas R . Role of the sympathetic nervous system in the genesis of ventricular arrhythmia. Circulation 1990; 82: I103–I113.
Romano M, Di Maro T, Carella G, Cotecchia MR, Ferro G, Chiariello M . Relation between heart rate and QT interval in exercise-induced myocardial ischemia. Am J Cardiol 1985; 56: 861–862.
Dean JW, Lab MJ . Effect of changes in load on monophasic action potential and segment length of pig heart in situ. Cardiovasc Res 1989; 23: 887–896.
Hansen DE . Mechanoelectrical feedback effects of altering preload, afterload, and ventricular shortening. Am J Physiol 1993; 264: H423–H432.
Lab MJ . Contraction-excitation feedback in myocardium. Physiological basis and clinical relevance. Circ Res 1982; 50: 757–766.
Okutucu S, Karakulak UN, Sahiner L, Aytemir K, Demiri E, Evranos B, Fatihoglu SG, Kaya EB, Kabakci G, Tokgozoglu L, Ozkutlu H, Oto A . The relationship between circadian blood pressure pattern and ventricular repolarization dynamics assessed by QT dynamicity. Blood Press Monit 2012; 17: 14–19.
Potucek P, Klimas J . Chronotherapy of hypertension with combination treatment. Pharmazie 2013; 68: 921–925.
Antonakis V, Tsioufis C, Tsiachris D, Andrikou I, Fantaki M, Dagres N, Vrachnis N, Stefanadis C . Associations of hemodynamic load and ventricular repolarization in patients with newly diagnosed essential hypertension: a long-term follow-up study. J Clin Hypertens (Greenwich) 2014; 16: 219–224.
Curtis LH, Ostbye T, Sendersky V, Hutchison S, Allen LaPointe NM, Al-Khatib SM, Usdin Yasuda S, Dans PE, Wright A, Califf RM, Woosley RL, Schulman KA . Prescription of QT-prolonging drugs in a cohort of about 5 million outpatients. Am J Med 2003; 114: 135–141.
Gowda RM, Khan IA, Wilbur SL, Vasavada BC, Sacchi TJ . Torsade de pointes: the clinical considerations. Int J Cardiol 2004; 96: 1–6.
Khan IA, Gowda RM . Novel therapeutics for treatment of long-QT syndrome and torsade de pointes. Int J Cardiol 2004; 95: 1–6.
Patel C, Antzelevitch C . Pharmacological approach to the treatment of long and short QT syndromes. Pharmacol Ther 2008; 118: 138–151.
Karpanou EA, Vyssoulis GP, Psichogios A, Malakou C, Kyrozi EA, Cokkinos DV, Toutouzas PK . Regression of left ventricular hypertrophy results in improvement of QT dispersion in patients with hypertension. Am Heart J 1998; 136: 765–768.
Fogari R, Zoppi A, Maffioli P, Monti C, Lazzari P, Mugellini A, Derosa G . Effects of aliskiren on QT duration and dispersion in hypertensive patients with type 2 diabetes mellitus. Diabetes Obes Metab 2012; 14: 341–347.
Malmqvist K, Kahan T, Edner M, Bergfeldt L . Cardiac repolarization and its relation to ventricular geometry and rate in reverse remodelling during antihypertensive therapy with irbesartan or atenolol: results from the SILVHIA study. J Hum Hypertens 2007; 21: 956–965.
Porthan K, Viitasalo M, Hiltunen TP, Vaananen H, Dabek J, Suonsyrja T, Hannila-Handelberg T, Virolainen J, Nieminen MS, Toivonen L, Kontula K, Oikarinen L . Short-term electrophysiological effects of losartan, bisoprolol, amlodipine, and hydrochlorothiazide in hypertensive men. Ann Med 2009; 41: 29–37.
Botchway AN, Turner MA, Sheridan DJ, Flores NA, Fry CH . Electrophysiological effects accompanying regression of left ventricular hypertrophy. Cardiovasc Res 2003; 60: 510–517.
Robertson JI . Diuretics, potassium depletion and the risk of arrhythmias. Eur Heart J 1984; 5 (Suppl A): 25–28.
Tsunoda M, Takezawa K, Santa T, Ina Y, Nagashima K, Ohmori K, Kobayashi S, Imai K . New approach for measurement of sympathetic nervous abnormality in conscious, spontaneously hypertensive rats. Jpn J Pharmacol 2000; 83: 39–45.
Dias da Silva VJ, Ferreira Neto E, Salgado HC, Fazan R Jr. . Chronic converting enzyme inhibition normalizes QT interval in aging rats. Braz J Med Biol Res 2002; 35: 1025–1031.
Black MJ, Bertram JF, Johnston CI . Effect of angiotensin-converting enzyme inhibition on myocardial vascularization in the adolescent and adult spontaneously hypertensive rat. J Hypertens 2001; 19: 785–794.
Crabos M, Roth M, Hahn AW, Erne P . Characterization of angiotensin II receptors in cultured adult rat cardiac fibroblasts. Coupling to signaling systems and gene expression. J Clin Invest 1994; 93: 2372–2378.
Morris SD, Yellon DM . Angiotensin-converting enzyme inhibitors potentiate preconditioning through bradykinin B2 receptor activation in human heart. J Am Coll Cardiol 1997; 29: 1599–1606.
van den Heuvel AF, van Gilst WH, van Veldhuisen DJ, de Vries RJ, Dunselman PH, Kingma JH . Long-term anti-ischemic effects of angiotensin-converting enzyme inhibition in patients after myocardial infarction. The Captopril and Thrombolysis Study (CATS) Investigators. J Am Coll Cardiol 1997; 30: 400–405.
Garg S, Narula J, Marelli C, Cesario D . Role of angiotensin receptor blockers in the prevention and treatment of arrhythmias. Am J Cardiol 2006; 97: 921–925.
Wang YH, Shi CX, Dong F, Sheng JW, Xu YF . Inhibition of the rapid component of the delayed rectifier potassium current in ventricular myocytes by angiotensin II via the AT1 receptor. Br J Pharmacol 2008; 154: 429–439.
Chopra N, Knollmann BC . Cardiac calsequestrin: the new kid on the block in arrhythmias. J Cardiovasc Electrophysiol 2009; 20: 1179–1185.
Bers DM, Despa S, Bossuyt J . Regulation of Ca2+ and Na+ in normal and failing cardiac myocytes. Ann N Y Acad Sci 2006; 1080: 165–177.
Hasenfuss G . Alterations of calcium-regulatory proteins in heart failure. Cardiovasc Res 1998; 37: 279–289.
Marx SO, Reiken S, Hisamatsu Y, Jayaraman T, Burkhoff D, Rosemblit N, Marks AR . PKA phosphorylation dissociates FKBP12.6 from the calcium release channel (ryanodine receptor): defective regulation in failing hearts. Cell 2000; 101: 365–376.
Flesch M, Schiffer F, Zolk O, Pinto Y, Stasch JP, Knorr A, Ettelbrück S, Böhm M . Angiotensin receptor antagonism and angiotensin converting enzyme inhibition improve diastolic dysfunction and Ca(2+)-ATPase expression in the sarcoplasmic reticulum in hypertensive cardiomyopathy. J Hypertens 1997; 15: 1001–1009.
Takeishi Y, Bhagwat A, Ball NA, Kirkpatrick DL, Periasamy M, Walsh RA . Effect of angiotensin-converting enzyme inhibition on protein kinase C and SR proteins in heart failure. Am J Physiol 1999; 276: H53–H62.
Dartsch T, Fischer R, Gapelyuk A, Weiergraeber M, Ladage D, Schneider T, Schirdewan A, Reuter H, Mueller-Ehmsen J, Zobel C . Aldosterone induces electrical remodeling independent of hypertension. Int J Cardiol 2013; 164: 170–178.
Delpon E, Caballero R, Gomez R, Nunez L, Tamargo J . Angiotensin II, angiotensin II antagonists and spironolactone and their modulation of cardiac repolarization. Trends Pharmacol Sci 2005; 26: 155–161.
Macdonald JE, Kennedy N, Struthers AD . Effects of spironolactone on endothelial function, vascular angiotensin converting enzyme activity, and other prognostic markers in patients with mild heart failure already taking optimal treatment. Heart 2004; 90: 765–770.
Akers WS, Cross A, Speth R, Dwoskin LP, Cassis LA . Renin-angiotensin system and sympathetic nervous system in cardiac pressure-overload hypertrophy. Am J Physiol Heart Circ Physiol 2000; 279: H2797–H2806.
van den Meiracker AH, Boomsma F . The angiotensin II-sympathetic nervous system connection. J Hypertens 2003; 21: 1453–1454.
Buschmann G, Schumacher W, Budden R, Kuhl UG . Evaluation of the effect of dopamine and other catecholamines on the electrocardiogram and blood pressure of rats by means of on-line biosignal processing. J Cardiovasc Pharmacol 1980; 2: 777–795.
Clementy J, Samoyeau R, Coste P, Bricaud H . Study of the electrophysiological properties of intravenous bisoprolol in patients with and without coronary artery disease by programmed stimulation. J Cardiovasc Pharmacol 1990; 16 (Suppl 5): S169–S174.
Mougenot N, Mediani O, Lechat P . Bisoprolol and hydrochlorothiazide effects on cardiovascular remodeling in spontaneously hypertensive rats. Pharmacol Res 2005; 51: 359–365.
Moss AJ, Zareba W, Hall WJ, Schwartz PJ, Crampton RS, Benhorin J, Vincent GM, Locati EH, Priori SG, Napolitano C, Medina A, Zhang L, Robinson JL, Timothy K, Towbin JA, Andrews ML . Effectiveness and limitations of beta-blocker therapy in congenital long-QT syndrome. Circulation 2000; 101: 616–623.
Kawamura M, Munetsugu Y, Tanno K, Kobayashi Y . Carvedilol is effective and safe in combination with bepridil for persistent atrial fibrillation and decreases the QT prolongation induced by bepridil therapy. J Cardiovasc Pharmacol 2013; 61: 77–82.
Zambruni A, Trevisani F, Di Micoli A, Savelli F, Berzigotti A, Bracci E, Caraceni P, Domenicali M, Felline P, Zoli M, Bernardi M . Effect of chronic beta-blockade on QT interval in patients with liver cirrhosis. J Hepatol 2008; 48: 415–421.
Zhou Q, Xiao J, Jiang D, Wang R, Vembaiyan K, Wang A, Smith CD, Xie C, Chen W, Zhang J, Tian X, Jones PP, Zhong X, Guo A, Chen H, Zhang L, Zhu W, Yang D, Li X, Chen J, Gillis AM, Duff HJ, Cheng H, Feldman AM, Song LS, Fill M, Back TG, Chen SR . Carvedilol and its new analogs suppress arrhythmogenic store overload-induced Ca2+ release. Nat Med 2011; 17: 1003–1009.
Maruyama M, Xiao J, Zhou Q, Vembaiyan K, Chua SK, Rubart-von der Lohe M, Lin SF, Back TG, Chen SR, Chen PS . Carvedilol analogue inhibits triggered activities evoked by both early and delayedafterdepolarizations. Heart Rhythm 2013; 10: 101–107.
El-Sherif N, Turitto G . Electrophysiologic effects of carvedilol: is carvedilol an antiarrhythmic agent? Pacing Clin Electrophysiol 2005; 28: 985–990.
Raine AE, Vaughan Williams EM . Adaptational responses to prolonged beta-adrenoceptor blockade in adult rabbits. Br J Pharmacol 1980; 70: 205–218.
Raine AE, Vaughan Williams EM . Adaptation to prolonged beta-blockade of rabbit atrial, purkinje, and ventricular potentials, and of papillary muscle contraction. Time-course of development of and recovery from adaptation. Circ Res 1981; 48: 804–812.
Vincent GM, Schwartz PJ, Denjoy I, Swan H, Bithell C, Spazzolini C, Crotti L, Piippo K, Lupoglazoff JM, Villain E, Priori SG, Napolitano C, Zhang L . High efficacy of beta-blockers in long-QT syndrome type 1: contribution of noncompliance and QT-prolonging drugs to the occurrence of beta-blocker treatment "failures". Circulation 2009; 119: 215–221.
Funck-Brentano C, Coudray P, Planellas J, Motte G, Jaillon P . Effects of bepridil and diltiazem on ventricular repolarization in angina pectoris. Am J Cardiol 1990; 66: 812–817.
Manouvrier J, Sagot M, Caron C, Vaskmann G, Leroy R, Reade R, Ducloux G . Nine cases of torsade de pointes with bepridil administration. Am Heart J 1986; 111: 1005–1007.
van Wijk LM, van den Toren WE, van Gelder I, Crijns HJ, Ruegg P, Lie KI . Electrophysiological properties of isradipine (PN200-110) in humans. J Cardiovasc Pharmacol 1989; 14: 492–495.
Lamarre-Cliche M, Lacourciere Y, de Champlain J, Poirier L, Larochelle P . Does QTc interval predict the response to beta-blockers and calcium channel blockers in hypertensives? Heart Dis 2003; 5: 244–252.
Peters FP, de Zwaan C, Kho L . Prolonged QT interval and ventricular fibrillation after treatment with sublingual nifedipine for malignant hypertension. Arch Intern Med 1997; 157: 2665–2666.
Grossman E, Messerli FH . Effect of calcium antagonists on plasma norepinephrine levels, heart rate, and blood pressure. Am J Cardiol 1997; 80: 1453–1458.
de Champlain J, Karas M, Nguyen P, Cartier P, Wistaff R, Toal CB, Nadeau R, Larochelle P . Different effects of nifedipine and amlodipine on circulating catecholamine levels in essential hypertensive patients. J Hypertens 1998; 16: 1357–1369.
Hjemdahl P, Wallen NH . Calcium antagonist treatment, sympathetic activity and platelet function. Eur Heart J 1997; 18 (Suppl A): A36–A50.
Leenen FH, Myers MG, Joyner CD, Toal CB . Differential effects of once-daily antihypertensive drugs on blood pressure, left ventricular mass and sympathetic activity: Nifedipine-GITS versus felodipine-ER versus enalapril. Can J Cardiol 2002; 18: 1285–1293.
Karas M, Lacourciere Y, LeBlanc AR, Nadeau R, Dube B, Florescu M, Lamarre-Cliche M, Poirier L, Larochelle P, de Champlain J . Effect of the renin-angiotensin system or calcium channel blockade on the circadian variation of heart rate variability, blood pressure and circulating catecholamines in hypertensive patients. J Hypertens 2005; 23: 1251–1260.
Takami T, Shigematsu M . Effects of calcium channel antagonists on left ventricular hypertrophy and diastolic function in patients with essential hypertension. Clin Exp Hypertens 2003; 25: 525–535.
Sakata K, Shirotani M, Yoshida H, Nawada R, Obayashi K, Togi K, Miho N . Effects of amlodipine and cilnidipine on cardiac sympathetic nervous system and neurohormonal status in essential hypertension. Hypertension 1999; 33: 1447–1452.
Takahara A, Nakamura Y, Wagatsuma H, Aritomi S, Nakayama A, Satoh Y, Akie Y, Sugiyama A . Long-term blockade of L/N-type Ca(2+) channels by cilnidipine ameliorates repolarization abnormality of the canine hypertrophied heart. Br J Pharmacol 2009; 158: 1366–1374.
Avdonin V, Shibata EF, Hoshi T . Dihydropyridine action on voltage-dependent potassium channels expressed in Xenopus oocytes. J Gen Physiol 1997; 109: 169–180.
Gotoh Y, Imaizumi Y, Watanabe M, Shibata EF, Clark RB, Giles WR . Inhibition of transient outward K+ current by DHP Ca2+ antagonists and agonists in rabbit cardiac myocytes. Am J Physiol 1991; 260: H1737–H1742.
Lind L, Lithell H, Johansson CG, Morlin C, Ljunghall S . Electrocardiographic changes during antihypertensive treatment with diltiazem in relation to the achieved serum levels of the drug. Scand J Clin Lab Invest 1994; 54: 337–340.
Fauchier L, Babuty D, Poret P, Autret ML, Cosnay P, Fauchier JP . Effect of verapamil on QT interval dynamicity. Am J Cardiol 1999; 83: A810–801.
Aiba T, Shimizu W, Inagaki M, Noda T, Miyoshi S, Ding WG, Zankov DP, Toyoda F, Matsuura H, Horie M, Sunagawa K . Cellular and ionic mechanism for drug-induced long QT syndrome and effectiveness of verapamil. J Am Coll Cardiol 2005; 45: 300–307.
Milberg P, Reinsch N, Osada N, Wasmer K, Monnig G, Stypmann J, Breithardt G, Haverkamp W, Eckardt L . Verapamil prevents torsade de pointes by reduction of transmural dispersion of repolarization and suppression of early afterdepolarizations in an intact heart model of LQT3. Basic Res Cardiol 2005; 100: 365–371.
Rabkin SW . Diltiazem and verapamil lower blood pressure in the unanaesthetized rat through CNS mechanisms involving endogenous opioids. Clin Exp Pharmacol Physiol 1991; 18: 431–438.
Balasubramaniam R, Grace AA, Saumarez RC, Vandenberg JI, Huang CL . Electrogram prolongation and nifedipine-suppressible ventricular arrhythmias in mice following targeted disruption of KCNE1. J Physiol 2003; 552: 535–546.
Thomas G, Gurung IS, Killeen MJ, Hakim P, Goddard CA, Mahaut-Smith MP, Colledge WH, Grace AA, Huang C . Effects of L-type Ca2+ channel antagonism on ventricular arrhythmogenesis in murine hearts containing a modification in the Scn5a gene modelling human long QT syndrome 3. J Physiol 2007; 578: 85–97.
Rabkin SW . Comparison of indapamide and hydrochlorothiazide on spontaneous contraction of cardiomyocytes in culture: the effect on alterations of extracellular calcium or potassium. Gen Pharmacol 1993; 24: 699–704.
Rabkin SW . Effect of indapamide on cyclic adenosine 3′,5′-monophosphate signal transduction system in isolated adult rat cardiomyocytes from normal myocardium and cardiac hypertrophy. J Cardiovasc Pharmacol 1993; 22 (Suppl 6): S35–S41.
Farquharson CA, Struthers AD . Increasing plasma potassium with amiloride shortens the QT interval and reduces ventricular extrasystoles but does not change endothelial function or heart rate variability in chronic heart failure. Heart 2002; 88: 475–480.
Wong KY, Wong SY, McSwiggan S, Ogston SA, Sze KY, MacWalter RS, Struthers AD . Myocardial fibrosis and QTc are reduced following treatment with spironolactone or amiloride in stroke survivors: a andomised placebo-controlled cross-over trial. Int J Cardiol 2013; 168: 5229–5233.
de Gregorio C, Morabito G, Cerrito M, Dattilo G, Oreto G . Citalopram-induced long QT syndrome and torsade de pointes: role for concomitant therapy and disease. Int J Cardiol 2011; 148: 226–228.
Letsas KP, Alexanian IP, Pappas LK, Kounas SP, Efremidis M, Sideris A, Kardaras F . QT interval prolongation and torsade de pointes associated with indapamide. Int J Cardiol 2006; 112: 373–374.
Mok NS, Tong CK, Yuen HC . Concomitant-acquired Long QT and Brugada syndromes associated with indapamide-induced hypokalemia and hyponatremia. Pacing Clin Electrophysiol 2008; 31: 772–775.
Wang CP, Guo GB . Indapamide induced syncope in a patient with long QT syndrome. Pacing Clin Electrophysiol 2002; 25: 1397–1399.
Read MI, Andreianova AA, Harrison JC, Goulton CS, Sammut IA, Kerr DS . Cardiac electrographic and morphological changes following status epilepticus: effect of clonidine. Seizure 2014; 23: 55–61.
Tsutsui K, Hayami N, Kunishima T, Sugiura A, Mikamo T, Kanamori K, Yamagishi N, Yamagishi S, Watanabe H, Ajiki K, Murakawa Y . Dexmedetomidine and clonidine inhibit ventricular tachyarrhythmias in a rabbit model of acquired long QT syndrome. Circ J 2012; 76: 2343–2347.
Yeragani VK, Tancer M, Uhde T . Heart rate and QT interval variability: abnormal alpha-2 adrenergic function in patients with panic disorder. Psychiatry Res 2003; 121: 185–196.
Biermann J, Wu K, Odening KE, Asbach S, Koren G, Peng X, Zehender M, Bode C, Brunner M . Nicorandil normalizes prolonged repolarisation in the first transgenic rabbit model with Long-QT syndrome 1 both in vitro and in vivo. Eur J Pharmacol 2011; 650: 309–316.
Akagi T, Sarazawa K, Inai Y, Kitagawa M, Takahashi N, Hamanaka I, Yamazaki T, Takebe M, Hama N, Hiraoka Y, Ueda K, Nakazawa K, Matsumoto N . Continuous administration of nicorandil decreases QT dispersion during the chronic phase of acute myocardial infarction. Int Heart J 2006; 47: 351–361.
Acknowledgements
This work was supported by the Grant of European Regional Development Fund—Project FNUSA-ICRC (No. CZ.1.05/1.1.00/02.0123), grant APVV-0887-11 Molecular aspects of drug induced heart failure and ventricular arrhythmias from the Slovak Research and Development Agency, and grant 1/0564/13 from the Science Grant Agency (VEGA), Slovak Republic. Grant of European Regional Development Fund—Project FNUSA-ICRC (No. CZ.1.05/1.1.00/02.0123), grant APVV-0887-11 Molecular aspects of drug induced heart failure and ventricular arrhythmias from the Slovak Research and Development Agency, and grant 1/0564/13 from the Science Grant Agency (VEGA), Slovak Republic.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Klimas, J., Kruzliak, P. & Rabkin, S. Modulation of the QT interval duration in hypertension with antihypertensive treatment. Hypertens Res 38, 447–454 (2015). https://doi.org/10.1038/hr.2015.30
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/hr.2015.30
Keywords
This article is cited by
-
Prevalence of prolonged QT interval in patients with HCV-related chronic liver disease
The Egyptian Heart Journal (2019)
-
Novel insight into arrhythmogenic remodeling: a target for reversal
Hypertension Research (2017)
-
Reverse electrical remodeling following pressure unloading in a rat model of hypertension-induced left ventricular myocardial hypertrophy
Hypertension Research (2017)
-
Wirkung von SGLT-2-Inhibitoren auf eine gestörte ventrikuläre Repolarisation
Der Diabetologe (2017)
-
Effect of selective vagal nerve stimulation on blood pressure, heart rate and respiratory rate in rats under metoprolol medication
Hypertension Research (2016)